Abstract
This study aimed to investigate the potential beneficial effects of estrogen on nigrostriatal dopaminergic neuron degeneration in postmenopausal drug-naïve Parkinson’s disease (PD). Based on the ratio of lifetime estrogen exposure length to the total length of the estrogen exposure and deprivation period, postmenopausal women with drug-naïve PD were divided into low (n = 31) and high (n = 31) estrogen exposure ratio groups. We performed a comparative analysis of the striatal dopamine transporter (DAT) availability between the two groups. Additionally, we evaluated the longitudinal change in the levodopa equivalent dose per month using a linear mixed model. The motor symptoms were more severe in the low estrogen exposure ratio group than in the high estrogen exposure ratio group (P = 0.016). PD patients in the two groups had significantly lower DAT availability on all striatal sub-regions except for ventral striatum than did age- and sex-matched normal controls. When comparing the two groups, PD patients in the low estrogen exposure ratio group exhibited significantly lower DAT availability in the posterior putamen (P = 0.024) and in the ventral putamen (P = 0.036) than those in the high estrogen exposure ratio group. The estimated monthly levodopa equivalent dose changes were 10.9 in the low estrogen exposure ratio group and 7.1 in the high estrogen exposure ratio group with a significant interaction between the two groups (P = 0.001). These in vivo data provide indirect evidence showing that estrogen may elicit a beneficial effect on nigrostriatal dopamine neurons in PD.
Similar content being viewed by others
Introduction
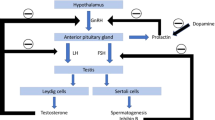
Epidemiologic studies have revealed that the prevalence of Parkinson’s disease (PD) is 1.5–2-fold lower in women than in men1,2, suggesting a possible protective influence of estrogen in predilection to the disease. Experimental evidences suggest that estradiol exerts neuroprotective effects on dopaminergic neurons and promotes dopaminergic activity in the striatum3,4. Observational studies have indicated that increased length of estrogen exposure period was correlated with less severe PD symptoms and that the risk of developing PD was increased in women with experience of surgical menopause5,6. These data suggest that estrogen may have a protective effect on dopaminergic neurons in PD.
However, several studies have demonstrated that the incidence of PD in women appears to vary depending on the formulation and dose of hormone therapy, timing and length of dosing period, and history of surgical or natural menopause7,8. Furthermore, one animal study reported that long-term estrogen deprivation by prolonged ovariectomy in primates might be an important determinant in maintaining nigrostriatal dopamine neurons9. This finding suggests that not only the total length of lifetime estrogen exposure, but also the total period of estrogen deprivation might be crucial factors to modulate the loss of dopaminergic neurons.
Until now, the potential beneficial effects of estrogen on nigrostriatal dopaminergic degeneration in patients with PD have not been investigated using in vivo neuroimaging. In this study, according to the ratio of estrogen exposure length to the total length of the estrogen exposure and deprivation period (Eratio), we performed a comparative analysis of the extent of presynaptic dopaminergic depletion by quantitatively measuring N-(3-[18F]fluoropropyl)-2β-carbon ethoxy-3β-(4-iodophenyl) nortropane (18F-FP-CIT) positron emission tomography (PET) in postmenopausal women with drug-naïve PD. Additionally, we evaluated the influence of Eratio on the longitudinal requirement of dopaminergic medications in these patients.
Methods
Subjects
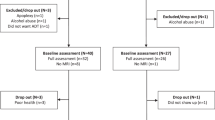
Subjects who visited the movement disorders clinic at Severance Hospital, Yonsei University from May 2010 to March 2018 were recruited from the database of the Yonsei Parkinson Center. The details of the enrollment of the study participants are illustrated in Fig. 1. Both 18F-FP-CIT PET scan and brain magnetic resonance imaging (MRI) were undergone within two months after initial visit to the clinic and before the start of dopaminergic medications. PD was diagnosed according to the clinical diagnostic criteria of the UK PD Society Brain Bank, and each subject exhibited decreased dopamine transporter (DAT) availability in the posterior putamen (PP) on the 18F-FP-CIT PET scans10. A total of 62 postmenopausal women out of 513 patients with drug-naïve PD were enrolled in this study.
To assess the severity of parkinsonian motor symptoms, the Unified Parkinson Disease Rating Scale Part III (UPDRS-III) was used. We calculated tremor and non-tremor scores for each patient according to the previously described method and classified patients into three subtypes: tremor dominant, akinetic-rigid, and mixed11. General cognition was scored using the K-MMSE score. The UPDRS-III and K-MMSE were measured at the initial visit to the clinic. Olfactory function was assessed with the Cross-Cultural Smell Identification Test (CCSIT), calculated as the sum of the correct responses. The Beck Depression Inventory (BDI) was administered to assess the severity of depression. The CCSIT and BDI were measured within one month after the first clinic visit. Age at menopause was estimated using the age at the last menstrual cycle or at the time of bilateral oophorectomy. Lifetime exposure to estrogen (estrogen exposure length) was the sum of difference between age at menopause and menarche (reproductive years) and years of estrogen replacement therapy (ERT) use12. The estrogen deprivation length was the difference between age at PD onset and estrogen exposure length. The estrogen exposure ratio (Eratio) was calculated by dividing estrogen exposure length by total length of estrogen exposure and deprivation period; (age at menopause − age at menarche + ERT duration)/(age at PD onset − age at menarche), as illustrated in Fig. S1. According to Eratio, patients were classified into two groups: the low Eratio group (n = 31) and the high Eratio group (n = 31) based on the distribution. The exclusion criteria included atypical parkinsonism, drug-induced parkinsonism, and evidence of focal brain lesions, diffuse white matter hyperintensities, or multiple lacunes in the basal ganglia as determined by MRI. In addition, we performed supplementary analysis to evaluate age-related decline in DAT availability. Considering the mean age at PD onset in the low and high Eratio groups (67.6 years and 55.9 years, respectively), female and male de novo PD patients whose mean age at PD onset was comparable to each Eratio group were obtained from our previous cohort group13. The female PD group comprised of two subgroups whose mean age at PD onset was 68.0 years (PD-old [PD-O], n = 81) and 55.8 years (PD-young [PD-Y], n = 83). Similarly, the male PD group included two subgroups whose mean age at PD onset was 67.4 years (PD-O, n = 86) and 55.9 years (PD-Y, n = 82). Normal controls consisted of 25 healthy female individuals with no neurological diseases. Considering the difference of mean ages at PD onset in low and high Eratio groups, we divided the control group (mean age, 61.4; n = 25) into two sub-groups whose mean ages were 67.4 years (control-old, n = 12) and 55.9 years (control-young, n = 13), which were comparable to mean ages at PD onset in the low and high Eratio groups. Then, we also performed a comparative analysis of DAT availability between the control-young and high Eratio groups as well as between the control-old and low Eratio groups, respectively. These subjects were selected from our institute and underwent 18F-FP-CIT PET and brain MRI. Eratio of the female PD-O and PD-Y groups was calculated by the information of reproductive factors obtained from the same questionnaire. This study was approved by the Yonsei University Severance Hospital ethical standards committee on human experimentation for experiments using human subjects and was therefore performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. Informed consent was waived because this study reviewed pre-existing data, which was approved by the Institutional Review Board of Yonsei University Severance Hospital (IRB No. 4-2014-0637).
Longitudinal assessment of the changes in the levodopa equivalent dose
PD medications were prescribed after confirming decreased DAT availability in PP on the 18F-FP-CIT PET scans. The starting point of PD medications was at second clinic visit, which was within three months after the first visit. After diagnosis of PD, two movement disorder specialists (P.H.L. and Y.H.S.) adjusted the doses of PD medications for symptom control at 3–6-month intervals. At each visit, the doses of PD medication were checked, and the levodopa equivalent dose (LED) was calculated based on previously described methodology14.
Quantitative analysis of the 18F-FP-CIT PET images
Image processing was performed using Statistical Parametric Mapping 12 (SPM12, Wellcome Department of Imaging Neuroscience, Institute of Neurology, London, England). For each patient, the reconstructed PET image was co-registered onto the corresponding MRI and normalized to the MNI152 template using normalization parameters defined from the corresponding MRI. Twelve volumes of interests (VOIs) of bilateral striatal sub-regions and one occipital VOI were drawn on the MNI152 template. The striatum was divided into right and left anterior/posterior caudate (AC/PC), anterior/ventral/posterior putamen (AP/VP/PP), and ventral striatum (VS) as previously described (Supplementary Methods)13. In the supplementary analysis performed by including our previous PD cohort group, we used the methodology for analyzing 18F-FP-CIT PET images that had been employed in a previous study10.
Statistical analysis
Data are expressed as means ± standard deviations. Demographic and reproductive characteristics were compared using a student’s t-test for continuous variables, and Pearson’s χ2 test for categorical variables. An analysis of covariance was performed to compare DAT availability in striatal sub-regions after adjusting for age at PD onset and education duration. When comparing the specific to non-specific binding ratios (SNBRs) of the striatal sub-regions between groups, P values were further corrected for multiple comparisons using a false discovery rate method. The LED changes were estimated using a linear mixed model that included the following covariates: Eratio group, age at PD onset, and baseline DAT availability in PP, time, and Eratio group × time interaction. Pearson’s partial correlation analysis controlling age at PD onset and education duration was conducted to evaluate the relationship between Eratio values and mean SNBRs on both sides of each striatal subregion. The statistical analyses were performed with SPSS (version 23.0; IBM Corporation, Armonk, NY, USA), and results with a two tailed P value of <0.05 were considered statistically significant.
Results
Demographic and reproductive characteristics
Demographic and reproductive characteristics of the low and high Eratio groups and normal controls are provided in Table 1. As expected, PD patients in the low Eratio group were older at PD onset (67.6 ± 1.3 vs 55.9 ± 1.3, P < 0.001) than those in the high Eratio group. The education duration was significantly longer in control-old and control-young groups than in the low and high Eratio groups. The low Eratio group had higher UPDRS-motor score (P = 0.016) than the high Eratio group. The proportion of PD subtypes did not differ between the two Eratio groups. The CCSI score was significantly lower in the low Eratio group than in the high Eratio group. Regarding reproductive characteristics, age at menopause in the low Eratio group was younger than in the high Eratio group (P = 0.002), whereas age at menarche, the proportion of ERT use, and surgical menopause did not differ between the two Eratio groups. The type of ERT formulation (estrogen-only or combined estrogen/progesterone therapy) was comparable between the two Eratio groups. Additionally, the timing at initiation of ERT was comparable between the groups. The estrogen exposure length was significantly shorter in the low Eratio group than in the high Eratio group (P < 0.001), and the total length of estrogen exposure and deprivation period was significantly longer in the low Eratio group than in the high Eratio group (P < 0.001).
SNBRs of PD subjects among the low and high Eratio groups and control groups
SNBRs for striatal sub-regions in patients with the two Eratio groups and control groups are presented in Table 2 and Fig. 2. When comparing DAT availabilities between the control-young and high Eratio groups and between the control-old and low Eratio groups, PD patients in the low or high Eratio groups also showed significantly lower SNBRs in all sub-striatal regions, except for ventral striatum (Table 2). In a direct comparison between the two Eratio groups, SNBRs in the low Eratio group were significantly lower in PP (P = 0.024) and VP (P = 0.036) than in the high Eratio group. This difference was pronounced in the more affected side of putamen than in the less affected side. However, SNBRs in other striatal sub-regions exhibited no significant difference between the two Eratio groups. In addition, when classifying two groups not based on Eratio but on estrogen exposure length itself, there was no significant difference in SNBRs on any striatal sub-regions between low and high Eratio groups.
Mean SNBRs of both sides of each striatal sub-region in PD subjects among the low and high Eratio groups and age- and sex-matched control groups. Mean SNBRs of both sides of each striatal sub-region in the low Eratio group were significantly lower in the PP and the VP compared to the high Eratio group. *P < 0.05. Abbreviations: SNBRs, Specific to non-specific binding ratios; PD, Parkinson’s disease; Eratio, estrogen exposure ratio; PP, posterior putamen; VP, ventral putamen.
SNBRs of male and female PD-Y and PD-O patients
To evaluate whether aging influences striatal DAT availability, we further analyzed striatal SNBRs in PD-Y and PD-O subgroups of both sexes. Demographic characteristics of these subgroups are provided in Table S1. Except for education duration, clinical parameters including symptom duration, UPDRS-motor score, and K-MMSE score were comparable between male and female PD-Y and PD-O subgroups. Reproductive factors including the age at menarche, age at menopause, duration of ERT use, and estrogen exposure length exhibited no difference between the female PD-Y and PD-O subgroups. As expected, the Eratio was higher in the female PD-Y group relative to the female PD-O group because of the difference in ages at PD onset. In contrast to sub-striatal DAT availabilities between the two Eratio groups, SNBRs in all striatal sub-regions did not differ between male PD-Y and PD-O subgroups. Similarly, SNBRs in all striatal sub-regions did not differ between the PD-Y and PD-O subgroups in female PD patients. SNBRs of all striatal sub-regions in the PD-Y and PD-O subgroups in both sexes are presented in Fig. 3 and Table S2.
Longitudinal changes in the LED during the follow-up period
The estimated monthly LED change was 10.9 in the low Eratio group and 7.1 in the high Eratio group, and there was significant interaction between the two groups and time in the linear mixed model, indicating that the monthly increase in the LED was significantly higher in the low Eratio group than in the high Eratio group (P = 0.001; Table 3).
Relationship between Eratio values and mean SNBRs on both sides of each striatal sub-region
Eratio values showed significant positive correlation with mean SNBRs on both sides of PP, VP, and AP (PP, P = 0.012; VP, P = 0.020; AP, P = 0.048). SNBRs in other striatal sub-regions exhibited no significant correlation with Eratio values (Table 4).
Discussion
The present study investigated whether estrogen exerts neuroprotective effect on nigrostriatal dopaminergic neurons in de novo PD patients using in vivo imaging. The major findings were as follows: (1) PD patients with higher Eratio exhibited less severe parkinsonian motor symptoms than those with lower Eratio. (2) PD patients in the high Eratio group had higher DAT availability in PP and VP than did those in the low Eratio group. Although the age at PD diagnosis was significantly lower in the high Eratio group relative to the low Eratio group, aging did not influence sub-striatal DAT availability in male or female PD subjects. (3) The longitudinal increase in the estimated monthly LED change was lower in the high Eratio group than in the low Eratio group. (4) Eratio values showed significant positive correlation with mean SNBRs on both sides of PP, VP, and AP.
In this study, we calculated Eratio based on not only lifetime estrogen exposure period, but also estrogen deprivation period. Previous studies have reported conflicting results on neuroprotective effects of estrogen exposure length on PD risk15,16. Several epidemiological studies reported that longer fertile life length is associated with decreased risk of PD in postmenopausal women17,18. On the other hand, it has been suggested that estrogen deprivation period after menopause could be a pivotal factor in the influence of estrogen effects on neurodegenerative disorders19. Likewise, our study showed that simple estrogen exposure period did not influence on DAT availability in any striatal sub-regions. Therefore, Eratio affected by both of the estrogen exposure and deprivation length could properly reflect estrogen-dependent pro-survival effect on dopaminergic neurons. Second, we performed a quantitative analysis of DAT availability using individual MRI co-registration to improve the accuracy of sub-striatal DAT patterns instead of a standard 18F-FP-CIT PET template acquired from healthy controls10.
The present study demonstrated that the high Eratio group had higher DAT availability in PP and VP than did the low Eratio group, and this difference was more pronounced in the more affected side than in the less affected side. It is well known that PP and VP, as the sensorimotor striatum20, receive projections from the dopaminergic neurons in the ventrolateral substantia nigra, and are connected to the primary motor and somatosensory cortices21. The results of the present study suggest that estrogen might exert a neuroprotective effect preferentially on nigrostriatal dopaminergic neurons that are primarily affected by PD pathology. Additionally, parkinsonian motor deficits were less severe in the high Eratio group relative to the low Eratio group, further supporting beneficial effect of estrogen on nigrostriatal dopaminergic system. Regarding pro-survival effects of estrogen, estrogen might suppress harmful inflammatory reactions, protect nigrostriatal dopaminergic neurons by regulating gene transcription, and target compensatory responses in surviving neurons to restore striatal functionality22,23,24.
Our data revealed an age discrepancy between the two Eratio groups, since age at PD onset was included as a parameter when grouping patients. Since the end point of estrogen deprivation period is age at PD onset according to the formula of Eratio, the low Eratio group with longer estrogen exposure and deprivation period had tendency to have older age at PD onset. This is the reason why the low Eratio group appeared to have older age at PD onset in this study. However, our study showed less motor severity and less dopamine depletion of sensorimotor striatum in the high Eratio group than in the low Eratio group, even though symptom durations before PD diagnosis were comparable. Generally, DAT availability in the striatum is decreased with age in normal subjects, at the mean rate of 5.3% per decade25,26. Considering that the difference in ages at PD onset between low and high Eratio groups is approximately ten years, our study showed 38.5% decline in mean SNBRs of posterior putamen and 19.3% decline in mean SNBRs of ventral putamen between the low and high Eratio groups. These percentages of decline in DAT availability between the groups were much greater than aging-related decline. Furthermore, in patients with PD, the effect of age on DAT availability in the striatum seems to be inconsistent; some reports demonstrated that the aging effects were significantly smaller in PD patients than in healthy subjects27, whereas others reported no aging effect on DAT availability in the putamen28,29,30. Therefore, to clarify whether beneficial effect of estrogen on dopaminergic neurons in the high Eratio group was secondary to the effect of age, we performed supplementary analysis by comparing DAT availability in PD-O and PD-Y groups of both sexes whose mean ages were comparable to each Eratio group. However, DAT availabilities in PD-O and PD-Y groups of both sexes did not differ in any striatal sub-regions. Especially, all reproductive factors including the estrogen exposure length were comparable between the female PD-O and PD-Y groups except for the mean ages at PD onset. Therefore, the difference of the estrogen exposure and deprivation length between the female PD-O and PD-Y groups was caused solely by the mean ages at PD onset, and subsequently resulted in the difference of Eratio values. Taken together, we can suggest that the effect of age alone does not lead to changes in DAT availabilities on the posterior and ventral putamen, but both estrogen exposure and deprivation length would have an impact on DAT availability selectively in the sensorimotor striatum.
Interestingly, the present study indicated that the longitudinal increase in the estimated monthly LED change during follow-up period was lower in the high Eratio group than in the low Eratio group. Although a longitudinal change in LED might not accurately reflect disease progression, LED appears to be indirectly associated with parkinsonian disability31. Regarding the prognostic role of estrogen, results of the present study may raise the possibility that estrogen can modulate longitudinal progression of parkinsonian motor deficits, although the difference in the longitudinal LED change may be secondary to the baseline difference in motor severity and sensorimotor striatal dopamine depletion.
There were several limitations in this study. First, this study was based on a questionnaire answered by postmenopausal participants who had to remember reproductive factors. Second, this study was based on a relatively small sample among a large cohort of more than 500 patients, which limits the generalization of our results possibly due to selection bias. However, we selected rigid inclusion and exclusion criteria to ascertain uniformity in clinical characteristics of enrolled patients, and strictly excluded those who could not remember their exact onset age of menarche and menopause to reduce recall bias as much as possible. In addition, because of small number of patients with surgical menopause or use of ERT, it was difficult to determine how these reproductive factors individually affect each striatal DAT availability. Especially, based on the difference in duration of ERT use between the two Eratio groups, a further study focusing on patients with PD who received ERT would be needed to uncover this issue. Third, nigral neuron counts are not associated with striatal DAT availability in PD32. Therefore, caution should be exerted in the interpretation of our result that striatal DAT availability may reflect axonal dysfunction or DAT expression rather than the number of viable neurons.
In conclusion, the present study demonstrated that Eratio was closely coupled with nigrostriatal DAT availability and longitudinal maintenance of dopaminergic medications in patients with postmenopausal de novo PD. These in vivo data provide indirect evidence that estrogen may have beneficial properties in PD.
Data Availability
For purposes of replicating procedures and results, any qualified investigator can request anonymized data after ethics clearance and approval by all authors.
References
Wooten, G. F., Currie, L. J., Bovbjerg, V. E., Lee, J. K. & Patrie, J. Are men at greater risk for Parkinson’s disease than women? J Neurol Neurosurg Psychiatry 75, 637–639 (2004).
Van Den Eeden, S. K. et al. Incidence of Parkinson’s disease: variation by age, gender, and race/ethnicity. Am J Epidemiol 157, 1015–1022 (2003).
Siani, F. et al. Influence of Estrogen Modulation on Glia Activation in a Murine Model of Parkinson’s Disease. Frontiers in neuroscience 11, 306 (2017).
Al Sweidi, S. et al. Oestrogen receptors and signalling pathways: implications for neuroprotective effects of sex steroids in Parkinson’s disease. Journal of neuroendocrinology 24, 48–61 (2012).
Cereda, E., Barichella, M., Cassani, E., Caccialanza, R. & Pezzoli, G. Reproductive factors and clinical features of Parkinson’s disease. Parkinsonism & related disorders 19, 1094–1099 (2013).
Rocca, W. A. et al. Increased risk of parkinsonism in women who underwent oophorectomy before menopause. Neurology 70, 200–209 (2008).
Popat, R. A. et al. Effect of reproductive factors and postmenopausal hormone use on the risk of Parkinson disease. Neurology 65, 383–390 (2005).
Liu, R. et al. Female reproductive factors, menopausal hormone use, and Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society 29, 889–896 (2014).
Leranth, C. et al. Estrogen is essential for maintaining nigrostriatal dopamine neurons in primates: implications for Parkinson’s disease and memory. J Neurosci 20, 8604–8609 (2000).
Oh, M. et al. Subregional patterns of preferential striatal dopamine transporter loss differ in Parkinson disease, progressive supranuclear palsy, and multiple-system atrophy. Journal of nuclear medicine: official publication, Society of Nuclear Medicine 53, 399–406 (2012).
Eggers, C., Kahraman, D., Fink, G. R., Schmidt, M. & Timmermann, L. Akinetic-rigid and tremor-dominant Parkinson’s disease patients show different patterns of FP-CIT single photon emission computed tomography. Movement disorders: official journal of the Movement Disorder Society 26, 416–423 (2011).
Park, H. K. et al. Lifetime exposure to estrogen and progressive supranuclear palsy: Environmental and Genetic PSP study. Movement disorders: official journal of the Movement Disorder Society 33, 468–472 (2018).
Yoo, H. S. et al. Presynaptic dopamine depletion determines the timing of levodopa-induced dyskinesia onset in Parkinson’s disease. European journal of nuclear medicine and molecular imaging 45, 423–431 (2018).
Tomlinson, C. L. et al. Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society 25, 2649–2653 (2010).
Rugbjerg, K., Christensen, J., Tjonneland, A. & Olsen, J. H. Exposure to estrogen and women’s risk for Parkinson’s disease: a prospective cohort study in Denmark. Parkinsonism & related disorders 19, 457–460 (2013).
Lv, M. et al. Reproductive factors and risk of Parkinson’s disease in women: A meta-analysis of observational studies. Behavioural brain research 335, 103–110 (2017).
Ragonese, P. et al. Risk of Parkinson disease in women: effect of reproductive characteristics. Neurology 62, 2010–2014 (2004).
Gatto, N. M. et al. Lifetime exposure to estrogens and Parkinson’s disease in California teachers. Parkinsonism & related disorders 20, 1149–1156 (2014).
Rodriguez-Perez, A. I., Borrajo, A., Valenzuela, R., Lanciego, J. L. & Labandeira-Garcia, J. L. Critical period for dopaminergic neuroprotection by hormonal replacement in menopausal rats. Neurobiology of aging 36, 1194–1208 (2015).
Parent, A. & Hazrati, L. N. Functional anatomy of the basal ganglia. I. The cortico-basal ganglia-thalamo-cortical loop. Brain Res Brain Res Rev 20, 91–127 (1995).
Lynd-Balta, E. & Haber, S. N. The organization of midbrain projections to the striatum in the primate: sensorimotor-related striatum versus ventral striatum. Neuroscience 59, 625–640 (1994).
Morale, M. C. et al. Estrogen, neuroinflammation and neuroprotection in Parkinson’s disease: glia dictates resistance versus vulnerability to neurodegeneration. Neuroscience 138, 869–878 (2006).
Sawada, H. & Shimohama, S. Neuroprotective effects of estradiol in mesencephalic dopaminergic neurons. Neuroscience and biobehavioral reviews 24, 143–147 (2000).
Gillies, G. E. & McArthur, S. Independent influences of sex steroids of systemic and central origin in a rat model of Parkinson’s disease: A contribution to sex-specific neuroprotection by estrogens. Hormones and behavior 57, 23–34 (2010).
Lavalaye, J., Booij, J., Reneman, L., Habraken, J. B. & van Royen, E. A. Effect of age and gender on dopamine transporter imaging with [123I]FP-CIT SPET in healthy volunteers. European journal of nuclear medicine 27, 867–869 (2000).
Kaasinen, V., Joutsa, J., Noponen, T., Johansson, J. & Seppanen, M. Effects of aging and gender on striatal and extrastriatal [123I]FP-CIT binding in Parkinson’s disease. Neurobiology of aging 36, 1757–1763 (2015).
Lee, C. S. et al. Uneven age effects of [(18)F]FP-CIT binding in the striatum of Parkinson’s disease. Annals of nuclear medicine 28, 874–879 (2014).
Lee, J. J., Ham, J. H., Lee, P. H. & Sohn, Y. H. Gender Differences in Age-Related Striatal Dopamine Depletion in Parkinson’s Disease. Journal of movement disorders 8, 130–135 (2015).
Booij, J., Bergmans, P., Winogrodzka, A., Speelman, J. D. & Wolters, E. C. Imaging of dopamine transporters with [123I]FP-CIT SPECT does not suggest a significant effect of age on the symptomatic threshold of disease in Parkinson’s disease. Synapse (New York, N.Y.) 39, 101–108 (2001).
Eising, E. G. et al. SPECT-evaluation of the monoamine uptake site ligand [123I](1R)-2-beta-carbomethoxy-3-beta-(4-iodophenyl)-tropane ([123I]beta-CIT) in untreated patients with suspicion of Parkinson disease. Journal of investigative medicine: the official publication of the American Federation for Clinical Research 45, 448–452 (1997).
Olanow, C. W. Levodopa: effect on cell death and the natural history of Parkinson’s disease. Movement disorders: official journal of the Movement Disorder Society 30, 37–44 (2015).
Saari, L. et al. Dopamine transporter imaging does not predict the number of nigral neurons in Parkinson disease. Neurology 88, 1461–1467 (2017).
Acknowledgements
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2016R1A2A2A05920131).
Author information
Authors and Affiliations
Contributions
The study was designed by Y.H. Lee and P.H. Lee. The statistical analysis was performed by Y.H. Lee, J.H. Cha, S.J. Chung, and H.S. Yoo. Drafting of the manuscript was performed by Y.H. Lee, J.H. Cha and P.H. Lee. The imaging data processing and analysis were carried out by J.H. Cha. The literature search, data interpretation, and revision of the manuscript were carried out by Y.H. Lee, J.H. Cha, S.J. Chung, H.S. Yoo, Y.H. Sohn, B.S. Ye, and P.H. Lee. P.H. Lee was involved in the final approval of the version to be published and takes responsibility for the integrity and accuracy of the data analyses.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, Y.H., Cha, J., Chung, S.J. et al. Beneficial effect of estrogen on nigrostriatal dopaminergic neurons in drug-naïve postmenopausal Parkinson’s disease. Sci Rep 9, 10531 (2019). https://doi.org/10.1038/s41598-019-47026-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-47026-6
This article is cited by
-
The identification of c-Abl inhibitors as potential agents for Parkinson’s disease: a preliminary in silico approach
Molecular Diversity (2024)
-
The impact of VPS35 D620N mutation on alternative autophagy and its reversal by estrogen in Parkinson's disease
Cellular and Molecular Life Sciences (2024)
-
A biological characterization of patients with postmenopausal Parkinson’s disease
Journal of Neurology (2024)
-
Mechanistic Insight into the role of Vitamin D and Zinc in Modulating Immunity Against COVID-19: A View from an Immunological Standpoint
Biological Trace Element Research (2023)
-
Sex and onset-age-related features of excessive daytime sleepiness and night-time sleep in patients with Parkinson’s disease
BMC Neurology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.