Abstract
Microbial activity is highly dependent on climatic factors (moisture and temperature) and edaphic characteristics in temperate ecosystems. Moreover, soil microbial community composition in high mountain areas is less known when compared to plant communities. In this study we investigated the soil microbial community from a functional perspective using PLFA (phospholipid fatty acid) methods in the four aspects of four summits (2,242 – 3,012 m above sea level) in the Spanish Central Pyrenees. Soil organic carbon (C), microbial biomass and nutrient dynamics (\(N{H}_{4}^{+}\) + \(N{O}_{3}^{-}\), N mineralization and nitrification potential) were also determined. Microbial biomass C was highest in the lowermost summit and decreased by approximately 50, 14 and 12% with increasing altitude. In each summit soil \(N{H}_{4}^{+}\) and \(N{O}_{3}^{-}\) concentrations differed significantly among summits and aspects. Soil nitrification potential varied significantly between the factors summit and aspects, e.g., southerly vs. northerly, easterly vs. westerly aspects. Gram negative bacteria and Actinobacteria functional groups dominated the microbial community, with almost 40% of the total PLFA. Non-metric multidimensional scale (NMS) analysis showed that most of the PLFA functional groups were present in all summits and aspects, although with specific biomarkers. A high abundance of biomarkers 16:1ω9c and 16:0 2OH (gram negative bacteria) were obtained in the lowermost summit, while the biomarkers 16.1ω7cDMA (anaerobes) and 19:3ω6c (Eukaryote) were only found in the uppermost summit. Linear mixed model (lmm) analysis was used with summit as fixed effect and aspect as random effect. In general, our results demonstrate a fundamental role for environment, principally moisture, temperature and organic matter in explaining the pattern observed for soil PLFA biomarkers. Under a global change scenario, we need to shed light on the relationships between soil microbial functional groups and soil nutrient-related variables in order to identify the associated patterns of decomposition rates and soil processes driven by microbial communities in mountain areas. The results could thus be used in global predictive models on climate change impact on C or N cycles in these environments.
Similar content being viewed by others
Introduction
Soil microorganisms are critical to C and N fluxes in terrestrial ecosystems1,2, as they thrive important processes related to nutrient cycling like mineralization of soil N for plant uptake. They are also highly dependent on climatic factors (moisture and temperature), edaphic characteristics such as clay mineralogy, and the physical and chemical characteristics of the decomposing resources3. Mountain ecosystems are fragile environments that play a role as early warning systems due to their sensitivity to global warming4. It is thus expected that soil functions like N mineralization and nitrification in these areas will be highly affected by global change (GC)5. Related GC factors like nitrogen deposition, elevated carbon dioxide (CO2) concentrations in the atmosphere, wind and water erosion, land use changes, and altered disturbance regimes are expected to affect plant community composition and cover, net primary productivity (NPP) and carbon (C) storage6,7. They will also affect soil decomposition rates by changing the composition and community structure of soil microorganism. Some studies have reported the effect of plant cover on microbial communities and/or their functions8 or on plant-soil interaction under GC context9,10,11,12, along altitudinal gradients, including arctic-alpine ecosystems13,14,15,16,17,18.
The ongoing reduction of snow cover in Alpine ecosystems by increasing temperatures due to global warming is predicted to affect plant dynamics and vegetation cover with progressive decline of cold mountain habitats and biota and a reshuffling of species on altitude gradients is to be expected as a consequence of individualistic species responses19. A warming effect called thermophilization, the process by which cold adapted plant species are progressively replaced by warm adapted species, has been described across a range of European mountainous areas19,20. The aspect of a slope has also been identified as a principal determinant of the pace of climate-induced plant migration processes21. In temperate regions of Europe, east- and south facing mountain slopes favour site-specific plant species richness, compared to the western and northern sides of the same mountains. Even at the highest altitudes plant productivity can be higher in the southern aspects compared to other aspects at same altitudes as a result of less cold temperatures, with shorter periods of snow cover and higher sunshine4. The upward migration of plants as a response to CC can be associated with concomitant changes in soil processes, such as decomposition, as an altered temperature regime can affect the turnover of soil nutrients and plant community structure. Consequently, the general reduction of plant productivity with increasing altitude occurring in mountain areas22 is expected to be counteracted in southern aspects through lower climatic constrains on organic matter (OM) decomposition.
In mountainous areas, abiotic factors such as erosive forces, thaw-freezing cycles prevail at high altitudes, while more anthropic driven factors like land husbandry are strong determinants of soil nutrient-related processes at lower altitudes. Nutrient-related processes in mountain soils with changes related to the forecasted temperature increase, rainfall pattern variations and plant cover dynamics in the next decades4 must be addressed.
The abundance of decomposer soil fungi, bacteria and actinobacteria is primarily driven by plant production, and hence mostly responsive to the quantity of plant inputs to soil23. Although some studies have documented the relationship among microbial biomass, microbial activity and N transformations in mountain summits24,25, in our research site no study has been done so far that relates microbial composition with soil environmental factors. Thus, more studies are needed to be used to inform predictive models on e.g. climate change impact on soil C or N cycles in these environments.
Baseline data are thus needed to be compared with future assessments of soil nutrient dynamics and related microbial community composition in mountain ecosystems. By so doing increased understanding of microbial mechanisms involved in SOM decomposition under changing climate will be achieved26,27,28. In four summits of Spanish Central Pyrenees we tested the hypothesis that (i) specific PLFA biomarkers varied across summits, (ii) available soil nutrient concentrations decreased with altitude and, (iii) microbial community composition and ecosystem processes like decomposition was influenced by a set of environmental factors across the accepted classical hierarchy of drivers8. Thus, the aim of this study was (i) to characterize the microbial community PLFA composition and its relation with soil N availability, N mineralization and nitrification potential.
Results
Soil properties
Total C, N and P decreased with altitude while pH increased (Table 1). The highest concentrations of total N were obtained in the two lower summits, while the lowest values were found in the two uppermost summits. Total P concentration was lowest in the two uppermost summits, i.e., less than 0.1 ppm. In the two lower summits, the lowest P concentration was found in ACU east and CUS north, while the highest values were found in ACU north and CUS west (Table 1). Soil pH was lowest in ACU west and highest in CUS north and OLA East. The analysis of soil elements showed that Ca2+, Mg2+ and K+ concentrations were highest at CUS west, while Na+ concentration was lowest. In general cation concentrations were lowest in the uppermost summit, OLA (Table 1).
SOC, MBC and MBN decreased with increasing altitude. Similarly, organic matter (OM) decreased with altitude, and the C:N ratio was lowest at TOB. The concentration of SOC ranged from 2.2 to 66.2 g C kg−1 at west aspects of OLA and ACU summits, respectively. MBC was highest at ACU west and CUS south, while MBN was highest in all aspects of ACU. Microbial biomass C decreased by approximately 50, 14 and 12% in CUS, TOB and OLA, respectively. The Cmic:Corg ratio did not follow a clear pattern. In fact, the lowest and highest Cmic:Corg values were obtained in the highest summit (Table 1). The significance of the lmm analysis for each soil variable is indicated in Table 2. The fixed effect summit yielded significant differences for most soil variables, except for Total P, C:N, inorganic C, percentage of sand and silt, and N:P and C:P ratios (Table 2).
Potential soil N mineralization rates
Significant differences were observed for \(N{H}_{4}^{+}\) concentration for the two factors considered (summits and aspect) and the interaction, while only the factor summit and the interaction was significant for \(N{O}_{3}^{-}\) concentration (Kruskal-Wallis ANOVA, p < 0.001; Table 3). Concentrations of \(N{H}_{4}^{+}\) were significantly higher in the two lowermost summits compared to the uppermost summits (Table 3), i.e. 11.9 and 9.5 mg kg−1 in the west aspects of ACU and CUS, respectively. The concentration of \(N{H}_{4}^{+}\) in the west aspect of ACU was significantly different to the other aspects (p < 0.05, Tukey HSD test), while it was lower in the west aspects of the uppermost summits. \(N{O}_{3}^{-}\) concentration was highest at CUS in the south aspect, i.e. 84.3 mg kg−1 (Table 3). In ACU, \(N{O}_{3}^{-}\) concentration was significantly highest in the west aspect (p < 0.05, Tukey HSD test). In TOB summit, no significant differences were found for \(N{O}_{3}^{-}\) concentration among aspects, and the lowest value was obtained at OLA, where it varied significantly among aspects (p < 0.05, Tukey HSD test).
Regarding short-term \(N{H}_{4}^{+}\) dynamics, low concentrations were observed and a flush of \(N{H}_{4}^{+}\) was detected at day 21, decreasing till day 60 (Fig. 1); this pattern was repeatedly observed for the rest of soil samples collected in the different aspects. A possible reason for the decreasing trend of \(N{H}_{4}^{+}\)-N content during the period of day 5 to 14 may be due to bacterial N immobilization or nitrification. On the contrary, \(N{O}_{3}^{-}\,\)increased rapidly up to 15 days in soils from ACU and CUS, the two lower summits, reaching a plateau that lasted from 30 to 60 days (Fig. 2). The patterns were slightly different for soils collected in the two uppermost summits, the \(N{O}_{3}^{-}\)-N decreased to day 7 and slightly increased from day 7 onwards at TOB, while \(N{O}_{3}^{-}\)-N concentration decreased until day 3 and then increased until day 60 (Fig. 2).
The potential net N mineralization, i.e. the net change in \(N{H}_{4}^{+}\) + \(N{O}_{3}\) between fresh and incubated samples, was calculated only for 21 days to avoid artifacts from the experimental setup. Soils collected in the uppermost summits had lower N cycling rates than those from the two lower summits as revealed by the potential net N mineralization and nitrification which decreased significantly with altitude (Table 3). Net N mineralization was significant for all summits, i.e. ACU (F3,11 = 88.56, p < 0.001), CUS (F3,11 = 12.86, p = 0.002), TOB (F3,11 = 14.70, p = 0.001) and OLA (F3,11 = 12.56, p = 0.002). The net potential nitrification was also significant in the four summits, i.e. ACU (F3,11 = 4.15, p = 0.048), CUS (F3,11 = 977.38, p < 0.001), TOB (F3,11 = 77.13, p < 0.001) and OLA (F3,11 = 17.39, p < 0.001). The cumulative inorganic N in the topsoil increased in the lowermost summits with higher cattle presence. Significant differences were found between aspects at each summit (Kruskal-Wallis ANOVA, p < 0.05).
Soil microbial community composition
Total PLFA biomass (total extracted lipid abundance) decreased with increasing elevation. Across summits it was noticeably higher in the lowermost summit (ACU) than in the rest of summits (Table 4), and ranged from 201.2 ± 110.7 nmol g−1 in the lowermost summit to 31.81 ± 17.5 nmol g−1 in the highest summit (average of all aspects). With regards to aspect, the highest PLFA biomass was observed in the western aspects in ACU, CUS and TOB, while in the uppermost summit (OLA), the highest PLFA biomass was found in the northern and eastern aspects (Table 4). In ACU west the functional groups Eukaryote, Gram negative, Gram positive, Actinobacteria, Fungi and arbuscular mycorrhizal fungi (AMF) had the highest abundance. Anaerobes were most abundant in ACU south. For all functional groups a general pattern was observed, a decrease in their abundance with increasing elevation (Table 4), with the highest values in the two lowermost summits, ACU and CUS.
When functional group ratios (fungal/bacteria and Gm+/Gm− bacteria) were correlated with soil variables no significant relationships appeared (Fig. 3). The fungal/bacteria ratio was lowest at TOB in the northern and highest in the western aspects, respectively. The Gm+/Gm− bacteria ratio was lowest in the southern aspect of OLA summit (0.26) than in the lowermost summit ACU, (0.62–0.84), reflecting the relative dominance of Gm−bacteria biomarkers at lower altitudes (not shown). Only PLFA biomass was positively correlated with nitrogen mineralization potential and nitrification and negatively correlated with soil pH (Fig. 3).
Fitted regression curves between PLFA biomass and Fungi/Bacteria ratio with selected soil variables: (a) correlation between PLFA biomass and nitrogen mineralization potential; (b) correlation for Na+ concentration in soil and Fungi/Bacteria ratio (not significant) (c) correlation between PLFA biomass and Nitrification potential, (d) idem for PLFA biomass and Nitrification potential and pH. Correlation coefficients (r), standard errors of estimates (sx) and F and P values are given. Initial soil variables and biomarkers were selected after multiple correlation analysis was performed (not shown). Fungal/biomass and Gram −/Gram+ ratios in soils collected in the four summits at “Ordesa and Monte Perdido” National Park.
The most abundant PLFA biomarkers for the different functional groups (Supplementary Table S1) were representatives of Gram positive (Gm+) bacteria with the branched group with iso or ante-iso methyl branching such as a15:0, i15:0, i16:0, a17:0 and 18:1ω9c; Gram negative (Gm−) bacteria biomarkers such as 16:1 ω7c, 17:0 cyclo ω7c, 18:1ω7c, 18:1ω5c, and 19:0 cyclo ω7c; the biomarker 18:2ω6c (fungi) and 16:1ω5 for AMF. There were also representatives of actinobacteria like 10-Me 16:0, 10-Me 17:1ω7c, 10-Me 18:1ω7c, 10-Me 18:0 and unspecified saturated lipids 16:0 and 18:0. In general, the microbial community was dominated by biomarkers of Gm – bacteria and actinobacteria (at least 40% of the total fatty acids in all summits and aspects). The percentage of fungi in these soils was less than 5% of the total fungal PLFA biomass. The largest amount of fungal PLFA biomass was measured in TOB western aspect while no fungi markers were found in the southern aspect of OLA (Table 4). AMF contributed to a small percentage of the total fungal PLFA biomass. Except for soils collected at the western aspect of TOB, more abundance of fatty acid biomarkers for bacteria than for fungi were present (the fungal/biomass ratio was below one).
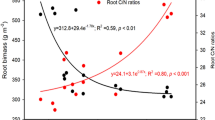
A significant relationship was found for some PLFA biomarkers and different soil variables across the altitudinal gradient (Fig. 4). We also explored the relationship between the microbial community composition at sampled sites and soil C concentration in different particle size fractions (<20, 20–53 and 53–250 µm) and mineralogy. This could provide further insights into decomposition processes in these areas. The data were taken from Jiménez and Villar29. Actually, a clear relationship was highlighted between Feo – Fep indicator, i.e. the content of ferrihydrite mineral30. An inverse relationship was observed between fungi other than AMF, only for 18:1ω9c, and soil pH (r = −0.626, p = 0.009; Fig. 4a) and positively with C concentration in < 20 µm size fraction (r = 0.620, p = 0.010; Fig. 4b). Soil C:N ratio was negatively associated with the abundance of the AMF biomarker 16:1ω5c (r = −0.557, p = 0.025; Fig. 4c), and also for the C:N ratio of the residues of 53–250 µm particle size fraction after hydrogen peroxide attack (r = −0.676, p = 0.010; Fig. 4d).
Fitted regression curves between fungal and AMF biomarkers 18:1ω9c, 16:1ω5c, 16:1ω7c and selected soil variables: (a) correlation between the abundance of 18:1ω9c biomarker and pH, (b) idem for 18:1ω9c biomarker and soil C concentration in the <20 µm size fraction, (c) idem for 16:1ω5c biomarker and C:N in the bulk soil, (d) idem for 16:1ω5c biomarker and the residues after peroxide attack of the 53–250 µm soil aggregates, (e) idem for 16:1ω5c biomarker and Feo – Fep mineral indicator (ferrihydrite) and (f) correlation between the abundance of 16:1ω7c biomarker and net nitrogen mineralization potential. Correlation coefficients (r), standard errors of estimates (sx) and F and P values are given. Initial soil variables and biomarkers were selected after multiple correlation analysis was performed (not shown).
Finally, the abundance of 16:1ω7c (biomarker for Gm− bacteria) was significantly correlated with N mineralization potential (Fig. 4f). The individual markers for Gm+ bacteria had a low predictive power when these were regressed together against N mineralization rates (data not shown).
Multivariate statistics ordination
The NMS yielded a final stress value (i.e., departure from monotonicity between the distance measure and the distance in ordination space) of 0.051 for PLFA biomarkers (Fig. 5), which is considered an excellent representation in reduced dimensions31.
Non-metric multidimensional scaling (NMS) of soil microbial PLFA community ordination and its significant relationship with soil variables. Axes were arbitrary and scaled in Bray-Curtis dissimilarity units. The significance was based on 999 permutations and only significant variables (p < 0.05) are shown. Arrow length refers to the strength of correlation.
The NMS ordination (Fig. 5) showed that soil microbial composition differed with summits, the majority of the functional groups were present in all summits and aspects, although with different PLFA biomarkers. Gram negative bacteria (16:1ω9c and 16:0 2OH) were abundant in the lowermost summit, ACU, while anaerobes (16.1ω7cDMA) and Eukaryote (19:3ω6c) biomarkers were characteristic in the highest summit, OLA. The most abundant biomarkers at TOB, the second uppermost summit, were 20:5ω3c, 20:1ω9c and 21.1ω3c (Fig. 5).
Soil variables that were significantly (corrected p after FDR procedure) associated with the NMS ordination are listed in Table 5. Most of the significant soil variables were grouped around ACU west: SOC, OM, nitrification potential and N mineralization potential, MBC, MBN, Soil moisture, June Tav15, June minTav15, June daily minimum T02–15, and AugT15, June daily minimum T15, June Tav2–15, Total N, pH, \(N{O}_{3}^{-}\) and K+. Soil pH and C:N were in opposition to the rest of soil variables. Actinobacteria were characteristic in the uppermost summit OLA together with specific PLFA biomarkers of anaerobes and eukaryotes.
Discussion
Soil microbial community structure and hierarchical pattern
Studies on the variations of soil microbial community composition and activity along altitudinal/climatic gradients and associated vegetation zones are not abundant. The soil microbial community structure varies with increasing elevation in mountain soils11,12,16. Decreasing activity of microorganisms with increasing altitude in an Alpine environment was observed32,33, and the fungal population and relative amount of Gm – bacteria increased with increasing elevation34. Djukic et al.4,25 found that neither microbial community size nor activity showed a consistent elevation trend in a climosequence from the montane to the subalpine and alpine climate/vegetation in the Austrian Alps (from 900 to 1900 m a.s.l.) They showed that the fungal PLFA biomarkers were significantly higher in the forest sites (between 9.2 and 6.7 mol%) compared to the shrubland and grassland sites (between 4.5 and 2.3 mol%). They concluded that with the expected upward migration of the treeline with climate warming, the abundance of fungi in the higher zones will increase. In a study conducted in disturbed sites of US logged forests, the observed decrease in fungal abundance with increasing altitude was correlated with decreased nitrogen mineralization35. Although soil microorganisms may be well adapted to the prevailing climate conditions in Alpine areas, no clear patterns are observed. In a recent study by Nottingham et al. (2018) the diversity of soil microorganisms was mean annual temperature along an altitudinal gradient in the Peruvian Andes and to a lesser extent soil pH and moisture. The most important drivers explaining the microbial community composition along the gradient are linked to soil humidity and soil temperature, pH and OM (Table 5). Our analysis showed that the best soil T indicator was minTav15 and dTmin2-15.
In our study, the effect of aspect was demonstrated for soil microorganism composition, i.e., total PLFA biomass was higher in the western aspects of ACU and TOB while for CUS and OLA the highest PLFA biomass was obtained in the southern and northern aspects, respectively (Table 4). No general pattern was observed in which the lowest values of PLFA biomarkers are found in the north aspects of each summit; in the lowest summit, ACU, values were lowest in north aspect compared to the rest, while in CUS these were found in the east aspect; fungi were lowest in TOB north, while eukaryote biomarkers were lowest in the east aspect, gram negative and actinobacteria were lowest in the south aspect and gram positive and AMF in the north aspect; in OLA the lowest values were found in the south aspect. Other studies, like Huang et al.36 reported an effect of slope orientation on soil microbial community composition, with higher relative abundance of Gm – bacteria and aerobes on the shady slopes of the Chinese plateau (1,400 m a.s.l.) In a forest of the Western Carpathians, the soil microbial community structure indices calculated from PLFA data1,37 did not vary along an altitudinal gradient (600–1200 m a.s.l.), similar to data reported by Winkler et al.21. In our study, on the contrary, the reasons may be due to the rocky structure in the southern facing slope of this summit and the low abundance of plants, especially in the southern and western facing slopes (Supplementary Table S2).
The composition of the soil microbial community is known to be controlled by a set of environmental factors, like soil pH, C:N ratio and plant community composition gradient, although not for fungal biomarkers25. In our study we found that climatic factors (soil moisture and T) was the main factor in the hierarchy structuring the microbial composition across the summits, followed by pH and OM. This is in agreement with Lavelle et al.38 who reported that climatic and edaphic determinants are likely to be more important in temperate ecosystems. In our study the abundance of PLFAs in the bulk soil was determined by soil pH and C:N ratio and the relative abundance of AMF biomarker 18:1ω9c was negatively correlated with soil pH.
In general, our results demonstrate a fundamental role for environment, principally moisture, temperature and OM (Table 4) in explaining the pattern observed for the community composition of soil PLFA biomarkers. In recent study undertaken in the Peruvian Andes Nottingham et al.39 found that the dominant factor explaining α-diversity for soil biota groups along an altitudinal gradient was mean annual temperature. The difference with our study is that we found that the best T indicator to explain the pattern observed was minTav15, i.e. June Tmin of daily average T recorded during 2015, when field work was performed. We would have expected a stronger effect of soil T, but it seems that in these summits soil moisture is the most important driver affecting soil biota. Whether these differences are the result of variations among temperate and tropical sites, more studies are thus needed to further address this hypothesis.
Microbial composition and activity has been demonstrated to shift along gradients in alpine ecosystems40. In the latter study, the shift in microbial community composition, including functional groups of Gm−, Gm+, and bacterial to fungal ratio, was linked to soil C:N ratio and pH, which is also in agreement with Yao et al.41. Soil C:N ratio was also found to be positively correlated with soil bacteria and fungal biomass37. Some authors have reported a positive correlation between Gm− and fungi and organic N, while anaerobes and actinobacteria were positively correlated with total N36. SOC and total N seemed to be the most important driver of microbial biomass and composition, similarly to results reported by Yao et al.41, who found a close relationship between microbial biomass, fatty acids and OM in Chinese soils. An increase in \(N{H}_{4}^{+}\)-oxidizing bacteria with increasing fertility of grassland soils was reported over a period of 40 years42.
Soil C availability is known to affect microbial activity and biomass differently43,44. Microbial biomass C is closely related to the growth of vegetation and to changes in the microorganism content of the soil36. Total N and \(N{O}_{3}^{-}\)-N levels were shown to be positively related to MBC and the amount of anaerobes and actinobacteria. Cmic:Corg ratio relates to the microbial activity and its potential to mineralize organic substances45. An increase in the microbial activity could result in increasing the MBC and soil organic N levels that are required for plant growth through the C and N cycles36. The change in substrate quality across vegetation succession has been hypothesized to presumably promote faster growing bacteria (particular Gm− bacteria) versus fungal mediated decomposition cycles46. In our study, however, AMF may have a key role, especially in the soils at the highest summit.
Soil chemical properties
The C:N and N:P ratios are considered important information source in ecosystems47. Nutrient limitation to plant growth, in particular, N and P availability has been shown to be major drivers of primary production. Plant productivity is limited in these alpine soils, i.e. the N:P or Redfield ratio, although difficult to interpret and rarely used in soil ecology, was below the threshold of 13.1 established by Cleveland and Liptzin48. Several studies with different plant communities have shown that N is limited when the N:P ratio is <14, and P is limited when the ratio is >1649. Plant productivity is reduced on northern aspects as temperature reduces, freezing periods and snow cover duration increase. This process determines C inputs to the soil, which can be low in alpine-nival environments50. In southern aspects, milder temperatures and shorter period of snow cover may allow slightly higher production of plant biomass.
A C:N ratio of <30 is considered as predictor for N mineralization while C:N ratios above this values can be used as predictor for initial net N immobilization51; in our study, soil N availability decreased in the uppermost summits. A decline of soil organic matter with increasing altitude was also reflected by decreasing concentrations of total C in soil that led to a narrowing of C:N ratios with increasing altitude, especially in TOB summit (Table 2).
N dynamics, mineralization and nitrification potential
Most plant available N (\(N{H}_{4}^{+}\)-N and \(N{O}_{3}^{-}\)-N) in natural ecosystems is the result of OM decomposition52. N mineralization or ammonification (\(N{H}_{4}^{+}\)-N) is the immediate product of this process, while nitrification (the conversion of \(N{H}_{4}^{+}\)-N to \(N{O}_{3}^{-}\)-N) is performed by specific groups of microorganisms53. Nitrification, is one of the key microbiological processes in the soil N cycle and is regulated mainly by temperature, soil moisture, input rates and quality of plant residues, as well as SOC availability54. Nitrification processes are altered by a set of abiotic and biotic factors, which can thus impact on N2O emissions to the atmosphere, which is why estimating nitrification potential is essential.
Some studies have reported that microbial biomass prefer \(N{H}_{4}^{+}\) to \(N{O}_{3}^{-}\,\)55,56, particularly when pH ranges between 6 and 7. The high variability observed in \(N{H}_{4}^{+}\)-N and \(N{O}_{3}^{-}\)-N concentrations and incubation data indicates a spatial and temporal dependence of soil nutrient dynamics in these summits, which are probably dependent of several factors (not specifically analysed in this study). Changes in ecosystem N dynamics along an altitudinal gradient in alpine-nival ecotone was demonstrated by Huber et al.24. They reported data from Caricion curvulae alpine pasture with nival plant groups of Androsacion alpinae on the southwest aspect of Mt. Schrankogel (Tyrol, Austria). The concentration of \(N{H}_{4}^{+}\)-N and \(N{O}_{3}^{-}\)-N ranged between 4 and 2 µg N g soil−1, respectively (from 2,906 to 3,079 m a.s.l., siliceous parent material). In our study, although the altitudinal variation is larger, almost 900 m between the lowest summit ACU and the highest summit OLA, similar values of \(N{H}_{4}^{+}\)-N concentration were obtained. The concentration was only higher in some aspects, between 9.1 and 11.9 µg N g−1 soil from the west facing slopes of ACU and CUS and the south facing slope of TOB (Table 2). In general, the two lower summits had higher concentrations of extractable N relative to the uppermost summits. In addition, low \(N{H}_{4}^{+}\)-N concentrations were found in the summits, resulting in limitations for mineral N uptake by plants, a limitation that can be linked to reduced microbial activity both in terms of biomass and number of PLFA biomarkers (Fig. 5). The lowest concentration of ions with increasing altitude shows that N mineralization rates decrease along the gradient, as observed by Huber et al.24. By contrast, the \(N{O}_{3}^{-}\)-N concentrations were much higher in our study, with a maximum of 84.3 µg N g−1 soil, than those obtained by the same authors, between 1 and 3 µg N g soil−1, and only in OLA summit differences were not markedly evident (Table 2). This indicates that nitrification is an important process in these summits, although this hypothesis should be further tested.
Under aerobic conditions \(N{H}_{4}^{+}\)-N can be rapidly used for microorganisms and be mineralized into \(N{O}_{3}^{-}\)-N for plant available N. The concentrations of \(N{H}_{4}^{+}\)-N in soils decreased with elevation reflecting decreasing rates of N mineralization with altitude. In our study, the highest \(N{H}_{4}^{+}\)-N concentration was 11.9 mg kg−1 in but in most summits and aspects the concentration was less than 5 mg kg−1, which might be related to the accessibility of \(N{H}_{4}^{+}\)-N by soil microorganisms. In the lowest summit no significant differences in the soil \(N{H}_{4}^{+}\)-N concentration were found between the northern, southern and eastern aspects (Table 4). As hypothesized, at the highest summits (TOB; OLA) the rates of net N mineralization and nitrification potential were lower than in the lowermost summits (ACU; CUS). Less available soil N was present at the higher summits.
C and N cycling processes like nitrification and N mineralization rates responded significantly to warming in mountain areas57,58. The expected increased temperatures can therefore profoundly influence soil processes in these areas by increasing nutrient availability59 and enhancing rates of N mineralization60,61, although climate manipulation studies have shown that microbial community structure was not affected at mid-term (4 years) in subalpine grassland soils62.
Conclusion
The data presented support the conclusion that concentration of elements decreased with increasing altitude, and that highest nutrient concentrations were obtained in the southern aspects of summits irrespective of altitude. Soil microbial biomass was highest in the lowermost summit. A set of hierarchically organized factors which regulate microbial composition and activity with climate (soil T) as the main factor structuring the microbial composition across the summits followed by OM and plant cover. Clear relationships were found between microbial community biomarkers and selected soil variables like pH, C:N soil mineralogy and N mineralization potential. The use of PLFA analysis in combination with NMS multi-variate statistics and linear mixed models has resulted in providing a clear fingerprint of the microbial community structure across summits. Although it is difficult to identify the individual influence of those biotic and abiotic parameters on the microbial community, the different summits and aspects were clearly separated in the multi-variate ordination.
As hypothesized in other studies, if the projected upward migration of plants in alpine environments due to thermophilization process is taking place, this will result in changes in the microbial community composition and therefore patterns of decomposition rates are expected to vary. The low abundances of functional groups (AMF, Eukaryote, Anaerobes, Fungi) in the uppermost summits may well be considered as indicators to follow changes linked to climate warming. Further studies are needed to explore the influence of above- and belowground biotic and abiotic factors on soil processes driven by microbial communities in these areas.
Methods
Site description
The study was conducted in Monte Perdido Massif, which is part of the Long-term Ecological Research (LTER) network, in four summits included in a pilot study area of GLORIA (Global Observation Research Initiative in Alpine Environments): “Punta Acuta” (ACU), 2,242 m; “Custodia” (CUS), 2,519 m; “Tobacor” (TOB), 2,779 m; and “Punta de las Olas” (OLA), 3,022 m a.s.l. (see Supplementary Fig. S1). All summits are within the area of the National Park, which is a Biosphere Reserve and World Heritage Site (UNESCO), and represented a gradient from the subalpine (2,242 m a.s.l.) to nival (3,012 m) vegetation belts and zonation with respect to altitude. Yearly average precipitation and temperature in the area are 1,720 mm and 5 °C, respectively (last 29 years at 2,200 m a.s.l.)63. The soil parent material comprised mainly calcareous substrates such as sandstone at ACU, CUS (the lowermost summits) and TOB and limestone at OLA (the uppermost ones). Soils are defined as humic Dystrocryept (Inceptisol) in the lowermost summits, and lithic Cryorthent (Entisol) in the uppermost summits.
Extensive domestic grazing (goat, sheep and cattle), primarily during the summer, has taken place for centuries, and is currently a permitted activity within the park. As a result, the upper tree line is ca. 300 m below its potential altitude as determined by climate64. Only scattered groups of Mountain pine (Pinus uncinata Ramond ex DC.) approach summit ACU at 2,180-2,200 m and no trees or shrubs are observed above.
The most dominant species in each summit are listed below:
ACU (lower alpine belt) – Festuca gautieri (Hack.), Festuca eskia Ramond ex DC.; Achillea millefolium L., Geranium cinereum Cav., Arenaria purpurascens Ramond ex DC. and Trifolium alpinum L.;
CUS (lower alpine belt) – Helictotrichon sedenense (Clarion ex DC.) Holub; F. gautieri (Hack.); G. cinereum Cav.; Poa alpina L; A. purpurascens Ramond ex DC.; Galium pyrenaicum Gouan, Lotus corniculatus L.; Trifolium thalii Vill. in CUS;
TOB (upper alpine belt) – Leucanthemopsis alpina (L.) Heywood; A. purpurascens Ramond ex DC.; G. pyrenaicum Gouan; Saxifraga oppositifolia L., H. sedenense (Clarion ex DC.) Holub; Thymus praecox Opiz; Saxifraga moschata Wulfen; Poa alpina L; G. cinereum Cav.; Festuca pyrenaica Reut.; T. thalii Vill.; Crepis pygmaea L.; Linaria alpina (L.) Mill.;
OLA (subnival belt) – Androsace ciliata DC.; S. oppositifolia L.; Saxifraga pubescens Pourr. subsp. iratiana (F.W. Schulz) Engl. & Irmscher; P. alpina L; Silene acaulis (L.) Jacq.; C. pygmaea L.; L. alpina (L.) Mill., Pritzelago alpina (L.) Kuntze; Minuartia cerastiifolia (Ramond ex DC.) Graebn.
While hiking activities and alpinism are normally present, there is little evidence of herbivory at the upper elevations.
Soil sampling
The sampling protocol is depicted in supplementary Fig. S2. All soil samples were taken in each aspect of the four summits (ACU, CUS, TOB, OLA) 15 m below the summit point, as vegetation inventories are conducted every 7 years within the GLORIA network65. Precautions were thus adopted to reduce human disturbance pressure in this nature reserve like avoiding excessive trampling in the area and reducing impact when soil cores were taken. Temperature loggers were installed in the topsoil (0–10 cm) of all summits and aspects in the summer of 2001.
Different soil samples were taken in this study based on the required explicative variables:
-
(a)
For classical soil physico-chemical analyses, a 500 g bulk soil sample was taken. Soil was first air dried and later sieved at 2-mm after visible root fragments and stones were manually removed.
-
(b)
Four soil samples (0–5 cm) were taken during the months of July in ACU,CUS and TOB summits and August in OLA (due to the presence of snow) of 2015 in each of the four cardinal points of each summit, i.e. North, South, East and West, following a transect of at least 10 m in each aspect (supplementary Fig. S2). This group of soil samples were transported to the lab in an ice cooler and stored at 4 °C to stop soil mineralization processes. These samples were used for microbial biomass carbon (MBC) and nitrogen (MBN) determinations and mineral N (\(N{H}_{4}^{+}\,\) and \(N{O}_{3}^{-}\)) dynamics.
-
(c)
Another set of four soil samples (0–5 cm) were taken within a radius of 10 m from which a composite sample was obtained and stored in plastic bags. These were preserved in a cooler in the field and transported the same day to the laboratory where they were sieved at < 2 mm mesh and then lyophilized (freeze-dried) and stored at −20 °C. These samples were used for microbial community characterization with phospholipid fatty acid (PLFA) analysis.
At the time of sampling an inventory of the present plant community was performed in each aspect of each summit. The inventory was performed 10 m below the established highest summit point to obtain a vegetation abundance/dominance index (Supplementary Material Table S2). More information about the vegetation sampling can be found in Pauli et al.65.
Soil physical and chemical properties
Soil pH in water (1:1 mass:volume ratio) was determined in air-dried < 2 mm sieved soil with a glass electrode. Soil texture was determined with the pipette method66 and total N with the Kjeldahl method67. Exchangeable base cations (Na+, K+, Ca2+ and Mg2+) and P (Mechlich) concentrations were analyzed by ICP-OES (Inductively Coupled Plasma Optical Emission Spectrometry, iCAP 6300 DUO, Thermo Electron Corporation, UK) after acid-digestion with hot HNO3. The gravimetric soil water content was calculated on a dry soil basis68 as:
The dry combustion method was used for assessment of C and N concentrations (0.5-g finely ground <200 μm subsamples) in a VarioMax CN Analyzer (Elementar GmbH, Hanau, Germany). Inorganic C was measured by CO2 evolution with HCl69, and soil organic carbon (SOC) was determined by subtracting the total inorganic C from total C. When inorganic C was not present total C was considered as total SOC.
The C:N and N:P ratios were also determined, the former depends on the plant community present56, while the second, the Redfield ratio, relates to plant productivity in a particular ecosystem70.
Microbial biomass C and N
The chloroform–fumigation extraction (CFE) method52 was used for determination of microbial biomass C and N. Total organic carbon (TOC) in fumigated and non-fumigated root-free soil subsamples was measured in 50 ml 0.5 M K2SO4 extracts of 25 g fresh soil (Shimadzu 500 TOC Analyzer). TOC in the non-fumigated extracts was assumed to be equal to the labile, extractable C pool. The difference in the flush of dissolved organic C between non-fumigated and fumigated samples (soil solution) allowed to estimate MBC after a proportionality constant (Kc = 0.38) was used. MBN was determined as the difference between extractable N in fumigated and non-fumigated samples using a correction factor (Kn) of 0.5471. Values were converted to a per gram soil dry weight (dw) basis52.
The Cmic:Corg ratio relates to the microbial activity and its potential to mineralize organic substances45.
Soil microbial community structure
Phospholipid fatty acids (PLFA) are used to characterize microbial community structure72,73,74,75,76. PLFAs were extracted from 2 g of lyophilized soil, separated and methylated77. The resulting fatty acids methyl esters (FAMEs) were separated by gas chromatography using an Agilent 7890 A GC System equipped with a HP-ULTRA 2 column (length 25 m, ID 0.20 mm; J&W Scientific Inc.) and a flame ionization detector. The individual FAME peaks were identified and quantified with the software Sherlock™ PLFA Method and Analysis Package (MIDI, Inc., Newark, DE) The internal standard 19:0 phosphatidylcholine (Avanti Polar Lipids, Alabaster, AL, USA) was used for quantification of FAMEs. Although most of bacterial PLFAs have acyl chain lengths of between 14 and 20 carbons, there are however fatty acids longer than 20 carbons that are considered to be predominantly of bacterial origin or from micro-eukaryotes78,79, like 21:0, 22:0, 22:5 ω3, 22:6 ω3, and 24:0). This software was designed to detect such fatty acids in soil samples, and therefore they were also taken into account in our study.
The viable microbial biomass was calculated by summing PLFAs concentrations and reported as nanomoles of PLFA per gram of soil. Specific PLFAs were used as biomarkers to quantify the relative abundances (mol%) of particular microbial groups80.
Several PLFAs may have various sources81. Fatty acids indicating fungi and arbuscular mycorrhizae fungi (AMF) were summed as total fungal biomass. The bacterial biomass was calculated from the residual fatty acids which could be assigned to bacterial groups. Biomass was expressed relative to dry weight of the freeze-dried soil. The ratios of fungal/bacterial (18:2ω6 for fungi) and Gram positive/Gram negative bacterial markers were also obtained. Detailed information on the PLFA pattern of soil microorganisms can be consulted76,82. We did not calculate diversity indices since there are flawed approaches and are not recommended81 although recent studies continue to use it83. There are essential reasons why diversity calculations should not be based on PLFA data. This is especially important when dealing with Fungi which have very few different PLFAs in their membranes. There can be thousands of different fungal species in an only be able to detect ten major types of fungal PLFAs81.
Mineral N and soil incubation for \(N{H}_{4}^{+}\,\)and \(N{O}_{3}^{-}\,\)determinations
Total N was determined with the Kjeldahl method. Soil mineral N content (plant available N, Navail) was extracted by shaking 4 g of subsamples of fresh soil stored at 4 °C with 40 ml of 1 m KCl solution for 30 min and after a 3-week incubation at room temperature while keeping soil moisture constant. The suspensions were later filtered and the extracts stored at −15 °C before analysis. The concentrations of ammonium (\(N{H}_{4}^{+}\)) and nitrate (\(N{O}_{3}^{-}\)) were determined in triplicate with standard colorimetric methods84. Water-soluble organic N was calculated as the difference between water-soluble N and inorganic N (\(N{H}_{4}^{+}\) plus \(N{O}_{3}^{-}\)) in deionized water extracts.
Potential net mineralization was calculated as the net change in \(N{H}_{4}^{+}\) and \(N{O}_{3}^{-}\) between fresh and incubated samples, while potential net nitrification was calculated as the net change in \(N{O}_{3}^{-}\), and time (t) the number of days in lab incubation. Accumulative net N mineralization and nitrification were calculated by summing the day-to-day accumulation of mineral N and concentrations in the incubated soils, respectively, and using the incubation days between the current sampling date and the previous sampling date, i.e., [\(N{H}_{4}^{+}\) + \(N{O}_{3}^{-}\)]0 and [\(N{H}_{4}^{+}\) + \(N{O}_{3}^{-}\)]21 are the respective concentrations of \(N{H}_{4}^{+}\) + \(N{O}_{3}^{-}\) (mg N kg soil−1) at t 0 and t 21 days. Soil fresh samples served as the initial samples for the course of incubation85.
Statistical analysis
In our study classical statistical analyses like ANOVAs were not possible for some variables. No independent samples were available since each soil sample was collected from a single monitoring unit, i.e. every aspect in each of the four summits and, thus, no degrees of freedom were left over for inference. Only for \(N{H}_{4}^{+}\) and \(N{O}_{3}^{-}\) determinations 4 field repetitions were available for each aspect of each summit. In this case, a two-way non-parametric Kruskal-Wallis ANOVA was performed with summit and aspect as main fixed factors between groups for soil \(N{H}_{4}^{+}\) and \(N{O}_{3}^{-}\) concentrations and specific incubation dates. When significant differences appeared the Tukey’s honestly significant difference (HSD) test at α = 0.05 was used for mean comparisons.
In mountain summits, there are rocks, cracks, fissures, etc. and this constraints the number of available areas for soil sampling. This pseudorreplication issue86 is not due to the study design but to the site conditions. In such case, for those samples that do not constitute real replicates, like in the case of PLFA determinations, a composite sample was used to capture the local variability. Linear mixed model analyses were used87 with summit as fixed effect and aspect as random effect (one datum for each aspect, 4 data for summit) for soil variables of which no true replicates were available. Aspect is nested within summit, i.e., within each summit there are 4 aspects (N, S, E, W).
Use of soil temperature (T) data
A correlation exists between a thermic vegetation indicator and habitat temperature across a range of European mountains19. They specifically showed that air temperature (T) in June was the best indicator. The strength of the correlation in our study area (ES-CPY) was in the limit of the significance (Spearman’s correlation rho = 0.6, p = 0.047). since dataloggers were installed on 31st July 2001 and recorded every hour we were able to use in the multivariate analysis 7 variables related to soil T in June (0–10 cm):
-
1.
Average T of June for the period 2002–2015 (Tav2–15);
-
2.
Average T of June in 2015 (Tav15);
-
3.
June Tmin of daily average T in 2015 (minTav15);
-
4.
June Tmin of daily average T for the period 2002–2015 (minTav2-15);
-
5.
Average of daily Tmin in 2015 (dTmin15);
-
6.
Average of daily Tmin for the period 2002–2015 (dTmin2-15);
-
7.
August mean soil T in 2015 (AugT15);
Data were averaged over June mean T values for the period 2002–2015 (available data), as this variable showed the best correlation. It is the temperature in the first part of the growing period which is most decisive for plant growth88. In the analysis we also used monthly mean T as two uppermost summits were not free of snow in June. Since we had all the data collected from the temperature loggers in all aspects and summits we wanted to precisely identify which variable of June temperatures could be the best indicator that influence microbial community composition and activity.
Multivariate ordination
Non-metric multidimensional scaling (NMS) is an iterative multivariate ordination technique that is suited to non-linear data that are not normally distributed31. The ordination is based on rank order information in a dissimilarity matrix to identify a space of minimum dimensionality where a satisfactory large number of the inter-point distances (corresponding to the dissimilarities) between every object is best represented geometrically89,90. These distances are regressed against the original distance matrix and the predicted ordination distances for each pair of samples is calculated. This regression fit (after 999 permutations) is called stress (scaled from 0 to 1) and reflects how well the ordination summarizes how far the distances in the reduced-space configuration are from the original distances91. A good rule of thumb is when stress values is <0.2, i.e., it provides an excellent representation in the reduced dimensions of the analysis.
NMS was performed on relative lipid abundance data (mol%) in order to eliminate the total PLFA biomass effect and focus our analysis on relative changes in microbial community structure. Only PLFAs with concentration higher than 0.5 mol% were used in the analysis74. PLFA relative abundances were used in NMS after arcsine transformations to meet normality assumption.
Probability correction
In both NMS analyses soil environmental and temperature variables (see below) were fitted to the PLFA ordination to search for correlations (after 999 permutations). Each test in the NMS yielded an initial p value which is then used to perform the corrections and search for significant differences at the corrected probability level. We used the false discovery rate (FDR) procedure92 to address multiple testing problems, i.e., as a mechanism to control the proportion of significant results that are in fact type I errors (‘false discoveries’) while simultaneously allowing type II errors to be reduced93. The procedure to adjust the significant α < 0.05 probability level is as follows:
where m is the number of tests and i is the test ranked in ascending order, i.e. P(1) ≤…..≤P(m). Starting with the highest p value each p is checked for this requirement; at the first p that meets the requirement its corresponding null hypothesis and all those having smaller p’s are rejected.
In our case, the final α’ corrected probability level was 0.026: Pcorr = 0.05*31/16 (the sixteenth ranked test yielded a p value lower than the initial p value).
All analyses were conducted with different packages included in the R statistical software94. The metaMDS function in the package vegan95 in R were used and the package mice was used to deal with NAs in the data matrix of soil temperatures. Graphical displays were performed with Sigmaplot software version 13.0 (Systat Software, Inc.).
References
Brookes, P. The soil microbial biomass: Concept, measurement and applications in soil ecosystem research. Microb & Environ. 16, 131–140 (2001).
Fraterrigo, J. M. et al. Nitrogen uptake and preference in a forest understory following invasion by an exotic grass. Oecologia 167, 781–791 (2011).
Garten, C. T. et al. Soil moisture surpasses elevated CO2 and temperature as a control on soil carbon dynamics in a multi-factor climate change experiment. Plant and Soil 319, 85–94 (2009).
IPCC. Summary for policymakers. In: Climate Change 2014. Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Field, et al. (eds)] Cambridge University Press, Cambridge. pp. 1–32 (2014).
Mills, R. T. E. et al. Diminished soil functions occur under simulated climate change in a sup-alpine pasture, but heterotrophic temperature sensitivity indicates microbial resilience. Sci. Tot. Environ. 473–474, 465–472 (2014).
Torn, M. S. et al. Storage and turnover of organic matter in the soil. In: Senesi, N., Xing, B. & Huang, P. M. (eds), Biophysico-Chemical Processes Involving Natural Nonliving Organic Matter in Environmental Systems, Cambridge University press, Cambridge. pp. 215–268 (2009).
Boutin, M. et al. Nitrogen deposition and climate change has increased vascular plant species richness and altered the composition of subalpine grasslands. J. Ecol. 105(5), 1199–1209 (2017).
Zinger, L. et al. Contrasting diversity patterns of crenarchaeal, bacterial and fungal soil communities in an alpine landscape. PLoS One 6(5), e19950 (2011).
Classen, A. T. et al. Direct and indirect effects of climate change on soil microbial and soil microbial‐plant interactions: What lies ahead? Ecosphere 6(8), 1–21 (2015).
Wookey, P. A. et al. Ecosystem feedbacks and cascade processes: understanding their role in the responses of Arctic and alpine ecosystems to environmental change. Glob. Change Biol. 15(5), 1153–1172 (2009).
Bragazza, L. et al. Linking soil microbial communities to vascular plant abundance along a climate gradient. New Phytologist 205(3), 1175–1182 (2015).
Wu, L. et al. Alpine soil carbon is vulnerable to rapid microbial decomposition under climate cooling. The ISME J 11(9), 2102–2111 (2017).
Roy, J. et al. Microbes on the cliff: alpine cushion plants structure bacterial and fungal communities. Frontiers Microb. 4, 64 (2013).
Siles, J. A. & Margesin, R. Abundance and diversity of bacterial, archaeal, and fungal communities along an altitudinal gradient in alpine forest soils: what are the driving factors? Microb. Ecol. 72(1), 207–220 (2016).
Qi, Q. et al. The biogeographic pattern of microbial functional genes along an altitudinal gradient of the Tibetan pasture. Frontiers Microb 8, 976 (2017).
Yuan, Y. et al. Bacterial community in alpine grasslands along an altitudinal gradient on the Tibetan Plateau. FEMS. Microb. Ecol. 87(1), 121–132 (2014).
Zak, D. R. & Kling, G. W. Microbial community composition and function across an arctic tundra landscape. Ecology 87(7), 1659–1670 (2006).
Zhang, B., Liang, C., He, H. & Zhang, X. Variations in soil microbial communities and residues along an altitude gradient on the northern slope of Changbai Mountain, China. PLoS One 8(6), e66184 (2013).
Gottfried, M. et al. Continent-wide response of mountain vegetation to climate change. Nat. Clim. Change 2(2), 111–115 (2012).
Pauli, H. et al. Recent plant diversity changes on Europe’s mountain summits. Science 336, 353–355 (2012).
Winkler, M. et al. The rich sides of mountain summits – a pan-European view on aspect preferences of alpine plants. J. Biogeog. 43, 2261–2273 (2016).
Hitz, C. et al. Below-ground and aboveground production of vegetational organic matter along a climosequence in alpine grasslands. J. Plant Nutr. & Soil Sci. 164, 389–397 (2001).
de Deyn, G. B., Quirk, H., Oakley, S., Ostle, N. & Bardgett, R. D. Rapid transfer of photosynthetic carbon through the plant-soil system in differently managed species-rich grasslands. Biogeosciences 8, 1131–1139 (2011).
Huber, E. et al. Shift in soil-plant nitrogen dynamics of an Alpine-nival ecotone. Plant Soil 301, 65–76 (2007).
Djukic, I., Zehetner, F., Mentler, A. & Gerzabek, M. H. Microbial community composition and activity in different Alpine vegetation zones. Soil Biol. Biochem. 42, 155–161 (2010).
Lipson, D. A. & Schmidt, S. K. Seasonal changes in an Alpine soil bacterial community in the Colorado Rocky Mountains. Appl. Environ. Microbiol. 70, 2867–2879 (2004).
Allison, S. D., Wallenstein, M. D. & Bradford, M. A. Soil-carbon response to warming dependent on microbial physiology. Nat. Geosci. 3, 336–340 (2010).
Budge, K., Leifeld, J., Egli, M. & Fuhrer, J. Soil microbial communities in (sub) alpine grasslands indicate a moderate shift towards new environmental conditions 11 years after soil translocation. Soil Biol. Biochem. 43, 1148–1154 (2011).
Jiménez, J. J. & Villar, L. Mineral controls on soil organic C stabilization in alpine and subalpine soils in the Central Pyrenees: insights from wet oxidation methods, mineral dissolution treatment and radiocarbon dating. Catena 149(1), 363–373 (2017).
Eusterhues, K. et al. Biodegradation of ferrihydrite-associated organic matter. Biogeochemistry 119, 45–50 (2014).
McCune, B. & Grace, J. B. Analysis of ecological communities. MjM Software Design, Gleneden Beach, Oregon, USA (2002).
Schinner, F. & Gstraunthaler, G. Adaptation of microbial activities to the environmental conditions in alpine soils. Oecologia 50, 113–116 (1981).
Schinner, F. Soil microbial activities and litter decomposition related to altitude. Plant and Soil 65, 87–94 (1982).
Margesin, R., Jud, M., Tscherko, D. & Schinner, F. Microbial communities and activities in alpine and subalpine soils. FEMS Microb. Ecol. 67, 208–218 (2009).
Fraterrigo, J. M., Balser, T. C. & Turner, M. G. Microbial community variation and its relationship with nitrogen mineralization in historically altered forests. Ecology 87(3), 570–579 (2006).
Huang, Y.-M., Liu, D. & An, S. -S. Effects of slope aspect on soil nitrogen and microbial properties in the Chinese Loess region. Catena 125, 135–145 (2015).
Klimek, B. Jelonkiewicz, Ł., Niklińska, M. Drivers of temperature sensitivity of decomposition of soil organic matter along a mountain altitudinal gradient in the Western Carpathians. Ecol. Res. 31, 609–615 (2016).
Lavelle, P. et al. A hierarchical model for decomposition in terrestrial ecosystems: Application to soils of the humid tropics. Biotropica 25(2), 130–150 (1993).
Nottingham, A. T. et al. Microbes follow Humboldt: temperature drives plant and soil microbial diversity patterns from the Amazon to the Andes. Ecology 99(11), 2455–2466 (2018).
Tscherko, D., Hammesfahr, U., Marx, M.-C. & Kandeler, E. Shifts in rhizosphere microbial communities and enzyme activity of Poa alpina across an alpine chronosequence. Soil Biol. Biochem. 36, 1685–1698 (2004).
Yao, H., He, Z., Wilson, M. J. & Campbell, C. D. Microbial biomass and community structure in a sequence of soils with increasing fertility and changing land use. Microb. Ecol. 40, 223–237 (2000).
Stienstra, A. W., Klein Gunnewiek, P. & Laanbroek, H. J. Repression of nitrification in soils under climax grassland vegetation. FEMS Microb. Ecol. 14, 45–52 (1994).
Allen, T. F. H. & Starr, T. B. Hierarchy, perspectives for ecological complexity. University of Chicago Press, Chicago, Illinois (1982).
Schmidt, M. W. I. et al. Persistence of soil organic matter as an ecosystem property. Nature 478, 49–56 (2011).
Anderson, T. H. & Domsch, K. H. Ratios of microbial biomass carbon to total organic carbon in arable soils. Soil Biol. Biochem. 21, 471–479 (1989).
Bardgett, R. D., Hobbs, P. J. & Frostegård, Å. Changes in fungal:bacterial biomass ratios following reductions in the intensity of management on an upland grassland. Biol. Fert. Soils 22, 261–264 (1996).
Wardle, D. A. et al. Ecological linkages between aboveground and belowground biota. Science 304, 1629–1633 (2004).
Cleveland, C. C. & Liptzin, D. C. N:P stochiometry in soil: is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 85, 235–252 (2007).
Koerselman, W. & Meuleman, A. F. M. The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. J. Appl. Ecol. 33(6), 1441–1450 (1996).
van Groenigen, K. J., Six, J., Hungate, B. A., de Graaff, M. A. & van Breemen, N. C. v. K. Element interactions limit soil carbon storage. Proc. Natl. Acad. Sci. USA 103, 6571–6574 (2006).
Stevenson, F. J. & Cole, M. A. Cycles of Soil: Carbon, Nitrogen, Phosphorus, Sulfur, Micronutrients, 2nd ed. Wiley, New York, 427 p. (1999).
Vance, E. D., Brookes, P. C. & Jenkinson, D. S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 19, 703–707 (1987).
Canfield, D. E., Glazer, A. N. & Falkowski, P. G. The evolution and future of Earth’s Nitrogen cycle. Science 330, 192–196 (2010).
Niklaus, P. A., Wardle, D. A. & Tate, K. R. Effects of plant species diversity and composition on nitrogen cycling and the trace gas balance of soils. Plant Soil 282, 83–98 (2006).
Geisseler, D., Horwath, W. R., Joergensen, R. G. & Ludwig, B. Pathways of nitrogen utilization by soil microorganisms – a review. Soil Biol. Biochem. 42, 2058–2067 (2010).
Fraterrigo, J. M., Turner, M. G., Pearson, S. M. & Dixon, P. Effects of past land use on spatial heterogeneity of soil nutrients in southern Appalachian forests. Ecol. Monog. 75, 215–230 (2005).
Grabherr, G., Gottfried, M. & Pauli, H. Climate effects on mountain plants. Nature 369, 448 (2009).
Jiang, L. et al. Alpine meadow restorations by non-dominant species increased soil nitrogen transformation rates but decreased their sensitivity to warming. J. Soils Sedim. 17(9), 2329–2337 (2016).
Michelsen, A. et al. Shoot biomass, δ13C, nitrogen and chlorophyll responses of two arctic dwarf shrubs to in situ shading, nutrient application and warming simulating climatic change. Oecologia 105, 1–12 (1996).
Hartley, A. E., Neill, C., Melillo, J. M., Crabtree, R. & Bowles, F. P. Plant performance and soil nitrogen mineralization in response to simulated climate change in subarctic dwarf shrub heath. Oikos 86, 331–343 (1999).
Shaw, M. R. & Harte, J. Response of nitrogen cycling to simulated climate change: differential responses along a subalpine ecotone. Global Change Biol. 7, 193–210 (2001).
Puissant, J. et al. Seasonal influence of climate manipulation on microbial community structure and function in mountain soils. Soil Biol. Biochem. 80, 296–305 (2015).
AEMET, Agencia Estatal de Meteorología [In Spanish].
Camarero, J. J. & Gutiérrez, E. Pace and pattern of recent treeline dynamics: Response of ecotones to climatic variability in Spanish Pyrenees. Climate Change 63, 181–200 (2004).
Pauli, H. et al. The GLORIA Field Manual – Multi-summit Approach. European Commission, Brussels (2004) (www.gloria.ac.at).
Bouyoucos, G. J. Hydrometer method improved for making particle size analysis of soil. Agron. J. 54, 464–465 (1962).
Kjeldahl, J. Neue Methode zur Bestimmung des Stickstoffs in organischen Korpern. Z. Anal. Chem. 22, 366–382 (1883).
Klute, A. Water retention: Laboratory methods. p. 635-662. In Klute, A. (ed.) Methods of soil analysis. Part 1. Physical and mineralogical methods. Agron. Monogr. 9. ASA and SSSA, Madison, WI (1986).
Nelson, D. W. & Sommers, L. E. Total carbon, organic carbon, and organic matter. In: Sparks, D. L. (ed.), Methods of Soil Analysis. Part 3: Chemical Methods: SSSA Book Series, No. 5, ASA-CSSA-SSSA. Madison, WI. pp. 961-1010 (1996).
Chapin, F. S., III, Matson P. A. & Mooney, H. A. Principles of Terrestrial Ecosystem Ecology. Springer, New York. 436 p. (2002).
Brookes, P. C., Landman, A., Pruden, G. & Jenkinson, D. S. Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol. Biochem. 17, 837–842 (1985).
Bradley, K., Drijber, R. A. & Knops, J. Increased N availability in grassland soils modifies their microbial communities and decreases the abundance of arbuscular mycorrhizal fungi. Soil Biol. Biochem. 38, 1583–1595 (2006).
Frostegård, A. & Bååth, E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol. Fertil. Soils 22, 59–65 (1996).
Moche, M. et al. Monthly dynamics of microbial community structure and their controlling factors in three floodplain soils. Soil Biol. Biochem. 90, 169–178 (2015).
Zelles, L. Phospholipid fatty acid profiles in selected members of soil microbial communities. Chemosphere 35, 275–294 (1997).
Zelles, L. Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: a review. Biol. Fertil. Soils 29, 111–129 (1999).
Buyer, J. S. & Sasser, M. High throughput phospholipid fatty acid analysis of soils. Appl. Soil Ecol. 61, 127–130 (2012).
Hangnail, D. & Getz, G. S. Phospholipids. In: Lipids and Biomembranes of Eukaryotic Microorganisms. Joseph A. Erwin (ed.) Academic Press, Inc. pp. 146–196 (1973).
Tunlid, A. & White, C. Biochemical analysis of biomass, community structure, nutritional status and metabolic activity of microbial communities in soil. In: Soil Biochemistry. Bollag, J. M., Stotzky, G. & Dekker, M. (eds) pp. 229–262 (1992).
Buckeridge, K. M., Banerjee, S., Siciliano, S. D. & Grogan, P. The seasonal pattern of soil microbial community structure in mesic low arctic tundra. Soil Biol. Biochem. 65, 338–347 (2013).
Frostegård, Å., Tunlid, A. & Bååth, E. Use and misuse of PLFA measurements in soils. Soil Biol. Biochem. 43, 1621–1625 (2011).
Ruess, L. & Chamberlain, P. M. The fat that matters: Soil food web analysis using fatty acids and their carbon stable isotope signature. Soil Biol. Biochem. 42, 1898–1910 (2010).
Li, X., Sun, J., Wang, H., Wang, J. & Zhang, H. Changes in the soil microbial phospholipid fatty acid profile with depth in three soil types of paddy fields in China. Geoderma 290, 69–74 (2017).
Maynard, D. G. & Kalra, Y. P. Nitrate and exchangeable ammonium nitrogen. In Martin R. Carter (ed.) Soil sampling and methods of analysis. Canadian Society of Soil Science. Lewis Publ., Boca Raton, FL. pp. 25–38 (1993).
Groffman, P. M., McDowell, W. H., Myers, J. C. & Merriam, J. L. Soil microbial biomass and activity in tropical riparian forests. Soil Biol. Biochem. 33, 1339–1348 (2001).
Davies, G. M. & Gray, A. Don’t let spurious accusations of pseudoreplication limit our ability to learn from natural experiments (and other messy kinds of ecological monitoring). Ecol. Evol. 5(22), 5295–5304 (2015).
Pinheiro, J. C. & Bates, D. M. Mixed Effects Models in S and S-Plus. Springer-Verlag, New York (2000).
Rammig, A., Jonas, T., Zimmermann, N. E. & Rixen, C. Changes in alpine plant growth under future climate conditions. Biogeosciences 7, 2013–2024 (2010).
Kruskal, J. G. Non-metric multi-dimensional scaling: a numerical method. Psychometrika 29, 115–129 (1964).
Gauch, H.G., Jr. Multivariate Analysis in Community Structure. Cambridge University Press, Cambridge (1982).
Legendre, P. & Legendre, L. Numerical ecology, 2nd ed. Elsevier, Amsterdam (1998).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate – a practical and powerful approach to multiple testing. J. Royal Stat. Soc. B 57, 289–300 (1995).
García, L. V. Controlling the false discovery rate in ecological research. Trends in Ecology and Evolution 18, 553–554 (2003).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria, ISBN 3-900051-07-0 http://www.R-project.org (2014).
Oksanen, J. et al. Vegan: Community Ecology Package. R Package Version. 2, 0–10 (2013).
Acknowledgements
This study is a contribution to the additional activities proposed within the framework of European network GLORIA (Global Observation Research Initiative in Alpine Environments). J. Azorín, S. Gutiérrez, S. Pérez and E. Ubieto are acknowledged for technical assistance in the lab and M. Gartzia for depicting study area map. The ARAID foundation is acknowledged for support to the first author. This manuscript benefited much from the useful and constructive comments raised by three anonymous referees in an earlier version.
Author information
Authors and Affiliations
Contributions
J.J.J. designed the study and collected soil samples together with L.V., J.M.I. performed PLFA analysis. J.L.B. provided soil T data and J.A. performed lab incubations. J.J.J. conducted statistical analyses together with J.M.I. and wrote the first draft of the paper. All authors worked on all subsequent versions of the paper and approved the final version.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiménez, J.J., Igual, J.M., Villar, L. et al. Hierarchical drivers of soil microbial community structure variability in “Monte Perdido” Massif (Central Pyrenees). Sci Rep 9, 8768 (2019). https://doi.org/10.1038/s41598-019-45372-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45372-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.