Abstract
The Patagonian Shelf Large Marine Ecosystem supports high levels of biodiversity and endemism and is one of the most productive marine ecosystems in the world. Despite the important role marine predators play in structuring ecosystems, areas of high diversity where multiple predators congregate remains poorly known on the Patagonian Shelf. Here, we used biotelemetry and biologging tags to track the movements of six seabird species and three pinniped species breeding at the Falkland Islands. Using Generalized Additive Models, we then modelled these animals’ use of space as functions of dynamic and static environmental indices that described their habitat. Based on these models, we mapped the predicted distribution of animals from both sampled and unsampled colonies and thereby identified areas where multiple species were likely to overlap at sea. Maximum foraging trip distance ranged from 79 to 1,325 km. However, most of the 1,891 foraging trips by 686 animals were restricted to the Patagonian Shelf and shelf slope, which highlighted a preference for these habitats. Of the seven candidate explanatory covariates used to predict distribution, distance from the colony was retained in models for all species and negatively affected the probability of occurrence. Predicted overlap among species was highest on the Patagonian Shelf around the Falkland Islands and the Burdwood Bank. The predicted area of overlap is consistent with areas that are also important habitat for marine predators migrating from distant breeding locations. Our findings provide comprehensive multi-species predictions for some of the largest marine predator populations on the Patagonian Shelf, which will contribute to future marine spatial planning initiatives. Crucially, our findings highlight that spatially explicit conservation measures are likely to benefit multiple species, while threats are likely to impact multiple species.
Similar content being viewed by others
Introduction
Large marine ecosystems (LMEs) are ecologically defined boundaries around the margins of continents that provide practical frameworks for ecosystem-based management at regional scales1. LMEs account for 75% of the global marine fish catches and contribute an estimated US$12.6 trillion in goods and services to the world’s economy2,3. They are, however, also hotspots of biodiversity loss and accelerated warming due to climate change3,4. Marine predators play important and diverse ecological roles in LMEs5,6. For example, seabirds and pinnipeds recycle and redistribute nutrients over large spatial scales, exert top-down control through predation and track the distribution of their prey5,7,8. Understanding the distribution of these taxa can therefore provide insights into processes that influence ecosystem structure and function that could otherwise be difficult to observe at regional scales. Moreover, marine predators often congregate in multi-species foraging aggregations, presumably a reflection of the spatiotemporal patchiness of their prey and perhaps the result of a higher incidence of generalism among marine predators compared to their terrestrial equivalents9,10. Consequently, localized threats are likely to impact multiple species, whilst spatially explicit conservation measures are likely to benefit multiple species. Hence, highlighting shared patterns in the spatial distribution of marine predators can identify ecologically significant areas that facilitates the conservation and management of not only marine predators, but also the wider ecosystem11,12. Advances in animal-borne tracking technology make this ever more practicable, enabling unprecedented insights into the space-use, foraging behavior and habitat preference of wide-ranging marine predators13. Although an explosion of movement ecology studies have facilitated the development of tracking datasets, integrated datasets from species and populations that exploit a range of habitats and ecosystem components (e.g., through diet and benthic/pelagic foraging mode) are still relatively rare, and knowledge of multi-species habitat use is typically poor, particularly in the context of LMEs9,10,11,12,14,15,16,17,18.
The Patagonian Shelf LME is one of the most productive marine ecosystems in the world19. Recognising its importance to biodiversity and regional economies, the Patagonian Shelf LME was the focus of a pioneering review of marine research and management20. The at-sea distribution of marine predators on the Patagonian Shelf has also received considerable attention and several studies have collated tracking datasets21,22. Nevertheless, early studies are heavily biased by the spatial usage of relatively few individuals, and until recently, limited tracking data were available for the Falkland Islands. This is a critical knowledge gap because, due to its proximity to the highly productive shelf edge and its many predator-free islands, the Falkland Islands are one of the most important locations on the Patagonian Shelf for colonial breeding marine predators. For example, the Falkland Islands are home to three quarters of the global population of black-browed albatross (Thalassarche melanophris), half of the global population of thin-billed prions (Pachyptila belcheri), half of the Atlantic population of South American fur seals (Arctocephalus australis), and one third of the global population of southern rockhopper penguins (Eudyptes chrysocome) and gentoo penguins (Pygoscelis papua)23,24,25,26,27. Despite the global significance of seabird and pinniped populations at the Falkland Islands, our understanding of shared patterns in their at-sea distribution is poor, recent advances notwithstanding12. Indeed, the movement ecology of many conspicuous species breeding at the Falkland Islands have only relatively recently been studied and the largest breeding colonies only recently tracked28,29,30,31,32. There is now an unprecedented opportunity to complement studies that have focussed on migration and individuals of unknown provenance12,21, and to quantify shared patterns in the at-sea distribution of colonial breeding marine predators at the Falkland Islands.
Given that many species selectively use habitats, habitat preference (selection relative to availability) can be defined by environmental variables, such as bathymetry and sea surface temperature (SST), which may provide useful proxies of resource availability10,33. In addition to habitat preference, colony location and density-dependent competition influence the at-sea distribution of colonial breeding marine predators26,29,34,35. For example, mutually exclusive colony specific foraging areas are common among colonial breeding marine predators in-part because the cost of foraging increases as a function of distance34,36,37,38. Accordingly, quantifying environmental and density-dependent factors will help to identify the drivers of spatial usage. In turn, this information can be used to predict the distribution of species more broadly, including from unsampled colonies37,39. In particular, Species Distribution Models (SDMs) can be used to quantify the dependence of space use on habitat (described by environmental variables) and accessibility. SDMs are increasingly being used to predict the distribution of animals around colonies that lack tracking data, using models fitted to data for observed colonies, and taking into account habitat accessibility and competition between animals from neighboring colonies26,33,37. Here, we aim to identify areas of high marine predator space use and diversity, which is an essential step in the management of dynamic marine ecosystems. To do so, we collated and modelled biotelemetry and biologging data for nine species of colonial breeding marine predators tracked from 21 breeding colonies at the Falkland Islands and predicted spatial usage for unsampled colonies. Given the diverse foraging guilds and foraging modes represented by these nine species, we expected that much of the area around the Falkland Islands would be used by at least some species.
Results
Biotelemetry and biologging data
In total, our dataset comprised locations from 1,891 foraging trips made by 686 animals (Table 1). Maximum foraging trip distance according to species ranged from 79 km for chick-rearing gentoo penguins to 1,325 km for incubating black-browed albatross (Table 1). Over 60% of sampled colonies (13 of 21) were in the north-east of the Falkland Islands (Fig. 1). The proportion of colonies from which animals were tracked for each species ranged from <10% for southern sea lions and gentoo penguins (3 of 71 and 6 of 81 colonies, respectively) to 30% for black-browed albatross (4 of 12 colonies). Rockhopper penguin and black-browed albatross foraging trips were significantly longer in distance during incubation than during chick rearing (rockhopper penguin Linear Mixed Model (LMM) F1,75 = 105, P < 0.001, black-browed albatross LMM F1,65 = 399, P < 0.001) (Table 1). Gentoo penguin foraging trips were significantly longer in distance during winter when compared to summer (LMM F1,30 = 19, P < 0.001).
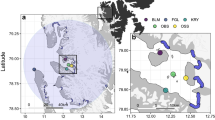
Panel (A) = locations of the 21 tracked breeding colonies (blue dots) at the Falkland Islands. Green dots are untracked colonies Panel (B) = important areas identified by overlap of 50% utilization distributions. Darker colours represent a higher number of overlapping utilization distributions. Thin black line is the 400 m bathymetric contour that marks the edge of the Patagonian Shelf, as well as the Burdwood Bank. Grey shading is the Falkland Islands Inner and Outer Conservation Zones, within which activities are managed and regulated by the Falkland Islands Government. Blue shading in the Argentinean Exclusive Economic Zone. SJI = Steeple Jason Island; NFUR = North Fur Island; SAU = Saunders Island; PBI = Pebble Island; CD = Cape Dolphin; CBG = Cape Bougainville; BSI = Big Shag Island; SEALB = Seal Bay; CB = Cow Bay; VPT = Volunteer Point; VRK = Volunteer Rocks; RHL = Rugged Hill; KDI = Kidney Island; DC = Diamond Cove; KELP = Kelp Island; BB = Berthas Beach; BLK = Bleaker Island; SLI = Sea Lion Island; BCI = Beauchene Island. BB = Burdwood Bank. Refer to Table 1 for details of the 13 groups for which utilization distributions are presented. Maps were created using R v3.5.2 and ArcMap v10.5.1.
Core areas used (50% UDs), extended as far north as 42°S, 1,000 km to the north of the Falkland Islands, and as far south as 56°S, 360 km to the south. Foraging trips were, however, predominantly confined to the Patagonian Shelf and the Burdwood Bank (Fig. 1). The highest overlap of core areas, both among species and groups, was the Patagonian Shelf to the north of the Falkland Islands (Fig. 1). In total, 83% of locations were associated with bathymetric depths less than 400 m (by species, the range was 67–99% of locations), which highlighted a preference for the Patagonian Shelf and Burdwood Bank for most species. The exceptions were southern elephant seals (Mirounga leonina), where 68% of foraging trip locations were associated with water depths between 400 m and 800 m (i.e., shelf slope waters), and rockhopper penguins, which foraged beyond the Patagonian Shelf and slope during incubation, in water >600 m deep (Figs 2, 3). Most foraging trip locations (74%) were within the Falkland Islands Conservation Zones. The remaining locations were within the Argentinean Exclusive Economic Zone (23%), and the high seas (3%). Our representativeness analysis indicated that curves failed to saturate for some species and some colonies, implying that the empirical 50% UDs underestimated the area used. Specifically, southern elephant seals, incubating black-browed albatross, chick rearing rockhopper penguins and Magellanic penguins (Spheniscus magellanicus) had representative scores <75% (Table 2). Representativeness scores ranged from 60 ± 12% to 96 ± 2% (Table 2).
Seabird predicted habitat use (probability of occurrence, proportional to the likelihood of absences) at the Falkland Islands and available biotelemetry and biologging data (blue line). MAG = Magellanic penguin, KP = king penguin during, RHP = rockhopper penguin, BBA = black-browed albatross, GEN = gentoo penguin, SSW = sooty shearwater. A = Chick rearing, B = Incubation for rockhopper penguin and black-browed albatross, and A = Summer, B = Winter for gentoo penguins. See Supplementary Material Fig S3 for species predictions weighted by colony.
Pinniped predicted habitat use (probability of occurrence, proportional to the likelihood of absences) at the Falkland Islands and available biotelemetry and biologging data (blue line). SAFS = South American fur seal A = winter, B = Spring, SES = southern elephant seal, SSL = southern sea lion. See Supplementary Material Fig S3 for species predictions weighted by colony.
Habitat modelling
Area Under the curve (AUC) scores ranged from 0.85 to 0.99 and indicated that our models adequately described the location data (Table 2). Moreover, specificity (range 0.67–0.97) and sensitivity values (range 0.72–0.99) indicated that models predicted space use accurately (Table 2). Of the seven candidate explanatory covariates considered, distance from the colony was retained in all models, capturing a negative relationship with space use (Table 2). Bathymetry was retained in all models except those for king penguins (Aptenodytes patagonicus) (Table 2). Occurrence decreased with distance from the breeding colony in all species and breeding stage/season (hereafter referred to as group, see Table 2), and typically decreased with bathymetric depth (Supplementary Information Fig S1). Black-browed albatross and Magellanic penguins had the most extensive predicted distribution, while the predicted distribution of chick rearing gentoo penguins and southern sea lions were comparatively small (Figs 2, 3). The predicted distribution of all species and groups, except rockhopper penguins during incubation, were concentrated on the Patagonian Shelf or shelf slope (Figs 2, 3). Models for species that travelled extended distances typically included dynamic environmental variables, in particular sea surface temperate. In contrast, for species that predominantly foraged near-shore, such as chick-rearing gentoo penguins, dynamic environmental variables were not retained in models. When all species and groups were combined, the probability of occurrence was high across a large area of the Patagonian Shelf (>1 million km2), which included the coastlines of Patagonia and Argentina, as far north as the Peninsula Valdés (Fig. 4). The Patagonian Shelf around the Falkland Islands and the Burdwood Bank had the highest areas of overlap in probability of occurrence and therefore, the highest predicted diversity (Fig. 4). Similar results were obtained when weighting the predictive outputs by colony size (Fig. 5). Individual species plots weighted by colony size are presented in Supplementary Material Fig S5.
As in Fig. 4, but weighted by colony size. Plots of (A) cumulative probability of occurrence (B) overlap in probability of occurrence.
Discussion
We analysed the most comprehensive tracking dataset compiled to date for wide-ranging marine predators breeding at the Falkland Islands. By modelling habitat selection as functions of colony distance, intra-specific competition and environmental indices, we predicted the at-sea distribution of animals from both sampled and unsampled colonies. In turn, this allowed us to identify not only high use areas for individual species but also areas likely to be used by multiple species. Our results therefore extend previous studies of marine predator distribution on the Patagonian Shelf that focus on migration or animals of unknown provenance and breeding status, and provide unprecedented insights into the distribution of colonial breeding marine predators on the Patagonian Shelf11,12,19,40. In particular, we provide novel insights into important areas on the Patagonian Shelf that transcend national boundaries and provide a more complete understanding of the distribution and co-occurrence of colonial breeding marine predators. For example, although the area to the east of the Burdwood Bank is highlighted as important habitat for migratory marine predators21,41, our predictive models highlighted the Burdwood Bank itself, which is known for its benthic biodiversity42, was also important habitat for marine predators breeding at the Falkland Islands. We also shed new light on the general ecology of our study species. In particular, gentoo penguin biotelemetry data revealed that individuals travelled extended distances during winter (480 km), challenging the widely held belief that gentoo penguins remain nearshore throughout their annual cycle43. In addition, the post-breeding movements of southern elephant seals were surprisingly short at the Falkland Islands, when compared to other breeding locations such as South Georgia (497 km versus 2,650 km, respectively) and thus they were included in our analysis of resident species44.
Although our predictive models generally performed well and our study improves knowledge of marine predator distribution on the Patagonian Shelf, caveats and constraints exist both with our modelling approach and the data available. Our models could have been extended by including additional terms to account for correlation structures inherent in the data, including (1) individual and colony-level slopes (random effects); (2) a term to model temporal/serial autocorrelation and (3) terms to describe spatial autocorrelation. However, including these terms has so far proved impracticable in studies that have analyzed datasets of similar size due to computational constraints and lack of convergence10,37,45. Even using parallel processing on multiple computers, Wakefield et al., (2017) found it feasible to only model colony-level random slopes. Bayesian methods such as integrated nested Laplace approximations (INLA) may soon make it possible to model spatiotemporal autocorrelation in such datasets, but at present such methods are not accessible to non-specialists. Given we used cross-validation and ROC curves, which are robust to violation of model assumptions, rather than likelihood ratio tests, we considered Generalized Additive Models (GAMs) to be a pragmatic and flexible modelling approach, suitable for our study aims that entailed predicting distributions, rather than hypothesis testing10,37,45. Furthermore, given our approach is relatively simple, it is accessible and easy to include more data as it becomes available, making it a potentially useful management tool when there is a need to combine ad-hoc tracking data.
In addition to model limitations, data were collected over different years, and for some species and some breeding colonies, the available tracking data were unlikely to fully represent core areas used based on our representativeness analysis. We do not know the degree to which inter-annual oceanographic variability influenced the at-sea spatial usage of our study species. Given data collection was opportunistic rather than systematic, inconsistencies in the temporal coverage of data between species could obscure potentially important shared areas of use. Ultimately, it would be ideal to track species at the same time, stratifying data collection across colonies within species, such that good geographical coverage was achieved, with more data collected from larger colonies. However, it is typically too costly to undertake simultaneous tag deployments on multiple species from multiple colonies. Finally, colony differences in habitat use are widely reported among marine predators, including those breeding at the Falkland Islands29,35,46,47. Spatial and temporal extrapolation are unavoidable and we cannot discount that within-species variability in foraging behaviour or habitat availability across relatively small spatial scales (tens to hundreds of km), could lead to poor model transferability31,48,49. Alternatively, model averages applied across a wide variety of habitats could lead to estimation bias49. While it would be useful for future studies to compare other modelling approaches, the spatial extent of the combined probability of occurrence, and the fact that distance was the most important variable in most models, implies that the species for which models were developed are broadly representative of the at-sea distribution of colonial breeding marine predators at the Falkland Islands.
Limitations notwithstanding, our results clearly demonstrated that the entire shelf area around the Falkland Islands, the Patagonian shelf slope and the Burdwood Bank, are important habitat for marine predators breeding at the Falkland Islands. Our findings extend that of a recent study that identified coastal waters around the Falkland Islands as important habitat for a suite of 36 seabird and pinniped species12. The large areas of shared spatial usage that we report, to some degree reflects the almost ubiquitous distribution of breeding colonies around the Falkland Islands for several study species, combined with long-distance movements of most study species. Furthermore, despite differences between species in foraging trip distance, dietary niches, foraging mode (e.g., benthic and pelagic), and breeding stage, most foraging trips were restricted to the Patagonian Shelf. The exceptions were southern elephant seal foraging trips, which were associated with the Patagonian shelf slope, and rockhopper penguins, which travelled beyond the Patagonian Shelf during incubation. The preference for shelf and slope waters is unsurprising given the Patagonian Shelf around the Falkland Islands is highly productive and is important habitat for marine predators migrating from distant breeding locations, such as South Georgia and New Zealand19,41,50. The region’s oceanography is dominated by the Falkland Current; a cold-water northward flowing current between 55°S and 37°S that originates from the Antarctic Circumpolar Current51. Shelf and shelf-break habitats likely provide predictable resources for marine predators breeding at the Falkland Islands, as is reported for marine predator communities in other regions such as the Kerguelen/Heard Plateau52. For example, the diet of albatrosses, petrels, pinnipeds, and penguins breeding at the Falkland Islands includes notothenid fish, ommastrephid squid, Falkland herring (Sprattus fuegensis) and lobster krill (Munida gregaria)43,53,54,55,56. The biomass of these species are typically an order of magnitude higher in neritic compared to oceanic waters, and the highest density of commercial fin-fish and squid catches also occurs on the Patagonian Shelf and slope57,58,59. Hence, the areas of overlap and high diversity that we identified are likely to represent high levels of marine predator and prey abundance and biodiversity for not only the species that were tracked, but for species such as gulls, cormorants and petrels, which were not tracked.
Given the preference for Patagonian Shelf waters around the Falkland Islands, it is unsurprising that distance and bathymetry were important explanatory covariates shared between species. The processes underlying the relationship between space use and colony distance are far from simple. For example, considerations include the energetic cost of travel to the adult, which could be greater on the return trip if animals are transporting food, the energetic cost of fasting/thermoregulating to the partner and offspring, competition (potential or realized) and social effects, such as network foraging which could be more effective close to the colony39. Nevertheless, while overly simplified, distance is an outcome of time and energy constraints that is common to all central place foragers, irrespective of their environment60. In contrast to static covariates, dynamic environmental covariates often only marginally improved model fit, or reduced model performance, as is reported for other colonial breeding marine predators during the central-place foraging period of their annual cycle37,39. This likely reflects the complexity of oceanography around the Falkland Islands, or ephemeral oceanographic features that aggregate species. For example, primary productivity is influenced by, among other factors, tidal movements, meso-scale fronts, quasi-stationary eddies and regions of upwelling in both summer and winter61. Hence, the spatial and temporal scales of dynamic environmental covariates may be too coarse to capture the environmental conditions that influence the distribution of marine predators, particularly when they are under strong central place foraging constraints10,62. Alternatively, understanding how dynamic environmental variables influence animal distribution may require a longer sampling period. In addition, space use and therefore habitat preference in relation to oceanography is, to some extent, decided by colony location in central place foragers, while competition can also shape the distribution of marine predators. Although we accounted for habitat availability and included an intra-specific density metric, density was not an important explanatory covariate in our models, and the influence of parapatric and sympatric competition is not always clear39. Nevertheless, colony location and competition with neighbouring colonies could further obscure direct links between dynamic environmental variables and the marine predators that we studied36,39,47.
In addition to being important habitat for marine predators, the Patagonian Shelf around the Falkland Islands also supports commercial industries including fisheries and ship-based tourism, and developing industries such as aquaculture and hydrocarbon exploration12. Indeed, the Falkland Islands economy is reliant on its marine environment (>60% GDP) and therefore it is essential that ecosystem-orientated approaches and marine conservation are based on robust empirical evidence and stakeholder inclusion12,63. In light of the loss of biodiversity within LMEs64, coupled with a need to address large-scale challenges with limited budgets, there has been considerable effort in recent years to generate, collate and synthesise the baseline data required for systematic and data driven ecosystem-orientated approaches at the Falkland Islands12,63. Our findings highlight that spatially explicit conservation measures are likely to benefit multiple species, but the suitability of measures will be scale dependent. Similarly, threats will impact multiple species. Given the importance of the Patagonian Shelf around the Falkland Islands and its complex hydrodynamics61,65, a more detailed understanding of finer scale oceanography and circulation within this region, could enable a better grasp of the dynamic environmental variables that influence marine predator distribution and may facilitate more precise predictions of distribution. Similarly, predictive models would benefit from longer-term research on the inherent variability of the relevant dynamic variables. Finally, our findings highlight that the distribution of marine predators on the Patagonian Shelf are not constrained by national jurisdictions. Clearly, the conservation and management of marine predators on the Patagonian Shelf would benefit from the continued development of strategic regional initiatives that integrate data and expertise from countries responsible for managing species throughout their Patagonian Shelf range22. Our study has extended prior knowledge of important shared areas of marine predator use for some of the largest populations of marine predators breeding on the Patagonian Shelf. Our findings will provide the baseline data required for future marine spatial planning initiatives at the Falkland Islands and therefore contribute to the management of the Patagonian Shelf LME.
Methods
Our focus was non-migratory movements, which included the winter movements of gentoo penguins and pinnipeds. We collated seabird and pinniped biotelemetry and biologging data from published and unpublished sources (Table 1; Supplementary Information Table S1). We also deployed biotelemetry and biologging tags (Argos satellite tags and FastlocGPS tags) on penguins (gentoo, Magellanic, rockhopper) and pinnipeds (fur seals, sea lions) between 2014 and 2017 (Supplementary Information Table S1).
Biotelemetry and biologging data analysis
We first identified individual foraging trips based on distance to land. While this was straight forward for most species, data exploration revealed large temporal gaps in gentoo penguin Argos location data, which made trip delineation difficult. Specifically, consecutive points could have represented separate foraging trips, leading to multiple trips being classed as a single foraging trip. To identify gentoo penguin foraging trips, we determined whether gentoo penguins could have returned to land between consecutive location points using a mean swim speed of 6 km/h. We were typically only able to identify multi-day foraging trips for gentoo penguins.
Location data were projected into Lambert Azimuthal Equal Area projection prior to analysis. Argos location data, Fastloc GPS and GPS data were first speed filtered (3 m/s for swimming animals, 20 m/s for flying seabirds), to remove obvious location outliers using the R package ‘Argosfilter’46,66. Argos locations have variable location errors, falling into six classes (3,2,1,0,A,B). In order to more accurately estimate the locations of each Argos-tracked animal along its trajectory, we fitted a continuous-time correlated random walk model, implemented through the R package ‘crawl’ (v2.1.1)67. Similarly, we also used ‘crawl’ to process data from Fastloc GPS tags, which provide both Argos and Fastloc GPS locations. To increase the temporal coverage of location data, we combined Fastloc GPS and Argos data, given these data are complementary46. Fastloc GPS locations associated with <4 satellites were removed prior to analysis46. We assumed Fastloc GPS locations were true locations within the model, given that error is typically <100 m46,68. The model estimated a best-fit track, with locations predicted hourly. GPS locations, which in the context of our study have negligible error, were linearly interpolated to hourly intervals.
To summarize the observed distribution of individuals at-sea, we split location data into groups according to species; colony; sex (if available); breeding status (breeding, not-breeding); and where appropriate, breeding stage. We used the R package adehabitatHR to estimate the utilization distribution (UD) of each group69. Specifically, we calculated the kernel density of locations for each individual and then normalized this to sum to one, using bathymetry as a mask to prevent UDs being estimated over land. We then averaged individual UDs by breeding colony, to create colony-level UDs. We excluded three colonies from which only one individual was tracked. To identify core areas of use, we calculated the 50% isopleth for each colony. We defined the kernel smoothing parameter h as the mean area-restricted search scale, which was calculated using First Passage Time analysis (assessed from 1 km to 150 km) averaged across all foraging trips for each group, and implemented using the fpt function in the R package adehabitatLT11. Finally, we identified shared areas of use across groups, by merging (adding) the 50% UDs of each colony, species and group11.
To obtain a measure of confidence in the data used for predictive modelling, we assessed whether UDs were representative of each breeding colony by calculating saturation curves based on 50% UD (i.e., core areas used). Specifically, for each species, group and breeding colony, we randomly selected an increasing number of foraging trips, with the mean area of the 50% UD and confidence interval for each step calculated from 500 iterations (a compromise between iterations and computing time). The mean area of the 50% UD was then modelled as a nonlinear asymptotic regression. We calculated a ‘representative value’ by taking the UD area of the species, and dividing it by the UD area at the asymptote11,14. High values (≥75%) were considered representative.
Habitat modelling
The procedure described above produced UD estimates for animals from colonies at which tags were deployed. These UDs are unlikely to adequately reflect the space use of animals from other colonies, especially distant colonies. For example, gentoo penguins and southern sea lions (Otaria flavescens) breed at over 70 sites around the Falkland Islands, but were tracked from only 6 and 3 sites, respectively (Supplementary Information, Fig S2). To assess habitat use and to predict the space use of animals from unobserved colonies, we modelled habitat selection using GAMs. Although tracking data are inherently correlated within individuals, the size of our dataset made it impractical to model individual random effects, and there is arguably little to be gained by including random effects, as is reported in other recent modelling studies (please refer to Supplementary Information for additional discussion on the use of GAMs)10,37,45. In brief, although GAMs assume independence of errors, the cross-validation method of model selection described below, is conservative to cope with non-independence from spatial autocorrelation. By not fitting random effects for individuals, repeated trips by individuals simply become another source of non-independence that is accommodated in precautionary model selection. Hence, for the purpose of predictions and in the context of our study, GAMs were a pragmatic modelling approach.
We modelled habitat selection using logistic regression, assuming a case-control design70. For each observed animal location, we generated 3 at-sea pseudo-locations for each true location, which were temporally matched (date randomly selected from the associated tracking data) but randomly placed in space, within the area assumed to be accessible to animals from each colony. This we defined as being within 1.1 times larger than the maximum distance travelled by each species and group using the R package ‘rgeos’33. We assigned four static and three dynamic environmental covariates to each location and pseudo-location (Table 3). We selected a limited number of dynamic environmental variables that have previously been shown to influence spatial usage of at least some of the study species33. We used the R package ‘rerddap’ to extract sea surface temperature (SST) data (Table 3). For the remaining variables (bathymetry, slope, sea level anomaly and eddy kinetic energy) we used the R package ‘raster’ to extract values for each location and pseudo-location (Table 3). Distance was extracted from a raster of geographic distance from the colony created using the R package ‘gdistance’, which accounted for the increased cost of travel with distance away from the breeding colony, while avoiding land71. In order to model the potential effect of intraspecific competition, we calculated a proxy for the potential competition of animals from each colony, namely Null density = population size/distance to breeding colony2,33. For each colony, we then iteratively summed the density rasters from all other colonies to represent the total potential competition from animals from all colonies at each location37.
We modelled the ratio of locations to pseudo-absences using binomial GAMs with up to 7 explanatory environmental covariates for each species, group, and colony (Table 3). Specifically, we created separate predictive models for rockhopper penguin and black-browed albatross during incubation and chick rearing stages, and separate models for gentoo penguins during winter (non-breeding) and summer (breeding), given that time and energy constraints typically differ between breeding stage and season, which in-turn may influence foraging behavior72 (Table 3). Differences in foraging trip distance between breeding stages were tested using LMM with individual as a random effect. We also created separate models for South American fur seals during winter and spring, given foraging behavior differs seasonally46. One predicative model was developed for each of the remaining species, including Magellanic penguins, which undertook both short and long foraging trips irrespective of breeding phase. Although colony information was not available for Magellanic penguins, they are distributed around much of the Falkland Islands coastline and breeding colonies often co-occur with gentoo penguins. Accordingly, we used gentoo penguin breeding colonies as a proxy for the distribution of Magellanic penguin breeding colonies. Colony predictions for southern sea lions and South American fur seals (Arctocephalus australis) were based on adult female data, given that the colony origin of males was unknown, and males often moved between breeding colonies (Table 3)30,73.
We checked for multicollinearity among the explanatory covariates using Variance Inflation Factors (VIF) and standardized covariates [(x - mean(x))/SD(x)]. To reduce the chances of overfitting, we specified cubic regression splines with shrinkage, and initially limited the number of knots to 3, increasing when fit was poor37. Model fit was inspected by plotting smooth terms and histograms of the raw data. We built models by forwards stepwise selection following Warwick-Evans et al.37. We assessed relative model performance by k-folds cross-validation, where k is the number of tracked breeding colonies. We also modelled distance and bathymetry as a tensor smooth to see whether this improved model fit10. To determine the order in which candidate covariates were added into the model, we first modelled our response variable as a function of each candidate explanatory covariate on its own, using data from n -1 tracked colonies. We evaluated models by predicting habitat use for the excluded breeding colony and generating a receiver operating curve (ROC) from which area under the curve (AUC), sensitivity (correctly predicted presences) and specificity (correctly predicted absences) were calculated using the R package ‘pROC’. Values of 0.5 indicated the fitted model was no better than random, whereas 1.0 indicated the model perfectly predicted space use. We added explanatory covariates to the main model in the order of AUC scores (highest to lowest). When two covariates had similar AUC values, we used each as the starting covariate, and the final model chosen was based on the model with the highest AUC values. For three species, data were only available for one breeding colony, which accounted for >90% of the population74,75. For these species - king penguins, southern elephant seals and sooty shearwaters (Ardenna grisea) - we performed cross-validation by randomly splitting datasets into training and testing datasets.
We used the best performing model for each group to predict the at-sea distribution of animals from all colonies. To estimate values that were proportional to the probability of habitat use we applied the following equation76:
Where Pa = the proportion of absences, Pu = the proportion of presences and β0… βp are model coefficients. To plot the results of the GAM predictions, we first standardized the probability of occurrence for each species and group, to produce a single raster of combined (cumulative) probability of occurrence using the mosaic function in the R package ‘raster’. To calculate overlap in the probability of occurrence, for each species and group we standardized each raster cell to equal 1 where occurance was predicted, and used the mosaic function to sum raster values. Finally, we weighted species outputs by colony size to highlight important areas adjacent to the largest breeding colonies – although both weighted and unweighted results are presented. Given that the environmental covariates for pseudo-points were extracted for the same period as the tracking data, predictions are limited to the period over which tracking data were collected (see Table 1).
Data Availability
Data are available on reasonable request made to the corresponding author, or via the South Atlantic Environmental Research Institute https://www.south-atlantic-research.org/research/data-science/data-services-metadata-catalogue/.
Change history
20 November 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Sherman, K. The Large Marine Ecosystem Concept: Research and Management Strategy for Living Marine Resources. Ecol. Appl. 1, 349–360 (1991).
Costanza, R. et al. The value of the world’s ecosystem services and natural capital. Nature 387, 253–260 (1998).
Sherman, K., Sevilla, N. P. M., Álvarez Torres, P. & Peterson, B. Sustainable development of Latin American and the Caribbean Large Marine Ecosystems. Environ. Dev. 22, 1–8 (2017).
Belkin, I. M. Rapid warming of Large Marine Ecosystems. Prog. Oceanogr. 81, 207–213 (2009).
Estes, J. A., Heithaus, M., McCauley, D. J., Rasher, D. B. & Worm, B. Megafaunal Impacts on Structure and Function of Ocean Ecosystems. Annu. Rev. Environ. Resour. 41, 83–116 (2016).
Reid, K. & Croxall, J. P. Environmental response of upper trophic-level predators reveals a system change in an Antarctic marine ecosystem. Proc. Biol. Sci. 268, 377–384 (2001).
Hunt, G. L. & McKinnell, S. Interplay between top-down, bottom-up, and wasp-waist control in marine ecosystems. Prog. Oceanogr. 68, 115–124 (2006).
Lynam, C. P. et al. Interaction between top-down and bottom-up control in marine food webs. Proc. Natl. Acad. Sci. 114, 1952–1957 (2017).
Hindell, M. A. et al. Foraging habitats of top predators, and Areas of Ecological Significance, on the Kerguelen Plateau. Kerguelen Plateau Mar. Ecosyst. Fish. 203–215, https://doi.org/10.1007/s003960050133 (2011).
Raymond, B. et al. Important marine habitat off east Antarctica revealed by two decades of multi-species predator tracking. Ecography (Cop.). 38, 121–129 (2015).
Lascelles, B. G. et al. Applying global criteria to tracking data to define important areas for marine conservation. Divers. Distrib. 22, 422–431 (2016).
Augé, A. A. et al. Framework for mapping key areas for marine megafauna to inform Marine Spatial Planning: The Falkland Islands case study. Mar. Policy 92, 61–72 (2018).
Hart, K. M. & Hyrenbach, K. D. Satellite telemetry of marine megavertebrates: The coming of age of an experimental science. Endanger. Species Res. 10, 9–20 (2010).
Reisinger, R. R. et al. Habitat modelling of tracking data from multiple marine predators identifies important areas in the Southern Indian Ocean. Divers. Distrib. 1–16, 535–550, https://doi.org/10.1111/ddi.12702 (2018).
Dias, M. P. et al. Using globally threatened pelagic birds to identify priority sites for marine conservation in the South Atlantic Ocean. Biol. Conserv. 211, 76–84 (2017).
Block, B. A. et al. Tracking apex marine predator movements in a dynamic ocean. Nature 475, 86–90 (2011).
Grecian, W. J. et al. Seabird diversity hotspot linked to ocean productivity in the Canary Current Large Marine Ecosystem. Biol. Lett. 12, 20160024 (2016).
Harrison, A. L. et al. The political biogeography of migratory marine predators. Nat. Ecol. Evol. 2, 1571–1578 (2018).
Croxall, J. P. & Wood, A. G. The importance of the Patagonian Shelf for top predator species breeding at South Georgia. Aquat. Conserv. Mar. Freshw. Ecosyst. 12, 101–118 (2002).
Osborne, M. J. & McIntyre, A. (eds). 2002. The south west Atlantic marine environment: research and management. Aquat. Conserv. Mar. Freshw. Ecosyst. Spec. Issue 12 (1): 1–164 (2002).
Falabella, V., Campagna, C. & Croxall, J. Atlas del Mar Patagónico. Especies y Espacios. Buenos Aires, Wildlife Conservation Society y BirdLife International (2009).
Campagna, C. et al. A species approach to marine ecosystem conservation. Aquat. Conserv. Mar. Freshw. Ecosyst. S122–S147 doi:10.1002/aqc (2008).
Baylis, A. M. M., Crofts, S. & Wolfaardt, A. C. Population trends of gentoo penguins Pygoscelis papua breeding at the Falkland Islands. Mar. Ornithol. 41, 1–5 (2013).
Baylis, A. M. M., Wolfaardt, A. C., Crofts, S., Pistorius, P. A. & Ratcliffe, N. Increasing trend in the number of Southern Rockhopper Penguins (Eudyptes c. chrysocome) breeding at the Falkland Islands. Polar Biol. 36, 1007–1018 (2013).
Catry, P., Campos, A., Segurado, P. & Strange, I. Population census and nesting habitat selection of thin-billed prion Pachyptila belcheri on New Island, Falkland Islands. Polar Biol. 26, 202–207 (2003).
Wakefield, E. D., Phillips, R. A. & Matthiopoulos, J. Habitat-mediated population limitation in a colonial central-place forager: The sky is not the limit for the black-browed albatross. Proc. R. Soc. B Biol. Sci. 281, 20132883 (2014).
Baylis, A. M. M. et al. Re-evaluating the population size of South American fur seals and conservation implications. Aquat. Conserv Mar. Freshw. Ecosyst (2019).
Baylis, A. M. M. et al. Disentangling the cause of a catastrophic population decline in a large marine mammal. Ecology 96, 2834–2847 (2015).
Ratcliffe, N. et al. Love thy neighbour or opposites attract? Patterns of spatial segregation and association among crested penguin populations during winter. J. Biogeogr. 41, 1183–1192 (2014).
Baylis, A. M. M., Tierney, M., Staniland, I. J. & Brickle, P. Habitat use of adult male South American fur seals and a preliminary assessment of spatial overlap with trawl fisheries in the South Atlantic. Mamm. Biol. 93, 76–81 (2018).
Catry, P. et al. Predicting the distribution of a threatened albatross: The importance of competition, fisheries and annual variability. Prog. Oceanogr. 110, 1–10 (2013).
Baylis, A. M. M. et al. Diving deeper into individual foraging specializations of a large marine predator, the southern sea lion. Oecologia 179, 1053–1065 (2015).
Wakefield, E. D. et al. Habitat preference, accessibility and competiton limit the global distribution of breeding black-browed albatrosses. Ecol. Monogr. 81, 141–167 (2011).
Robson, B. W. et al. Separation of foraging habitat among breeding sites of a colonial marine predator, the northern fur seal (Callorhinus ursinus). Fish. Sci. 29, 20–29 (2004).
Masello, J. F., Kato, A., Sommerfeld, J., Mattern, T. & Quillfeldt, P. How animals distribute themselves in space: Variable energy landscapes. Front. Zool. 14, 1–14 (2017).
Wakefield, E. D. et al. Space Partitioning Without Territoriality in Gannets. Science 341, 68–70 (2013).
Warwick-Evans, V. et al. Using habitat models for chinstrap penguins Pygoscelis antarctica to advise krill fisheries management during the penguin breeding season. Divers. Distrib. 1–16, 1756–1771, https://doi.org/10.1111/ddi.12817 (2018).
Cairns, A. D. K. The Regulation of Seabird Colony Size: A Hinterland Model. Am. Nat. 134, 141–146 (1989).
Wakefield, E. D. et al. Breeding density, fine-scale tracking, and large-scale modeling reveal the regional distribution of four seabird species. Ecol. Appl. 27, 2074–2091 (2017).
Gonzalez Carman, V. et al. Distribution of megafaunal species in the Southwestern Atlantic: key ecological areas and opportunities for marine conservation. ICES J. Mar. Sci. 73, 1579–1588 (2016).
Nicholls, D. G. et al. Foraging niches of three Diomedea albatrosses. Mar. Ecol. Prog. Ser. 231, 269–277 (2002).
Schejter, L. et al. Namuncura Marine Marine Protected Area: an oceanic hot spot Namuncura of benthic biodiversity at Burdwood Bank, Argentina. Polar Biol. 2373–2386, https://doi.org/10.1007/s00300-016-1913-2 (2016).
Clausen, A. P. & Pütz, K. Winter diet and foraging range of gentoo penguins (Pygoscelis papua) from Kidney Cove, Falkland Islands. Polar Biol. 26, 32–40 (2003).
McConnell, B. J., Chambers, C. & Fedak, M. A. Foraging ecology of southern elephant seals in relation to the bathyrnetry and productivity of the Southern Ocean. Antarct. Sci. 4, 393–398 (1992).
Trathan, P. N. et al. Managing fishery development in sensitive ecosystems: identifying penguin habitat use to direct management in Antarctica. Ecosphere 9 (2018).
Baylis, A. M. M., Tierney, M., Orben, R. A., Staniland, I. J. & Brickle, P. Geographic variation in the foraging behaviour of South American fur seals. Mar. Ecol. Prog. Ser. 596, 233–245 (2018).
Masello, J. F. et al. Diving seabirds share foraging space and time within and among species. Ecosphere 1, 1–28 (2010).
Torres, L. G. et al. Poor transferability of species distribution models for a pelagic predator, the grey petrel, indicates contrasting habitat preferences across ocean basins. PLoS One 10, 1–18 (2015).
Paton, R. S. & Matthiopoulos, J. Defining the scale of habitat availability for models of habitat selection. Ecology 97, 1113–1122 (2016).
Phillips, R. A., Silk, J. R. D., Croxall, J. P. & Afanasyev, V. Year-round distribution of white-chinned petrels from South Georgia: Relationships with oceanography and fisheries. Biol. Conserv. 129, 336–347 (2006).
Peterson, R. G. & Whitworth, T. III The subantarctic and polar fronts in relation to deep water masses through the southwestern Atlantic. J. Geophys. Res. 94, 10817–10838 (1989).
Thiers, L., Delord, K., Bost, C. A., Guinet, C. & Weimerskirch, H. Important marine sectors for the top predator community around Kerguelen Archipelago. Polar Biol. 40, 365–378 (2017).
Handley, J. M., Connan, M., Baylis, A. M. M., Brickle, P. & Pistorius, P. Jack of all prey, master of some: Influence of habitat on the feeding ecology of a diving marine predator. Mar. Biol. 164, 82 (2017).
Baylis, A. M. M., Arnould, J. & Staniland, I. J. Diet of South American fur seals at the Falkland Islands. Mar. Mammal Sci. 30, 1210–1219 (2014).
Thompson, D., Duck, C. D., McConnell, B. J. & Garrett, J. Foraging behaviour and diet of lactating female southern sea lions (Otaria flavescens) in the Falkland Islands. J. Zool. 246, 135–146 (1998).
Cherel, Y., Pütz, K. & Hobson, K. Summer diet of king penguins (Aptenodytes patagonicus) at the Falkland Islands, southern Atlantic Ocean. Polar Biol. 25, 898–906 (2002).
Arkhipkin, A., Brickle, P. & Laptikhovsky, V. Links between marine fauna and oceanic fronts on the Patagonian Shelf and Slope. Arquipel. - Life Mar. Sci. 30, 19–37 (2013).
Jennings, S. et al. Global-scale predictions of community and ecosystem properties from simple ecological theory. Proc. R. Soc. B Biol. Sci. 275, 1375–1383 (2008).
Agnew, D. J. Critical aspects of the Falkland Islands pelagic ecosystem: distribution, spawning and migration of pelagic animals in relation to oil exploration. Aquat. Conserv. Mar. Freshw. Ecosyst. 12, 39–50 (2002).
Orians, G. & Pearson, N. On the theory of central place foraging. In Analysis of Ecological Systems, eds D. J. Horn, R. D. Mitchell, and G. R. Stairs (Ohio Univ Press, Athens, OH), 155–177 (1979).
Arkhipkin, A. I., Brickle, P., Laptikhovsky, V. & Winter, A. Dining hall at sea: feeding migrations of nektonic predators to the eastern Patagonian Shelf. J. Fish Biol. 81, 882–902 (2012).
Scales, K. L. et al. Scale of inference: on the sensitivity of habitat models for wide-ranging marine predators to the resolution of environmental data. Ecography (Cop.). 40, 210–220 (2017).
Blockley, D. & Tierney, M. Addressing priority gaps in understanding ecosystem functioning for the developing Falkland Islands offshore hydrocarbon industry – the ‘Gap Project’. Phase I Final Report, September 2016. Report prepared for the Falkland Islands Offshore Hydrocarbons Env. (2017).
Worm, B. et al. Impacts of biodiversity loss on ocean ecosystem services. Science. 314, 787–790 (2006).
Combes, V. & Matano, R. P. The Patagonian shelf circulation: Drivers and variability. Prog. Oceanogr. 167, 24–43 (2018).
Freitas, C., Lydersen, C., Fedak, M. A. & Kovacs, K. M. A simple new algorithm to filter marine mammal Argos locations. Mar. Mammal Sci. 24, 315–325 (2008).
Johnson, D., London, J., Lea, M. & Durban, J. Continuous-time correlated random walk model for animal telemetry data. Ecology 89, 1208–1215 (2008).
Costa, D. P. et al. Accuracy of ARGOS locations of Pinnipeds at-sea estimated using Fastloc GPS. PLoS One 5, e8677 (2010).
Calenge, C., Dray, S. & Royer-Carenzi, M. The concept of animals’ trajectories from a data analysis perspective. Ecol. Inform. 4, 34–41 (2009).
Aarts, G., MacKenzie, M., McConnell, B., Fedak, M. & Matthiopoulos, J. Estimating space-use and habitat preference from wildlife telemetry data. Ecography (Cop.). 31, 140–160 (2008).
Matthiopoulos, J. The use of space by animals as a function of accessibility and preference. Ecol. Modell. 159, 239–268 (2003).
Huin, N. Foraging distribution of the black-browed albatross,Thalassarche melanophris, breeding in the Falkland Islands. Aquat. Conserv. Mar. Freshw. Ecosyst. 12, 89–99 (2002).
Baylis, A. M. M. et al. Habitat use and spatial fidelity of male South American sea lions during the nonbreeding period. Ecol. Evol. 1–11, https://doi.org/10.1002/ece3.2972 (2017).
Pistorius, P. A., Baylis, A. M. M., Crofts, S. & Pütz, K. Population development and historical occurrence of king penguins at the Falkland Islands. Antarct. Sci. 6, 1–6 (2012).
Galimberti, F., Sanvito, S., Boitani, L. & Fabiani, A. Viability of the southern elephant seal population of the Falkland Islands. Anim. Conserv. 81–88 (2001).
Manly, B., McDonald, L., Thomas, D., McDonald, T. & Erickson, W. Resource Selection by Animals: Statistical Design and Analysis for Field Studies. (Kluwer Academic Publishers, 2004).
Acknowledgements
This manuscript forms part of the South Atlantic Environmental Research Institute’s GAP project, an initiative of the Falkland Islands Offshore Hydrocarbons Environmental Forum, funded by the Falkland Islands Government and Falkland Islands Petroleum Licensees Association. We are grateful to the many colleagues who assisted with field work, David Blockley for his contribution to the management of the GAP project and Amélie Augé and Ben Lascelles who assisted with data compilation. All animal handling was approved by the Falkland Islands Environmental Committee and research was undertaken under permit issued by the Falkland Islands Government.
Author information
Authors and Affiliations
Contributions
P.B., M.T. and A.M.M.B. conceived and designed the research. M.T. coordinated a workshop attended by all authors and collated existing tracking data. All authors contributed data. A.M.M.B. analyzed the data and M.T., V.W.-E., R.A.O., E.W., P.T., N.R., W.J.G. and R.R., contributed R code, methods or provided advice. A.M.M.B. wrote the manuscript, with input from E.W., N.R., M.T., V.W.-E., R.A.O., P.T., R.R., J.C. and P.B. All authors commented on manuscript drafts.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Baylis, A.M.M., Tierney, M., Orben, R.A. et al. Important At-Sea Areas of Colonial Breeding Marine Predators on the Southern Patagonian Shelf. Sci Rep 9, 8517 (2019). https://doi.org/10.1038/s41598-019-44695-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-44695-1
This article is cited by
-
Evidence of colony- and individual-level variation in habitat usage of Atlantic Puffins (Fratercula arctica)
Marine Biology (2024)
-
Year-round behavioural time budgets of common woodpigeons inferred from acceleration data using machine learning
Behavioral Ecology and Sociobiology (2023)
-
How animals distribute themselves in space: energy landscapes of Antarctic avian predators
Movement Ecology (2021)
-
Pelagic and benthic ecosystems drive differences in population and individual specializations in marine predators
Oecologia (2021)
-
A critical assessment of marine predator isoscapes within the southern Indian Ocean
Movement Ecology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.