Abstract
Pegiviruses belong to the family Flaviviridae and have been found in humans and other mammalian species. To date eleven different pegivirus species (Pegivirus A-K) have been described. However, little is known about the tissue tropism and replication of pegiviruses. In 2016, a so far unknown porcine pegivirus (PPgV, Pegivirus K) was described and persistent infection in the host, similar to human pegivirus, was reported. In this study, qRT-PCR, phylogenetic analyses and fluorescence in situ hybridization (FISH) were implemented to detect and quantify PPgV genome content in serum samples from domestic pigs from Europe and Asia, in tissue and peripheral blood mononuclear cell (PBMC) samples and wild boar serum samples from Germany. PPgV was detectable in 2.7% of investigated domestic pigs from Europe and China (viral genome load 2.4 × 102 to 2.0 × 106 PPgV copies/ml), while all wild boar samples were tested negative. Phylogenetic analyses revealed pairwise nucleotide identities >90% among PPgVs. Finally, PPgV was detected in liver, thymus and PBMCs by qRT-PCR and FISH, suggesting liver- and lymphotropism. Taken together, this study provides first insights into the tissue tropism of PPgV and shows its distribution and genetic variability in Europe and China.
Similar content being viewed by others
Introduction
Pegiviruses comprise a group of positive-sense, single-stranded RNA viruses, with a genome size of 9–13 kb, that were recently classified into eleven species (Pegivirus A-K) within the genus Pegivirus in the Flaviviridae family1. They can infect humans as well as a range of mammalian species, including primates, bats, rodents, horses and pigs2,3,4,5,6,7,8,9. While pegiviruses are known to cause persistent infections in humans and horses, their pathogenicity remains largely unknown1,4,10,11,12. Though a pegivirus was identified in horses with Theiler’s Disease in the USA5, recent studies imply that viruses of the copiparvovirus group are associated with serum hepatitis in horses13,14. Human pegiviruses (HPgV) are distributed globally and viral RNA is present in roughly 750 million people, making it one of the most prevalent human RNA viruses15.
Though HPgV was initially thought to be hepatotropic and a possible agent of Non-A-E hepatitis, evidence of viral replication in the liver of infected patients is missing or inconclusive16,17,18. Rather, as HPgV replication has been shown in peripheral blood mononuclear cells (PBMCs) ex vivo for several weeks, the virus appears to be lymphotropic19,20,21. Additionally, HPgV RNA has been found in serum microvesicles, which have successfully delivered viral RNA to uninfected PBMCs that then supported HPgV replication ex vivo22. Interestingly, pegivirus infection in humans may have a beneficial effect on the outcome of human immunodeficiency virus type 1 (HIV-1) infections in individuals co-infected with both viruses, including reduced retroviral loads, slower progression to AIDS and improved survival rates. These benefits are attributed to immune-modulating effects as well as direct and indirect antagonistic mechanisms of HPgV on HIV-1 infection23.
Porcine pegiviruses (PPgV) were first described in domestic pigs from Germany in 20169. The study reported a PPgV detection rate of 2.2% (10 of 455) in porcine serum samples and described persistent infection for up to 22 months in three pigs that did not display any clinical signs of disease. Apart from Germany, presence of PPgV has been investigated in North America, where a recent study revealed a PPgV detection rate of 15.1% (24 of 159 samples) in the USA24. Additionally, a recent study investigated 469 porcine serum samples from China, 34 (7.25%) of which were found PPgV positive. Samples originated from different age groups and proved an ascending trend in the PPgV positive rate from suckling piglets (1.61%) and nursing piglets (1.85%) to finishing pigs (6.56%) and sows (11.34%)25.
In this study we analyzed the presence of PPgV genome in pigs from Europe and Asia. To clarify whether wild boar might play a role in the epidemiology of PPgV, as seen in infections with, for example, classical swine fever virus26,27, African swine fever virus28 and atypical porcine pestivirus (APPV)29, we also investigated the presence of PPgV genome in wild boar serum samples from Germany. To date the primary permissive cell type(s) of HPgV and other pegiviruses remain unknown. For this reason, we analyzed the tissue and cell tropism of PPgV through detection and quantification of viral RNA in tissues and PBMCs from PPgV positive pigs using qRT-PCR and fluorescence in situ hybridization (FISH).
Results
PPgV RNA in serum samples from Europe and Asia
The in vitro transcribed RNA copy standard evidenced a highly efficient qRT-PCR assay that was able to detect ten viral genome copies per reaction at Cq values around 36. PPgV genome was detectable in 47 of 1,736 (2.7%) serum samples from domestic pigs corresponding to 20 out of 132 herds (15.2%) (Table 1). Highest detection rates were found in individual animals from Great Britain (10.3%) and in herds from China (58.3%). In the different age groups investigated here, the PPgV positive rates were 1.9% in animals under 4 weeks of age, 1.2% in fattening pigs over 4 weeks of age, 3.4% in sows and boars, and 10.1% in pigs of unknown age and use (Table 2). Viral loads varied between 2.4 × 102 and 2.0 × 106 PPgV RNA copies/ml serum, with an overall average of 3.8 × 105 copies/ml. For individual countries on average, lowest genome loads were detected in Poland (1.9 × 104 copies/ml) and highest in Italy (7.1 × 105 copies/ml). All 800 wild boar samples were negative for PPgV RNA.
Phylogenetic analyses
Altogether 31 PPgV partial NS3 sequences were obtained from domestic pigs, of which nine were identical to one or more other sequences. In total, ten sequences from Germany, three sequences from Italy, four sequences from Poland, nine sequences from Great Britain and five sequences from China were acquired. Sequences GER/SA/13, GER/SA/91, and PL/159 were identical to one additional sequence each, while IT/77, GB/16, and GB/23 were identical to two further sequences each. In Germany, Poland, and Italy, all identical sequences originated from samples from the same farms, while herd affiliation was unknown for samples from Great Britain.
Twenty-two distinct sequences shown here (Fig. 1) were submitted to GenBank. They displayed nucleotide sequence identities of >90%. According to phylogenetic analysis, PPgV formed a separate branch in the tree of pegiviruses and viral sequences segregated into two main clusters, one of which contained only sequences from Europe (Germany, Great Britain and Poland). Within the second main cluster, some branches contained sequences recovered from animals in Europe (GER/NDS/T72 and IT/77) in close proximity to variants from China (i.e. CN/6/5) and USA (i.e. 33/ND/2017)24.
Phylogenetic analysis of porcine pegiviruses from different countries and other mammalian pegiviruses. Numbers along branches represent percentage bootstrap values (bootstrap values < 80% are not given). GenBank accession numbers are in parentheses. Scale bar indicates nucleotide substitutions per site. PPgV sequences are marked with a circle and the circle color indicates the country of origin. Pegivirus species A-K are indicated on the right.
Overall, the most closely related pegivirus sequence found in other species when compared to PPgV was bat pegivirus sequence PDB-1715 (GenBank KC796088), which had a nucleotide sequence identity of 58.1% with PPgV GB/30. A human pegivirus type 2 sequence, ABT0070P.US (GenBank KT427411) had the lowest nucleotide identity (47.1%) compared to PPgV sequences. When comparing PPgV sequences with pegivirus sequences originating from horses, nucleotide identities ranged from 53.7% to 55.7%. The sequence identities between PPgV and rodent pegivirus were around 54%, while the identities with simian pegiviruses ranged from 50.0% to 55.8%.
PPgV RNA in tissue samples
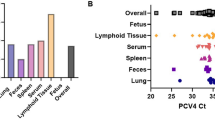
In tissue samples of PPgV positive pigs, PPgV RNA was most abundant in the liver (Table 3). Liver samples of animals A and B contained 343.9 and 142.5 viral RNA copies/mg tissue, respectively, while 119.3 copies/mg tissue were found in the liver of animal C using qRT-PCR. Serum samples of these animals contained 2,051.1 copies/µl (animal A), 388.6 copies/µl (animal B) and 157.0 copies/µl (animal C). PBMCs were only available from animal A and contained 46 copies/µl whole blood used for isolation (Table 3).
FISH was used to investigate the liver, thymus, PBMCs and different lymph nodes of animal A, as well as the liver and thymus of animal B, and respective tissues of negative control pigs. PPgV specific signals were detected in the liver of both PPgV positive pigs (Table 3; Fig. 2). Furthermore, several cells of the medullary and cortical region of the thymus of animals A and B were observed to be virus positive using the PPgV specific probe. Additionally, PBMCs of animal A were found to be virus positive in FISH, while lymph nodes, spleen, tonsils, bone marrow and pancreas of animal A tested virus negative. The non-probe incubation as well as the PPgV PCR-negative pigs showed no detectable positive area in the same tested organs, respectively. During necropsy of animal A, multifocal, mild, subendocardial hemorrhages were present. Histopathology showed a mild, portally accentuated, lymphohistiocytic hepatitis, a mild, diffuse infiltration of eosinophils within the thymus, tonsils and lymph nodes and single multinucleated giant cells within the medullary part of the thymus. Furthermore, lymph nodes revealed a mild, diffuse sinus histiocytosis. A moderate, focal, perivascular, lymphoplasmahistiocytic, partially eosinophilic dermatitis was present at the pinna. Additionally, a mild endocardiosis, a mild, lymphohistiocytic epicarditis and a mild to moderate, focal, follicular, lymphocytic conjunctivitis were observed.
Fluorescence in situ hybridization of porcine pegivirus (PPgV) positive and negative pigs using a PPgV specific probe; overlay phase contrast and immunofluorescence; bar = 100 µm. (A) Single hepatocytes of the liver of a PPgV positive pig showed an intracytoplasmic positive signal for PPgV using a PPgV specific probe, also shown at higher magnification in the insert; arrows: nuclei of hepatocytes surrounded by intracytoplasmic, red, positive signals. (B) The liver of a PPgV negative pig lacked a PPgV specific signal. (C) Within the thymus of a PPgV positive pig, scattered cells showed an intracytoplasmic red positive signal for PPgV, also shown at higher magnification in the insert. (D) Within the thymus of a PPgV negative pig, all cells were negative for PPgV using a PPgV specific probe. (E) Several PBMCs from a PPgV positive pig showed a red positive signal using a PPgV specific probe, also shown at higher magnification in the insert. (F) PBMCs from a PPgV negative pig were negative for PPgV.
Discussion
The genus Pegivirus has grown in recent years, as new viruses were identified in different hosts. Yet little is known about their pathogenicity and the impact on the host’s immune response. In this study, our aim was to gain detailed insights into the distribution of PPgV in different parts of the world and the genetic diversity of PPgV. Viral RNA was detected in serum samples from domestic pigs from various European countries and China, with an overall individual detection rate of 2.7%.
Investigation of three different age groups from Europe and Asia showed a lower PPgV positive rate in younger animals such as piglets (1.9%) and fattening pigs (1.2%) than in adult animals (3.4%). This observation is concordant with the results from a recently published study from China; however, the increase in PPgV positive rate was more prominent there (1.6–11.3%). Focusing on samples from China, we found similar results: 1.0% detection rate in piglets and 9.7% detection rate in sows and boar25.
The PPgV positive rates found in this study differ between countries. While no samples were PPgV positive from Switzerland, Serbia and Taiwan, samples from Germany, Poland and Italy have a positive rate similar to the one described previously for German domestic pigs (2.2%)9. High detection rates in China (7.8%) and Great Britain (10.3%) found here are nonetheless lower than the positive rate observed in the USA (15.1%) in a previous study24. In humans, HPgV prevalence ranges from 0.5 to 5% in healthy blood donors from developed countries, but is higher in blood donors from developing countries (5–18.9%), and in individuals co-infected with blood borne or sexually transmitted diseases, like hepatits C virus or HIV-130,31,32. Equine pegivirus (EPgV) has been found in 12 of 328 horses (3.7%) from Europe and 7 of 74 horses (9.5%) from USA4,12, thus showing similar detection rates as PPgV. The divergence in PPgV detection rates suggests uneven distribution of virus infection and local spread of PPgV. This may be caused by the occurrence of other infectious diseases in pig populations, similar to observations in humans with co-infection, and needs to be studied further. The viral loads determined here (2.4 × 102 to 2 × 106 copies/ml) are similar to EPgV RNA loads described in one study, which ranged from 3.2 × 104 to 3.2 × 106 RNA copies/ml4. However, another study found higher EPgV viral loads (4.1 × 105 to 2.0 × 109 RNA copies/ml)12, and the mean RNA load of HPgV in human plasma typically reaches >1 × 107 copies/ml. This may suggest lower replication of PPgV in vivo compared to HPgV and EPgV.
Although HPgV does not appear to be hepatotropic, high amounts of PPgV RNA in the porcine liver shown by qRT-PCR and in situ techniques suggest that viral RNA may accumulate in the liver or even that PPgV has the ability to replicate in hepatocytes. However, this hypothesis will have to be investigated in future studies, as well as whether PPgV infections might be the cause of histopathological changes in the liver, as seen in animal A. Moreover, presence of PPgV RNA in PBMCs and in the thymus supports lymphotropism analogical to HPgV22. Positive FISH results in primary but not secondary lymphoid organs, such as spleen or lymph nodes, imply that the virus might replicate in the thymus and spread to other tissues (e.g. the liver) via PBMCs, but successfully evades recognition by the immune system, which could lead to a persistent infection in the host. Despite significant amounts of viral RNA detected in cells and tissues, highest viral loads were present in the serum of infected animals. With regard to this, low amounts of PPgV RNA in further organs and tissues can most probably be attributed to blood residues. Possible presence of viral RNA in serum microvesicles and associated virus uptake by PBMCs, as seen for HPgV, remain to be determined22.
Phylogenetic analyses showed close genetic relationships among PPgV sequences from different countries, like sequences GER/NDS/T72 and CN/6/5. This could suggest virus spread by international trade with pigs or pig products, such as feed. While all wild boar samples were tested negative for PPgV RNA in this study, other porcine viruses from the family Flaviviridae, such as APPV, have been shown to be present at a higher rate in wild boar (19%) than in domestic pigs from Germany (6.2%)29,33. For APPV, virus transmission between wild boar and domestic pigs appears likely, as strains originating from wild and domestic animals show genetic distance of as little as 6.6%29. However, due to the comparatively low prevalence of PPgV in Germany, transmission of the virus from domestic pigs to wild boar and vice versa may be limited. Only samples from wild boar hunted in northern Germany entered the present study. Future studies with extended sampling will reveal whether PPgV is also absent in wild boar from other geographical regions. As genome detection alone may result in underestimation of virus dissemination, upcoming investigations of samples from domestic pigs, wild boar and other species will also address serological reactions upon infection with PPgV.
These results manifest that PPgV, like other pegiviruses, is distributed over several continents. It can be hypothesized that putative immune modulatory effects of PPgV infections are implicated in pig health worldwide. Detection of PPgV RNA in lymphoid cells suggests that the virus has the potential to affect the immune system of pigs. First insights into the cell- and organ tropism of PPgV suggest that the virus may be hepatotropic and/or lymphotropic. Future studies will clarify the pathogenic potential and immune modulatory effects of this newly discovered, widely distributed virus.
Methods
1,736 serum samples from domestic pigs from different countries in Europe (Germany, Great Britain, Poland, Switzerland, Italy and Serbia) and Asia (mainland China and Taiwan) originating from 132 different herds were analyzed in this study. For samples collected in Great Britain, the number of herds was unknown. Samples included 108 piglets up to four weeks old, 923 fattening pigs over four weeks old, 557 sows and boar, and 148 pigs of unknown age and use. Samples were taken between 2014 and 2018, originated from apparently healthy domestic pigs and were taken within the framework of national veterinary health management in concordance with national legal and ethical regulations. Residual volumes of these samples were provided for use in the current study, therefore no ethical approval was required for use of these samples. In addition, 800 serum samples from hunted wild boar from Lower Saxony, Germany, were included. 456 of these wild boar samples were collected during the hunting seasons of 2015/2016 and 2016/2017 and were used in a previous study investigating APPV prevalence29. 344 additional wild boar samples were collected during the hunting season of 2017/2018. Furthermore, blood and post-mortem tissue samples originated from apparently healthy PPgV positive pigs (n = 3, animals A, B, and C) from the Clinic for Swine, Small Ruminants, Forensic Medicine and Ambulatory Services (University of Veterinary Medicine, Hannover) and PPgV negative control pigs (n = 2). To rule out presence of co-infections with APPV and porcine reproductive and respiratory syndrome virus (PRRSV), PPgV positive animals were also tested using RT-PCR and found negative for both viruses (data not shown). One pig (animal A) was submitted to the Department of Pathology, University of Veterinary Medicine Hannover. A full necropsy was performed and samples of 40 different tissues were collected and stored at −80 °C or fixed in 10% neutral buffered formalin and embedded in paraffin wax. For histopathological examination, 3 µm thick sections were stained with hematoxylin and eosin. Different organ and tissue samples and a liver sample originated from two further PPgV positive pigs, animal B and animal C, respectively. Control samples for FISH were taken from PPgV negative pigs. PBMCs from animal A and one negative control animal were isolated from ~1 ml blood by density gradient centrifugation with Histopaque (Merck, Darmstadt, Germany). Euthanasia and sampling were approved by Lower Saxony’s official authorities (LAVES AZ 15A602 and 17A195) and were carried out in accordance with German legislation (TierSchVersV).
RNA was isolated from 140 µl of serum using the QIAmp Viral RNA Mini Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. Isolation of RNA from preweighed tissue samples was achieved using the NucleoSpin RNA kit (Macherey-Nagel, Düren, Germany) or the RNeasy Mini Kit (QIAGEN) according to the manufacturer’s instructions and RNA samples were stored at −80 °C until testing. For PPgV genome quantification, a TaqMan based qRT-PCR targeting the highly conserved NS3 encoding region with primers PPgV/fwd/7 (5′-GTCTATGCTGGTCACGGA-3′), PPgV/rev/8 (5′-CACTCATCGCAAATGACCAC-3′) and probe PPgV/pro/11 (5′-[6FAM]-CCATTTCGCGAACCACTGATTCCA-[BHQ1]-3′) was developed and verified using samples that were PPgV positive in a SYBR Green qRT-PCR (QIAGEN) described in an earlier study9. For the new PCR assay, an in vitro transcribed RNA copy standard was developed using MEGAscript Kit (ThermoFisher Scientific, Germany) to allow for absolute quantification of genome copies. Real-time RT-PCR was performed using the Mx3005P QPCR System (Agilent Technologies, Santa Clara, USA) and the QuantiTect Probe RT-PCR Kit (QIAGEN) according to the manufacturer’s instructions on samples and RNA standard dilutions. Briefly, 12.5 µl RT-PCR master mix, 0.25 µl reverse transcriptase, 0.8 pmol of each primer and 0.2 pmol of the probe, 5.25 µl water and 5 µl sample RNA were used in each reaction of 25 µl with the following temperature profile: 50 °C for 30 minutes, 95 °C for 15 minutes and 40 cycles of 94 °C for 15 seconds and 60 °C for 1 minute. Serum samples were initially screened in pools containing three to ten individual samples; subsequently samples from positive pools were tested individually.
For phylogenetic analysis, amplicons corresponding to a partial NS3 coding sequence were generated by one of the following two methods: a) RT-PCR with SuperScript III One-Step RT-PCR System with Platinum TaqDNA Polymerase (Life technologies, Germany) with primers PPgV/fwd/G1 (5′-CACCGGGCTGTTTCTGCTA-3′) and PPgV/rev/G4 (5′-TTCCTTCCACACCAACCCAT-3′), or b) cDNA synthesis with SuperScript II Reverse Transcriptase (Invitrogen, Germany) using random hexamers followed by nested PCR with outer primers PPgV/fwd/G1 and PPgV/rev/G4, and inner primers PPgV/fwd/G3 (5′-CGGGCTGTTTCTGCTAGGT-3′) and PPgV/rev/G2 (5′-CACCAACCCATCGAGGATCA-3′) using Taq polymerase included in the Maxima Hot Start Green PCR Master Mix (2X) (ThermoFisher Scientific) and the following cycling parameters: 95 °C for 4 min, 40 cycles of 95 °C for 30 s, 52 °C for 30 s, 72 °C for 75 s, and 72 °C for 10 min. PCR products with an expected length of 1,290 (method a) and 1,278 (method b) were purified using the GeneJET PCR Purification Kit (ThermoFisher Scientific) according to the manufacturer’s instructions and submitted to Sanger sequencing (FlexiRun, LGC Genomics, Germany) with primers PPgV/fwd/G3 and PPgV/rev/G2. Sequences were trimmed to a final length of 1041 base pairs and a multiple sequence alignment was performed with ClustalW implemented in BioEdit 7.034. Phylogenetic trees were calculated in MEGA7 using the Maximum-likelihood method and the Kimura 2-parameter substitution model35 with 500 replicates for statistical evaluation.
FISH was performed on formalin-fixed, paraffin-embedded organ sections of two qRT-PCR positive pigs (animal A and B) and on the PBMC pellet of one pig (animal A) using a PPgV specific RNA probe covering parts of the PPgV NS3. The probe set (ViewRNA TYPE 1 Probe Set, ThermoFisher Scientific) covered positions 2–816 of a target sequence with 1172 nucleotides that overlapped with the partial PPgV sequence of animal A (GenBank MH979651). The procedure was carried out according to the manufacturer´s protocol with minor variations as previously described (ViewRNA TYPE 1 Probe Set; ViewRNA™ ISH Tissue Assay Kit (1-plex) and ViewRNA Chromogenic Signal Amplification Kit; ThermoFisher Scientific;)36. Briefly, sections were deparaffinized, boiled in pretreatment solution® at 90 °C for 10 minutes, digested by a protease QF® at 40 °C for 10 minutes and afterwards fixed. Hybridization to the specific probe was performed for 6 hours. Following preamplification and amplification steps, sections were stained with Fast Red Substrate and counterstained with Mayer´s hemalum® (Carl Roth GmbH, Karlsruhe, Germany). Images were acquired with an inverted fluorescence microscope (Olympus IX-70; Olympus Life Science Europe GmbH, Hamburg, Deutschland). The specificity of the probe was confirmed by including a non-probe incubation which served as system negative control and organ sections and cells of PPgV RT-PCR-negative pigs, respectively.
Accession codes
The obtained DNA sequences were deposited in GenBank (accession numbers: MH979651-MH979672).
References
Smith, D. B. et al. Proposed update to the taxonomy of the genera Hepacivirus and Pegivirus within the Flaviviridae family. J Gen Virol 97, 2894–2907, https://doi.org/10.1099/jgv.0.000612 (2016).
Simons, J. N. et al. Identification of two flavivirus-like genomes in the GB hepatitis agent. Proc Natl Acad Sci USA 92, 3401–3405 (1995).
Quan, P. L. et al. Bats are a major natural reservoir for hepaciviruses and pegiviruses. Proc Natl Acad Sci USA 110, 8194–8199, https://doi.org/10.1073/pnas.1303037110 (2013).
Kapoor, A. et al. Identification of a pegivirus (GB virus-like virus) that infects horses. J Virol 87, 7185–7190, https://doi.org/10.1128/JVI.00324-13 (2013).
Chandriani, S. et al. Identification of a previously undescribed divergent virus from the Flaviviridae family in an outbreak of equine serum hepatitis. Proc Natl Acad Sci USA 110, E1407–1415, https://doi.org/10.1073/pnas.1219217110 (2013).
Epstein, J. H. et al. Identification of GBV-D, a novel GB-like flavivirus from old world frugivorous bats (Pteropus giganteus) in Bangladesh. PLoS Pathog 6, e1000972, https://doi.org/10.1371/journal.ppat.1000972 (2010).
Firth, C. et al. Detection of zoonotic pathogens and characterization of novel viruses carried by commensal Rattus norvegicus in New York City. MBio 5, e01933–01914, https://doi.org/10.1128/mBio.01933-14 (2014).
Kapoor, A. et al. Identification of rodent homologs of hepatitis C virus and pegiviruses. MBio 4, e00216–00213, https://doi.org/10.1128/mBio.00216-13 (2013).
Baechlein, C. et al. Pegivirus Infection in Domestic Pigs, Germany. Emerg Infect Dis 22, 1312–1314, https://doi.org/10.3201/eid2207.160024 (2016).
Stapleton, J. T., Foung, S., Muerhoff, A. S., Bukh, J. & Simmonds, P. The GB viruses: a review and proposed classification of GBV-A, GBV-C (HGV), and GBV-D in genus Pegivirus within the family Flaviviridae. J Gen Virol 92, 233–246, https://doi.org/10.1099/vir.0.027490-0 (2011).
Theze, J., Lowes, S., Parker, J. & Pybus, O. G. Evolutionary and Phylogenetic Analysis of the Hepaciviruses and Pegiviruses. Genome Biol Evol 7, 2996–3008, https://doi.org/10.1093/gbe/evv202 (2015).
Lyons, S. et al. Viraemic frequencies and seroprevalence of non-primate hepacivirus and equine pegiviruses in horses and other mammalian species. J Gen Virol 95, 1701–1711, https://doi.org/10.1099/vir.0.065094-0 (2014).
Lu, G. et al. Identification and genetic characterization of a novel parvovirus associated with serum hepatitis in horses in China. Emerg Microbes Infect 7, 170, https://doi.org/10.1038/s41426-018-0174-2 (2018).
Divers, T. J. et al. New Parvovirus Associated with Serum Hepatitis in Horses after Inoculation of Common Biological Product. Emerg Infect Dis 24, 303–310, https://doi.org/10.3201/eid2402.171031 (2018).
Chivero, E. T. & Stapleton, J. T. Tropism of human pegivirus (formerly known as GB virus C/hepatitis G virus) and host immunomodulation: insights into a highly successful viral infection. J Gen Virol 96, 1521–1532, https://doi.org/10.1099/vir.0.000086 (2015).
Berg, T. et al. Dynamics of GB virus C viremia early after orthotopic liver transplantation indicates extrahepatic tissues as the predominant site of GB virus C replication. Hepatology 29, 245–249, https://doi.org/10.1002/hep.510290121 (1999).
Fan, X. et al. Is hepatitis G/GB virus-C virus hepatotropic? Detection of hepatitis G/GB virus-C viral RNA in liver and serum. J Med Virol 58, 160–164 (1999).
Laskus, T., Radkowski, M., Wang, L. F., Vargas, H. & Rakela, J. Lack of evidence for hepatitis G virus replication in the livers of patients coinfected with hepatitis C and G viruses. J Virol 71, 7804–7806 (1997).
Fogeda, M. et al. In vitro infection of human peripheral blood mononuclear cells by GB virus C/Hepatitis G virus. J Virol 73, 4052–4061 (1999).
George, S. L., Xiang, J. & Stapleton, J. T. Clinical isolates of GB virus type C vary in their ability to persist and replicate in peripheral blood mononuclear cell cultures. Virology 316, 191–201 (2003).
Rydze, R. T., Bhattarai, N. & Stapleton, J. T. GB virus C infection is associated with a reduced rate of reactivation of latent HIV and protection against activation-induced T-cell death. Antivir Ther 17, 1271–1279, https://doi.org/10.3851/IMP2309 (2012).
Chivero, E. T. et al. Human pegivirus RNA is found in multiple blood mononuclear cells in vivo and serum-derived viral RNA-containing particles are infectious in vitro. J Gen Virol 95, 1307–1319, https://doi.org/10.1099/vir.0.063016-0 (2014).
Schwarze-Zander, C., Blackard, J. T. & Rockstroh, J. K. Role of GB virus C in modulating HIV disease. Expert Rev Anti Infect Ther 10, 563–572, https://doi.org/10.1586/eri.12.37 (2012).
Yang, C. et al. Detection and genetic characterization of porcine pegivirus in pigs in the United States. Transbound Emerg Dis, https://doi.org/10.1111/tbed.12844 (2018).
Lei, D. et al. Detection and genetic characterization of porcine pegivirus from pigs in China. Virus Genes 55, 248–252, https://doi.org/10.1007/s11262-018-1624-6 (2019).
Moennig, V. The control of classical swine fever in wild boar. Front Microbiol 6, 1211, https://doi.org/10.3389/fmicb.2015.01211 (2015).
Postel, A., Austermann-Busch, S., Petrov, A., Moennig, V. & Becher, P. Epidemiology, diagnosis and control of classical swine fever: Recent developments and future challenges. Transbound Emerg Dis 65(Suppl 1), 248–261, https://doi.org/10.1111/tbed.12676 (2018).
Guinat, C. et al. Transmission routes of African swine fever virus to domestic pigs: current knowledge and future research directions. Vet Rec 178, 262–267, https://doi.org/10.1136/vr.103593 (2016).
Cagatay, G. N. et al. Frequent infection of wild boar with atypical porcine pestivirus (APPV). Transbound Emerg Dis, https://doi.org/10.1111/tbed.12854 (2018).
Mohr, E. L. & Stapleton, J. T. GB virus type C interactions with HIV: the role of envelope glycoproteins. J Viral Hepat 16, 757–768, https://doi.org/10.1111/j.1365-2893.2009.01194.x (2009).
Alter, H. J. G-pers creepers, where’d you get those papers? A reassessment of the literature on the hepatitis G virus. Transfusion 37, 569–572 (1997).
Stapleton, J. T. GB virus type C/Hepatitis G virus. Semin Liver Dis 23, 137–148, https://doi.org/10.1055/s-2003-39943 (2003).
Postel, A. et al. High Abundance and Genetic Variability of Atypical Porcine Pestivirus in Pigs from Europe and Asia. Emerg Infect Dis 23, 2104–2107, https://doi.org/10.3201/eid2312.170951 (2017).
Hall, T. A. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser (Oxf), 95–98 (1999).
Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16, 111–120 (1980).
Postel, A. et al. Presence of atypical porcine pestivirus (APPV) genomes in newborn piglets correlates with congenital tremor. Sci Rep 6, 27735, https://doi.org/10.1038/srep27735 (2016).
Acknowledgements
We thank Vesna Milićević (Belgrade, Serbia), Ming-Chung Deng and Chia-Yi Chang (New Taipei City, Taiwan), Serum Bank of the Federal Food Safety and Veterinary Office (Bern, Switzerland), Animal and Plant Health Agency (Weybridge, UK), Francesco Feliziani and Gian Mario De Mia (Perugia, Italy), Katarzyna Podgórska and Katarzyna Stępniewska (Puławy, Poland), Hua-Ji Qiu and Yuan Sun (Harbin, China), Sarah Derking and Jörg Tenhündfeld (Vreden, Germany) and Thomas Große Beilage (Essen/Oldenburg, Germany) for providing porcine serum samples. We highly appreciate the help of Inga Grotha concerning sample database set-up and RNA preparation. This study was supported by the German Center for Infection Research (DZIF), Thematic Translational Unit “Emerging Infections”, grant number 8002801801 as well as by the European Union’s Horizon 2020 research and innovation program under grant agreement No. 643476 (COMPARE) and the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – 398066876/GRK 2485/1. This publication was further supported by the Deutsche Forschungsgemeinschaft and University of Veterinary Medicine Hannover, Foundation within the funding programme “Open Access Publishing”.
Author information
Authors and Affiliations
Contributions
P.B., J.K. and C.B. designed the study. Methods were developed by J.K., V.M.P. and C.B. RNA extraction, quantification, and phylogenetic analyses were performed and interpreted by J.K., A.P., P.B. and C.B. Necropsies, histological investigations and FISH were performed and interpreted by V.M.P., M.G., and W.B. Sampling of animals and sample preparation was performed by D.H., A.P. and O.K. All authors contributed to the manuscript and reviewed the final version.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kennedy, J., Pfankuche, V.M., Hoeltig, D. et al. Genetic variability of porcine pegivirus in pigs from Europe and China and insights into tissue tropism. Sci Rep 9, 8174 (2019). https://doi.org/10.1038/s41598-019-44642-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-44642-0
This article is cited by
-
The Prevalence, Genetic Characterization, and Evolutionary Analysis of Porcine Pegivirus in Guangdong, China
Virologica Sinica (2021)
-
The Role of Emerging and Neglected Viruses in the Etiology of Hepatitis
Current Infectious Disease Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.