Abstract
Previous studies investigating HIV-infected patients suggested a direct link between underweight and the mortality rate of AIDS. However, there was a lack of evidence showing the optimal range of initial body mass index (BMI) patients maintain during antiretroviral therapy (ART). We aimed to evaluate associations of the BMI values pre-ART and during the entire ART duration with mortality among HIV-positive individuals. In total, 5101 HIV/AIDS patients, including 1439 (28.2%) underweight, 3047 (59.7%) normal-weight, 548 (10.7%) overweight and 67 (1.3%) obese patients, were included in this cohort. The cumulative mortality of underweight, normal-weight, and overweight were 2.4/100 person-years (95% CI 1.9–2.9), 1.1/100 person-years (95% CI 0.9–1.3), and 0.5/100 person-years (95% CI 0.1–0.9), respectively. Cumulative mortality was lower in both the normal-weight and overweight populations than in the underweight population, with an adjusted hazard ratio (AHR) of 0.5 (95% CI 0.4–0.7, p < 0.001) and 0.3 (95% CI 0.1–0.6, p = 0.002), respectively. Additionally, in the 1176 patients with available viral load data, there was significant difference between the underweight and normal-weight groups after adjustment for all factors, including viral load (p = 0.031). This result suggests that HIV-infected patients in Guangxi maintaining a BMI of 19–28 kg/m2, especially 24–28 kg/m2, have a reduced risk of death.
Similar content being viewed by others
Introduction
Since the human immunodeficiency virus (HIV)/acquired immunodeficiency syndrome (AIDS) epidemic crisis in 1981, HIV/AIDS has been one of the leading causes of death worldwide1. As of December 31, 2017, there were 985,034 reported cases of HIV/AIDS in China, with 232,290 reported death, resulting in a mortality rate of 23.6%2. Guangxi, the province with the second-highest HIV prevalence rate in Southwest China, according to the report of the Guangxi Health and Family Planning Commission3, had approximately 118 thousand total HIV/AIDS patients as of January 31, 2017, with 40,500 deaths, resulting in a mortality rate (34.3%) higher than the national average (23.8%) during the same period. Currently, the most frequently reported risk factors for HIV-related mortality in people living with HIV/AIDS (PLWHA) were not receiving antiretroviral therapy (ART)4,5 and having delayed ART initiation6. Clearly, due to the implementation of ART, the morbidity and mortality rates in PLWHA have significantly decreased7,8. Globally, an estimated 18.2 million PLWHA had received ART as of mid-2016, approximately 1.1 million patients had died of AIDS-related causes at the end of 2015, and the AIDS-related mortality rate decreased by approximately 28% from 2000 to 20159.
PLWHA on ART remain at higher risk of death than the general population10. In China, the National Free Antiretroviral Treatment Program (NFATP), initiated in 2002, and scaled up in 2003, has successfully reduced the overall HIV mortality rate5. However, in spite of the expansion of antiviral treatment coverage, China is still facing an enormous challenge from the high rate of AIDS-related death11. Wang et al. examined the temporal trend in HIV/AIDS-related deaths in China and found that the mortality rate increased significantly from 2000–2012, with an average annual percentage change of 22.3%12.
The average mortality rate among all treated patients was 2.63/100 person-years in Guangxi, China from 2010 to 201513. Studies have reported many independent factors related to death among ART patients, including late diagnosis14, advanced WHO disease stage15,16,17 and lower CD4 cell count5,15,17,18. In addition to these factors, many studies have shown that a low body mass index (BMI), mostly a BMI of <18.5 kg/m2, seems to be a risk factor for mortality in PLWHA on ART15,17,19,20,21.
Historically, studies have used BMI as the general obesity index. The BMI classification is defined in China as follows22,23: underweight (<18.5 kg/m2), normal weight (18.5–23.9 kg/m2), overweight (24–27.9 kg/m2), and obese (≥28 kg/m2). The full spectrum of weight categories is seen in HIV patients currently, and different levels of BMI seem to be closely related to AIDS progression in PLWHA. A longitudinal study of the Miami HIV-1-infected drug abusers cohort suggests that mild-to-moderate obesity in HIV-1-infected chronic drug users does not impair immune function and is associated with improved HIV-1-related survival rates24. A low baseline BMI and a decreasing BMI during follow-up were independently predictive of progression to AIDS, meaning that overweight patients may progress more slowly to AIDS25. In addition, many other studies similarly reported that a low baseline BMI and a BMI decrease were associated with a high risk of HIV progression and opportunistic infections during follow-up26,27,28,29. However, in Kim et al.’s study, overweight and obese HIV patients had a higher risk of multimorbidity than normal or underweight patients30. In another study, weight gain among underweight or normal-weight HIV ART participants predicted reduced inflammation and improved survival, but this effect did not apply to overweight or obese participants31.
Zhang et al. conducted a survival analysis of PLWHA on ART in Ningbo, China and found that a BMI of <18.5 was one of the risk factors for mortality19. Two studies in Guangxi showed that a BMI of <18 kg/m2 was associated with an increased risk of asymptomatic pulmonary tuberculosis among HIV patients32 and that HIV patients with an initial BMI of <18.5 kg/m2 had increased mortality33. In this study, we retrospectively collected data from China’s National Free Antiretroviral Treatment Program (NFATP) in Nanning city and Guigang city in Guangxi, China, aiming to ascertain the BMI variance status and to investigate the association between BMI and mortality in Chinese HIV patients on ART.
Results
Baseline characteristics
Data were collected from May 19, 2005 to June 30, 2016. A total of 10,111 HIV-positive patients received combination ART in the study settings. Of these patients, we excluded 4816 without a recorded height, 6 without recorded baseline weight, and 190 without recorded follow-up weight, leaving 5101 individuals enrolled (12,458.9 person-years) (Fig. 1). Among these patients, 1439 (28.2%) were underweight, 3047 (59.7%) had a normal weight, 548 (10.7%) patients were overweight and 67 (1.3%) were obese. Table 1 shows the baseline characteristics of the participants. Among the participants, 1281 (25.1%) patients were aged 30–39 years; the mean age of the patients was 46.86 years (32.79–60.93), and the median age was 45 years (IQR 36–58). Among the eligible patients, 3538 (69.4%) were male, and 3524 (69.1%) were married or living with a partner. Most (63.2%) patients’ CD4 T cell count were less than 200 cells/μL. The main route of HIV infection was heterosexual intercourse (85.1%), and 1286 (25.2%) patients’ initial antiretroviral regimen was EFV + 3TC + TDF (Efavirenz, Lamivudine, Tenofovir disoproxil fumarate). A total of 1903 (37.3%) patients were in WHO clinical stage I before ART. The four groups were significantly different in age, sex, marital status, CD4 T cell count, route of HIV infection, initial antiretroviral regimen and WHO clinical stage before ART. The median CD4 T cell count was 51 (20–172) cells/μL in the underweight group, 149 (41–274) cells/μL in the normal-weight group, 221(106–322) cells/μL in the overweight group and 281 (200–368) cells/μL in the obese group.
Chart of the inclusion and exclusion criteria in this study. A total of 10,111 HIV-positive patients received combination ART in the study settings, but 4816 had no height data and 6 had no weight data at baseline, and 190 had no weight data during follow-up; these patients were excluded, leaving a total of 5,101 individuals enrolled in this study.
The mortality rate of HIV-infected patients who received ART in the different BMI groups
The mortality rate was 1.4/100 person-years (95% CI 1.2–1.6) in Guangxi. The mortality rates in the four groups were significantly different (p < 0.001). The mortality rate was 2.4/100 person-years (95% CI 1.9–2.9) in the underweight group, 1.1/100 person-years (95% CI 0.9–1.3) in the normal-weight group, 0.5/100 person-years (95% CI 0.1–0.9) in the overweight group and 2.4/100 person-years (95% CI −0.2–4.9) in the obese group (Table 2). Figure 2 shows that the cumulative mortality rate in the underweight group was significantly higher than that in the overweight and normal-weight groups and that the mortality rate in the obese group was higher than that in the normal-weight and overweight groups.
Cumulative mortality rates of patients in the different BMI groups. Figure 2 shows that the cumulative mortality rate in the underweight group was significantly higher than that in the overweight and normal-weight groups and that the mortality rate in the obese group was higher than that in the normal-weight and overweight groups.
Associations between BMI and time to death
We used a Cox proportional hazards regression model to control for confounding factors. Table 3 shows that patients in the normal-weight group had a lower risk of death than those in the underweight group, while patients in the overweight group had the lowest risk of death. With respect to the underweight group, the hazard ratio of overweight adjusted for all factors except viral load was 0.3 (95% CI 0.1–0.6, p = 0.002). In addition, there was a significant difference in the mortality rate (p < 0.001) between the underweight and normal-weight groups when adjusted for all factors except viral load. There was also a significant difference between the underweight and normal-weight groups after adjustment for all factors, including viral load (p = 0.031).
Stratified analysis
We further controlled for mixed factors by stratified analysis. The mortality rates were significantly different among the underweight, overweight and obese groups, as stratified by the baseline characteristics of age (30–39 and ≥60), sex (male), marital status (married or living with a partner), WHO clinical stage before ART (stages 1, 2 and 3), CD4 T cell count (<200 cells/μL) and initial antiretroviral regimen (EFV + 3TC + TDF). There was no significant difference in other baseline characteristics (Supplementary Table S1).
We further analysed the cumulative mortality rates in the four groups stratified into different CD4 T cell count groups. Figure 3 shows that there was a significant difference in the Kaplan-Meier analysis results between the CD4 < 100 cells/μL group and the other three CD4 groups. In the CD4 < 100 cells/μL group, the cumulative mortality rate was the highest in the underweight group and was higher in the BMI < 18.5 kg/m2 group than in the normal-weight and obese groups; the overweight group had the lowest mortality rate, which was similar to that in the group with 200 < CD4 < 350 cells/μL. In the 100 < CD4 < 200 cells/μL group, the cumulative mortality was inversely related to the BMI with respect to the time of follow-up (years). In the CD4 > 350 cells/μL group, the mortality rates in the underweight and normal-weight groups were higher than those in the other two groups. We observed the effect of BMI on the mortality rate in the different CD4 groups: patients with a lower BMI had a higher risk of mortality when the CD4 cell count was less than 350cells/μL, similar to the results without stratification by the CD4 T cell count.
Kaplan-Meier Survival Curves for the four weight groups in each stratum of CD4 T cell count. (a) Cumulative mortality rate of HIV-infected patients in the “CD4 < 100/µL” subgroup. (b) Cumulative mortality rate of HIV-infected patients in the “100 ≤ CD4 < 200/µL” subgroup. (c) Cumulative mortality rate of HIV-infected patients in the “200 ≤ CD4 < 350/µL” subgroup. (d) Cumulative mortality rate of HIV-infected patients in the “CD4 ≥ 350/µL” subgroup.
BMI changes during ART and mortality rate changes by BMI
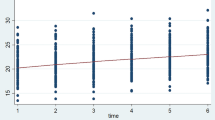
Figure 4 shows the average BMI with respect to the time of follow-up in the four baseline BMI groups. Among the different BMI groups, there was almost no change in the patients’ BMI values from baseline to the end of follow-up. Figure 5 indicates the change in the mortality rate with respect to BMI; the mortality rate decreased with increases in the baseline and final BMI. This figure demonstrates that the mortality rate was relatively lower for a BMI of 19–28 kg/m2 and was lowest for a BMI of 24–28 kg/m2.
Mean BMI and standard deviation with respect to the time of follow-up in the four baseline BMI groups. (a) Average BMI with respect to follow-up time in the underweight group. (b) Average BMI with respect to follow-up time in the normal-weight group. (c) Average BMI with respect to follow-up time in the overweight group. (d) Average BMI with respect to follow-up time in the obese group.
Discussion
This study was a retrospective cohort study over an 11-year period (2005–2016) in HIV-infected patients who received ART in Guangxi, China. The mortality rate was 1.4/100 person-years (95% CI 1.2–1.6). In an earlier study in Guangxi, the average mortality rate among all treated patients was 2.6/100 person-years from 2010 to 201513. In our study, the mortality rate of HIV-infected patients who received ART in the different BMI groups was 1.42/100 person-years. Possible reasons for the difference in the mortality rate between our study and that of Tang et al.13 include: (1) The earlier study13 included HIV patients from all cities of Guangxi, including the two cities that we studied in the current study. (2) The eligible patients in our study were PLWHA with ART who had available BMI data. Therefore, the characteristics of the subjects were different from those in the earlier study. In a meta-analysis conducted in resource-limited countries of Africa, Asia, and South and Central America, mortality rates ranged from 2.6% to 29.7%34, higher than those observed in our study. The highest rate was reported in a multi-regional South American cohort and the lowest was in sub-Saharan Africa34.
In our study, higher baseline BMI (19–28 kg/m2) was associated with a lower mortality rate than that in the underweight group. We compared the baseline characteristics among the four groups and found significant differences among these four groups; in addition, we found that the patients in the normal and overweight groups had a lower risk of death than those in the underweight group (the hazard ratios adjusted for all factors were 0.5 (95% CI 0.4–0.7) and 0.3 (95% CI 0.1–0.6) for normal-weight and overweight, respectively). When we controlled for the baseline characteristics by stratified analysis, we found similar results—the mortality rate in the normal-weight group was lower than that in the underweight and overweight groups. Similarly, Troiano et al.35 showed an U-shaped association between BMI and mortality. Studies in Southeast Asia36,37 and sub-Saharan Africa38,39,40 reported that a low baseline BMI predicts early mortality in resource-limited settings. In another study, the mortality rate of underweight PLWHA prior to ART was almost two-fold that of normal-weight PLWHA in the 1st two years of ART41. A meta-analysis showed that compared to normal-weight, overweight was related to lower all-cause mortality, while the outcome was opposite for obesity42. We also found that BMI changed little from baseline to the end of follow-up, which further demonstrated that the baseline BMI and mortality rate are closely related.
In our study, the CD4 T cell count was a key differential factor among the four groups. Therefore, we controlled for the CD4 T cell count via stratified analysis. We found that the mortality rate in the underweight group was higher than that in the normal-weight group, which was higher than that in the overweight group, for patients with CD4 < 350 cells/μL but was slightly different for patients with CD4 > 350 cells/μL; the reason may be the small sample size, indicating that in the different CD4 T cell count groups, the effect of BMI on the mortality rate of patients in this study is generally similar. Regarding the change in the CD4 T cell count during ART, our findings are consistent with other findings that showed an enhanced increase in the CD4 T cell count of 1 year after ART initiation43. Similarly, in other study, there was no difference associated with weight in the undetectable viral load or the increase in the CD4 T cell count at 3–9 months after ART initiation44. Therefore, we suggest that underweight HIV patients in Guangxi who received ART may be at greater risk of mortality and that patients maintaining a BMI of 19–28 kg/m2, especially 24–28 kg/m2, have a relatively lower risk of mortality. However, a study showed that weight may affect immune cell counts over the course of ART45. In other studies, increased BMI values were associated with increased CD4 counts and improved survival24,25. Similarly, several previous studies have shown that increases in BMI were positively associated with increased CD4+ T cell counts in HIV-negative children and women46,47,48.
Adipose tissue is an active endocrine and paracrine organ that regulates energy storage, immunity and inflammation48. Adipocytes secrete cytokines and adipokines, including adiponectin and leptin. The plasma leptin concentration is positively related to the percentage of adipose tissue49. Leptin is a hormone that is similar to cytokines in both function and structure50. Both thymic development and peripheral lymphocyte count and function are reduced in leptin-deficient mice51. In some studies, leptin replacement therapy reversed the defects in lymphocyte numbers and the decrease in function in humans52,53. Therefore, adipokines may be the possible mechanism underlying the effect of BMI on mortality in HIV-infected patients.
There were a few limitations in this study. First, this study is a retrospective cohort study, so there may be bias in subject selection, leading to a significant difference in the baseline characteristics. However, we controlled for and adjusted the factors by stratified analysis and multivariate analysis. Second, due to the small number of obese patients in the study, the results for the obese group should be considered with caution. Third, most of the viral load data were missing, and we were not able to assess the real influence of the viral load on the results. However, despite these limitations, the study provides meaningful epidemiological data on decreasing the mortality risk and prolonging the survival time of HIV-infected patients who receive ART. Additionally, to our knowledge, this study is the first retrospective cohort study over an 11-year period in China—even in Asia—and has great public health significance.
In conclusion, compared to normal weight, underweight increased the risk of death among PLWHA who received ART, while overweight decreased the risk of death. Our study provides further evidence that underweight may contribute to increased mortality in HIV-positive patients and suggests the optimal range of BMI in PLWHA in Guangxi, China.
Methods
Study design and included patients
This HIV treatment retrospective cohort study was conducted in Guigang and Nanning cities, Guangxi, China. PLWHA who received antiretroviral treatment (ART) were included. Beginning in 2002, the criteria to receive free combination ART were as follows: (1) a CD4 T cell count of <200 cells/μL, (2) a total lymphocyte count of less than 1200 cells/μL, or (3) a WHO disease stage of 3 or 422. In 2008, the criteria were changed to the following: (1) a CD4 count of ≤350 cells/μL or a WHO disease stage of 3 or 423. The inclusion criteria for this study were as follows: PLWHA ≥18 years of age who received standard ART and had baseline height and weight data and follow-up weight data. The exclusion criteria were as follows: absence of baseline weight or height data or of follow-up weight data, presence of severe liver or kidney disease, and pregnancy (in women). All included patients had weight records, and most had records of the CD4 T cell count, viral load clinical index, other diseases, ART regimen and survival status at fixed time points (0.5, 1, 2, and 3 months after initiation of ART and followed up thereafter every three months). However, if the treatment programme was changed, the follow-up period was restarted. Pre-ART BMI data was not changed even if the treatment programme changed.
Ethics statement
In this study, every patient provided written informed consent for our specific retrospective study before enrolment. The study procedures were clearly explained to the participants, and they were offered the opportunity to ask questions. All methods were performed in accordance with the relevant guidelines and regulations, and all methods were approved by the Ethics and Human Subjects Committee of Guangxi Medical University (Ethical Review No. 20130305-17).
Data collection and definitions
Data were collected from the Fourth People’s Hospital of Nanning city and from the Guigang Center for Disease Control and Prevention (CDC). These data were collected from May 19, 2005 to June 30, 2016. In the study, baseline information was collected at initiation of ART. BMI was calculated according to the following formula: “BMI = weight (kg)/height (m)2” and the grade of BMI was calculated following the criteria established by the Health Industry Standards of the People’s Republic of China: underweight (BMI < 18.5 kg/m2), normal weight (18.5–24.0 kg/m2), overweight (24.0–28.0 kg/m2), and obese (>28.0 kg/m2)22,23. The CD4 T cell count stage was classified as follows: stage 1 (≥350cells/μL), stage 2 (200–350 cells/μL), stage 3 (200–349 cells/μL), and stage 4 (<200cells/μL). We collected baseline and follow-up weight records to calculate BMI values. The outcomes included censored data and death. June 30, 2016 was established as the last date of follow-up.
Definition of clinical diseases and clinical test indexes
All clinical diseases includes the following: TB infection, skin lesion, thrush, oral hairy leukoplakia, persistent diarrhoea, continuous or intermittent fever, recurrent severe bacterial infections, disseminated non-tuberculosis bacillus infection, oesophageal candidiasis, extrapulmonary cryptococcal infection, Yersinia pneumocystis pneumonia, disseminated fungal disease, cytomegalovirus infection, extrapulmonary tuberculosis, repeated severe bacterial pneumonia, chronic herpes simplex virus infection, herpes zoster, toxoplasma encephalopathy, Kaposi sarcoma, and brain lymphoma. Clinical test indexes included white blood cell count, lymphocyte count, blood platelet count, haemoglobin, serum creatinine, blood urea nitrogen, triglyceride, total cholesterol, blood glucose, blood amylase, aspartate aminotransferase, alanine aminotransferase and total bilirubin.
Statistical analysis
The mortality of HIV-positive patients was calculated by the number of annual deaths divided by the total number of 100 person-years. We used a Chi-square test (for categorical variables) to compare the characteristics among the four BMI groups (underweight, normal-weight, overweight, and obese). Kaplan-Meier analysis was used to calculate survival probabilities for cumulative mortality, and Cox proportional hazards ratios were used to evaluate the related factors among the four groups. We compared the four groups using Cox proportional hazards regression analyses stratified by age, sex, CD4 T cell count, marital status, WHO clinical stage before ART, route of HIV infection, and initial antiretroviral regimen to estimate hazard ratios (HRs; reported with 95% CIs) and adjusted hazard ratios (AHRs; reported with 95% CIs). To ensure that the characteristics did not affect the results among the four groups, we used a Chi-square test for each baseline characteristic and clinical disease; we then used stratified analysis to control for and balance the baseline characteristics between the normal-weight and overweight groups and Cox proportional hazards models to evaluate and adjust the mortality results among the underweight, overweight, and obese groups. The data were analysed using Statistical Package for the Social Sciences (SPSS) version 20.0 (SPSS Inc., Chicago, USA) and GraphPad Prism version 6.0 (GraphPad Software, San Diego, California, USA).
Ethical statement
In this study, every patient provided written informed consent for our specific retrospective study before enrolment. The study procedures were clearly explained to the participants, and they were offered the opportunity to ask questions. The study was approved by the Ethics and Human Subjects Committee of Guangxi Medical University (Ethical Review No. 20130305-17).
Data Availability
Since the data contain information of HIV-infected patients, which involve patient privacy, they cannot be disclosed.
References
Rubens, M., Ramamoorthy, V., Saxena, A., Shehadeh, N. & Appunni, S. HIV Vaccine: Recent Advances, Current Roadblocks, and Future Directions. Journal of immunology research 2015, 560347, https://doi.org/10.1155/2015/560347 (2015).
Fox, M. P. et al. Initiating patients on antiretroviral therapy at CD4 cell counts above 200 cells/microl is associated with improved treatment outcomes in South Africa. Aids 24, 2041–2050, https://doi.org/10.1097/QAD.0b013e32833c703e (2010).
http://www.gxhfpc.gov.cn/xxgks/yqxx/yqyb/2017/0213/34174.html.
Cuong do, D. et al. Survival and causes of death among HIV-infected patients starting antiretroviral therapy in north-eastern Vietnam. Scandinavian journal of infectious diseases 44, 201–208, https://doi.org/10.3109/00365548.2011.631937 (2012).
Zhang, F. et al. Effect of earlier initiation of antiretroviral treatment and increased treatment coverage on HIV-related mortality in China: a national observational cohort study. The Lancet. Infectious diseases 11, 516–524, https://doi.org/10.1016/S1473-3099(11)70097-4 (2011).
Lesko, C. R. et al. The effect of antiretroviral therapy on all-cause mortality, generalized to persons diagnosed with HIV in the USA, 2009–11. International journal of epidemiology 45, 140–150, https://doi.org/10.1093/ije/dyv352 (2016).
Collaboration, H.-C. et al. The effect of combined antiretroviral therapy on the overall mortality of HIV-infected individuals. Aids 24, 123–137, https://doi.org/10.1097/QAD.0b013e3283324283 (2010).
Wada, N. et al. Cause-specific life expectancies after 35 years of age for human immunodeficiency syndrome-infected and human immunodeficiency syndrome-negative individuals followed simultaneously in long-term cohort studies, 1984–2008. American journal of epidemiology 177, 116–125, https://doi.org/10.1093/aje/kws321 (2013).
Mupere, E. et al. Lean tissue mass wasting is associated with increased risk of mortality among women with pulmonary tuberculosis in urban Uganda. Annals of epidemiology 22, 466–473, https://doi.org/10.1016/j.annepidem.2012.04.007 (2012).
Zhao, Y., Poundstone, K. E., Montaner, J. & Wu, Z. Y. New policies and strategies to tackle HIV/AIDS in China. Chinese medical journal 125, 1331–1337 (2012).
Wang, C. W., Chan, C. L. & Ho, R. T. HIV/AIDS-related deaths in China, 2000–2012. AIDS care 27, 849–854, https://doi.org/10.1080/09540121.2015.1005568 (2015).
Tang, Z. et al. Effects of high CD4 cell counts on death and attrition among HIV patients receiving antiretroviral treatment: an observational cohort study. Sci Rep 7, 3129, https://doi.org/10.1038/s41598-017-03384-7 (2017).
Chen, L. et al. HIV cause-specific deaths, mortality, risk factors, and the combined influence of HAART and late diagnosis in Zhejiang, China, 2006–2013. Sci Rep 7, 42366, https://doi.org/10.1038/srep42366 (2017).
Ladep, N. G. et al. Hepatitis B Co-Infection is Associated with Poorer Survival of HIV-Infected Patients on Highly Active Antiretroviral Therapy in West Africa. Journal of AIDS & clinical research Suppl 3, https://doi.org/10.4172/2155-6113.S3-006 (2013).
Wen, Y. et al. Baseline factors associated with mortality within six months after admission among hospitalized HIV-1 patients in Shenyang, China. Internal medicine 53, 2455–2461 (2014).
Damtew, B., Mengistie, B. & Alemayehu, T. Survival and determinants of mortality in adult HIV/Aids patients initiating antiretroviral therapy in Somali Region, Eastern Ethiopia. The Pan African medical journal 22, 138, https://doi.org/10.11604/pamj.2015.22.138.4352 (2015).
Bisson, G. P. et al. Risk factors for early mortality on antiretroviral therapy in advanced HIV-infected adults. Aids 31, 2217–2225, https://doi.org/10.1097/QAD.0000000000001606 (2017).
Zhang, J. H. et al. [Survival analysis of HIV/AIDS patients with access to highly antiretroviral therapy in Ningbo during 2004–2015]. Zhonghua liu xing bing xue za zhi = Zhonghua liuxingbingxue zazhi 37, 1262–1267, https://doi.org/10.3760/cma.j.issn.0254-6450.2016.09.016 (2016).
Woodd, S. L. et al. Risk factors for mortality among malnourished HIV-infected adults eligible for antiretroviral therapy. BMC infectious diseases 16, 562, https://doi.org/10.1186/s12879-016-1894-3 (2016).
Liu, E. et al. Nutritional status and mortality among HIV-infected patients receiving antiretroviral therapy in Tanzania. The Journal of infectious diseases 204, 282–290, https://doi.org/10.1093/infdis/jir246 (2011).
Zhang, B. et al. Maternal Prepregnancy Body Mass Index and Small for Gestational Age Births in Chinese Women. Paediatric and perinatal epidemiology 30, 550–554, https://doi.org/10.1111/ppe.12315 (2016).
Yang, W. et al. Association between Body Mass Index and All-Cause Mortality in Hypertensive Adults: Results from the China Stroke Primary Prevention Trial (CSPPT). Nutrients 8, https://doi.org/10.3390/nu8060384 (2016).
Shor-Posner, G. et al. When obesity is desirable: a longitudinal study of the Miami HIV-1-infected drug abusers (MIDAS) cohort. Journal of acquired immune deficiency syndromes 23, 81–88 (2000).
Shuter, J., Chang, C. J. & Klein, R. S. Prevalence and predictive value of overweight in an urban HIV care clinic. Journal of acquired immune deficiency syndromes 26, 291–297 (2001).
Ayele, T., Jarso, H. & Mamo, G. Clinical Outcomes of Tenofovir Versus Zidovudine-based Regimens Among People Living with HIV/AIDS: a Two Years Retrospective Cohort Study. The open AIDS journal 11, 1–11, https://doi.org/10.2174/1874613601711010001 (2017).
Feigl, A. B. et al. The Effect of HIV and the Modifying Effect of Anti-Retroviral Therapy (ART) on Body Mass Index (BMI) and Blood Pressure Levels in Rural South Africa. PLoS One 11, e0158264, https://doi.org/10.1371/journal.pone.0158264 (2016).
Martinez, S. S. et al. Effect of BMI and fat mass on HIV disease progression in HIV-infected, antiretroviral treatment-naive adults in Botswana. The British journal of nutrition 115, 2114–2121, https://doi.org/10.1017/S0007114516001409 (2016).
Reid, M. J. A. et al. Higher Body Mass Index Is Associated With Greater Proportions of Effector CD8+ T Cells Expressing CD57 in Women Living With HIV. Journal of acquired immune deficiency syndromes 75, e132–e141, https://doi.org/10.1097/QAI.0000000000001376 (2017).
Kim, D. J. et al. Multimorbidity patterns in HIV-infected patients: the role of obesity in chronic disease clustering. Journal of acquired immune deficiency syndromes 61, 600–605, https://doi.org/10.1097/QAI.0b013e31827303d5 (2012).
Mave, V. et al. Inflammation and Change in Body Weight With Antiretroviral Therapy Initiation in a Multinational Cohort of HIV-Infected Adults. The Journal of infectious diseases 214, 65–72, https://doi.org/10.1093/infdis/jiw096 (2016).
Zhang, Y. et al. Diagnosis of pulmonary tuberculosis among asymptomatic HIV+ patients in Guangxi, China. Chinese medical journal 123, 3400–3405 (2010).
Spillane, H. et al. Incidence, risk factors and causes of death in an HIV care programme with a large proportion of injecting drug users. Tropical medicine & international health: TM & IH 17, 1255–1263, https://doi.org/10.1111/j.1365-3156.2012.03056.x (2012).
Gupta, A. et al. Early mortality in adults initiating antiretroviral therapy (ART) in low- and middle-income countries (LMIC): a systematic review and meta-analysis. PLoS One 6, e28691, https://doi.org/10.1371/journal.pone.0028691 (2011).
Troiano, R. P., Frongillo, E. A. Jr., Sobal, J. & Levitsky, D. A. The relationship between body weight and mortality: a quantitative analysis of combined information from existing studies. International journal of obesity and related metabolic disorders: journal of the International Association for the Study of Obesity 20, 63–75 (1996).
Argemi, X. et al. Impact of malnutrition and social determinants on survival of HIV-infected adults starting antiretroviral therapy in resource-limited settings. Aids 26, 1161–1166, https://doi.org/10.1097/QAD.0b013e328353f363 (2012).
Paton, N. I., Sangeetha, S., Earnest, A. & Bellamy, R. The impact of malnutrition on survival and the CD4 count response in HIV-infected patients starting antiretroviral therapy. Hiv Med 7, 323–330, https://doi.org/10.1111/j.1468-1293.2006.00383.x (2006).
Johannessen, A. et al. Predictors of mortality in HIV-infected patients starting antiretroviral therapy in a rural hospital in Tanzania. Bmc Infect Dis 8, 52, https://doi.org/10.1186/1471-2334-8-52 (2008).
Maskew, M. et al. Gender differences in mortality and CD4 count response among virally suppressed HIV-positive patients. Journal of women’s health 22, 113–120, https://doi.org/10.1089/jwh.2012.3585 (2013).
May, M. et al. Prognosis of patients with HIV-1 infection starting antiretroviral therapy in sub-Saharan Africa: a collaborative analysis of scale-up programmes. Lancet 376, 449–457, https://doi.org/10.1016/S0140-6736(10)60666-6 (2010).
Sharma, A. et al. Relationship between Body Mass Index and Mortality in HIV-Infected HAART Users in the Women’s Interagency HIV Study. PLoS One 10, e0143740, https://doi.org/10.1371/journal.pone.0143740 (2015).
Flegal, K. M., Kit, B. K., Orpana, H. & Graubard, B. I. Association of all-cause mortality with overweight and obesity using standard body mass index categories: a systematic review and meta-analysis. Jama 309, 71–82, https://doi.org/10.1001/jama.2012.113905 (2013).
Koethe, J. R., Jenkins, C. A., Shepherd, B. E., Stinnette, S. E. & Sterling, T. R. An optimal body mass index range associated with improved immune reconstitution among HIV-infected adults initiating antiretroviral therapy. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 53, 952–960, https://doi.org/10.1093/cid/cir606 (2011).
Mossdorf, E. et al. Improved antiretroviral treatment outcome in a rural African setting is associated with cART initiation at higher CD4 cell counts and better general health condition. BMC infectious diseases 11, 98, https://doi.org/10.1186/1471-2334-11-98 (2011).
Crum-Cianflone, N. F. et al. Impact of weight on immune cell counts among HIV-infected persons. Clinical and vaccine immunology: CVI 18, 940–946, https://doi.org/10.1128/CVI.00020-11 (2011).
Nowicki, M. J. et al. Correlates of CD4+ and CD8+ lymphocyte counts in high-risk immunodeficiency virus (HIV)-seronegative women enrolled in the women’s interagency HIV study (WIHS). Human immunology 68, 342–349, https://doi.org/10.1016/j.humimm.2007.01.007 (2007).
Womack, J. et al. Obesity and immune cell counts in women. Metabolism: clinical and experimental 56, 998–1004, https://doi.org/10.1016/j.metabol.2007.03.008 (2007).
Zaldivar, F. et al. Body fat and circulating leukocytes in children. International journal of obesity 30, 906–911, https://doi.org/10.1038/sj.ijo.0803227 (2006).
Considine, R. V. et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. The New England journal of medicine 334, 292–295, https://doi.org/10.1056/NEJM199602013340503 (1996).
Tilg, H. & Moschen, A. R. Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nature reviews. Immunology 6, 772–783, https://doi.org/10.1038/nri1937 (2006).
Lord, G. M. et al. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 394, 897–901, https://doi.org/10.1038/29795 (1998).
Farooqi, I. S. et al. Beneficial effects of leptin on obesity, T cell hyporesponsiveness, and neuroendocrine/metabolic dysfunction of human congenital leptin deficiency. J Clin Invest 110, 1093–1103, https://doi.org/10.1172/JCI15693 (2002).
Oral, E. A. et al. Leptin replacement therapy modulates circulating lymphocyte subsets and cytokine responsiveness in severe lipodystrophy. The Journal of clinical endocrinology and metabolism 91, 621–628, https://doi.org/10.1210/jc.2005-1220 (2006).
Acknowledgements
We would like to express our gratitude to all of staffs from Fourth People’s Hospital of Nanning city and Guigang Centers for Disease Control and Prevention (CDC) in Guangxi, China, for their help in collecting, verifying, and cleaning the data used in this study. We are also thankful to the Guangxi Center for Disease Control and Prevention (Guangxi, China) for their support in gathering data. The study was supported by National Natural Science Foundation of China (NSFC, 81560326, 81460511), Guangxi Scientific and Technological Key Project (Gui Ke Gong 14124003-1), Guangxi Bagui Scholar (to Junjun Jiang), Guangxi University “100-Talent” Program & Guangxi university innovation team and outstanding scholars program (Gui Jiao Ren 2014[7]).
Author information
Authors and Affiliations
Contributions
H.L., F.Y.W. and L.Y. designed the study. X.L.Q., S.R.M., J.P.H., C.W.Q., Y.F.L. and Y.X.H. collected the data. J.J.J. and H.F.L. analyzed the data. J.G.H., N.Z., B.Y.L. and C.Y.N. interpreted the data. F.X.Q. and Y.Y.L. designed the figures. J.J.J. and H.F.L. drafted the report. A.S.A. provided critical comments on the 1st draft of the manuscript. All authors reviewed, revised, and approved the final draft of the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiang, J., Qin, X., Liu, H. et al. An optimal BMI range associated with a lower risk of mortality among HIV-infected adults initiating antiretroviral therapy in Guangxi, China. Sci Rep 9, 7816 (2019). https://doi.org/10.1038/s41598-019-44279-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-44279-z
This article is cited by
-
Effects of undernutrition on mortality and morbidity among adults living with HIV in sub-Saharan Africa: a systematic review and meta-analysis
BMC Infectious Diseases (2021)
-
Effect of tuberculosis infection on mortality of HIV-infected patients in Northern Tanzania
Tropical Medicine and Health (2020)
-
Effect of AIDS-defining events at initiation of antiretroviral therapy on long-term mortality of HIV/AIDS patients in Southwestern China: a retrospective cohort study
AIDS Research and Therapy (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.