Abstract
Extensive regions of tropical forests are subjected to high rates of deforestation and forest regrowth and both are strongly affect soil nutrient cycling. Nitrogen (N) dynamics changes during forest regrowth and the recovery of forests and functioning similar to pristine conditions depends on sufficient N availability. We show that, in a chronosequence of Amazonian forests, gross nitrification and, as a result, nitrate-to-ammonium (NO3−: NH4+) ratio were lower in all stages of regrowing forests (10 to 40 years) compared to pristine forest. This indicates the evolution of a more conservative and closed N cycle with reduced risk for N leaking out of the ecosystem in regrowing forests. Furthermore, our results indicate that mineralization and nitrification are decoupled in young regrowing forests (10 years), such as that high gross mineralization is accompanied by low gross nitrification, demonstrating a closed N cycle that at the same time maintains N supply for forest regrowth. We conclude that the status of gross nitrification in disturbed soil is a key process to understand the mechanisms of and time needed for tropical forest recovery.
Similar content being viewed by others
Introduction
In the Brazilian Amazon region, almost 800 000 km2 of land has been deforested, mainly for soya bean cultivation, logging and cattle ranching1. The high rate of tropical deforestation led to global concern since these areas are a hot spot of biodiversity and have direct influence on the global climate through hydrology and exchange of greenhouse gases2,3,4,5. However, a large area of approximately 167 000 km2 previously deforested land has been abandoned after exploitation6 and secondary forests have established on that land7. The regrowth area in the Amazon is increasing6, but our current knowledge about nutrient availability, biogeochemical processes, and how the post-disturbance regeneration influences these processes is poorly understood8. Likewise, nutrient shortage in deforested areas is expected9, but the influence and magnitude of limitation, which can drive the recovery trajectory, on regrowth forest are still uncertain10.
Early secondary forests have high growth rates with rapidly increasing forest biomass11, even when N is apparently limited12. This indicates that feedback mechanisms on soil N availability exist, providing sufficient plant available N to maintain forest regrowth. Microbial processes, such as mineralization and nitrification, drive the soil N cycle and thereby control the amount of organic and inorganic N forms in soil13,14. Mineralization of soil organic matter (SOM) is responsible for inorganic N production in terrestrial ecosystems, which is important for plant N uptake that occurs mainly in inorganic form. The NH4+ released by mineralization also supports nitrification15, the oxidation of NH4+ to NO3−. These two inorganic N forms may have different fates in soils, as immobilization in biomass, leaching and gas losses16, and the occurrence and magnitude of these pathways might influence the forest growth17.
Davidson et al.8 investigated the N cycling recovery in secondary forest age chronosequences after agricultural abandonment in the Amazon region. These authors found indications for a conservative N cycling in soils of young successional tropical forests based on N and phosphorus (P) contents in leaves, litterfall and soils, low NO3−: NH4+ ratios as well as low nitrous oxide (N2O) emissions. However, the mechanistic changes in the soil N cycle during forest regrowth have not been studied in the Amazon Region. The actual dynamic of labile N in soils is best represented by gross soil N cycle dynamics, such as gross N mineralization and nitrification, since the gross transformations directly control the inorganic N availability for plants growth. Therefore, quantifying the gross N transformations in tropical regrowth forest soils is an important step in managing and enhancing abandoned managed areas, which also provides valuable information for model implementation.
We evaluated the gross soil N cycling in four forests, including one pristine forest and one regrowth forest (40 years old) located inside and two regrowth forests (10 and 20 years old) near the Ecological Station of Cuniã in the state of Rondônia, Western Amazonia, with focus on gross N mineralization and gross nitrification. The slash-and-burn practice was applied in all three regrowth areas studied here. The 15N pool dilution technique using the “virtual soil core” approach18 was used to quantify in situ gross N processes rates. Predominant soil type of the investigated forests is Plinthosol19, soil texture in the pristine forest is sandy loam with 55.4% (±4.4) sand, 39.1% (±4.8) silt and 4.9% (±0.7) clay (mean ± SD; N = 7). The vegetation is dominated by hardwood with abundance of palms20.
Results and Discussion
Sustained production of plant available N in tropical regrowth forests
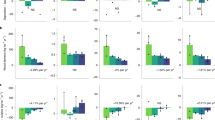
Changes in the internal soil N cycle as consequence of reforestation reflect alterations in the microbial and plant community during regrowth stage21. Rates of gross mineralization in the pristine forest at Cuniã (7.8 ± 4.7 µg N g−1 d−1; Fig. 1) are within the range of gross mineralization reported in other pristine tropical forests22,23. In a study in Eastern Amazon forest24 during the dry season, gross mineralization was measured in situ with a rate of 13.9 ± 3.8 and 7.2 ± 1.8 µg N g−1 d−1 from clay and sandy soils24 respectively, similar to the gross rate in the pristine forests in our study.
Gross N mineralization (gray bars) and nitrification (white bars) rates (µg N g−1 SDW d−1; Mean ± SE) in four forest soils at Cuniã Ecological Station, Rondônia, with one pristine forest (set to t = 0 years) and three regrowth forests (10, 20 and 40 years old). (a) For gross N mineralization, the 10 years old regrowth forest was significantly higher than the 40 years old forest (One way ANOVA with Tukey’s post hoc test P < 0.05) and slightly higher than the 20 years old forest (One way ANOVA with Tukey’s post hoc test P = 0.055). F value was 1.327 with degree of freedom of 3. (b) For gross nitrification, the pristine forest was significantly higher than all three regrowth forests (One way ANOVA with Tukey’s post hoc test P < 0.05). F value was 1.629 with degree of freedom of 3.
Along the chronosequence of forest regrowth gross N mineralization was nearly doubled in the youngest forest (10 years old; 14.8 ± 6.5 µg N g−1 d−1) but only half in the older regrowth forests (3.8 ± 2.1 µg N g−1 d−1) compared to the pristine forest (Fig. 1). A similar pattern was also observed in subtropical Australia, where gross N mineralization was 2 to 3 times higher in early monospecific forest plantation (5 years) than pristine forest and older (53 years) plantation25. In general, the main pattern seen in early successional forests is high rate of NH4+ release through mineralization25,26, although fewer contrasting results have also been reported27,28,29.
The observed change in gross N mineralization is not caused by SOM content. Across the chronosequence, the SOM content in pristine forest was significantly higher (P < 0.05) than in 10 years old regrowth, but not different from 20 and 40 years old regrowth forest (Table 1). This pattern is exactly the opposite as found for gross mineralization, hence the lowest SOM content was found in the forest with the highest gross N mineralization rate (10 years old regrowth forest). Instead, the quality of SOM30,31 might be more important for controlling N mineralization24,32,33. The C: N ratio, an indicator of the SOM quality and its degradation rate34, confirmed that, since the 10 years old regrowth had the lowest C: N ratio of 17.7 of the investigated forest soils (Table 1). Gross mineralization in early regrowth forests can be high due to the recent disturbance that redistribute SOM stored in deeper soil layers to soil surface26. Furthermore, the previous management, as slash-and-burn, degraded the SOM, releasing labile compounds that are easier to mineralize21,35. Subsequently, gross mineralization decreases over time, possibly due to depleting in labile SOM and are lower than in pristine forests due to reduced root exudation and rhizosphere priming36,37,38. After a fire event, soil texture might change, usually showing a decrease of clay and increase of sand content39. In addition, clay aggregates can change in terms of size and distribution in the soil. We only measured soil texture in the pristine forest, which had low clay content (4.9 ± 0.7%). Therefore, we expect small changes in soil texture along the chronosequence caused by the slash-and-burn practice, consequently hence having minimal effects on the measured microbial processes rates.
Our results indicate that plant available N is sustained during forest regrowth due to enhanced gross N mineralization. High N mineralization in the early successional stage provides plant available N, overcoming a potential N limitation of forest regrowth. With time, N demand will decrease, which is also reflected in the decrease in gross N mineralization over time of forest regrowth found by us (Fig. 1) and others25.
Conservative N cycling in tropical regrowth forests through decreased nitrification
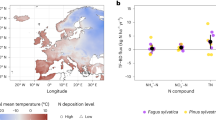
Secondary forests exhibit a more conservative N cycle compared to pristine forests in the Amazon region, indicated by the shift in the dominant inorganic N form in the soil towards NO3− (refs8,40), which is also observed at the chronosequence at Cuniã (Fig. 2). We show here that the underlying process is a change in gross nitrification, which was significantly lower in all stages of forest regrowth than the pristine forest in our chronosequence (Fig. 1). Gross nitrification rates of 3.27 ± 1.14 µg N g−1 d−1 in the pristine forest at Cuniã are in accordance with rates reported in earlier studies, which reported in situ gross nitrification in the range of 0.5 to 5.2 µg N g−1 d−1 (e.g. refs22,24,41).
Content of soil NH4+ and NO3− as well as NO3−: NH4+ ratio in pristine forest (set to t = 0 years) and three regrowth forests (10, 20 and 40 years) at the Ecological Station of Cuniã, Rondônia (Brazil). The contents were calculated from the first extraction after 15N labelling by subtracting the amount of tracer recovered (based on 15N enrichment). The black circle represents NH4+ content, the empty circle represents NO3− content and the symbol X represents the NO3−: NH4+ ratio. The unit of the N contents is µg N g−1 SDW and the values represent mean ± standard error.
Gross nitrification rates were lower in all regrowth stages, than in the pristine forest (Fig. 1), justifying the measured low soil NO3− content (Fig. 2), which confirms the idea of N retention and conservation during ecosystems succession8. This could be related to the alteration of the soil microbial community. In the Amazon region a higher abundance of nitrifiers was found in a pristine forest in comparison to regrowth forest soils42, explaining the higher rates in pristine forest. In addition, an enhanced plant N demand, competing with the nitrifiers for NH4+, could also contribute to a low gross nitrification in regrowing forests.
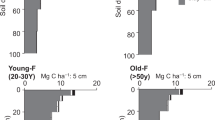
The pattern of gross nitrification observed in this study, based on pseudo-replicated field experiment, is consistent and in agreement with studies from other chronosequences of tropical and sub-tropical forests22,25,28,29,43,44 (Fig. 3), which enables generalizations. Although the magnitude of gross nitrification varies between the different studies, consistently higher gross nitrification rate in pristine than secondary forests has been found, corroborating gross nitrification as the most suitable N process to evaluate recovery of tropical forest ecosystems.
Relative gross nitrification rates in different tropical and subtropical pristine and secondary forests around the world. The figure compiles relative nitrification rates from pristine forest soil (black bar representing the highest nitrification rate in percentage) and secondary forests (plantation or regrowth) of different age (bars with different tones of gray). Data from refs22,25,28,43,44 and the present study.
Nitrogen cycling along a tropical forest chronosequence
Tropical regrowing forests are characterized by a closed N cycle with low risk for N losses, indicated by decreased NO3−: NH4+ ratios7,8, which was also observed in this study (Fig. 2). Here, we provide a mechanistic understanding of the biogeochemical processes responsible for the evolution of the N cycle under forest regrowth. The consistent pattern reported for gross nitrification (Figs 1 and 3) explains the observed pattern of low NO3−: NH4+ ratios in regrowing forests. The relative excess in NO3− in pristine forests promotes N losses by leaching and gaseous emission23,24,45,46. Regrowing forests, on the other hand, have a tighter N cycle with decreased N losses and enhanced N retention47.
Particularly the results from an investigation in a pristine sub-tropical forest and two forest plantations of different age in Australia are strikingly similar to our study25 (Fig. 3). Early regrowth forests are in both studies characterized by high rates of gross N mineralization and low rates of gross nitrification (Fig. 2), showing a decoupling of these two processes. As a consequence, inorganic N in the young forests is mainly in the form of NH4+, which leads to reduced N losses24,48, but maintains availability of N for plant uptake. Older regrowing forests, have a lower N demand49 and not only nitrification but also mineralization rates are low25 (Fig. 1).
Although we did not directly investigate this, our results infer that plants are probably crucial in regulating the observed pattern of dominant N pathways during the forest regrowth in this part of the Amazon (Fig. 4). Root exudation and plant N uptake control the availability of inorganic N by affecting N cycling processes. The root exudation of labile organic compounds in the pristine forest provides not only a substrate for N mineralization but can stimulate gross mineralization further by rhizosphere priming37,38,50 (Fig. 4c). In regrowing forests with lower tree biomass, root exudation is lower, thereby reducing the effects on gross mineralization (Fig. 4a,b). In the youngest regrowth forest, this negative effect is though more than compensated for by the presence of labile SOM from the slash-and-burn practices21,35. The N assimilation in biomass is larger in regrowth in comparison to pristine forests, which have more N loss from litterfall than regrowth forests40. Because of that, the net uptake (gross N uptake minus N loss) is higher in regrowth (Fig. 4a,b) in comparison to pristine51 (Fig. 4c), decreasing the availability of NH4+ for nitrifiers, leading to a decrease in nitrification. As a consequence, the NO3−: NH4+ ratio will vary according to the forest status.
Conceptual model of N cycling along a forest chronosequence in Amazon region, (a,b). Nitrogen pathways in regrowing forest soils of different ages after one time disturbance by slash-and-burn. In the early regrowth forest (10 years; a) a new source of labile N from the burning of biomass stimulates gross mineralization and, as consequence of investment in forest growth, higher N uptake by plants. Nitrification and N uptake receiving support from N mineralization in 20 and 40 years old regrowth forests (b). In pristine forest (c) root exudations stimulates mineralization, which supports nitrification. See text for more details.
Relevance of gross nitrification as an indicator of forest recovery
Gross N mineralization and gross nitrification are sensitive to environmental changes and ecosystem disturbance. Our findings suggest that 20 years after slash-and-burn disturbance gross N mineralization process decreases to closer rates to pristine stage. On the other hand, gross nitrification did not recovery even after 40 years (Fig. 1). This result indicates that the time for gross nitrification to recover to pristine conditions is much longer, suggesting that this process is more sensitive to disturbances. As a consequence, N is retained in the soil as NH4+, a plant available form, which is less prone to leaching processes. It is important to highlight the small range of environmental conditions evaluated here, such as season of the year associated to the precipitation variability, limited spatial replication, and absence of some characteristics of soil (i.e. soil texture), which can be a source of variation and spatial limitations. In the Eastern Amazon, Sotta et al.24 did not find differences in gross soil N cycling between clay and sand soil, neither between seasons. Moreover, the environmental factors probably have stronger influence on the magnitude than the patterns of the N cycle rates. Our findings have shown similar gross nitrification patterns as other tropical chronosequence forests (Fig. 3), which gives confidence in the robustness of the observed pattern. These findings combined demonstrate the sensibility of nitrification to disturbances in different tropical forests around the world and highlights the importance of gross nitrification as being the best mechanism to evaluate the evolution and recovery of N cycling in soils of secondary succession forests.
Methods
Study area
The study was carried out at and near the Ecological Station of Cuniã, Porto Velho municipality, Rondônia, Brazil (08°06′23″ S and 63°28′59″ W). The station was established for conservation and scientific research in 2001 in one of the Brazilian states with highest deforestation rate in the Amazonian region between 1980 and 199052. The area of the station corresponds to 125,849.23 hectares of open rainforest dominated by hardwood with abundance of palms20. The soils studied were classified as Plinthosols, iron-rich and humus-poor and predominance of kaolinitic clay19. Inside the station, there is an area of 2500 ha previously defined for sampling and used in long-term monitoring. The mean annual precipitation in this region is 2500 mm, the rain season occurs from October to April, and the dry season from June to August. The mean annual temperature is around 26 °C53.
To investigate the in situ gross N transformations in intact soils of pristine and regrowth forest, one pristine forest and three regrowth forests with an age of 10, 20 and 40 years after slash-and-burn practice were chosen. The pristine forest was inside a grid of 1 km2 and was within the long-term monitoring site, as was the 40 years old regrowth forest (3–4 km from the pristine plot). The other two regrowth forests (approximately 10 and 20 years old; personal communication by local farmer) were located in the surrounding area, 10–12 km away.
In situ 15N labelling
To investigate the in situ gross N transformations in intact soil, with an intact rhizosphere, a 15N labelling using the “virtual soil core” approach18 was conducted at the beginning of the dry season in April 2013. Earlier studies on tropical forest soils24,41 found no differences in gross N rates between dry and wet season.
In the pristine forest seven plots in two straight lines, 1 km apart, were established with 10 m distance between plots. In the regrowing forests, three plots were randomly chosen with a distance of 10 m either in a straight line (40 years old) or in a triangle (10 and 20 years old), which was mainly governed by accessibility. Each plot was a pseudoreplication and, in each of them, two sets of a paired labelling spots were establish receiving a solution containing NH4+ and NO3− with one of the N species enriched with 15N at 99% (Supplementary Fig. 1a).
Each spot received eleven 1 mL injections of 15N solution in a circular area of 7 cm in diameter, homogenously distributed into the soil underneath the litter to a depth of 9 cm using a 1 mL syringe and 9 cm spinal needle18 (Supplementary Fig. 1b). The total amount added corresponded to 1.73 µg NH4+-N and 0.86 µg NO3−-N per gram dry soil. One of the paired labelling spots was sampled immediately after labelling (t0) and the second one 24 hours (t24) after labelling. Soil sampling was conducted in the inner 4 cm labeling spot. The larger labeling area provides a buffer zone around the sampling18.
The intact soil samples were immediately transported to the field laboratory, where they were gently broken by hand to remove stones, leaves and large roots by tweezers. After sieving, 50 grams of each soil sample was added to a brown plastic bottle together with 100 mL of 1 M KCl, placed on a shaker for 1 hour, and lastly filtered through MN 615 filter paper (Macherey-Nagel).
The remaining soil was dried later in the laboratory to measured physicochemical soil properties; gravimetric water content (GWC) by drying at 100 °C, the soil organic matter content (SOM) by loss-on-ignition, and the total C and N (TC and TN) was measured on an elemental analyser coupled to an Isotope Ratio Mass Spectrometer (IRMS) (20–22, Sercon Ltd., Cheshire, UK). The pH was measured in the 1 M KCl extracts with a pH meter (691, Metrohm AG, Herisau, CH). Concentrations of NH4+ and NO3− in KCl extracts were measured on flow injection analyser (FIAstar 5000, Foss Tecator AB, Brazil). The soil texture was determined in the pristine soil using a laser type granulometer (Malvern Mastersizer 2000, Malvern Instruments SA, Orsay cedex, France). The soil properties are showed in Table 1.
For analysis of 15N abundance, NO3− in extracts was measured using the automatic measuring method Sample Preparation of Inorganic N compounds Mass Spectrometry (SPINMAS)54 at UFZ Halle. The 15N abundance of NH4+ was analysed using the micro-diffusion technique55, in which NH4+ is trapped in acidified glass fibre filters and analysed using an elemental analyser (ANCA-GSL, PDZ Europa, UK) coupled to the same IRMS as above, conducted at the Stable Isotope Facility at the University of California, Davis.
Data analysis
Gross N mineralization and nitrification rates were calculated for each plot using the analytical 15N tracing model56, using data from the 15NH4+ labelling for gross mineralization and 15NO3− labeling for gross nitrification:
where N0 and Nt are soil NH4+ or NO3− content at time zero and t, respectively, t is the time in days. The a′0 and a′t are the excess 15N fractions of NH4+ or NO3− at time zero and t, respectively. All raw data used in the equation 1 to calculate gross rates are presented Supplementary Table 1. Average gross rates were calculated per forest type and are presented on soil dry weight (SDW). A one-way analysis of variance (ANOVA) with Tukey’s post-test (P < 0.05) was carried out to examine the differences between the four forest sites.
The Normality test (Shapiro-Wilk) was used to examine the normality of soil properties. As some of our data, such as GWC, TN, soil NH4+ and NO3− content were not normally distributed, the Kruskal-Wallis test with Dunn’s post-test (P < 0.05) was conducted to examine the difference between the four forest sites. Data of pH, SOM and TC showed a normal distribution and one-way analysis of variance (ANOVA) was conducted. All the analyses were conducted using GraphPad Prism (Version 5.01, GraphPad Software, Inc.).
References
Davidson, E. A. et al. Nitrogen and Phosphorus Limitation of Biomass Growth in a Tropical Secondary Forest. Ecological Applications. 14(4), 150–163 (2004).
Davidson, E. A. et al. Recuperation of Nitrogen Cycling in Amazonian Forests Following Agricultural Abandonment. Nature. 447(7147), 995–98 (2007).
Barona, E., Ramankutty, N., Hyman, G. & Coomes, O. T. The role of pasture and soybean in deforestation of the Brazilian Amazon. Environmental Research Letters. 5, 024002 (2010).
McGrath, D. A., Smith, C. K., Gholz, H. I. & Oliveira, F. A. Effects of Land-Use Change on Soil Nutrient Dynamics in Amazônia. Ecosystems. 4(7), 625–645 (2001).
Laurance, W. F. et al. The Future of the Brazilian Amazon. Ecology. 291(5503), 438–439 (2001).
Kirby, K. R. et al. The future of deforestation in the Brazilian Amazon. Futures. 38(4), 432–453 (2006).
Aguiar, A. P. D. et al. Land use change emission scenarios: anticipating a forest transition process in the Brazilian Amazon. Global Change Biology. 22, 1821–1840 (2016).
INPE (2015) Amazon Program - Monitoring the Brazilian Amazon by satellite: The PRODES, DETER, DEGRAD and TerraClass Systems. Available at www.inpe.br (accessed 01 February 2017).
Hietz, P. et al. Long-term change in the nitrogen cycle of tropical forests. Science. 334, 664–666 (2011).
Nagy, R. C., Rastetter, E. B., Neill, C. & Porder, S. Nutrient limitation in tropical secondary forests following diffferent management practices. Ecological Applications. 27(3), 734–755 (2017).
Martin, P. A., Newton, A. C. & Bullock, J. M. Carbon pools recover more quickly than plant biodiversity in tropical secondary forests. Proc. R. Soc. B. 280, 20132236 (2013).
Amazonas, N. T., Martinelli, L. A., Piccolo, M. C. & Rodrigues, R. R. Nitrogen dynamics during ecosystem development in tropical forest restoration. Forest Ecology and Management. 262(8), 1551–1557 (2011).
Kowalchuk, G. A. & Stephen, J. R. Ammonia-oxidizing bacteria: a model for molecular microbial ecology. Annual Review of Microbiology. 55, 485–529 (2001).
Schimel, J. P. & Bennett, J. Nitrogen mineralization: challenges of a changing paradigm. Ecology. 85(3), 591–602 (2004).
Booth, M. S., Stark, J. M. & Edward, R. Controls on nitrogen cycling in terrestrial ecosystems: A synthetic analysis of literatura data. Ecological Monographs. 75(2), 139–157 (2005).
Templer, P. H., Silver, W. L., Pett-Ridge, J., DeAngelis, K. M. & Firestone, M. K. Plant and microbial controls on nitrogen retention and loss in a humid tropical forest. Ecology. 89(11), 3030–3040 (2008).
Schmidt, S. K. et al. Biogeochemical Consequences of Rapid Microbial Turnover. Ecology 88(6), 1379–1385 (2007).
Rütting, T., Boeckx, P., Müller, C. & Klemedtsson, L. Assessment of the importance of dissimilatory nitrate reduction to ammonium for the terrestrial nitrogen cycle. Biogeosciences. 8, 1779–1791 (2011).
IUSS Working Group WRB World Reference Base for Soil Resources 2014, update 2015 International soil classification system for naming soils and creating legends for soil maps. World Soil Resources Reports No. 106. FAO, Rome (2015).
Pansini, S. et al. Riqueza e seletividade de palmeiras ao longo de gradientes ambientais na região do interflúvio Purus-Madeira em Porto Velho, RO. Biota Amazônica. 6(2), 93–100 (2016).
González-Pérez, J. A., González-Vila, F. J., Almendros, G. & Knicker, H. The effect of fire on soil organic matter – a review. Environmental International. 30, 855–870 (2004).
Silver, W. L., Thompson, A. W., Reich, A., Ewel, J. J. & Firestone, M. K. Nitrogen cycling in tropical plantation forests: Potential controls on nitrogen retention. Ecological Applications. 15(5), 1604–1614 (2005).
Allen, K., Corre, M. D., Tjoa, A. & Veldkamp, E. Soil nitrogen-cycling responses to conversion of lowland forests to oil palm and rubber plantations in Sumatra, Indonesia. PlosOne. 10(7), e0133325 (2015).
Sotta, E. D., Corre, M. D. & Veldkamp, E. Differing N status and N retention processes of soils under old-growth lowland forest in Eastern Amazonia, Caxiuanã, Brazil. Soil Biology and Biochemistry. 40(3), 740–50 (2008).
Burton, J., Chen, C., Xu, Z. & Ghadiri, H. Gross nitrogen transformations in adjacent native and plantation forests of subtropical Australia. Soil Biology & Biochemistry. 39, 426–433 (2007).
Aidar, M. P. M., Schmidt, S., Moss, G., Stewart, G. R. & Joly, C. A. Nitrogen use strategies of neotropical rainforest trees in threatened Atlantic Forest. Plant, Cell & Environment. 26(1996), 389–399 (2003).
Zou, X. M., Valentine, D. W., Sanford, R. L. & Binkley, D. Resin-core and buried-bag estimates of nitrogen transformations in Costa Rican lowland rainforests. Plant and Soil. 139, 275–283 (1992).
Scowcroft, P. G., Haraguchi, J. E. & Hue, N. V. Reforestation and topography affect mountain soil properties, nitrogen pools, and nitrogen transformations in Hawaii. Soil Science Society of America Journal. 68, 959–968 (2004).
Kita, S., Agus, C., Toda, H. & Haibara, K. Effects of cutting and regeneration on microbial activity of natural forest in east Kalimantan. Journal of the Japanese Forestry Society. 87, 217–224 (2005).
Zhang, J., Cai, Z. & Zhu, T. N2O production pathways in the subtropical acid forest soils in China. Environmental Research. 111, 643–649 (2011).
Lehmann, J. & Kleber, M. The contentious nature of soil organic matter. Nature. 528, 60–68 (2015).
Mooshammer, M. et al. Stoichiometric controls of nitrogen and phosphorus cycling in decomposing beech leaf litter. Ecology. 93, 770–782 (2012).
Wang, H. et al. Quality of fresh organic matter affects priming of soil organic matter and substrate utilization patterns of microbes. Scientific Reports. 5, 10102 (2015).
Lu, M. et al. Minor stimulation of soil carbon storage by nitrogen addition: A meta-analysis. Agriculture, Ecosystems and Environmental. 140, 234–244 (2011).
Steiner, C. et al. Nitrogen retention and plant uptake on a highly weathered central Amazonian Ferralsol amended with compost and charcoal. Journal of Plant Nutrition and Soil Science. 171, 893–899 (2008).
Dijkstra, F. A., Cheng, W. & Johnson, D. W. Plant biomass influences rhizosphere priming effects on soil organic matter decomposition in two differently managed soils. Soil Biology and Biochemistry. 38, 2519–2526 (2006).
Bengtson, P., Braker, J. & Grayston, S. J. Evidence of a strong coupling between root exudation, C and N availability, and stimulated SOM decomposition caused by rhizosphere priming effects. Ecology and Evolution. 2(8), 1843–1952 (2012).
Dijkstra, F. A., Carrilo, Y., Pendall, E. & Morgan, J. A. Rhizosphere priming: a nutrient perspective. Frontiers in Microbiology. 4 (216) (2013).
Ketterings, Q. M., Bigham, J. M. & Laperche, V. Changes on soil mineralogy and texture caused by slash-and-burn in Sumatra, Indonesia. Soil Sci. Soc. Am. J. 64, 1108–1117 (2000).
Markewitz, D., Davidson, E., Moutinho, P. & Nepstad, D. Nutrient loss and redistribution after forest clearing on a highly weathered soil in Amazonia. Ecological Applications. 14(4), 177–199 (2004).
Corre, M. D., Veldkamp, E., Arnold, J. & Wright, A. J. Impact of elevated N input on soil N cycling and losses in old-growth lowland and montane forests in Panama. Ecology. 91(6), 1715–1729 (2010).
Paula, F. S. et al. Land use change alters functions gene diversity, composition and abundance in Amazon forest soil microbial communities. Molecular Ecology. 23, 2988–2999 (2014).
Corre, M. F., Dechert, G. & Veldkamp, E. Soil Nitrogen Cycling following Montane Forest Conversion in Central Sulawesi, Indonesia. Soil Science Society of America. 70, (359–366 (2006).
Ueda, M. U. et al. Soil properties and gross nitrogen dynamics in old growth and secondary forest in four types of tropical forest in Thailand. Forest Ecology and Management. 398, 130–139 (2017).
Neill, C., Piccolo, M. C., Melillo, J. M., Steudler, P. A. & Cerri, C. C. Nitrogen Dynamics in Amazon Forest and Pasture Soils Measured by 15N Pool Dilution. Soil Biology and Biochemistry. 31(4), 567–572 (1999).
Huygens, D. et al. Soil Nitrogen Conservation Mechanisms in a Pristine South Chilean Nothofagus Forest Ecosystem. Soil Biology and Biochemistry. 39(10), 2448–2458 (2007).
Compton, J. E., Hooker, T. D. & Perakis, S. S. Ecosystem N distribution and δ15N during a century of forest regrowth after agricultural abandonment. Ecosystems. 10, 1197–1208 (2007).
Huygens, D. et al. Mechanisms for retention of bioavailable nitrigen in volcanic rainforest soils. Nature Geoscience. 1, 543–548 (2008).
Brown, S. & Lugo, A. E. Tropical secondary forests. Journal of Tropical Ecology. 6(1), 1–32 (1990).
Holz, M. et al. Gross nitrogen dynamics in the mycorrhizosphere of an organic forest soil. Ecosystems. 19, 284–295 (2015).
Waring, B. G., Becknell, J. M. & Powers, J. S. Nitrogen, phosphorus, and cation use efficiency in stands of regenerating tropical dry forests. Oecologia. 178, 887–897 (2015).
ICMBio - Instituto Chico Mendes de Conservação da Biodiversidade. Esec de Cuniã Available in: http://www.icmbio.gov.br/portal/biodiversidade/unidades-deconservacao/biomas-brasileiros/amazonia/unidades-de-conservacao-amazonia/1911 [accessed em August 17 2017].
Alvares, C. A. et al. Köppen’s climate classification map for Brazil. Meteorologische Zeitschrift. 22(6), 711–728 (2013).
Stange, C. F. et al. Automated and rapid online determination of 15N abundance and concentration of ammonium, nitrite, or nitrate in aqueous samples by the SPINMAS technique. Isotopes in Environmental Health Studies. 43, 227–236 (2007).
Brooks, P. D. et al. Diffusion method to prepare soil extracts for automated nitrogen-15 analysis. Soil Science Society of America Journal. 53, 1707–1711 (1989).
Kirkham, D. & Bartholomew, W. V. Equations for following nutrient transformations in soil, utilizing tracer data. Soil Sci. Soc. Am. Proc. 18, 33–34 (1954).
Acknowledgements
We thank Karina Tôsto, Silvia Rivera and Anders Primé for assistance in the field, Ângelo Manzatto, Márcio Miranda and Wanderlay Bastos for logistic helping, and Ricardo Pollery for soil texture measurement. V.F. and A.E.P. thanks CNPq, CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), FAPERJ (Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro) and STINT (The Swedish Foundation for International Cooperation in Research and Higher Education) for travel support from Brazil to Sweden for sample analyses. V.F. has a post doctoral fellowship from FAPERJ NOTA 10 program. A.E.P. is a research fellow from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) and Cientista do Estado from FAPERJ (Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro). T.R. is supported by the Strategic Research Area BECC (Biodiversity and Ecosystems services in a Changing Climate; www.becc.lu.se).
Author information
Authors and Affiliations
Contributions
This work was originally conceived by V.F., A.E.P. and T.R.; fieldwork was done by V.F., T.R. and A.E.P.; laboratory analyses were conducted by V.F. and T.R.; data analyses were performed by V.F. and T.R. All authors contributed to the writing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Figueiredo, V., Enrich-Prast, A. & Rütting, T. Evolution of nitrogen cycling in regrowing Amazonian rainforest. Sci Rep 9, 8538 (2019). https://doi.org/10.1038/s41598-019-43963-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-43963-4
This article is cited by
-
Does climate change alter the nutrient trends of Cedrela fissilis Vell. trees in the southern Brazilian Amazon?
Ecological Processes (2023)
-
Converting acidic forests to managed plantations reduces soil nitrogen loss by inhibiting autotrophic nitrification while inducing nitrate immobilization in the tropics
Biology and Fertility of Soils (2023)
-
Trajectories in nitrogen availability during forest secondary succession: illustrated by foliar δ15N
Ecological Processes (2022)
-
Effects of long-term nitrogen addition and seasonal variation on soil faunal community structure in a temperate natural secondary forest
European Journal of Forest Research (2022)
-
Conservative N cycling despite high atmospheric deposition in early successional African tropical lowland forests
Plant and Soil (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.