Abstract
Interaction between childhood trauma and genetic factors influences the pathophysiology of posttraumatic stress disorder (PTSD). This study examined the interaction effect of childhood trauma and brain-derived neurotrophic factor (BDNF) Val66Met polymorphism on PTSD symptoms and brain cortical thickness. A total of 216 participants (133 healthy volunteers and 83 PTSD patients) were recruited. T1-weighted structural magnetic resonance imaging, BDNF rs6265 genotyping through blood sampling, and clinical assessments including the childhood trauma questionnaire (CTQ) and posttraumatic stress disorder Checklist (PCL) were performed. A moderated regression analysis, two-way multivariate analysis of covariance, and correlation analysis were conducted. An interaction between the CTQ and the BDNF polymorphism significantly influenced PTSD symptom severity. In fact, people with rs6265 Val/Val genotype and higher CTQ scores showed higher PCL scores. Additionally, this interaction was significant on both left fusiform and transverse temporal gyri thickness. Furthermore, the thickness of both brain regions was significantly correlated with psychological symptoms including depression, anxiety, rumination, and cognitive emotion regulation methods; yet this was mainly observed in people with the Val/Val genotype. The interaction between childhood trauma and BDNF polymorphism significantly influences both PTSD symptoms and cortical thickness and the Val/Val genotype may increase the risk in Korean population.
Similar content being viewed by others
Introduction
Posttraumatic stress disorder (PTSD), a highly debilitating condition with severe symptoms, is influenced by both environmental and genetic factors1. Among many traumatic experiences, people who faced childhood trauma are more likely to develop PTSD symptoms2,3,4. Furthermore, childhood trauma is identified as an environmental factor which could contribute to PTSD symptoms when a genetic predisposition to childhood trauma is present, commonly referred to a gene-environment interaction5. The brain-derived neurotrophic factor (BDNF) is known to be one of the key candidate genes for PTSD6,7,8,9,10.
BDNF and its receptors play important roles in the regulation, differentiation, and maintenance of both the peripheral and central nervous systems11. The BDNF gene contains a functional single-nucleotide polymorphism (rs6265) that is associated with regulation of both transportation and secretion of BDNF in neurons12. Additionally, BDNF is renowned for its role in neuronal survival and growth-promoting actions in the central nervous system13. Specifically, the rs6265 polymorphism results in a valine (Val) to methionine (Met) substitution at codon 66 (Val66Met)14. Interestingly, the rs6265 was hypothesized to play an important role in fear learning15.
Although increasing evidence indicates that the rs6265 is involved in various psychiatric disorders including PTSD16,17,18,19, the results are inconsistent. In fact, several studies suggested that the Met allele may be related to increased PTSD susceptibility8,10,20,21,22, while the Val/Val genotype may have a protective role towards PTSD development16. However, other studies did not report any significant correlation between the rs6265 polymorphism and PTSD23,24,25,26. Further studies even suggested the Met allele to have a protective function and participants with the Val/Val genotype to show symptoms such as anxiety27, neuroticism28, obsessive-compulsive disorder29,30,31,32, and depression27,33.
Furthermore, up to date, the findings related to the interaction effect of BDNF rs6265 polymorphism and childhood traumatic experiences on brain structure are also contradictory. In fact, previous studies found that Met allele carriers to present thinner and smaller brain cortical structures. Additionally, Met carriers with a history of childhood adversity had significantly less gray matter in their subgenual anterior cingulate cortices34, smaller amygdala volumes, and less gray matter35,36 when compared to both Met carriers without any childhood adversity and Val/Val homozygotes with childhood adversity. However, other studies found the Val/Val genotype to be related to thinner and smaller brain cortical structures; maltreated Val/Val participants showed thinner rostral anterior cingulate cortices than non-maltreated Val/Val participants37, and Val/Val children with a history of traumatic experiences presented decreased hippocampal volume when compared to the Met allele children38. Some researchers claimed the reason behind such mixed to be racial differences26 and the relative rarity of the Met allele19. Only one Korean study reporting the interaction between BDNF and childhood trauma to have an effect on the severity of anxiety symptoms in healthy participants exists39. However, a study focusing on the interaction effect of BDNF and childhood trauma on cortical structures in Korean participants is yet to be conducted. Moreover, there are no studies on discovering the relationship between brain regions, which are affected by the interaction of childhood trauma and rs6265, and other psychological symptoms that are likely to comorbid with PTSD or cognitive styles that could affect the onset of PTSD in Korean participants.
The purpose of this study was to examine the interaction effect of childhood trauma and BDNF rs6265 on both PTSD symptoms and cortical thickness in Korean participants. In addition, this study was aimed to investigate the relationship of cognitive styles and psychological symptoms with cortical thickness of regions affected by the interaction of childhood trauma and rs6265. We hypothesized that the effect of childhood trauma on both PTSD symptoms and cortical thickness might differ depending on the BDNF rs6265genotype.
Results
Descriptive Statistics
The comparisons of demographic, genetic, and psychological characteristics between healthy and PTSD participants are provided in Table 1. The years of education, the childhood trauma questionnaire (CTQ), posttraumatic stress disorder checklist (PCL), posttraumatic growth inventory (PTGI), lifetime events checklist (LEC), hospital anxiety and depression scale (HADS), insomnia severity index (ISI), and ruminative response scale (RRS) scores, and some subscales of the cognitive emotion regulation questionnaire (CERQ) were significantly different between healthy participants and PTSD participants. However, since all variables, including both the CTQ and PCL scores, were normally distributed even for the total participants pool, further analyses with such a pool were executed, based on the assumption of normal distribution to represent the phenomenon in the general population.
Moderated Regression Analyses
To examine the interaction effect of childhood trauma and rs6265 on the PCL score, moderated regression analyses were executed. The CTQ score was set as the independent variable since it represents genetic factors, whereas the rs6265 genotype (Val/Val vs. Val/Met + Met/Met) was set as a moderator. Firstly, the effect of demographic factors such as gender, age, and years of education, and the effect of the experience of traumatic events other than childhood trauma (LEC) were controlled for as covariates to reveal the pure effect of CTQ and rs6265 on PTSD symptoms. In the total participants pool, both the moderation model (R² = 0.355, p < 0.001) and the moderation effect were significant, given that the R² was increased due to the interaction (ΔR² = 0.020, ΔF = 6.492, p = 0.012). Although the rs6265 coefficient was not significant (B = 12.375, p = 0.060), coefficients of both the CTQ (B = 0.480, p < 0.001) and the interaction between the CTQ and rs6265 (B = −0.326, p = 0.012) were significant. Separately, this moderation effect was significant in PTSD participants (B = −0.353, p = 0.022), but not in healthy participants (B = −0.119, p = 0.466).
Secondly, another analysis was executed by including the effect of other psychological symptoms such as anxiety, depression, insomnia, and alcohol problems for additional covariates to exclude the effect of comorbid symptoms of PTSD. In the total participants pool, both the moderation model (R² = 0.757, p < 0.001) and the moderation effect were significant, given that the R² was increased due to the interaction (ΔR² = 0.005, ΔF = 3.954, p = 0.048). Although rs6265 coefficient was not significant (B = 7.690, p = 0.063), coefficients of both the CTQ (B = 0.236, p < 0.001) and the interaction between the CTQ and rs6265 (B = −0.161, p = 0.048) were significant. Separately, however, this moderation effect was marginally significant in PTSD participants (B = −0.212, p = 0.057), but not in healthy participants (B = −0.135, p = 0.302).
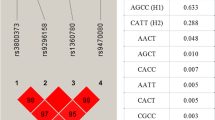
Results of both moderation analyses are described in Table 2. Although both analyses showed similar results, the second moderation model had much higher R² than that of the first moderation model (R²Model1 = 0.335; R²Model2 = 0.757) even with more covariates. Therefore, the second moderation model was accepted and displayed in Fig. 1.
As described in Table 2 and Fig. 1, the effect of the CTQ on the PCL was significant in those with the Val/Val (CC) genotype (B = 0.236, p < 0.001), whereas not in those with the Met allele (CT + TT) (B = 0.076, p = 0.157). As shown in Fig. 1, people with the Val/Val (CC) genotype showed lower CTQ score than those with the Met allele (CT + TT) when the CTQ score was below 47.500, while people with the Val/Val (CC) genotype showed higher CTQ score than those with the Met allele (CT + TT) when the CTQ score exceeded 47.500.
Two-way MANCOVA
For further analyses, the total participants pool was used to represent the phenomenon in the general population, instead creating a division between the healthy and the patient groups. Among the total participants pool, the CTQ score was partitioned into two groups, at the point in which the interaction effect of the CTQ and rs6265 was observed, i.e. the score of 47.500. Subsequently, participants were divided into four following groups: (1) Group 1: 36 participants with low CTQ (below 47.500) and rs6562 Val/Val (CC) genotype, (2) Group 2: 99 participants with low CTQ and rs6265 Met allele (CT + TT), (3)Group 3: 31 participants with high CTQ (above 47.500) and rs6265 Val/Val (CC) genotype, and (4) Group 4: 50 participants with low CTQ and rs6265 Met allele (CT + TT).
The two-way MANCOVA was executed on cortical thickness in the total participants pool to examine the effect of low vs. high CTQ, rs6265 Val/Val vs. Met allele, and the interaction effect of the CTQ and rs6265. The demographic factors such as gender, age, and years of education were controlled for as covariates since they could affect cortical thickness, while the use of medication was not considered as a covariate since it was not associated with cortical thickness in previous studies40,41. The effect of the experience of traumatic events (LEC) was also controlled for as additional covariate to see the pure effect of the childhood trauma experience on cortical thickness. Considering that several cortical areas were analyzed as dependent variables simultaneously, the Bonferroni correction was used for multiple comparisons (padj < 0.00125).
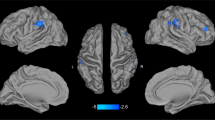
The interaction effect of the CTQ and rs6265 was significant on the thickness of the left fusiform gyrus [F(1, 208) = 10.817, p = 0.001]. Although it was significantly thinner in participants with the Val/Val (CC) genotype and with high CTQ than in those with low CTQ [F(1, 61) = 8.252, p = 0.006], it was not significantly different regardless of the CTQ level those with Met allele (CT + TT) [F(1, 143) = 1.728, p = 0.191]. In addition, left fusiform gyrus thickness was not significantly different regardless of the genotype in people with low CTQ [F(1, 129) = 1.001, p = 0.319], although it was significantly thicker in participants with high CTQ and with the Met allele (CT + TT) than in those with the Val/Val (CC) genotype [F(1, 75) = 11.746, p = 0.001].
Additionally, the interaction effect of the CTQ and rs6265 was significant on the thickness of the left transverse temporal gyrus [F(1, 208) = 10.604, p = 0.001]. In fact, the left transverse temporal gyrus was marginally thinner in participants with the Val/Val (CC) genotype and with high CTQ than in those with low CTQ [F(1, 61) = 3.873, p = 0.054]. In contrast, it was not significantly different regardless of the CTQ level those with Met allele (CT + TT) [F(1, 143) = 1.728, p = 0.191]. Furthermore, the thickness of the left fusiform gyrus was not significantly different regardless of the genotype in people with low CTQ [F(1, 129) = 3.100, p = 0.081], although it was significantly thicker in those with high CTQ and with the Met allele (CT + TT) than in those with the Val/Val (CC) genotype [F(1, 75) = 11.746, p = 0.001]. These results are presented in Fig. 2.
Correlation Analysis
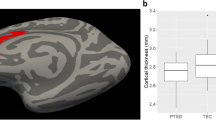
A partial correlation analysis was executed on both left fusiform and transverse temporal gyri thickness to examine their relationship with psychological symptoms by generating 1000 bootstrapped samples for multiple comparisons42. Age was controlled for as a covariate since it was significantly correlated with both the thicknesses of the left fusiform (r = −0.217, p = 0.001) and transverse temporal (r = −0.470, p < 0.001) gyri, while gender (r = −0.036, p = 0.597; r = −0.006, p = 0.928, respectively) and education years (r = −0.057, p = 0.405; r = −0.002, p = 0.978, respectively) were not. As shown in Fig. 3, both the left fusiform and left transverse temporal gyri were significantly associated with the PCL score, both the anxiety and depression scores of the HADS, the RRS score, and the score describing catastrophizing in the CERQ. In addition, left transverse temporal gyrus thickness was significantly correlated with the score describing blaming others in the CERQ and the ISI score.
Further partial correlation analyses were executed on both the thicknesses of the left fusiform and transverse temporal gyri among people with the Val/Val (CC) genotype and the Met allele (CT + TT) for comparison. Again, age was controlled for as a covariate. As shown in Table 3, the correlations between cortical thickness and psychological symptoms differed among genotypes. In fact, the left fusiform gyrus thickness showed a significant negative correlation with PCL, HADS, RRS, and CERQ only in people with the Val/Val (CC) genotype. Similarly, the left transverse temporal gyrus thickness reported significant negative correlations with PCL, HADS, RRS, and catastrophizing in CERQ and a significant positive correlation with positive reappraisal in CERQ and PTGI only in people with the Val/Val (CC) genotype. In contrast, it indicated significant negative correlation with HAD - anxiety, blaming others and catastrophizing in CERQ in people with the Met allele (CT + TT).
Discussion
The current study aimed at examining the interaction effect of childhood trauma and BDNF rs6265 polymorphism on both the PTSD symptoms and cortical thickness among Korean population. People with both the Val/Val genotype of BDNF rs6265 and a CTQ score over 47.500 showed severe PTSD symptoms. Furthermore, the interaction between this gene and childhood trauma also affected both the thickness of the left fusiform and transverse temporal gyri. Thickness, in turn, significantly correlated with symptoms severity, although the relationship differed between genotypes.
Our results revealed that the experience of childhood trauma could predict the severity of PTSD symptoms; however, this relationship was only significant in people with Val/Val (CC) genotype, but not in people with the Met allele (CT + TT) of the BDNF rs6265. In contrast, the BDNF rs6265 genotype did not affect PTSD symptom severity itself. Although previous studies revealed the effect of the rs6265 polymorphism not to be significantly related with PTSD, they did not consider the environmental effect16,19,23,25,26,43,44. Therefore, our results validated the hypothesis stating that gene-environment interaction may be the key to understanding the development of PTSD symptoms.
This moderation effect was significant both in the total participants pool (B = −0.161, p = 0.048), marginally in PTSD participants (B = −0.212, p = 0.057), but not in healthy participants (B = −0.135, p = 0.302). These group differences could be a result of dissimilarities in both the pathological characteristics and the genetic predisposition of our groups. For example, PTSD participants reported a higher CTQ mean score with a smaller standard deviation than healthy participants, which could imply the possibility of the floor effect. In addition, the frequency of Met carriers, the protective allele, was slightly higher in healthy participants (73.1%) than in PTSD participants (61.9%).
This interaction effect of childhood trauma and BDNF polymorphism was also found on the cortical thickness of both the left fusiform and left transverse temporal gyri. These regions were in fact thinner in participants with Val/Val genotype and with higher CTQ scores than in those with lower CTQ. Furthermore, both regions resulted thinner in participants with higher CTQ scores and with the Val/Val genotype than in those with the Met allele. Moreover, cortical thickness of both brain areas was negatively associated with scores of PTSD symptoms severity. These results suggest that the gene-environment interaction could influence the thickness of the left temporal regions, which are also related to PTSD symptoms.
Previous studies discovered a significant correlation between the left fusiform gyrus and PTSD45,46,47. The left fusiform gyrus is generally known to be associated with cognition, learning processes48,49,50, negative cognition of PTSD such as dysfunction of memory, dissociative symptoms51, and threat appraisal47. The current research is in line with these studies since the thickness of the left fusiform gyrus was significantly correlated with PTSD symptoms and other negative cognition (e.g., rumination, blaming others, and catastrophizing). Additionally, the left transverse temporal gyrus, also referred to as Heschl’s gyrus, was described to be linked to PTSD in several studies52,53,54,55,56. The left transverse temporal gyrus appears to be involved in assigning a spectral order to pitch information57 and language comprehension58, which could be related to the low-level perceptual deficits observed in PTSD59. Although this study did not evaluate low-level perception, our results highlight the importance of the left transverse temporal gyrus in PTSD.
Other than PTSD symptoms, this study found the relation of left fusiform and transverse gyri with diverse psychological symptoms including anxiety, depression, rumination, insomnia, and some subtypes of cognitive emotional regulation, which is in line with various other studies. The reduced fusiform gyrus thickness is known as a trait marker for vulnerability to depression60 and social anxiety61. The fusiform gyrus is also considered to be related with the retrieval of memories and rumination62. In addition, the thin transverse temporal gyrus was related with depression63, and the decreased connectivity in the transverse temporal gyrus was found in high anxiety people64, in insomnia patients65, and in depressive patients with rumination66. Although transverse temporal gyrus is known to be related with exteroceptive sensory perception, a frontoparietal network including this area could be associated with cognitive control as well67. Therefore, results of this study support the possibility that these brain regions could act as key roles regarding psychopathology including PTSD and other various symptoms.
Results from this study suggest the BDNF rs6265 Met allele assumes a protective genotype when interacting with childhood trauma; in contrast, the Val/Val genotype may increase the risk of developing PTSD symptoms and cortical thickness. However, previous studies showed mixed results on this matter, as some indicated the Met allele to be risky8,10,20,21,22, while others proposed the Val/Val genotype to be the risky one instead27,28,33. A possible reason behind such a difference may be participants’ racial differences26. For example, our participants reported higher rs6265 Met allele (66.8%) than did other studies on western participants (approximately 31–40%)68,69. This said, the allele frequency found in our study participants was similar to the one reported by other studies (63.64–71.64%) conducted on Korean participants23,70,71.
A study with Korean participants revealed an absence of association between the rs6265 and PTSD23, while others found the Met allele to act as an allele increasing the risk of developing PTSD71,72. However, when considered the interaction of rs6265 and childhood maltreatment, the Val/Val genotype was reported to be associated with a higher anxiety than the Met/Met genotype in Korean females39. Furthermore, the participants with the Val/Val genotype showed high harm avoidance traits after recent negative stressors in Korean participants73.
Another possible reason behind the mixed results related to the genotype effect may be gene-gene interaction. A few studies suggested in fact that the BDNF genotype may interact with the catechol O-methyltransferase (COMT) and the serotonin transporter gene linked promoter region (5-HTTLPR) to produce differences in resilience74 and onset age of depressive disorder75, respectively. Further studies investigating such a gene-gene interaction are needed, given that PTSD is known to be related to resilience76, depression77, and other psychological factors.
Despite significant findings, there are some limitations in this study. Firstly, childhood trauma was assessed retrospectively. Although the CTQ is known to present a stable measurement over time78, additional longitudinal studies would be helpful to determine the effect of childhood trauma with accuracy. Furthermore, this study considered the BDNF polymorphism only. Multi gene effects should be explored for better define the gene-environment interaction in future studies, including genome wide association studies. Lastly, neither the severity nor frequency of the traumatic events in PTSD patients were considered as a possible covariate in this study. Further studies considering these effects should be executed in future.
This is one of the first studies to examine the interaction effect of BDNF rs6265 and childhood trauma on both PTSD symptoms and cortical thickness in Korean population. We found that people with both the Val/Val genotype of rs6265 and high CTQ score showed severe PTSD symptoms. Furthermore, this interaction affected the thickness of both the left fusiform and transverse temporal gyri, which significantly correlated with psychological symptoms, although such relationship differed between genotypes. Finally, these results indicate the possibility that the Val/Val genotype may increase the risk of such effect in our Korean population.
Methods
Participants
A total of 255 Korean volunteers were initially included in the present study. However, 12 participants were excluded due to missing data from psychological measurements; nine additional participants were omitted from the analyses due to missing MRI and rs6265 data; and 18 participants who showed high denial scores in the childhood trauma questionnaire were also excluded, leaving a final sample of 216 volunteers. While the healthy participants were 133 (61.57%) and recruited through community advertisements, the PTSD patients were 83 (38.43%) and were recruited through notices on the bulletin board in the hospital instead. The PTSD patients were diagnosed based on the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) by a psychiatrist, and healthy participants were also assessed using the DSM-5 by a psychiatrist. The PTSD patients were mainly exposed to the following traumatic events: 57 (68.67%) to severe motor vehicle accidents, 7 (8.43%) to defect from North Korea, other 7 (8.43%) to physical or sexual violence, further 7 (8.43%) to relationship issues, 4 (4.82%) to death of a family member, and 1 (1.20%) to fire. The total participants pool was comprised of 72 (33.33%) men and 144 (66.67%) women, with a mean age of 45.67 years (SD = 13.45) and a mean for years of education of 13.16 (SD = 3.18). Specially, healthy participants consisted of 40 (30.07%) men and 93 (39.93%) women, with a mean age of 46.88 years (SD = 13.54). The mean years of education were 13.79 (SD = 2.91). PTSD patients consisted of 32 (38.55%) men and 51 (61.45%) women with a mean age of 43.88 years (SD = 13.18) and a mean for years of education of 12.16 (SD = 3.36). Each participant signed a written form of informed consent, approved by the Institutional Review Board at Inje University Ilsan Paik Hospital prior to the start of the research (IRB no. 2015-07-025), and all measurements and experiments were executed in accordance with guidelines and regulations of the board.
Psychological measures
Childhood trauma was examined using a Korean validated version of childhood trauma questionnaire (CTQ) which consists of five subscales of various childhood traumas, including emotional abuse, physical abuse, sexual abuse, emotional neglect, and physical neglect, and another scale for detecting minimization and denial79. The CTQ is comprised of 28 items and it is assessed with a 5-point Likert scale, ranging from 1 (“never true”) to 5 (“very often true”).
To examine the severity of PTSD symptoms, the posttraumatic stress disorder checklist for DSM-5 (PCL-5) was administered. The PCL is a self-report rating scale for assessing symptoms of PTSD80 and was well-validated in Korea81. It consists of 20 items and is rated using a 5-point Likert scale, ranging from 1 (“not at all”) to 5 (“extremely”). In addition to PTSD symptoms, The Korean validated version of the posttraumatic growth inventory (PTGI) was conducted to measure the degree of reported positive changes experienced after traumatic events82. It comprise four subscales, namely changes of self-perception, increase of interpersonal depth, finding new possibilities, and increase of spiritual interest83. It consists of 21 items and is rated using a 6-point Likert scale, ranging from 0 (“I did not experience this change as a result of my crisis”) to 5 (“I experienced this change to a very great degree as a result of my crisis”).
To control the exposure to traumatic events other than childhood trauma, the Korean validated version of Life Events Checklist (LEC) was used to assess the experience of potentially traumatic events84. The LEC comprised of 17 items of PTEs and the responses include experiencing, witnessing, and learning about it. This study analyzed responses of experience since other responses could be confusing to some respondents.
Other psychological symptoms that could possibly comorbid with PTSD were also assessed. The Korean validated version of the hospital anxiety and depression scale (HADS) was used to evaluate anxiety and depression symptoms. It consists of 7 items for describing anxiety and 7 items for determining depression85, and it is assessed with a 4-point Likert scale, ranging from 0 (no problems) to 3 (maximum distress). Moreover, the Korean version of the insomnia severity index (ISI) was used to evaluate the difficulty in falling and staying asleep, problems in waking up too early, the satisfaction with current sleep patterns, the interference with daily functions, the perception of impairment attributed to sleep problems, and the distress caused by the sleep problem86. It comprises seven items assessed with a 5-point Likert scale ranging from 0 to 4, with a higher score indicating greater insomnia severity. Additionally, the Alcohol Use Disorders Identification Test (AUDIT) was used to assess alcohol consumption, drinking behaviors, and alcohol-related problems before and after experiencing traumatic events. The AUDIT is a 10-item screening tool developed by the World Health Organization, and well-validated in Korea87. The AUDIT is assessed with a 5-point Likert scale ranging from 0 (“never”) to 4 (“4 or more times a week”).
Questionnaires that measures cognitive styles which could affect the onset of PTSD were assessed as well. The Korean validated version of the ruminative response scale (RRS) was conducted to measure ruminative responses. It is comprised of three subscales, specifically evaluating self-reproach, reflection, and depressive rumination88. It consists of 19 items and is rated using a 4-point Likert scale, ranging from 1 (“almost never”) to 4 (“almost always”). Furthermore, the Korean validated version of the cognitive emotion regulation questionnaire (CERQ) was applied to measure the specific cognitive emotion regulation strategies used by participants in response to stressful life events. It consists of nine subscales, evaluating putting into perspective, refocus of planning, positive refocusing, self-blame, blaming other, focus on thought rumination, acceptance, catastrophizing, and positive reappraisal89. It consists of 36 items and is assessed with a 5-point Likert scale, ranging from 1 (“almost never”) to 5 (“almost always”).
BDNF genotyping
All participants had their blood sampled to extract their DNA using a NanoDrop® ND-1000UV-Vis Spectrophotometer. Thereafter, their genomic DNA was diluted to a 5 ng/\(\mu \ell \) concentration on 96 well PCR plates. TaqMan SNP Genotyping Assays were obtained from Applied Biosystems. Following, the probes were labeled with either the FAM or the VIC dye at the 5′ end and with a minor-groove binder and a non-fluorescent quencher at the 3′ end. PCR was performed in a 5 μl of a mixture containing 2 μl of a DNA sample, 0.125 μl of each TaqMan™ SNP Genotyping Assay (Thermo Fisher Scientific, USA), 2.5 μl of the TaqMan™ Genotyping Master Mix (Thermo Fisher Scientific, USA), and 0.375 μl of distilled water. Amplification and detection were completed with a detection system (QuantStudio 12 K Flex Real-Time PCR System, Thermo Fisher Scientific, USA), using the following profile: 50 °C for 2 min, 95 °C for 10 min followed by 60 cycles of 95 °C for 15 sec, and 60 °C for 1 min. Successively to PCR amplification, allelic discrimination, an endpoint plate read, is performed at the same machines (QuantStudio 12 K Flex Real-Time PCR System). In fact, the QuantStudio 12 K Flex Software calculates the fluorescence measured during the plate read and plots Rn values based on the signals coming from each well. Subsequently, automatic or manual allele calls were performed on the analyzed plates.
Three positive and one negative control samples were present on each plate. We confirmed the clustering image with positive controls, which consisted of. Intra genomic DNA (gDNA) samples of known genotypes are used for positive control.
Sixty-seven participants had the Val/Val (CC) genotype, whereas 149 participants reported the had Met allele (CT + TT) in the rs6265.
MRI acquisition and processing
A 1.5 T scanner (Magneton Avanto, Siemens, Erlangen, Germany) with restraining foam pads to reduce head motions was used for MRI acquisition. Parameters for MRI images were same as our previous study: a 227 × 384 acquisition matrix, a 210 × 250 field-of-view, 0.9 × 0.7 × 1.2 voxel size, a total of 87,168 voxels, a TE of 3.42 ms, a TR of 1,900 ms, 1.2 mm slice thickness, and a flip angle of 15°90.
After visual inspection for artifacts, the surface-based morphometry (SBM) analysis was executed using the Computational Anatomy Toolbox (CAT12, http://www.neuro.uni-jena.de/cat/) toolbox in SPM12 (Wellcome Department of Cognitive Neurology, London, UK). The images were regularized with the International Consortium for Brain Mapping template for East Asian brains and normalized using the Diffeomorphic Anatomical Registration using Exponentiated Lie algebra (DARTEL) algorithm91. The images were segmented into gray matter, white matter, and cerebrospinal fluid92 using Jacobian-transformed tissue probability maps.
The cortical thickness was estimated with the projection-based thickness method93 and extracted using the Desikan-Killany Atlas (DK40), which contains 35 cortical areas in each hemisphere94. The cortical thickness value at each vertex was calculated as the closest distance from the gray/white boundary to the gray/CSF boundary95.
Statistical Analysis
Normality was tested using the skewness and kurtosis measures. Skewness over 2.0 and kurtosis over 7.0 are considered to present moderately non-normal distribution96. All variables in our results were within the range of normal distribution. After checking for normality, a regression analysis using the SPSS Macro PROCESS for SPSS 2.16.397 was performed to examine the interaction between childhood trauma and rs6265 on the PCL score while controlling for the effect of gender, age, and years of education as covariates. Subsequently, the CTQ score was partitioned into two, at the point in which the moderation effect was first observed. A two-way multivariate analysis of covariance (MANCOVA) was used to find the interaction effect of childhood trauma and rs6265 on cortical thickness, whereas a correlation analysis was conducted to find the relation between cortical thickness and psychological symptoms. All statistical analyses were performed with the SPSS 25 (SPSS, Inc., Chicago, IL, USA). The order and the process of statistical analyses are presented in Table 4. All measurements and all experiment protocols were approved by the Inje University Ilsan Paik Hospital Institutional Review Board (IRB no. 2015-07-025) and were executed in accordance with guidelines and regulations of the board.
Data Availability
The data that support the findings of this study are available from the corresponding author, S.-H.L., upon reasonable request.
References
Association, A. P. Diagnostic and statistical manual of mental disorders (DSM-5®). (American Psychiatric Pub, 2013).
Spertus, I. L., Yehuda, R., Wong, C. M., Halligan, S. & Seremetis, S. V. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child abuse & neglect 27, 1247–1258 (2003).
Sullivan, T. P., Fehon, D. C., Andres‐Hyman, R. C., Lipschitz, D. S. & Grilo, C. M. Differential relationships of childhood abuse and neglect subtypes to PTSD symptom clusters among adolescent inpatients. Journal of Traumatic Stress: Official Publication of The International Society for Traumatic Stress Studies 19, 229–239 (2006).
Yehuda, R., Halligan, S. L. & Grossman, R. Childhood trauma and risk for PTSD: relationship to intergenerational effects of trauma, parental PTSD, and cortisol excretion. Development and psychopathology 13, 733–753 (2001).
Uher, R. Gene–environment interactions in common mental disorders: an update and strategy for a genome-wide search. Social psychiatry and psychiatric epidemiology 49, 3–14 (2014).
Bountress, K. E. et al. The Effects of a BDNF Val66Met Polymorphism on Posttraumatic Stress Disorder: A Meta-Analysis. Neuropsychobiology 76, 136–142, https://doi.org/10.1159/000489407 (2017).
Broekman, B. F. P., Olff, M. & Boer, F. The genetic background to PTSD. Neuroscience & Biobehavioral Reviews 31, 348–362 (2007).
Dai, W. et al. Brain-derived neurotropic factor Val66Met polymorphism and posttraumatic stress disorder among survivors of the 1998 Dongting Lake Flood in China. BioMed research international 2017 (2017).
Kim, T. Y. et al. Epigenetic alterations of the BDNF gene in combat‐related post‐traumatic stress disorder. Acta Psychiatrica Scandinavica 135, 170–179 (2017).
Zhang, L. et al. PTSD risk is associated with BDNF Val66Met and BDNF overexpression. Molecular psychiatry 19, 8 (2014).
Yogeetha, B. S. et al. BDNF and TNF-α polymorphisms in memory. Molecular biology reports 40, 5483–5490 (2013).
Schindowski, K., Belarbi, K. & Buee, L. Neurotrophic factors in Alzheimer’s disease: role of axonal transport. Genes, Brain and Behavior 7, 43–56 (2008).
Binder, D. K. in Recent advances in epilepsy research 34–56 (Springer, 2004).
de Araujo, C. M. et al. Effects of the brain-derived neurotropic factor variant Val66Met on cortical structure in late childhood and early adolescence. Journal of psychiatric research 98, 51–58 (2018).
Andero, R. & Ressler, K. J. Fear extinction and BDNF: translating animal models of PTSD to the clinic. Genes, Brain and Behavior 11, 503–512 (2012).
Bruenig, D. et al. A case-control study and meta-analysis reveal BDNF Val66Met is a possible risk factor for PTSD. Neural plasticity 2016 (2016).
Gratacòs, M. et al. Brain-derived neurotrophic factor Val66Met and psychiatric disorders: meta-analysis of case-control studies confirm association to substance-related disorders, eating disorders, and schizophrenia. Biological psychiatry 61, 911–922 (2007).
Verhagen, M. et al. Meta-analysis of the BDNF Val66Met polymorphism in major depressive disorder: effects of gender and ethnicity. Molecular psychiatry 15, 260 (2010).
Wang, T. Does BDNF Val66Met Polymorphism Confer Risk for Posttraumatic Stress Disorder? Neuropsychobiology 71, 149–153, https://doi.org/10.1159/000381352 (2015).
Frielingsdorf, H. et al. Variant brain‐derived neurotrophic factor Val66Met endophenotypes: implications for posttraumatic stress disorder. Annals of the New York Academy of Sciences 1208, 150–157 (2010).
Li, R. H., Fan, M., Hu, M. S., Ran, M. S. & Fang, D. Z. Reduced severity of posttraumatic stress disorder associated with Val allele of Val66Met polymorphism at brain‐derived neurotrophic factor gene among Chinese adolescents after Wenchuan earthquake. Psychophysiology 53, 705–711 (2016).
Pivac, N. et al. The association between brain-derived neurotrophic factor Val66Met variants and psychotic symptoms in posttraumatic stress disorder. The World Journal of Biological Psychiatry 13, 306–311 (2012).
Lee, H. J. et al. No association between the brain‐derived neurotrophic factor gene Val66Met polymorphism and post‐traumatic stress disorder. Stress and Health: Journal of the International Society for the Investigation of Stress 22, 115–119 (2006).
Olff, M., Langeland, W. & Gersons, B. P. R. The psychobiology of PTSD: coping with trauma. Psychoneuroendocrinology 30, 974–982 (2005).
Valente, N. L. M. et al. Candidate-gene approach in posttraumatic stress disorder after urban violence: association analysis of the genes encoding serotonin transporter, dopamine transporter, and BDNF. Journal of Molecular Neuroscience 44, 59–67 (2011).
Zhang, H. et al. Brain derived neurotrophic factor (BDNF) gene variants and Alzheimer’s disease, affective disorders, posttraumatic stress disorder, schizophrenia, and substance dependence. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics 141, 387–393 (2006).
Ribeiro, L. et al. The brain-derived neurotrophic factor rs6265 (Val66Met) polymorphism and depression in Mexican-Americans. Neuroreport 18, 1291–1293, https://doi.org/10.1097/WNR.0b013e328273bcb0 (2007).
Terracciano, A. et al. BDNF Val66Met is associated with introversion and interacts with 5-HTTLPR to influence neuroticism. Neuropsychopharmacology 35, 1083 (2010).
Alonso, P. et al. Extensive genotyping of the BDNF and NTRK2 genes define protective haplotypes against obsessive-compulsive disorder. Biological psychiatry 63, 619–628 (2008).
Hall, D., Dhilla, A., Charalambous, A., Gogos, J. A. & Karayiorgou, M. Sequence variants of the brain-derived neurotrophic factor (BDNF) gene are strongly associated with obsessive-compulsive disorder. The American Journal of Human Genetics 73, 370–376 (2003).
Mj, R. J. T. et al. BDNF gene and obsessive compulsive disorder risk, symptom dimensions and treatment response. Asian journal of psychiatry (2017).
Timpano, K. R., Schmidt, N. B., Wheaton, M. G., Wendland, J. R. & Murphy, D. L. Consideration of the BDNF Gene in relation to two phenotypes: Hoarding and Obesity. Journal of abnormal psychology 120, 700–707, https://doi.org/10.1037/a0024159 (2011).
Krishnan, V. et al. Molecular adaptations underlying susceptibility and resistance to social defeat in brain reward regions. Cell 131, 391–404 (2007).
Gerritsen, L. et al. BDNF Val66Met genotype modulates the effect of childhood adversity on subgenual anterior cingulate cortex volume in healthy subjects. Molecular psychiatry 17, 597 (2012).
Carballedo, A. et al. Brain‐derived neurotrophic factor Val66Met polymorphism and early life adversity affect hippocampal volume. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics 162, 183–190 (2013).
Harrisberger, F. et al. BDNF Val66Met polymorphism and hippocampal volume in neuropsychiatric disorders: a systematic review and meta-analysis. Neuroscience & Biobehavioral Reviews 55, 107–118 (2015).
van Velzen, L. S. et al. Effect of childhood maltreatment and brain-derived neurotrophic factor on brain morphology. Social cognitive and affective neuroscience 11, 1841–1852 (2016).
Marusak, H. A. et al. Interactive effects of BDNF Val66Met genotype and trauma on limbic brain anatomy in childhood. European child & adolescent psychiatry 25, 509–518, https://doi.org/10.1007/s00787-015-0759-4 (2016).
Min, J. A. et al. Gender-specific effects of brain-derived neurotrophic factor Val66Met polymorphism and childhood maltreatment on anxiety. Neuropsychobiology 67, 6–13, https://doi.org/10.1159/000342384 [doi] (2013).
Dickie, E. W., Brunet, A., Akerib, V. & Armony, J. L. Anterior cingulate cortical thickness is a stable predictor of recovery from post-traumatic stress disorder. Psychological medicine 43, 645–653 (2013).
Sadeh, N. et al. SKA2 methylation is associated with decreased prefrontal cortical thickness and greater PTSD severity among trauma-exposed veterans. Molecular psychiatry 21, 357 (2016).
Westfall, P. H. On using the bootstrap for multiple comparisons. Journal of Biopharmaceutical Statistics 21, 1187–1205 (2011).
Solovieff, N. et al. Genetic association analysis of 300 genes identifies a risk haplotype in SLC18A2 for post-traumatic stress disorder in two independent samples. Neuropsychopharmacology 39, 1872 (2014).
van den Heuvel, L., Suliman, S., Malan-Müller, S., Hemmings, S. & Seedat, S. Brain-derived neurotrophic factor Val66met polymorphism and plasma levels in road traffic accident survivors. Anxiety, Stress, & Coping 29, 616–629 (2016).
Bremner, J. D. et al. Neural correlates of memories of childhood sexual abuse in women with and without posttraumatic stress disorder. American Journal of Psychiatry 156, 1787–1795 (1999).
Daniels, J. K. et al. Neural and behavioral correlates of peritraumatic dissociation in an acutely traumatized sample. The Journal of clinical psychiatry 73, 420–426, https://doi.org/10.4088/JCP.10m06642 [doi] (2012).
Harricharan, S. et al. fMRI functional connectivity of the periaqueductal gray in PTSD and its dissociative subtype. Brain and behavior 6, e00579 (2016).
Bi, T., Chen, J., Zhou, T., He, Y. & Fang, F. Function and structure of human left fusiform cortex are closely associated with perceptual learning of faces. Current Biology 24, 222–227 (2014).
McCandliss, B. D., Cohen, L. & Dehaene, S. The visual word form area: expertise for reading in the fusiform gyrus. Trends in cognitive sciences 7, 293–299 (2003).
Mion, M. et al. What the left and right anterior fusiform gyri tell us about semantic memory. Brain 133, 3256–3268 (2010).
Sui, S. G. et al. Abnormal grey matter in victims of rape with PTSD in Mainland China: a voxel-based morphometry study. Acta neuropsychiatrica 22, 118–126 (2010).
Chao, L., Weiner, M. & Neylan, T. Regional cerebral volumes in veterans with current versus remitted posttraumatic stress disorder. Psychiatry Research: Neuroimaging 213, 193–201 (2013).
Hunter, M. et al. Lateralized abnormalities in auditory M50 sensory gating and cortical thickness of the superior temporal gyrus in post-traumatic stress disorder: preliminary results. Psychiatry research 191, 138–144, https://doi.org/10.1016/j.pscychresns.2010.09.012 [doi] (2011).
Kroes, M. C., Rugg, M. D., Whalley, M. G. & Brewin, C. R. Structural brain abnormalities common to posttraumatic stress disorder and depression. Journal of psychiatry & neuroscience: JPN 36, 256–265, https://doi.org/10.1503/jpn.100077 [doi] (2011).
Wang, T. et al. Altered resting-state functional activity in posttraumatic stress disorder: A quantitative meta-analysis. Scientific reports 6, 27131 (2016).
Zhang, L. et al. Increased white matter integrity of posterior cingulate gyrus in the evolution of post-traumatic stress disorder. Acta neuropsychiatrica 24, 34–42 (2012).
Johnsrude, I. S., Penhune, V. B. & Zatorre, R. J. Functional specificity in the right human auditory cortex for perceiving pitch direction. Brain 123, 155–163 (2000).
De Bellis, M. D. et al. Superior temporal gyrus volumes in maltreated children and adolescents with PTSD. Biological psychiatry 51, 544–552 (2002).
Shang, J. et al. Alterations in low-level perceptual networks related to clinical severity in PTSD after an earthquake: a resting-state fMRI study. PLoS One 9, e96834 (2014).
Papmeyer, M. et al. Cortical thickness in individuals at high familial risk of mood disorders as they develop major depressive disorder. Biological Psychiatry 78, 58–66 (2015).
Syal, S. et al. Grey matter abnormalities in social anxiety disorder: a pilot study. Metabolic Brain Disease 27, 299–309 (2012).
Burkhouse, K. L. et al. Neural correlates of rumination in adolescents with remitted major depressive disorder and healthy controls. Cognitive, Affective, & Behavioral Neuroscience 17, 394–405 (2017).
Jaworska, N. et al. A preliminary study of the influence of age of onset and childhood trauma on cortical thickness in major depressive disorder. BioMed research international 2014 (2014).
Modi, S., Kumar, M., Kumar, P. & Khushu, S. Aberrant functional connectivity of resting state networks associated with trait anxiety. Psychiatry Research: Neuroimaging 234, 25–34 (2015).
Jespersen, K. et al. Reduced structural connectivity in Insomnia Disorder. bioRxiv, 510784 (2019).
Kokonyei, G. et al. Resting states correlates of depressive symptoms and rumination about them: are they different? European Neuropsychopharmacology 26, S343–S344 (2016).
Lindquist, K. A. & Barrett, L. F. A functional architecture of the human brain: emerging insights from the science of emotion. Trends in cognitive sciences 16, 533–540 (2012).
Egan, M. F. et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112, 257–269 (2003).
Stanne, T. M. et al. Genetic variation at the BDNF locus: evidence for association with long-term outcome after ischemic stroke. PLoS One 9, e114156 (2014).
Daily, J. W. & Park, S. Interaction of BDNF rs6265 variants and energy and protein intake in the risk for glucose intolerance and type 2 diabetes in middle-aged adults. Nutrition 33, 187–194 (2017).
Lyoo, I. K. et al. The neurobiological role of the dorsolateral prefrontal cortex in recovery from trauma: longitudinal brain imaging study among survivors of the South Korean subway disaster. Archives of General Psychiatry 68, 701–713 (2011).
Kim, S. N. et al. Impact of the BDNF Val66Met polymorphism on regional brain gray matter volumes: relevance to the stress response. Psychiatry investigation 10, 173–179 (2013).
Kim, S. J. et al. Interaction between brain-derived neurotrophic factor Val66Met polymorphism and recent negative stressor in harm avoidance. Neuropsychobiology 61, 19–26, https://doi.org/10.1159/000258639 [doi] (2010).
Kang, J. I., Kim, S. J., Song, Y. Y., Namkoong, K. & An, S. K. Genetic influence of COMT and BDNF gene polymorphisms on resilience in healthy college students. Neuropsychobiology 68, 174–180, https://doi.org/10.1159/000353257 [doi] (2013).
Lee, K. Y. et al. Genetic role of BDNF Val66Met and 5-HTTLPR polymorphisms on depressive disorder. Psychiatry investigation 11, 192–199 (2014).
Streb, M., Haller, P. & Michael, T. PTSD in paramedics: resilience and sense of coherence. Behavioural and cognitive psychotherapy 42, 452–463 (2014).
Stander, V. A., Thomsen, C. J. & Highfill-McRoy, R. M. Etiology of depression comorbidity in combat-related PTSD: a review of the literature.
Paivio, S. C. Stability of retrospective self-reports of child abuse and neglect before and after therapy for child abuse issues*. Child abuse & neglect 25, 1053–1068 (2001).
Yu, J. H., Park, J. S., Park, D. H., Ryu, S. H. & Ha, J. H. Validation of the Korean childhood trauma questionnaire: the practical use in counselling and therapeutic intervention. Korean Journal of Health Psychology 14, 563–578 (2009).
Weathers, F. W. et al. The ptsd checklist for dsm-5 (pcl-5). Scale available from the National Center for PTSD at www. ptsd. va. gov (2013).
Park, S. et al. Reliability and validity of the Korean version of the post-traumatic stress disorder checklist in public firefighters and rescue workers. Korean Journal of Biological Psychiatry 23, 29–36 (2016).
Tedeschi, R. G. & Calhoun, L. G. The Posttraumatic Growth Inventory: Measuring the positive legacy of trauma. Journal of traumatic stress 9, 455–471 (1996).
Song, S. H., Lee, H. S., Park, J. H. & Kim, K. H. Validity and reliability of the Korean version of the posttraumatic growth inventory. Korean J Health Psychol 14, 193–214 (2009).
Bae, H., Kim, D., Koh, H., Kim, Y. & Park, J. S. Psychometric properties of the life events checklist-Korean version. Psychiatry investigation 5, 163–167 (2008).
Oh, S. M., Min, K. J. & Park, D. B. A study on the standardization of the hospital anxiety and depression scale for Koreans: a comparison of normal, depressed and anxious groups. Journal of Korean Neuropsychiatric Association 38, 289–296 (1999).
Cho, Y. W., Song, M. L. & Morin, C. M. Validation of a Korean version of the insomnia severity index. Journal of clinical neurology 10, 210–215 (2014).
Lee, B., Lee, C., Lee, P., Choi, M. & Namkoong, K. Development of Korean version of alcohol use disorders identification test (AUDIT-K): Its reliability and validity. J Korean Acad Addict. Psychiatry 4, 83–92 (2000).
Kim, S. J., Kim, J. H. & Youn, S. C. Validation of the Korean-Ruminative Response Scale (K-RRS). Korean J Clin Psychol 29, 1–19 (2010).
Ahn, H. N., Lee, N. B. & Joo, H. S. Validation of the cognitive emotion regulation questionnaire in a Korean population. Korean J Couns 14, 1773–1794 (2013).
Kim, S. et al. Mismatch Negativity and Cortical Thickness in Patients With Schizophrenia and Bipolar Disorder. Schizophrenia bulletin (2018).
Ashburner, J. A fast diffeomorphic image registration algorithm. NeuroImage 38, 95–113 (2007).
Ashburner, J. & Friston, K. J. Unified segmentation. NeuroImage 26, 839–851 (2005).
Dahnke, R., Yotter, R. A. & Gaser, C. Cortical thickness and central surface estimation. NeuroImage 65, 336–348 (2013).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 31, 968–980 (2006).
Fischl, B. & Dale, A. M. Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proceedings of the National Academy of Sciences of the United States of America 97, 11050–11055, https://doi.org/10.1073/pnas.200033797 [doi] (2000).
Curran, P. J., West, S. G. & Finch, J. F. The Robustness of Test Statistics to Nonnormality and Specification Error in Confirmatory Factor Analysis. Vol. 1 (1996).
Hayes, A. F. & Rockwood, N. J. Regression-based statistical mediation and moderation analysis in clinical research: Observations, recommendations, and implementation. Behaviour research and therapy 98, 39–57 (2017).
Acknowledgements
This study was supported by a grant from the Brain Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2015M3C7A1028252), and a grant from the Korea Science and Engineering Foundation (KOSEF), funded by the Korean government (NRF-2018R1A2A2A05018505). The authors thank Sunhae Jeon, Han Na Seo, and Hee Jung Jeon for the assistance on this project.
Author information
Authors and Affiliations
Contributions
M.J.J. suggested the idea, conducted the experiment, analyzed the results, and wrote the manuscript. H.J. analyzed the MRI data. M.H.H. edited the manuscript. S-H.L. designed the study and edited the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jin, M.J., Jeon, H., Hyun, M.H. et al. Influence of childhood trauma and brain-derived neurotrophic factor Val66Met polymorphism on posttraumatic stress symptoms and cortical thickness. Sci Rep 9, 6028 (2019). https://doi.org/10.1038/s41598-019-42563-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-42563-6
This article is cited by
-
Interaction of childhood trauma with BDNF and FKBP5 gene polymorphisms in predicting burnout in general occupational groups
European Archives of Psychiatry and Clinical Neuroscience (2024)
-
Childhood maltreatment history and attention bias variability in healthy adult women: role of inflammation and the BDNF Val66Met genotype
Translational Psychiatry (2021)
-
The BDNF Val66Met polymorphism affects negative memory bias in civilian women with PTSD
Scientific Reports (2020)
-
Cellular mechanisms and molecular signaling pathways in stress-induced anxiety, depression, and blood–brain barrier inflammation and leakage
Inflammopharmacology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.