Abstract
Species delimitation in minute freshwater snails is often difficult to perform using solely shell morphology. The problem intensifies when invasive species spread within the distribution range of morphologically similar native species. In Chile, the Truncatelloidean snails are represented by the native genera Heleobia and Potamolithus plus the invasive mudsnail Potamopyrgus antipodarum, which can easily be confused. Using an integrative approach, we performed molecular phylogenetic analysis and studied reproductive and morphological features to identify superficially similar forms inhabiting the central area of the country. Truncatelloidean snails were identified in 40 of 51 localities sampled, 10 containing Potamopyrgus antipodarum, 23 Heleobia and 7 Potamolithus. Based on these results and previously published data, the known distribution of the mudsnail in Chile encompasses 6 hydrological basins, including 18 freshwater ecosystems. The finding of the mudsnails in several type localities of native species/subspecies of “Heleobia” that were not find in situ suggests species replacement or significant extinction of native fauna, a hypothesis supported by the restudy of type material that shows that endemic forms belong to the genus Potamolithus. This study shows the usefulness of integrative taxonomy not only resolving complex taxa with cryptic morphology but also measuring the extent of an ongoing invasion.
Similar content being viewed by others
Introduction
Non-native species can generate a range of effects on ecosystems invaded. One of the most serious impacts on the environment is the threat to endemic biodiversity, which may be affected through population demise, rarefaction, species displacement and even extinction1,2,3,4,5,6,7. In some cases, biological invasions may be difficult to recognize due to the introduction of cryptic species8, a problem that intensifies when invasive species colonize ecosystems located within the known distribution range of morphologically similar native species, which may lead, in turn, to unfortunate misidentifications9.
The gastropods of the superfamily Truncatelloidea include minute freshwater snails that are difficult to identify due to small size, substantial intraspecific variability and convergence of external shell features9,10,11,12. In Chile, these snails are represented by two native genera: Heleobia Stimpson, 1865 (Cochliopidae), with about 30 species, and Potamolithus Pilsbry, 1896 (Tateidae species), composed by Potamolithus australis Biese, 1944 and Potamolithus santiagensis (Biese, 1944)13,14,15,16,17,18,19,20,21,22. A third representative is the mudsnail Potamopyrgus antipodarum (Gray, 1843) (Tateidae), native to New Zealand23,24, a highly invasive species the morphology of which closely resembles snails of the two Chilean endemic genera25,26. Besides these taxa, the mudsnail can also be confused with other Tateidae as well as other species belonging to different gastropod families, including Cochliopidae, Amnicolidae, Bithyniidae, Hydrobiidae, and Lithoglyphidae27,28,29,30,31,32.
The morphological similarity of the mudsnail with native snails has led to misidentifications in invaded habitats. For instance, in Garden Lakes, USA, the native Tryonia porrecta (Mighels, 1845) was incorrectly identified as the mudsnail31 whereas in Nordic areas from Europe, some species of Bithynia Leach, 1818 are also superficially similar in morphology to this species29. The mudsnail also resembles the snail Pyrgophorus platyrachis Thompson, 1968, native to Florida and subsequently introduced in Singapore and the Middle East9,11,12. Identification errors can even lead to the description of a cryptic invasive species as a new species, making the correct identification of the invasive species, and consequently, the invasion, overlooked25,33,34. Perhaps one of the more striking examples of this occurred at the end of the 19th century when the mudsnail was described from the Thames estuary (UK) as Potamopyrgus jenkinsi (Smith, 1889), an overlooked biological invasion that was finally confirmed almost a century later23,33,35. Similarly, in Australia and Tasmania the mudsnail was also erroneously recognized as Potamopyrgus niger (Quoy and Gaimard, 1835) for a long time33. In Chile, the presence of the mudsnail was first reported in 2014 and later in 2016 but the species was introduced earlier since it was confused with native species of the genus Heleobia in 201118,25,26. Accurate identification of freshwater snails in the Southern Cone of South America becomes even more difficult considering that native species of Potamolithus have been confused with Heleobia18,22,36,37,38, for example “Heleobia” hatcheri (Pilsbry, 1911) and “Heleobia” santiagensis.
The invasive populations of the mudsnail can reach very high densities in the invaded ecosystems39,40,41,42,43. Investigations of the effect of this species on these environments have shown both positive and negative relationships with the invertebrate taxa44. For example, some studies have demonstrated the absence of significant changes in local communities with respect to biomass and biodiversity, as well as decrease in population abundances and local extinctions of some species22,41,45,46,47,48,49. However, other studies have found an increase in densities of macroinvertebrates, as well as an increase in family richness and diversity44,50,51.
Molecular analyses based on DNA sequences are a powerful tool for accurate identification of invasive species52,53, including freshwater snails25,31,34,54,55,56. These studies can be coupled to traditional morphological investigations in an integrative way. The objective of this study was to identify cryptic native and invasive snails of the superfamily Truncatelloidea distributed in central Chile, including samples collected from six type localities of native species, using molecular and morphological analysis. We also reexamined the type specimens of the taxa originally described under the genus Littoridina Souleyet, 185210,13,14,15,16,17,18 in the study area to investigate if this represents original misidentifications or possible extinctions of native fauna.
Results
Taxa identification
Of 51 localities sampled, the preliminary morphological examination identified truncatelloidean snails in 40 of them (Table S1). The shell, superficially similar among populations (Fig. 1a,d,g), precluded an objective and prompt identification of taxa in the field. A close examination of the protoconch (Fig. 1b,e,h) and operculum (Fig. 1c,f,i) allowed us to differentiate the mudsnail from Heleobia and Potamolithus snails and also to distinguish between these two endemic genera, but with greater difficulty. The protoconch showed differences in length among genera, being longer in the mudsnail. The operculum, corneous, paucispiral and with eccentric nucleus in all populations studied, showed differences among genera regarding the color and the presence/absence of a white smear (Fig. 2a). Reproductive features showed some particular differences in secondary characters, sexuality and reproductive strategies that also allowed differentiating among genera (Fig. 2b–h). In all populations examined the radula is taenioglossan (Fig. 3a–f), with seven teeth on each row, including two marginal teeth (external and internal) plus a lateral tooth placed on each side of the central (rachidian) tooth (Fig. 3a,c,e); the general formula is 3–1–3. This structure allowed identifying snails to the family level according to differences provided by the central and external marginal teeth. A summary with the main morphological differences of the samples studied are shown in Table 1.
Operculum and soft body of invasive and native Chilean truncatelloidean snails. (a–d) Potamopyrgus antipodarum. (a) Operculum (external view) of an adult specimen showing a conspicuous white smear (asterisk). (b,c) Soft body of an adult specimen (right and left side, respectively) after dissection to show shelled juvenile within brood pouches. (d) Anatomy of the anterior region of the body. (e–h) Native snails. (e,f) Females of Potamolithus sp. from Lo Carreño. (g) Female of Heleobia sp. from El Yali. (h) Male of Heleobia sp. from Los Molles. Abbreviations: e, eye; f, foot; h, head; j, juvenile; l, larvae; m, mantle; n, nuchal node; o, operculum; p, penis; s, snout, t, tentacles; v, visceral mass. Scale bar: (a) = 200 µm; (b,c) = 1 mm; (d–h) = 500 µm. (Plate of photographs: Gonzalo A. Collado)
Radular morphology of cryptic truncatelloidean freshwater snails found in central Chile. Portion of the radular ribbon (left) and central radular teeth (right). (a,b) Potamopyrgus antipodarum. (c,d) Potamolithus sp. (e,f) Heleobia sp. Note the external marginal tooth (e), flange (f), internal marginal tooth (i), lateral tooth (l) and rachidian tooth (r). (Images: Gonzalo A. Collado).
Specimens of Potamopyrgus antipodarum were found in 10 sites, Heleobia spp. in 23 and Potamolithus spp. in 7. In no locality were all three genera found co-distributed. In all the localities where Heleobia occurs, no other truncatelloidean snail was found. Potamopyrgus antipodarum and Potamolithus live in syntopy in two localities. Heleobia populations were not identified at the species level due to the variability of morphological characters, the finding of putative new species and the fact that only mudsnails were found in six type localities of endemic species ascribed to this genus (Table S1). With the exception of Potamolithus santiagensis from El Yeso Spring and Lo Carreño22 native populations of this genus could not be identified to the species level because the samples are morphologically variable, potentially representing new species. In addition, our samples of Potamolithus populations were morphologically differentiable of the holotype of Potamolithus australis, a species that besides inhabit more than 800 km south of the southernmost site of the present study13.
Taxonomic Account
Class Gastropoda Cuvier, 1797
Superfamily Truncatelloidea Gray, 1840
Family Tateidae Thiele, 1925
Genus Potamopyrgus Stimpson, 1865
Potamopyrgus antipodarum (Gray, 1843)
Diagnosis
Shell shape ovate to elongated conical (Fig. 1a), with up to six to seven whorls, smooth, suture deep. Shell colors vary from grayish and dark brown to light brown. Aperture oval, sometimes with external lip thickened. Protoconch large, 11/4 whorls (including nucleus) (Fig. 1b). Body colors vary from grayish-white or gray to grayish-black or black. Operculum bright orange-brown, ellipsoid, relatively thick, with a central muscle insertion area to the right of the nucleus and another long and narrow attachment region extending close to the inner margin (Fig. 1c); the central area encloses a conspicuous white smear (Fig. 2a). Females (Fig. 3b–d) ovoviviparous, containing embryos or shelled juveniles in the brood pouches (Fig. 2b,c). Males were not observed. Central tooth of the radula with three to five lateral cusps on each side of a larger median cusp and three pairs of basal cusps, which decrease in size distally (Fig. 3a,b). Flange well developed on the external marginal teeth, leaving free only the distal section of them.
Distribution
Eighteen localities in Central Chile, considering the 10 sites reported in the present study (Table S1) plus eight documented previously22,25,26. Región de Coquimbo: Huentelauquén (in the northern sector of the town), La Brunina, Consuelo Stream, Illapel River, Choapa River, Camisas Stream, Limahuida Stream; Salamanca (Chalinga River), Cunlagua (Chalinga River), Zapallar (Chalinga River), Canal Zapallar; Región de Valparaíso: Canal La Laja, Romeral Stream, Chincolco; Región Metropolitana: Dehesa Stream, Parque O’Higgins Spring, El Yeso Spring; Región de O’Higgins: Canal Lo Carreño. In El Yeso Spring and Lo Carreño mudsnails coexist with Potamolithus santiagensis22.
Genus Potamolithus Pilsbry, 1896.
Diagnosis
Shell shape ovate to elongated conical (Fig. 1d), relatively thick, with up to five to six whorls, with very thin axial ribs, suture deep. Shell colors vary from light to dark brown. Aperture oval-rounded, outer lip slightly thick. Protoconch is 11/8 whorls (Fig. 1e). Body colors vary from white to grayish-white or grayish-black. Operculum oval (Fig. 1f), light brown, translucent, thin, without white smear. Females (Fig. 2e,f) of several populations with nuchal node (Fig. 2f), without brood pouches (oviparous). Males having simple penis (without ornamentations), grayish-white. Radula of Potamolithus (Fig. 3c,d), including external marginal teeth, as in mudsnail.
Distribution
Potamolithus snails were collected in seven localities in the present study (Table S1). Although we could not find topotype specimens of Heleobia choapaensis (Biese, 1944), Heleobia choapaensis albolabris (Biese, 1944), Heleobia bruninensis (Biese, 1944), Heleobia compacta (Biese, 1944) and Heleobia choapaensis minor (Biese, 1944) in ecosystems from the Choapa River basin, where these forms were originally described under the genus Littoridina13,14, the protoconcha of respective type specimens housed at the MNNHCL suggests that they belong to Potamolithus. The same was revealed for Heleobia santiagensis (Biese, 1944) from Dehesa Stream in the Maipo River basin. In Chile, the distribution records of the genus encompass from the Región de Coquimbo to the Región de Los Lagos according to our results and previously published data13,14,22 (Table S1). Potamolithus santiagensis occurs in Dehesa Stream, El Yeso Spring, Lo Carreño and El Colorado according to the present study, Biese13,14 and Collado et al.22. Potamolithus australis inhabits Puerto Chico in Llanquihue Lake, its type locality13, but this need to be confirmed57.
Family Cochliopidae Tryon, 1866
Genus Heleobia Stimpson, 1865.
Diagnosis
Shell shape ovate to elongated conical (Fig. 1g), thin, with up to six to seven whorls, with very thin axial ribs, suture deep. Shell colors vary from light brown-translucent to grayish. Aperture oval to ellipsoid, outer lip thin. Protoconch ca. 1 whorl (Fig. 1h). Body colors vary from light brownish-yellow to gray, grayish-white or grayish-black. Operculum oval to ellipsoid (Fig. 1i), thin, translucent, with a faint light brown central area, without white smear. Females (Fig. 2g) without brood pouches (oviparous), nuchal node absent. Males having penis with apocrine glands or lobes (Fig. 2h). Central tooth of the radula with four to seven lateral cusps on each side of a larger median cusp and one pair of basal cusps (Fig. 3e,f). External marginal teeth large, thin and free almost from the base (with flange poorly developed).
Distribution
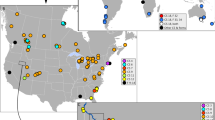
In Chile the known distribution of the genus Heleobia encompass from the Región de Arica y Parinacota to the Strait of Magellan13,14,15,16,17,18,19,20,21,58,59. In the present study, snails were collected in 23 localities (Table S1). No case of syntopy was detected with the other taxa collected in the localities studied. The known distribution records of the truncatelloidean taxa in central Chile22,25,26, are shown in Fig. 4.
Known distribution records of the truncatelloidean taxa in central Chile according to the present study and other sources22,25,26. The red circles correspond to records of the invasive mudsnail Potamopyrgus antipodarum. Records of the native populations of the genera Heleobia and Potamolithus are indicated by green squares and blue pentagon, respectively. The map was created using ArcGIS software (www.arcgis.com/index.html). (Map: G.A. Collado).
Molecular analysis
The Bayesian analysis using the COI gene recovered our original sequences into two main strongly supported clades, Cochliopidae (BPP = 1.00), including populations assigned to the genus Heleobia, and Tateidae (BPP = 1.00), composed by species/populations assigned to the genera Potamolithus (BPP = 0.98) and Potamopyrgus (BPP = 1.00), the latter represented by Potamopyrgus antipodarum (BPP = 0.99) (Fig. 5). Using our original COI sequences, divergence levels (p-distance) among groups were 15.1% between P. antipodarum and Potamolithus, 19.9% between the former and Heleobia and 17.8% between the latter and Potamolithus. The average p-distance within groups were 0.0% in P. antipodarum, 0.6% in Potamolithus and 2.5% in Heleobia.
The Bayesian tree of Chilean truncatelloidean snails. Original sequences of specimens used in the present study are highlighted in bold. Posterior probabilities (>0.94) values are indicated at the nodes. Numbers after taxa names indicate GenBank access numbers20,21,22,25,32,38,87,101,102,103,104,105,106 (Table S2).
Discussion
In the present study, molecular, morphological and microstructural data were used to evaluate the occurrence of cryptic populations of native and non-native freshwater snails of the superfamily Truncatelloidea in central Chile. The data obtained show that the mudsnail Potamopyrgus antipodarum is widespread in the sampling area. This is a significant finding regarding conservation since the known distribution range of this species encompasses type localities of five native species or subspecies of Heleobia described from the Choapa River basin13,14, which are: Heleobia choapaensis (Choapa River), Heleobia choapaensis albolabris (Consuelo Stream), Heleobia bruninensis (La Brunina), Heleobia compacta and Heleobia choapaensis minor (Zapallar). Regarding these localities, it is important to emphasize two aspects, one is that considering the types specimens reexamined, Biese (1944) probably misidentified Potamolithus specimens and named the species under the genus Littoridina (currently assigned to Heleobia), and second, we showed that the mudsnail invaded these localities and apparently now there would be no native species inhabiting them. Similarly, in Dehesa Stream, type locality of Heleobia santiagensis13,14,18,22, only mudsnails have been found in different years of sample collections (2011, 2014, 2015, 2017)18,22,25, including the present study. The finding that the Biese’s “Heleobia”13,14 above-mentioned are Potamolithus raises the possibility that the mudsnail has replaced or extinguished native snails in several type localities. The occurrence of Physella acuta (Draparnaud, 1805) in the Choapa River basin34 and Dehesa Stream (unpublished data), another non-native freshwater snail in Chile34, could have contributed to the decline (or extinction) of the native snail populations. In El Yeso Spring and Lo Carreño, where Physella acuta has not been found, mudsnails are syntopic with Potamolithus, and although not evaluated, much more abundant so it is likely competition in some niche dimensions between them. Species displacement, or even extinction of native fauna in central Chile is plausible considering that after the introduction of the mudsnail to the Snake River drainage (U.S.), five species of native mollusks were officially listed as “threatened” or “endangered”, in part due to the proliferation of the mudsnail41. Besides, this species has also been associated with decline in settlement of native invertebrates41,46,49.
Due to its high invasive power, the mudsnail has been considered as one of the most widespread invasive aquatic invertebrates in the world60,61. In line with this status, ongoing research on this species have shown a rapid expansion in central Chile from 201118,25, which can lead to serious consequences for the natural heritage of the country. It is clear that more sampling and molecular work is required in the future, especially outside of our study area, to better determine the prevalence, distribution and taxonomy of native taxa. Moreover, the present study also allowed extending the distribution of the genus Potamolithus hundreds of kilometers northward. Similarly, although mudsnail occurs in typical environments from which “Heleobia” species have been previously recorded in Chile13,14,18, several localities of occurrence were added here. This is also significant for conservation purposes since several species of this genus have been classified as vulnerable or critically endangered in Chile62,63,64,65. On the other hand, it is important to note that the mudsnail is an intermediate host of digenetic trematodes66,67,68, being able to transport parasites to non-native ecosystem68 whereby they could potentially affect native final vertebrate hosts, a potential problem not studied in Chile. Non-native disease vector, in addition to direct competition and displacement of endemic fauna could be one mechanism by which the invasive species places these taxa at risk. However, the mudsnail tend to finely partition the habitat regarding vegetation zones and depth66,67,68,69 so competition with endemic invertebrate could be, in part, diminished.
Different traits may explain the invasive success of the mudsnail70. The species is a generalist, can tolerate a wide range of environmental conditions39,71,72 and may achieve an astonishingly high reproductive rate. The latter is favored by the fact that invasive populations are composed almost exclusively by parthenogenetic females that can brood and release a lot of snails several times a year23,41,73,74. It has been estimated that under ideal conditions, a single female can produce 3.125 × 108 offspring in six broods per year beginning with 50 offspring produced per female41. Besides, since a single female may establish a new invasive population71,75, it is not surprising that the species has spread quickly in Chile because until now only females have been found in the country25,26, present study. This may be also favored by a high dispersal capacity of the species, being able to use natural and non-natural vectors27,40,71,76,77,78. However, the mechanism of introduction and spreading of the species in Chile remains unknown.
The early detection of invasive species is fundamental to develop measures to mitigate its invasion and subsequent establishment in new ecosystems79. The method used here to evaluate the presence of the mudsnail and morphologically similar cryptic truncatelloidean taxa may be applied in other geographical areas since members of this superfamily are widespread around the world10,80. This would constitute the first step towards taking appropriate control measures to treat a new invasion or at best achieve timely eradication.
Methods
Specimen collection
We prospected 51 freshwater ecosystems in Central Chile between 2015 and 2017 (Table S1), including brackish waters, swamps, reservoirs, rivers, streams, springs, ponds and irrigation canals. Snail specimens were sampled using a manual sieve (1 mm mesh) and preserved in absolute ethanol. A few specimens with uncertain taxonomic status sampled previously18,22, in press were further studied to assign the species to genera. The first author was authorized to collect freshwater snails in Chile (Subsecretaría de Pesca y Acuicultura, Ministerio de Economía, Fomento y Turismo, República de Chile, Resolution No. 3285).
Morphological analyses
The sex was determined according to the presence/absence of a penis. Genus Heleobia is dioecious, males develop a conspicuous penis on the right side of the head, ornamented with apocrine glands or accessory lobes10,18,19,20,21,81,82,83. In the mudsnail the penis is simple (i.e., devoid of apocrine glands or lobes23,32,71,84), colorless or whitish, mostly with an elongate spot of black pigment. However, non-native mudsnail populations are composed typically of parthenogenetic females, with males being rare or absent23,43,85,86. Genus Potamolithus is dioecious, the penis is simple, variable in pigmentation among taxa87,88,89,90,91,92; the females of some species develop a nuchal node88,89,90. In contrast to Heleobia and Potamolithus, whose species are oviparous18,19,82,93,94, the mudsnail is ovoviviparous, with females brooding small juvenile in the pallial oviduct25,26,32,33,41. To determine the presence of embryos or juveniles in the brood pouches, the oviduct wall was dissected and examined using a stereomicroscope. To observe the radula, the buccal mass was put in a dilute sodium hypochlorite solution for 3–10 minutes to isolate the structure and remove the organic material to subsequently be mounted on specimen stubs and observed using scanning electron microscopy (SEM) (Hitachi SU3500; Jeol JSM–6610LV). Similarly, the operculum was isolated from the foot and treated in the same way as the radula. The protoconch of specimens collected was also immersed in a sodium hypochlorite solution and observed using SEM. The shell of the type specimens of Truncatelloidean taxa described from the studied area housed at the MNHNCL13,14 were examined using stereomicroscope; the protoconch was observed under light microscope. The observations were compared with data from the literature10,23,32,89,92,95,96.
Molecular analysis
DNA extraction, amplification and sequencing of partial DNA sequences of the mitochondrial gene cytochrome c oxidase subunit I (COI) ranging from 511 to 638 bp were performed following Collado25. The sequences were edited and aligned in BioEdit97 and then analyzed together with homologous sequences of related taxa accessed through GenBank. The COI matrix was used to generate a Bayesian tree with the MrBayes v. 3.1.2 program98 based on the HKY + I + G model of sequence evolution identified as the best-fitting substitution model in jModelTest99 using the Akaike Information Criterion. The analysis was run for 5,000,000 generations, sampling every 1,000 generations, and setting a burn-in period of 20%. The node support was estimated as Bayesian posterior probabilities (BPP). The species Ascorhis tasmanica (Martens, 1858) was used to root the tree. Average p-distances among sequences were obtained in Mega 7100.
Data Availability
The sequences obtained in the present study were deposited in GenBank (accession numbers MH729593-MH729628) (Table S2). Voucher specimens were deposited in the Museo Nacional de Historia Natural (MNHNCL), Santiago, Chile, Museo de Zoología de la Universidad de Concepción (MZUC–UCC), Concepción, Chile, and Museo de Ciencias Naturales y Arqueología “Profesor Pedro Ramírez Fuentes”, Chillán, Chile (Table S3).
References
Brenchley, G. A. & Carlton, J. T. Competitive displacement of native mud snails by introduced periwinkles in the New England intertidal zone. Biol. Bull. 165, 543–558 (1983).
Byers, J. E. Competition between two estuarine snails: implications for invasions of exotic species. Ecology 81, 1225–1239 (2000).
Mack, R. N. et al. Biotic invasions: causes, epidemiology, global consequences, and control. Ecol. Appl. 10, 689–710 (2000).
Gurevitch, J. & Padilla, D. K. Are Invasive Species a Major Cause of Extinctions? Trends. Ecol. Evol. 19, 470–474 (2004).
Sagoff, M. Do Non-native Species Threaten the Natural Environment? J. Agric. Environ. Ethics. 18, 215–236 (2005).
White, E. M., Wilson, J. C. & Clarke, A. R. Biotic indirect effects: a neglected concept in invasion biology. Divers. Distrib. 12, 443–455 (2006).
Mouthon, J. & Daufresne, M. Long-term changes in mollusc communities of the Ognon river (France) over a 30-year period. Fundam. Appl. Limnol. 178, 67–79 (2010).
Morais, P. & Reichard, M. Cryptic invasions: A review. Sci. Total Environ. 613–614, 1438–1448 (2018).
Ng, T. H., Liew, J. H., Song, J. Z. E. & Yeo, D. C. J. First record of the cryptic invader Pyrgophorus platyrachis Thompson, 1968 (Gastropoda: Truncatelloidea: Cochliopidae) outside the Americas. Bioinvasions Rec. 5, 75–80 (2016).
Hershler, R. & Thompson, F. G. A review of the aquatic gastropod subfamily Cochliopinae (Prosobranchia: Hydrobiidae). Malacol. Rev. 5(Suppl), 1–140 (1992).
Mienis, H. K., Rittner, O. & Vaisman, S. Another riddle from Israel: How can we explain the presence of a Pyrgophorus species in the Tanninim River Basin? Ellipsaria 13, 17–18 (2011).
Nasarat, H., Amr, Z. & Neubert, E. Two invasive freshwater snails new to Jordan (Mollusca: Gastropoda). Zool. Middle East. 60, 46–49 (2014).
Biese, W. A. Revisión de los moluscos terrestres de agua dulce provistos de concha de Chile. Parte I. Familia Amnicolidae. Bol. Mus. Hist. Nat. Chile. 22, 169–190 (1944).
Biese, W. A. Revisión de los moluscos terrestres y de agua dulce provistos de concha de Chile. Parte II, Familia Amnicolidae (continuación). Bol. Mus. Hist. Nat. Chile. 23, 63–77 (1947).
Stuardo, J. Contribución a un catálogo de los moluscos chilenos de agua dulce. Gayana 1, 7–32 (1961).
Valdovinos, C. Biodiversidad de moluscos chilenos: Base de datos taxonómica y distribucional. Gayana 63, 111–164 (1999).
Valdovinos, C. Estado de conocimiento de los gastrópodos dulceacuícolas de Chile. Gayana 70, 88–95 (2006).
Collado, G. A., Méndez, M. A., Letelier, S., Veliz, D. & Sabando, M. C Morfología peniana y taxonomía del género Heleobia Stimpson, 1865 en Chile junto a una revisión de los ejemplares tipo del Museo Nacional de Historia Natural de Chile. Amici Molluscarum (Número especial), 49–58 (2011).
Collado, G. A. A new freshwater snail (Caenogastropoda: Cochliopidae) from the Atacama Desert, northern Chile. Zootaxa 3925, 445–449 (2015).
Collado, G. A., Valladares, M. A. & Méndez, M. A. A new species of Heleobia (Caenogastropoda: Cochliopidae) from the Chilean Altiplano. Zootaxa 4137, 277–280 (2016a).
Collado, G. A., Valladares, M. A. & Méndez, M. A. Unravelling cryptic species of freshwater snails (Caenogastropoda, Truncatelloidea) in the Loa River basin, Atacama Desert. Syst. Biodivers. 14, 417–429 (2016b).
Collado, G. A. et al. Systematic evaluation of cryptic freshwater snails from central Chile, including the enigmatic Littoridina santiagensis (Gastropoda, Truncatelloidea). Eur. J. Taxon. 524, 1–15 (2019).
Winterbourn, M. J. The New Zealand species of Potamopyrgus (Gastropoda: Hydrobiidae). Malacologia 10, 283–321 (1970).
Winterbourn, M. J. Morphological variation of Potamopyrgus jenkinsi (Smith) from England and a comparison with the New Zealand species, Potamopyrgus antipodarum (Gray). P. Malac. Soc. London. 40, 133–145 (1972).
Collado, G. A. Out of New Zealand: molecular identification of the highly invasive freshwater mollusk Potamopyrgus antipodarum (Gray, 1843) in South America. Zool. Stud. 53, 70 (2014).
Collado, G. A. Filling the gap: New records of the invasive New Zealand mudsnail Potamopyrgus antipodarum in Central Chile. J. Zool. St. 3, 13–19 (2016).
Zaranko, D. T., Farara, D. G. & Thompson, F. G. Another exotic mollusc in the Laurentian Great Lakes: the New Zealand native Potamopyrgus antipodarum (Gray 1843) Gastropoda, Hydrobiidae. Can. J. Fish. Aquat. Sci. 54, 809–814 (1997).
Levri, E. P., Kelly, A. A. & Love, E. The invasive New Zealand mud snail (Potamopyrgus antipodarum) in lake Erie. J. Great Lakes Res. 33, 1–6 (2007).
Jensen, K. R. NOBANIS – Invasive alien species fact sheet – Potamopyrgus antipodarum – From: Identification key to marine invasive species in Nordic waters – NOBANIS, http://www.nobanis.org Accessed 27 May 2017 (2010).
Kistner, E. J. & Dybdahl, M. F. Parallel variation among populations in the shell morphology between sympatric native and invasive aquatic snails. Biol. Invasions. 16, 2615–2626 (2014).
Hershler, R. et al. New discoveries of introduced and cryptogenic fresh and brackish water gastropods (Caenogastropoda: Cochliopidae) in the western United States. Aquat. Invasions 10, 147–156 (2015).
Haase, M. The radiation of hydrobiid gastropods in New Zealand: a revision including the description of new species based on morphology and mtDNA sequence information. Syst. Biodivers. 6, 99–159 (2008).
Ponder, W. F. Potamopyrgus antipodarum, a molluscan colonizer of Europe and Australia. J. Moll. Stud. 54, 271–286 (1988).
Collado, G. A. Unraveling cryptic invasion of a freshwater snail in Chile based on molecular and morphological data. Biodivers. Conserv. 26, 567–578 (2017).
Städler, T., Frye, M., Neiman, M. & Lively, C. M. Mitochondrial haplotypes and the New Zealand origin of clonal European Potamopyrgus, an invasive aquatic snail. Mol. Ecol. 14, 2465–2473 (2005).
Martín, P. R. Evidence for parthenogenesis and natural imposex in the Patagonian freshwater snail Heleobia hatcheri (Gastropoda: Hydrobiidae). J. Moll. Stud. 68, 291–295 (2002).
Collado, G. A. & Méndez M. A. Estrategias reproductivas y tipos de desarrollo en especies endémicas del género Heleobia Stimpson, 1865 (Caenogastropoda: Cochliopidae) de Chile. Amici Molluscarum (Número especial), 67–71 (2011).
Koch, E., Martin, S. M. & Ciocco, N. F. A molecular contribution to the controversial taxonomical status of some freshwater snails (Caenogastropoda: Rissooidea, Cochliopidae) from the Central Andes desert to Patagonia. Iheringia Ser. Zool. 105, 69–75 (2015).
Dorgelo, J. Density fluctuations in populations (1982–1986) and biological observations of Potamopyrgus jenkinsi in two trophically differing lakes. Hydrobiol. Bull. (Amsterdam). 21, 95–110 (1987).
Richards, D. C., Cazier, L. D. & Lester, G. T. Spatial distribution of three snail species, including the invader Potamopyrgus antipodarum, in a freshwater spring. Western North Am. Nat. 61, 375–380 (2001).
Richards, D. C. The New Zealand mudsnail invades the Western United States. Aquat. Nuis Species. 4, 42–44 (2002).
Hall, R. O., Dybdahl, M. F. & Vanderloop, M. C. Extremely high secondary production of introduced snails in rivers. Ecol. Appl. 16, 1121–1131 (2006).
Kerans, B. L., Dybdahl, M. F., Gangloff, M. M. & Jannot, J. E. Potamopyrgus antipodarum: distribution, density, and effects on native macroinvertebrate assemblages in the Greater Yellowstone ecosystem. J. N. Am. Benthol. 24, 123–138 (2005).
Rakauskas, V., Šidagytė, E., Butkus, R. & Garbaras, A. Effect of the invasive New Zealand mud snail (Potamopyrgus antipodarum) on the littoral macroinvertebrate community in a temperate mesotrophic lake. Mar. Freshwater. Res. 69, 155–166 (2017).
Cada, C. Interactions between the invasive New Zealand mudsnail, Potamopyrgus antipodarum, mayflies, and fish predators. M.Sc. Thesis, Montana State University, Bozeman, M. T., USA (2004).
Richards, D. Competition between the threatened Bliss Rapids snail, Taylorconcha serpenticola (Hershler et al.), and the invasive aquatic snail, Potamopyrgus antipodarum (Gray). Ph.D. Thesis, Montana State University, Bozeman, M. T., USA (2004).
Kerans, B. L., Cada, C. A. & Zickovich, J. Asymmetrical behavioural interactions between the New Zealand mud snail, Potamopyrgus antipodarum, and scraping, collector-gathering and collector-filtering macroinvertebrates. J. Freshwater. Ecol. 25, 657–666 (2010).
Riley, L. A., Dybdahl, M. F. & Hall, J. R. O. Invasive species impact: asymmetric interactions between invasive and endemic freshwater snails. J. N. Am. Benthol. 27, 509–520 (2008).
Schmidlin, S., Schmera, D. & Baur, B. Alien molluscs affect the composition and diversity of native macroinvertebrates in a sandy flat of Lake Neuchàtel, Switzerland. Hydrobiologia 679, 233–249 (2012).
Schreiber, E. S. G., Lake, P. S. & Quinn, G. P. Facilitation of native stream fauna by an invading species? Experimental investigation of the interaction of the snail, Potamopyrgus antipodarum (Hydrobiidae) with native benthic fauna. Biol. Invasions. 4, 317–325 (2002).
Moore, J. W., Herbst, D. B., Heady, W. N. & Carlson, S. M. Stream community and ecosystem responses to the boom and bust of an invading snail. Biol. Invasions. 14, 2435–244 (2012).
Armstrong, K. F. & Ball, S. L. DNA barcodes for biosecurity: invasive species identification. Philos. T. Roy. Soc. B. 360, 1813–1823 (2005).
Darling, J. A. & Michael, J. B. DNA-based methods for monitoring invasive species: a review and prospectus. Biol. Invasions. 9, 751–765 (2007).
Letelier, S. et al. The highly invasive freshwater apple snail Pomacea canaliculata (Gastropoda: Ampullariidae) in Northern Chile: morphological and molecular confirmation. J. Zool. Stud. 3, 119–128 (2016).
Perez, B. J., Segrest, A. H., Campos, S. R., Minton, R. L. & Burks, R. L. First record of japanese mystery snail Cipangopaludina japonica (von Martens, 1861) in Texas. Check List. 12, 1973 (2016).
David, A. A., Zhou, H., Lewis, A., Yhann, A. & Verra, S. DNA barcoding of the banded mystery snail, Viviparus georgianus in the adirondacks with quantification of parasitic infection in the species. Am. Malacol. Bull. 35, 175–180 (2017).
López Armengol M. F. Estudio sistemático y bioecológico del género Potamolithus (Hydrobiidae) utilizando técnicas de taxonomía numérica. Tesis Doctoral N° 455, Facultad Ciencias Naturales y Museo, Universidad Nacional de La Plata, La Plata, Argentina (1985).
Preston, H. B. List of the Mollusca, with descriptions of six new species. J. Conchol. 14, 347–352 (1915).
Collado, G. A., Valladares, M. A. & Méndez, M. A. Hidden diversity in spring snails from the Andean Altiplano, the second highest plateau on Earth, and the Atacama Desert, the driest place in the world. Zool. Stud. 52, 50 (2013).
Son, M. O. Rapid expansion of the New Zealand mud snail Potamopyrgus antipodarum (Gray, 1843) in the Azov-Black Sea Region. Aquat. Invasions. 3, 335–340 (2008).
Butkus, R., Šidagytė, E. & Arbačiauskas, K. Two morphotypes of the New Zealand mud snail Potamopyrgus antipodarum (J. E. Gray, 1843) (Mollusca: Hydrobiidae) invade Lithuanian Lakes. Aquat. Invasions. 7, 211–218 (2012).
Collado, G. A. Heleobia atacamensis (Philippi, 1860). In: 10° Proceso de Clasificación de Especies. Ministerio del Medio Ambiente, Chile. Available from, http://www.mma.gob.cl/clasificacionespecies/listado_especies_10o_Pac.htm (2013).
Collado, G. A. Heleobia chimbaensis (Biese, 1944). In: 10° Proceso de Clasificación de Especies. Ministerio del Medio Ambiente, Chile. Available from, http://www.mma.gob.cl/clasificacionespecies/listado_especies_10o_Pac.htm (2013).
Collado, G. A. Heleobia ascotanensis (Courty, 1907). In: 11° Proceso de Clasificación de Especies. Ministerio del Medio Ambiente, Chile. Available from, http://www.mma.gob.cl/clasificacionespecies/listado_especies_11o.htm (2014).
Collado, G. A. Heleobia transitoria (Biese, 1947). In: 11° Proceso de Clasificación de Especies. Ministerio del Medio Ambiente, Chile. Available from, http://www.mma.gob.cl/clasificacionespecies/listado_especies_11o.htm (2014).
Jokela, J. & Lively, C. M. Spatial variation in infection by digenetic trematodes in a population of freshwater snails (Potamopyrgus antipodarum). Oecologia 103, 509–517 (1995).
McKone, M. J. et al. Fine-scale association between parasites and sex in Potamopyrgus antipodarum within a New Zealand lake. New Zeal. J. Ecol. 40, 330–333 (2016).
Gérard, C. et al. A native-range source for a persistent trematode parasite of the exotic New Zealand mudsnail (Potamopyrgus antipodarum) in France. Hydrobiologia 785, 115–126 (2017).
Fox, J. A., Dybdahl, M. F., Jokela, J. & Lively, C. M. Genetic structure of coexisting sexual and clonal subpopulations in a freshwater snail (Potamopyrgus antipodarum). Evolution 50, 1541–1548 (1996).
Alonso, A. & Castro-Diez, P. What explains the invading success of the aquatic mud snail Potamopyrgus antipodarum (Hydrobiidae, Mollusca)? Hydrobiologia 614, 107–116 (2008).
Proctor, T. et al. National management and control plan for the New Zealand mudsnail (Potamopyrgus antipodarum). US Fish and Wildlife Service Report, http://www.anstaskforce.gov/Documents/NZMS_MgmtControl_Final.pdf, Accessed 27 May 2017 (2007).
Haynes, A. & Taylor, B. J. R. Food finding and food preference in Potamopyrgus jenkinsi (E. A. Smith) (Gastropoda: Prosobranchia). Arch. Hydrobiol. 100, 479–491 (1984).
Lassen, H. H. Reproductive effort in Danish mudsnails (Hydrobiidae). Oecologia 40, 365–369 (1979).
Gangloff, M. M. The New Zealand mud snail in western North America. Aquat. Nuis Species. 2, 25–30 (1998).
Cheng, Y. W. & LeClair, L. L. A quantitative evaluation of the effect of freezing temperatures on the survival of New Zealand mudsnails (Potamopyrgus antipodarum Gray, 1843), in Olympia Washington’s Capitol Lake. Aquat. Invasions. 6, 47–54 (2011).
Bowler, P. The rapid spread of the freshwater Hydrobiid snail Potamopyrgus antipodarum (Gray) in the Middle Snake River, Southern Idaho. Proc. Desert Fishes Council. 21, 173–182 (1991).
Vareille-Morel, C. Les mouvements journaliers du mollusque prosobranche Potamopyrgus jenkinsi Smith, etude sur le terrain et en laboratoire. Haliotis 13, 31–34 (1983).
Ribi, G. Within-lake dispersal of the prosobranch snails, Viviparus ater and Potamopyrgus jenkinsi. Oecologia. 69, 60–63 (1986).
Pluess, T. et al. Which factors affect the success or failure of eradication campaigns against alien species? PLoS ONE 7, e48157 (2012).
Kabat, A. R. & Hershler, R. The prosobranch family Hydrobiidae (Gastropoda: Rissooidea): review of classification and supraspecific taxa. Smithson. Contrib. Zool. 547, 1–94 (1993).
Gaillard, M. C. & de Castellanos, Z. J. A. Mollusca, Gasteropoda, Hydrobiidae. In: Ringuelet RA (Dir), Fauna de agua dulce de la República Argentina, 15, pp 1–39. Buenos Aires: Fundación para la Educación, la Ciencia y la Cultura (FECIC) (1976).
Cazzaniga, N. Nota sobre los hidróbidos argentinos. I (Gastropoda: Rissoidea), Acerca de Littoridina occidentalis (Doering, 1884). Neotropica 26, 187–191 (1980).
Cazzaniga, N. Nota sobre los hidróbidos argentinos. II (Gastropoda: Rissoidea), Una Littoridina del “grupo parchappii” en Península Valdés (Chubut). Rev. Mus. La Plata (Nueva Serie) 13. Sección Zool. 129, 11–16 (1982).
Radea, C., Louvrou, I. & Economou-Amilli, A. First record of the New Zealand mud snail Potamopyrgus antipodarum J. E. Gray 1843 (Mollusca: Hydrobiidae) in Greece—Notes on its population structure and associated microalgae. Aquat. Invasions. 3, 341–344 (2008).
Wallace, C. Parthenogenesis, sex, and chromosomes in Potamopyrgus. J. Moll. Stud. 58, 93–107 (1992).
Richards, D. C. & Lester, G. T. Comparison of the number of Potamopyrgus antipodarum neonates produced seasonally, between habitats, and in two freshwater springs, Idaho and Montana: a preliminary investigation. Report to Idaho Power Company, Boise, ID. pp 18 (2000).
de Lucía, M. & Gutiérrez Gregoric, D. E. The genus Potamolithus Pilsbry, 1896 (Gastropoda, Tateidae) on the Somuncurá Plateau, Patagonia, Argentina. Moll. Res. 37, 202–211 (2017b).
Davis, G. M. & Pons da Silva, M. C. Potamolithus: morphology, convergence, and relationships among hydrobioid snails. Malacologia 25, 73–108 (1984).
Pons da Silva, M. C. & Veitenheimer-Mendes, I. L. Redescrição de Potamolithus catharinae com base em topotipos (Gastropoda, Hydrobiidae), rio Hercílio, Santa Catarina, Brasil. Iheringia Ser. Zool. 94, 83–88 (2004).
López Armengol, M. F. Taxonomic revision of Potamolithus agapetus Pilsbry, 1911, and Potamolithus buschii (Frauenfeld, 1865) (Gastropoda: Hydrobiidae). Malacologia 38, 1–17 (1996).
Simone, L. R. L. & Moracchioli, N. Hydrobiidae (Gastropoda: Hydrobioidea) from the Ribeira valley, SE Brazil, with descriptions of two new cavernicolous species. J. Moll. Stud. 60, 445–459 (1994).
Núñez, V. Redescription of Potamolithus paranensis (Pilsbry, 1911) and Potamolithus simplex (Pilsbry, 1911) (Gastropoda: Tateidae). Moll. Res. 37, 17–30 (2017).
Marcus, E. & Marcus, E. On brazilian supralittoral and brackish water snails. Bol. Inst. Oceanogr. (São Paulo) 13, 41–52 (1963).
Marcus, E. & Marcus, E. On brazilian supralittoral and estuarine snails. Bol. Fac. Filos. Ciên. Let. Univ. São Paulo (Zool.) 25, 19–82 (1965).
Pons da Silva, M. C. & Thomé, J. W. Uma nova Heleobia (Prosobranchia, Hyrobidae) do “rio” Guaíba, Rio Grande do Sul. Rev. Bras. Zool. 45, 515–534 (1985).
Jokela, J., Niederegger, S., Negovetic, S. & Mutikainen, P. Mode of reproduction, ploidy and fluctuating asymmetry: comparison of coexisting sexual and asexual freshwater snails. Evol. Ecol. Res. 3, 969–984 (2001).
Hall, T. A. BioEdit: a user-friendly biological sequence alignment editor and analyses. Nucl. Acids Symp. Ser. 41, 95–98 (1999).
Ronquist, F. & Huelsenbeck, J. P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19, 1572–1574 (2003).
Posada, D. jModelTest: phylogenetic model averaging. Mol. Biol.Evol. 25, 1253–1256 (2008).
Kumar, S., Stecher, G. & Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 33, 1870–1874 (2016).
Hershler, R., Liu, H.-P. & Mulvey, M. Phylogenetic relationships within the aquatic snail genus Tryonia: implications for biogeography of the North American Southwest. Mol. Phyl. Evol. 13, 377–391 (1999).
de Lucía, M. & Gutiérrez Gregoric, D. E. Redescripción de Potamolithus supersulcatus Pilsbry, 1896 (Gastropoda, Tateidae) del sur de la cuenca del Plata. Pap. Avulsos Zool., São Paulo 57, 207–219 (2017a).
Kroll, O. et al. The endemic gastropod fauna of Lake Titicaca, correlation between molecular evolution and hydrographic history. Ecol. Evol. 2, 1517–1530 (2012).
Hamada, K., Tatara, Y. & Urabe, M. Survey of mitochondrial DNA haplotypes of Potamopyrgus antipodarum (Caenogastropoda: Hydrobiidae) introduced into Japan. Limnology 14, 223–228 (2013).
Wilke, T. et al. Pushing short DNA fragments to the limit: phylogenetic relationships of ‘hydrobioid’ gastropods (Caenogastropoda: Rissooidea). Mol. Phyl. Evol. 66, 715–736 (2013).
Haase, M. Rapid and convergent evolution of parental care in hydrobiid gastropods from New Zealand. J. Evol. Biol. 18, 1076–1086 (2005).
Acknowledgements
We are especially grateful to Gabriela Hinojosa and Professor Oscar Gálvez of the MNHNCL for their help in the study of type specimens. We also thank Francis Miño, Nicolás Villalobos and Sebastián Pozo for fieldwork assistance and Cristian Suarez for his invaluable work in take SEM images. To CONICYT-FONDEQUIP Program (No EQM-140088) for the acquisition of Hitachi Scanning Electron Microscope (SEM). This study was supported by the Project DIUBB 153309 2/R, ECOS-SUD program 150003 (Project ECOS-CONICYT C15B02) and CONICYT-FONDECYT 11130697.
Author information
Authors and Affiliations
Contributions
G.A.C. designed the study and collected samples. G.A.C. and K.P.A. carried out molecular analyses. G.A.C. and F.J.C. performed morphological analyses and took the photographs. G.A.C. and N.P. wrote the paper, with input from M.A.V., M.A.M., M.A.V. and D.E.G.G. All authors reviewed and discussed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Collado, G.A., Vidal, M.A., Aguayo, K.P. et al. Morphological and molecular analysis of cryptic native and invasive freshwater snails in Chile. Sci Rep 9, 7846 (2019). https://doi.org/10.1038/s41598-019-41279-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-41279-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.