Abstract
The integrin αvβ3 has been shown to play an important role in osteocyte mechanotransduction. It has been reported that there are fewer β3 integrin-containing cells in osteoporotic bone cells. Osteocytes cultured in vitro under estrogen deficient conditions demonstrate altered mechanotransduction. However, it is unknown whether the altered mechanotransduction in estrogen deficient osteocytes is directly associated with defective αvβ3 expression or signalling. The objective of this study is to investigate the role of estrogen deficiency for regulating MLO-Y4 cell morphology, αvβ3 expression, focal adhesion formation and mechanotransduction by osteocytes. Here, we report that estrogen withdrawal leads to a smaller focal adhesion area and reduced αvβ3 localisation at focal adhesion sites, resulting in an increased Rankl/Opg ratio and defective Cox-2 responses to oscillatory fluid flow. Interestingly, αvβ3 antagonism had a similar effect on focal adhesion assembly, Rankl/Opg ratio, and Cox-2 responses to oscillatory fluid flow. Taken together, our results provide the first evidence for a relationship between estrogen withdrawal and defective αvβ3-mediated signalling. Specifically, this study implicates estrogen withdrawal as a putative mechanism responsible for altered αvβ3 expression and resultant changes in downstream signalling in osteocytes during post-menopausal osteoporosis, which might provide an important, but previously unidentified, contribution to the bone loss cascade.
Similar content being viewed by others
Introduction
Osteocytes are the most abundant cells in bone and are responsible for mediating the balance between bone formation and resorption1. It has been proposed that osteocytes detect mechanical stimuli using mechanosensitive proteins, include stretch activated ion channels, gap junctions, primary cilia and integrins2,3,4 and transduce them into biochemical responses5,6. Molecular factors produced by osteocytes regulate osteoclasts and osteoblasts, in particular RANKL and sclerostin, which promote osteoclast formation and inhibit osteoblastogenesis respectively, and OPG, which acts as a decoy receptor for RANK and thereby prevents osteoclast formation7,8,9,10.
Integrins are heterodimeric transmembrane proteins, comprised of α and β subunits, which connect the intracellular cytoskeleton to the extracellular matrix through protein complexes known as focal adhesions (FA), which also comprise proteins such as vinculin, α-actinin, talin, and paxillin11,12. Focal adhesions are involved in Focal Adhesion Kinase (FAK) and shc signalling13,14 and are widely understood to play a role in mechanosensation for many distinct cell types15,16,17,18,19,20,21. Osteocytes express both β1 and β3 integrins4,11 and it has been shown that β1 integrins localise around osteocyte cell bodies, whereas osteocyte cell processes have αvβ3 integrins and both interact with the surrounding pericellular matrix4,22. It has been proposed integrin based adhesions and pericellular matrix tethers together facilitate strain amplification4,23,24,25. In vitro studies have shown that Ca2+ response to a fluid stimulus was highly polarised along osteocyte cell processes but this Ca2+ response was compromised when cultured with a small molecule inhibitor of αvβ317. Moreover, when αvβ3 was blocked using an antagonist, Cox-2 expression and PGE2 release were reduced and cell morphology was altered, whereby the cells had a reduced cell area and fewer cell processes15.
Post-menopausal osteoporosis is a disease characterised by a decrease in circulating estrogen levels and an imbalance in bone cell remodelling, which causes bone loss and an increased susceptibility to fracture26. Estrogen acts as a regulator to maintain the balance of osteoblasts and osteoclasts27 and enhances the response of bone cells to mechanical stress28. It has been reported that the estrogen receptors, ERα and ERβ, play a role in this mechanobiological response by osteoblasts and osteocytes29,30. In osteoblasts, estrogen was shown to increase Opg expression and augment Cox-2 (via β1 integrins and ERs) response to fluid shear stress31,32 and decrease Rankl and Sost expression33,34. In osteocytes, supraphysiological levels of estrogen (100 nM) were shown to have a protective role against apoptosis35,36, to induce an intracellular Ca2+ response37 and increase connexion 43 gap junction expression and mechanosensitivity38. In vitro supplementation of culture media with levels of estrogen (10 nM) within the range of estrogen in healthy humans (pre-menopausal), was shown to led to increased osteogenic signalling by MLO-Y4 osteocytes39. However, most in vitro studies of osteocyte biology use culture media without exogenous estrogen, and thus there is a limited understanding of pre-menopausal levels of estrogen on osteocyte biology.
Human osteoblastic bone cells derived from osteoporotic patients have been shown to exhibit an impaired biochemical response (PGE2) to mechanical stress compared to those derived from healthy patients40. Estrogen deficiency in vitro can be achieved by pre-treatment with 17β-estradiol followed by estrogen withdrawal or addition of an estrogen receptor antagonist39,41. Estrogen withdrawal in osteocytes was shown to attenuate fluid flow-induced intracellular calcium signalling, thus altering osteocyte mechanosensitivity39, and lead to higher levels of osteocyte apoptosis, compared to estrogen treated cells41. Estrogen deficiency induced by ovariectomy (OVX) has been shown to lead to an altered tissue composition and mineral distribution within bone, altered mechanical environment of osteocytes and a reduction in β3-positive cells in cortical bone compared to controls42,43,44. However, it is unknown whether such changes arise as a direct response to reduced estrogen or the ability of the β3 integrins to facilitate mechanotransduction.
In this study, we test the hypothesis that altered osteocyte mechanosensitivity during estrogen deficiency is associated with an impairment in the mechanotransduction by β3 integrins. Specifically, we investigate (1) the role of pre-menopausal levels of estrogen for regulating MLO-Y4 cell morphology, focal adhesion formation and mechanotransduction response to fluid flow, (2) changes in αvβ3 expression and spatial organisation in osteocytes during estrogen deficiency, and (3) whether altered mechanosensitivity of osteocytes under estrogen deficiency correlate to defective αvβ3 expression and functionality.
Results
Estrogen supplementation, to mimic pre-menopausal levels, leads to more mature FA assembly and actin cytoskeleton, and increased Opg expression compared to osteocytes cultured in standard media
In vitro studies of MLO-Y4 osteocyte biology commonly use α-MEM culture media without the addition of estrogen. However, in vivo estrogen is important for normal bone cell function and so, to more closely mimic the physiological environment of osteocytes in vivo, in this study we first investigated the effect of supplementing culture media with pre-menopausal levels of estrogen (10 nM) on MLO-Y4 osteocytes under static and oscillatory flow conditions in vitro.
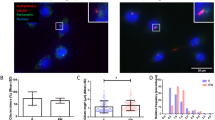
A robust cytoskeleton with distinct focal adhesion sites can be seen in both control and estrogen treated cells under static culture conditions (Fig. 1A). Quantification of these images showed that estrogen supplementation leads to the development of larger cells (p = 0.0525), which have more focal adhesions (p < 0.05) and most interestingly, these cells also have a significantly larger focal adhesion area (p < 0.01) (N = 3, n ≥ 111 cells per group), see Figs 1B,C and S1A. The actin fluorescence intensity was greater and the cells exhibited a higher degree of organisation (anisotropy) of the actin cytoskeleton in estrogen treated cells compared to controls (p < 0.0001 and p < 0.0001 respectively, N = 3, n ≥ 111 cells per group) (Fig. 1D,E). Moreover, estrogen treated osteocytes were shown to express higher levels of Opg (p < 0.01, n = 7–10), but Rankl expression was unaffected, and this ultimately resulted in a lower Rankl/Opg ratio (p = 0.0645, N = 5–7) (Fig. 1F–H). There was no difference in Cox-2 expression in the estrogen treated group compared to controls (Fig. 1I). Thus, estrogen supplementation leads to larger osteocytes with more mature FA assembly and a more robust actin cytoskeleton, and these changes are accompanied by a reduction in gene expression associated with osteoclast differentiation (Rankl/Opg).
The effect of estrogen treatment and oscillatory fluid flow on MLO-Y4 cell morphology and downstream signalling. (A) Immunocytochemistry images showing actin fibres and vinculin staining (N = 3, n ≥ 111 cells per group). Quantification of the images showing (B) cell area, (C) % focal adhesion area/cell area, (D) actin fluorescent intensity, and (E) anisotropy of actin fibres. RT-PCR results of (F) Rankl expression (N = 5–10), (G) Opg expression (N = 7–10), (H) Rankl/Opg ratio (N = 5–7), and (I) Cox-2 expression (N = 7–8) (Student’s t-test, *p < 0.05, **p < 0.01, ****p < 0.0001).
The application of oscillatory fluid flow resulted in an increase in actin fluorescent intensity for both control and estrogen treated cells, when compared to their static counterparts (p < 0.0001 and p < 0.01 respectively, N = 3, n ≥ 111 cells per group). However, fluid flow had no effect on cell area, focal adhesion area, or actin anisotropy for either cohort. Fluid shear stress led to a higher Opg expression in the control group (p < 0.05, N = 7–10), but had no effect on Rankl expression, and therefore, led to a lower Rankl/Opg ratio in that same group (p = 0.051, N = 5–7). There was no effect of fluid shear stress on Rankl or Opg expression in the estrogen treated group. Both control and estrogen treated cells showed a higher Cox-2 response to fluid shear stress (p < 0.05 and p < 0.05 respectively, N = 5–7).
In summary, treatment of osteocytes in vitro with endogenous levels of estrogen, to mimic the pre-menopausal status of osteocyte biology, significantly alters cell behaviour, resulting in a more established cytoskeleton and a shift towards a more osteogenic phenotype when compared to widely used culture media for studies of MLO-Y4 osteocyte biology. Therefore, the estrogen treated groups were used as a control for studies of estrogen withdrawal, which are presented hereafter.
Estrogen withdrawal alters osteocyte morphology, and this affect is further augmented by mechanical stimulation
Next we wished to understand what role estrogen withdrawal had on osteocyte morphology under both static and mechanical loading conditions.
Under static conditions, cells cultured under estrogen withdrawal were observed to be smaller by immunostaining, and while they had focal adhesion sites, these were disordered, which was in contrast to estrogen treated cells that had distinct focal adhesion sites (Fig. 2A). Quantitative analysis of the actin and vinculin staining confirmed that cells cultured under estrogen withdrawal had a reduced cell and focal adhesion area, when compared to cells cultured under continuous estrogen conditions (p < 0.0001 and p < 0.05 respectively, N = 3, n ≥ 115 cells per group) (Fig. 2B,C). The actin staining in the estrogen withdrawal cells was less intense and there were fewer stress fibres, when compared to the estrogen treated cells, which had noticeable and numerous stress fibres (Fig. 2A). Quantitative analysis of the actin staining confirmed that the intensity and anisotropy of the actin fibres were lower following estrogen withdrawal in comparison to estrogen conditions (p < 0.0001 and p < 0.01 respectively, N = 3, n ≥ 115 cells per group) (Fig. 2D,E), demonstrating a less robust and ordered actin cytoskeleton for estrogen deficient osteocytes under static conditions.
The effect of estrogen withdrawal and oscillatory fluid flow on MLO-Y4 cell morphology. (A) Immunocytochemistry images showing actin fibres and vinculin staining (N = 3, n ≥ 115 cells per group). Quantification of the images showing (B) cell area, (C) % focal adhesion area/cell area, (D) actin fluorescent intensity and (E) anisotropy of actin fibres. (Student’s t-test, *p < 0.05, **p < 0.01, ****p < 0.0001).
Fluid shear stress led to a lower cell area in estrogen withdrawal cells when compared to its static counterpart (p < 0.01, N = 3, n ≥ 115 cells per group), but there was no difference in cell area or focal adhesion area following flow in the estrogen supplemented cells. Oscillatory fluid flow led to more intense actin staining in estrogen cells, compared to static groups (p < 0.01, N = 3, n ≥ 115 cells per group), but this was not observed for estrogen withdrawal cells, which also exhibited had a lower anisotropy following fluid flow (p < 0.0001, N = 3, n ≥ 115 cells per group), indicating a less ordered actin fibre organisation. Taken together, this data points to a defect in the formation of mechanosensitive focal adhesion sites following estrogen withdrawal.
Estrogen withdrawal resulted in reduced αvβ3 localisation at focal adhesion sites
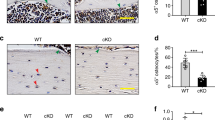
Given the perturbed focal adhesion assembly seen in estrogen withdrawal conditions, we next investigated the effect of estrogen withdrawal on αvβ3 quantity and organisation. ELISA measurements of total αvβ3 content within MLO-Y4 cells showed no statistical differences following estrogen withdrawal (p = 0.0612, N = 6) (Fig. 3B). However, analysis of immunocytochemistry imaging showed significantly lower αvβ3 intensity per cell in estrogen withdrawal cells, compared to estrogen supplemented cells (p < 0.05, N = 3, n ≥ 115 cells per group) (Fig. 3C). At focal adhesion (FA) sites, this lowered αvβ3 intensity in estrogen withdrawal cells was even more pronounced (p < 0.01, N = 3, n ≥ 115 cells per group) (Fig. 3D). A less ordered αvβ3 organisation at FA sites was also qualitatively seen in the immunocytochemistry images for estrogen withdrawal osteocytes (Fig. 3A).
The effect of estrogen withdrawal and oscillatory fluid flow on αvβ3 quantity and organisation in MLO-Y4 cells. (A) Immunocytochemistry images showing αvβ3 and vinculin staining (N = 3, n ≥ 115 cells per group). (B) ELISA measurement of total αvβ3 quantity normalised to DNA content (N = 6). Quantification of the images showing (C) αvβ3 intensity per cell, and (D) αvβ3 intensity at focal adhesion (FA) sites (Student’s t-test, *p < 0.05, **p < 0.01, ****p < 0.0001).
The application of oscillatory fluid flow led to higher αvβ3 content in both estrogen and estrogen withdrawal groups compared to their static counterparts (p < 0.01 and p < 0.05 respectively, N = 6). This relationship was also seen in the αvβ3 intensity across the whole cell area (p < 0.01 and p < 0.0001 respectively, N = 3, n ≥ 115 cells per group), but not at FA sites. Overall, these results demonstrate that estrogen withdrawal led to a lower αvβ3 quantity and impaired organisation at a cell-level and, more importantly, at mechanosensitive focal adhesion sites.
Estrogen withdrawal upregulated expression of genes associated with paracrine osteoclastogenic signalling (Rankl/Opg ratio), but perturbed the Cox-2 response to fluid shear stress
Given the alterations in MLO-Y4 cell morphology and αvβ3 organisation during estrogen withdrawal, we next investigated the effect of estrogen deficiency on genes associated with mechanotransduction (Cox-2) and osteoclastogenesis (Rankl and Opg). Under static culture conditions, estrogen withdrawal led to an upregulation in Rankl expression, in comparison to cells cultured under continuous estrogen culture, (p = 0.0587, N = 7–8), as well as a significant downregulation in Opg expression (p < 0.05, N = 8–9) (Fig. 4A,B). This resulted in a higher Rankl/Opg ratio in estrogen withdrawal conditions in comparison to the E group (p < 0.05, N = 5–7) (Fig. 4C).
After application of oscillatory fluid flow, Rankl expression was lower in the estrogen withdrawal group, compared to its static counterpart (p < 0.05, N = 7–8). However, there was no noticeable effect of flow on Opg expression, and ultimately there was no significant difference in the Rankl/Opg ratio for cells that underwent estrogen withdrawal and were mechanically stimulated (Fig. 4A–C).
For cells cultured under static conditions, estrogen withdrawal had no significant effect on Cox-2 expression, when compared to the estrogen supplemented cells (Fig. 4D). The application of fluid shear stress led to a higher Cox-2 response in the estrogen supplemented group, when compared to its static counterpart (p < 0.05, N = 7–8). However, this response to fluid flow was lost in estrogen withdrawal cells.
Taken together, these results demonstrate that estrogen withdrawal perturbed the normal osteogenic responses to mechanical stimulation by osteocytes, and also lead to the alteration in genes associated with paracrine osteoclastogenic signalling.
αvβ3 antagonism led to an altered cell morphology, as seen with estrogen withdrawal
Due to the fact that we showed a disorganised αvβ3 organisation under estrogen withdrawal conditions, we wished to investigate whether the perturbed cell function and responses to flow following estrogen withdrawal were associated with an altered αvβ3 function. Therefore, we used a small molecular inhibitor to block αvβ3 function.
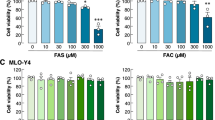
Osteocytes that underwent αvβ3 antagonism demonstrated a perturbed morphology, with a visually smaller cell area and fewer distinct focal adhesion sites visible in the estrogen group (Fig. 5A), in comparison to unblocked counterparts, see Fig. 2A. Quantification of the images confirmed a lower cell area (p < 0.0001), focal adhesion area (p < 0.01), and focal adhesion number (p < 0.05) in the estrogen cells following αvβ3 antagonism (N = 3, n ≥ 90 cells per group) (Figs 5B,C and S1C). However, there was no statistical difference in cell area, focal adhesion area or number between blocked and unblocked cells in the estrogen withdrawn cells. Actin was also shown to be affected by αvβ3 antagonism; whereby there was a lower actin fluorescent intensity and anisotropy in estrogen supplemented cells following αvβ3 antagonism (p < 0.01, p < 0.0001, p < 0.0001 and p < 0.0001 respectively, N = 3, n ≥ 90 cells per group) (Fig. 5D,E). In estrogen withdrawal cells, αvβ3 antagonism led to lower actin intensity in cells subjected to oscillatory fluid flow, and a lower actin anisotropy between the static groups (p < 0.0001 and p < 0.001 respectively, N = 3, n ≥ 90 cells per group).
The effect of αvβ3 antagonism and oscillatory fluid flow on MLO-Y4 cell morphology. (A) Immunocytochemistry images showing actin fibres and vinculin staining (N = 3, n ≥ 90 cells per group). Quantification of the images showing (B) cell area, (C) % focal adhesion area/cell area, (D) actin fluorescent intensity, and (E) anisotropy of actin fibres. (Student’s t-test, ##p < 0.01 compared to static, ###p < 0.001 compared to static, ####p < 0.0001 compared to static, $$$$p < 0.0001 compared to flow).
A key finding was that the estrogen cells that underwent αvβ3 antagonism showed a similar morphology to the estrogen withdrawal cells (unblocked). Quantitative analysis of the images confirmed no difference in cell area, focal adhesion area, or actin cytoskeleton intensity and organisation between estrogen cells following αvβ3 antagonism and estrogen withdrawal cells (unblocked). These results clearly underpin the importance of αvβ3 on overall cell function and implicate the dysregulation of the integrin αvβ3 for the impaired cell morphology during estrogen withdrawal.
αvβ3 antagonism led to a lower αvβ3 intensity per cell and at FA sites
To confirm the efficacy of the αvβ3 antagonism, we investigated the αvβ3 quantity following antagonism. We investigated the intensity of αvβ3 per cell and at FA sites using immunostaining. As expected, we saw a lower αvβ3 intensity per cell and at FA sites in the estrogen cells in comparison to their unblocked counterparts (p < 0.05, p < 0.0001, p < 0.01, and p < 0.05 respectively, N = 3, n ≥ 90 cells per group) (Fig. 6C,D). In the estrogen withdrawal groups, we saw no change in αvβ3 intensity per cell and at FA sites following antagonism, indicating that the estrogen withdrawal groups already had a lowered αvβ3 quantity prior to antagonism. Further to this, there was no statistical difference in the intensity of αvβ3 between αvβ3 blocked estrogen cells and unblocked estrogen withdrawal cells. These results further confirm that estrogen withdrawal has a negative effect on αvβ3 integrin quantity and organisation. ELISA measurements of αvβ3 antagonism showed a higher αvβ3 content compared to unblocked counterparts (p < 0.05, p < 0.05 and p < 0.05 respectively, N = 6) (Fig. 6B).
The effect of αvβ3 antagonism and oscillatory fluid flow on αvβ3 quantity and organisation in MLO-Y4 cells. (A) Immunocytochemistry images showing αvβ3 and vinculin staining (N = 3, n ≥ 90 cells per group). (B) ELISA measurement of total αvβ3 quantity normalised to DNA content (N = 6). Quantification of the images showing (C) αvβ3 intensity per cell, and (D) αvβ3 intensity at focal adhesion (FA) sites (Student’s t-test, *p < 0.05, #p < 0.05 compared to static, ##p < 0.01 compared to static, $p < 0.05 compared to flow, $$$$p < 0.0001 compared to flow).
αvβ3 antagonism affected the Rankl/Opg response to estrogen withdrawal and resulted in a perturbed Cox-2 response to fluid flow
Finally, we wished to delineate the role of the integrin αvβ3 in the altered Rankl/Opg and Cox-2 responses, seen during estrogen withdrawal. αvβ3 antagonism attenuated the higher Rankl response and lower Opg response seen previously in estrogen withdrawal conditions (Fig. 7A,B) and in turn resulted in no change in Rankl/Opg ratio in estrogen withdrawal cells (Fig. 7C). This lack of Rankl/Opg response to the estrogen withdrawal conditions following antagonism may implicate the integrin αvβ3 as an important transducer of estrogen-related signalling. Moreover, following αvβ3 antagonism, the Cox-2 response to flow was absent in all groups (Fig. 7D). Taken together, these results show the importance of changes in the localisation of the integrin αvβ3 in the estrogen deficient conditions of post-menopausal osteoporosis.
Discussion
Given the importance of the integrin αvβ3 in osteocyte mechanotransduction, we sought to investigate the effect of estrogen withdrawal on the integrin αvβ3 and αvβ3-mediated signalling. Here, we report for the first time that, under static conditions, cell area, focal adhesion area, and the distribution of αvβ3 at mechanosensitive focal adhesion sites were all negatively affected by estrogen withdrawal conditions. Moreover, downstream mechanotransduction signalling associated with the integrin αvβ3 (Cox-2 gene expression) was negatively affected following estrogen withdrawal. Osteocytes, in static conditions, that underwent estrogen withdrawal also increased expression of the Rankl gene and downregulated Opg expression, which are known to regulate osteoclastogenesis in a paracrine fashion. To investigate whether the integrin αvβ3 was responsible for the changes seen in estrogen withdrawal conditions, we blocked the integrin αvβ3 with an antagonist. Most interestingly, we report similar changes in cell morphology and downstream signalling in the absence of functional αvβ3 mechanoreceptors, as we had observed for cells that underwent estrogen withdrawal. Therefore, our results implicate a link between the estrogen withdrawal conditions of post-menopausal osteoporosis and altered αvβ3 functionality (Fig. 8).
Previous studies have clearly demonstrated defective osteocyte mechanotransduction during estrogen withdrawal in vitro39,42. In vivo, the number of β3-containing bone cells in cortical bone was lower compared to controls in an estrogen deficient rat model of post-menopausal osteoporosis42. Here, we demonstrate an altered osteocyte morphology, specifically by means of a lower cell area and altered actin cytoskeleton, following estrogen withdrawal, which is in keeping with previous research39. We report for the first time impaired focal adhesion assembly in cells that are cultured under estrogen withdrawal conditions, along with a lower αvβ3 intensity at these compromised focal adhesion sites. Disperse cytosolic staining of vinculin was clearly visible in estrogen withdrawal cells. However, only distinct focal adhesion sites were quantified. The αvβ3 staining in our estrogen treated cells showed similarities to studies of MLO-Y4 cells in culture media (without estrogen supplementation)15,45, but the spatial relationship of αvβ3 to other FA proteins, such as vinculin, has not been previously investigated. Recently, β3 integrins were found along mouse osteocyte cells processes in vivo within specialised structures containing pannexin1, P2X7R, and CaV3.2-1. However, in that study the larger vinculin containing-focal adhesion complexes were found around the cell body and not the tightly packed cell processes, due to space constraints22. In our in vitro study, cells do not have space constraints, and we show that vinculin co-localises with αvβ3 integrin. While it is known that the β3 integrin binding sites along osteocyte cell processes are key sites in osteocyte mechanotransduction in vivo, particularly due to strain amplification, computational modelling has shown that osteocytes subjected to fluid shear stress in vitro, as seen in in this study, do transduce this mechanical stimulation through integrin binding sites found along the cell processes and cell body46. As such, we have measured focal adhesion sites found along the cell processes and cell body in our study.
Together our findings of an increase in Rankl/Opg ratio, under static conditions, and abrogation of the Cox-2 response to oscillatory fluid flow demonstrate an altered response to mechanical stimulation in estrogen withdrawal conditions. RANKL is a cytokine produced by osteocytes, which binds to RANK receptors on osteoclast progenitors and regulates osteoclast differentiation and activity, whereas OPG is also produced by osteocytes but acts a decoy receptor for RANKL47,48,49,50,51. It has been established that the application of oscillatory fluid shear stress to MLO-Y4 cells results in a decrease in the Rankl/Opg ratio16, which indicates that mechanically stimulated osteocytes reduce paracrine signalling of osteoclastogenesis. We showed that estrogen withdrawal led to an increased Rankl/Opg ratio. Estrogen withdrawal has been shown to induce osteocyte apoptosis41,52, which, in turn, leads to changes in RANKL expression53. Interestingly, it has previously been reported that in osteoblasts, estrogen treatment led to higher Opg expression in a dose-dependent manner, compared to untreated control cells31. COX-2 is an enzyme that produces prostaglandins to promote inflammation, and expression of the Cox-2 is upregulated in osteocytes following oscillatory fluid flow2,16. Given the importance of Cox-2 for normal fracture repair54 and an increased Rankl/Opg ratio for inducing osteoclast activity7,9,26, we propose that under estrogen withdrawal, osteocyte signalling would contribute to the increased bone resorption seen in post-menopausal osteoporosis26,55.
The importance of integrin αvβ3 in osteocyte mechanotransduction has been recently demonstrated15,17, and, in keeping with a previous study15, our results show that αvβ3 antagonism resulted in a smaller cell area. Interestingly, αv-integrin antagonism in melanoma cells was shown to lead to rearrangement and disassembly of focal adhesion proteins56 and we propose that integrin antagonism may have a similar effect on focal adhesion in osteocytes. It is interesting to note that while focal adhesion area was lower following αvβ3 antagonism, some distinct focal adhesion sites were present. If we assume that the antagonist blocked all αvβ3-containing focal adhesion sites, it is likely that these remaining focal adhesion sites contained other integrin subunits, such as β1 integrins4. αvβ3 antagonism resulted in a lower αvβ3 fluorescent intensity. Moreover, αvβ3 antagonism resulted in an attenuated Rankl and Opg response following estrogen withdrawal. As the integrin αvβ3 was shown to mediate OPG protein production in a human endothelial-breast cancer cell co-culture57, we propose that the integrin αvβ3 plays a similar role in regulating Rankl and Opg signalling in osteocytes. Previous research studying the role of αvβ3 in Rankl and Opg expression, saw no significant changes following fluid flow, and therefore it was unclear what role the integrin αvβ3 played in the Rankl/Opg response to flow in this instance15. Following αvβ3 antagonism, we saw a diminished Cox-2 response to oscillatory fluid flow. Abrogation of this Cox-2 response to flow following αvβ3 antagonism has been demonstrated previously for MLO-Y4 cells cultured under control conditions (no added estrogen)15. Other integrin subunits, notably β1 integrins, play an important role in osteocyte mechanotransduction and have been shown to be necessary for normal Cox-2 response to flow16,58. While the role of β1 integrins was not investigated in this study, it is interesting to note that β1 integrins were not fully able to recover Cox-2 responses to flow following αvβ3 antagonism. This may indicate that β1 and β3 integrins are closely linked in function in vitro. By showing an altered focal adhesion assembly, αvβ3 localisation at FA sites, and abrogated Cox-2 response to flow following estrogen withdrawal or αvβ3 antagonism, our study is the first to demonstrate a link between altered osteocyte mechanobiology during estrogen withdrawal and altered αvβ3 functionality.
Following mechanical stimulation of osteocytes, αvβ3 has been shown to activate MAP kinase pathways leading to an upregulation in c-fos, IGF-1, and Cox-245. It has been proposed that upregulation of c-fos is associated with the Ca2+ influx pathway45, which has separately been shown to be mediated by αvβ317. In osteoblasts, the αvβ3 pathway has been studied more extensively, with the application of fluid flow leading to a synergistic activation of FAK and shc, resulting in the activation of PI3-K and Akt/mTOR/p70S6K pathway, and ultimately increases in Cox-2 expression14. In osteocytes, FAK, PI3-K, and Akt have been shown to be activated in response to fluid shear stress, but it is unknown whether this is mediated by αvβ359. Estrogen treatment has been shown to induce FAK phosphorylation, and activation of PI3-K in breast cancer cells60 and in HUVECs61. The breast cancer cells also activated Akt and formed focal adhesion sites in response to estrogen treatment60. Thus we propose that Cox-2 expression by osteocytes following oscillatory fluid flow may arise because αvβ3 integrins activate FAK, PI3-K and Akt pathways. Moreover, due to the role of estrogen in regulating FAK and PI3-K pathways in other cell types, we propose that estrogen withdrawal may result in a defect in this proposed αvβ3 pathway and could account for the abrogated Cox-2 response to fluid flow.
There are a number of limitations that must be considered. Firstly, this study involved the use of MLO-Y4 osteocyte-like cells, which were cultured in a monolayer within a parallel plate flow bioreactor. While this cannot fully mimic the mechanical stimulation of osteocytes within the lacunar-canalicular system in vivo, both the MLO-Y4 cells and the bioreactor have been used a model extensively in vitro to understand the mechanobiology of osteocytes during health and disease15,16,39,62,63,64. Secondly, our regime involved immediate withdrawal of estrogen, but the exact timeline of changes in serum estradiol levels during the menopause in vivo is unknown. In humans, serum estradiol levels deplete over a four year period65, whereas in mice, serum estradiol levels were shown to be lower than normal controls one week after ovariectomy66. Thirdly, it was surprising that ELISA measurements of αvβ3 antagonism showed a higher αvβ3 content compared to unblocked counterparts. We believe that these higher readings may be due to the fact that the αvβ3 antagonist was detected by the ELISA along with MLO-Y4 αvβ3 integrins. However, it should be noted that the immunocytochemistry staining confirmed the effectiveness of the αvβ3 antagonist.
Our results show that the mechanosensor, integrin αvβ3, and downstream gene expression is negatively affected by estrogen withdrawal and therefore highlights the importance of integrin αvβ3 in post-menopausal osteoporosis. As β3 integrin expression is affected by estrogen deficiency in OVX animals, this study implicates estrogen withdrawal as the mechanism responsible for altered αvβ3 expression and resultant downstream signalling in osteocytes during post-menopausal osteoporosis. Further study to determine a means of protecting the normal expression and signalling of these integrins may offer a potential novel therapy for post-menopausal osteoporosis. Given the altered morphology and gene expression in both estrogen withdrawal conditions and following αvβ3 antagonism, it is clear that the integrin αvβ3 is affected by estrogen withdrawal and this in turn affects αvβ3-mediated signalling. However, it is unlikely that the integrin αvβ3 is the only mechanosensor affected by estrogen withdrawal. Many other mechanosensors including β1 integrins16, the primary cilium2, and stretch activated ion channel, TRPV43, have all been shown to be important in osteocyte mechanotransduction. To date, the effect of estrogen withdrawal on the ability of these other mechanosensors to transduce mechanical stimulation has not been reported. Given the multitudinous effects of estrogen on cell function, studying the effects of estrogen withdrawal on these other osteocyte mechanosensors may provide an exciting avenue of future research.
Conclusions
Our results show that osteoporotic conditions induced by estrogen withdrawal negatively affected mechanosensor function, specifically through alterations in αvβ3 quantity at focal adhesion sites, and that also resulted in impaired mechanobiological responses to flow (Cox-2 and Rankl/Opg gene expression). These altered responses to flow were also seen following αvβ3 antagonism. We propose that due to the estrogen deficient conditions of post-menopausal osteoporosis, osteocytes undergo altered αvβ3 organisation and resultant changes in downstream signalling, particularly those that govern osteoclastogenesis. This research thus uncovers a previously unknown element of the bone loss cascade, whereby estrogen deficiency alters the mechanosensory ability of osteocytes and they in turn contribute to paracrine signalling to govern osteoclast resorption.
Materials and Methods
Cell Culture, and Estrogen Treatment Regimes
MLO-Y4 mouse osteocyte-like cells were cultured on type I collagen (0.15 mg/ml in 0.02 M acetic acid and phosphate buffered saline (PBS)) coated T-75 flasks in α-minimum essential media (α-MEM) supplemented with 5% fetal calf serum (FCS), 5% fetal bovine serum (FBS), 2 mM L-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin at 37 °C in a humidified environment at 5% CO2. The effect of estrogen treatment and estrogen withdrawal on MLO-Y4 cells was studied using the following groups: (1) standard culture media with no added 17β-estradiol (Ctrl), (2) continuous treatment with 10 nM 17 β-estradiol for 5 days (E), and (3) pre-treatment with 10 nM 17β-estradiol for 3 days and withdrawal for 2 further days (EW), following previous approaches developed in our laboratory39. On day 3 (two days prior to the induction of oscillatory fluid flow/static conditions), cells were seeded onto collagen coated glass slides, at a density of 200,000 cells/slide, and cultured for two days in accordance with their treatment groups.
Integrin αvβ3 Antagonism
The integrin αvβ3 was blocked using a small molecule inhibitor for αvβ3 integrins, IntegriSense 750 (PerkinElmer)15,17. After removal of culture medium, 1 mL of media containing 0.5 µM IntegriSense 750 was added to each slide for 30 min prior to flow/static conditions. Following this, slides were washed twice with PBS.
Oscillatory Fluid Flow
Laminar oscillatory fluid flow was applied to a cell monolayer using a parallel plate bioreactor system, which consisted of a syringe pump (NE-1600, New Era Pump Systems, Farmingdale, NY), polycarbonate plate chambers, media reservoirs connected by gas-permeable and platinum-cured silicone tubing (Cole-Parmer, Vernon Hills, IL)15,39. The fluid flow regime subjected the cell monolayer to a shear stress of 1 Pa at 0.5 Hz for 1 h, which is within the range of shear stresses experiences by osteocytes in vivo25,67,68,69,70. For comparison, static counterparts in each treatment group were not subjected to laminar oscillatory fluid flow conditions for the same time-period.
DNA Content
DNA content was measured using a Hoechst assay. Briefly, 1 mL of sterile deionised (DI) water was carefully placed on each slide and the cells were lysed via the freeze-thaw lysis method in a −80 °C freezer. DNA samples were added to wells with the working dye solution consisting of Hoechst dye: 1X Hoechst Buffer (1:1,000) (Sigma Aldrich). The fluorescence intensity was read using a spectrophotometer for an excitation of 360 nm and at an emission of 460 nm. Calf thymus DNA (Sigma Aldrich) was used as a standard.
Integrin αvβ3 Quantification
αvβ3 integrin concentration was quantified by means of an αvβ3 ELISA (Emelca Bioscience) as per the supplier’s protocol. Briefly, cells were lysed used the freeze-thaw lysis method, as above. The sample lysate was placed in the ELISA wells and an antibody against αvβ3 was added to each well. Biotin labelled antibodies and horseradish peroxidase were added to further label the samples for αvβ3 integrins. The well plate was incubated for 37 °C and washed with a buffer between each of these steps, according to the supplier’s protocol. Reagents to aid in colour visualisation were added and the plate absorbance was read at 450 nm using a spectrophotometer (Synergy HT, BioTek). αvβ3 integrin concentration readings were normalised to DNA content.
Immunofluorescence
Immunofluorescence was used to study and quantify the spatial distribution of αvβ3 integrins in cells from each treatment group cultured statically or following application of flow. Briefly, the cells were washed in PBS and fixed in 3.8% formaldehyde solution, and then permeabilised in 0.1% Triton-X. The cells were incubated in 10% BSA to prevent non-specific binding occurring during the staining process. Integrin αvβ3 staining was performed using an anti-αvβ3 antibody directly conjugated to Alexa Fluor® 488 (1:100) (Santa Cruz). Vinculin staining was performed using a mouse anti-vinculin primary antibody (1:800) (Sigma Aldrich) and goat anti-mouse Alexa Fluor® 647 secondary antibody (1:200) (Abcam) to label focal adhesion and facilitate the investigation of αvβ3 integrin co-localisation (See Supplementary Table 1 for further information on antibodies used). The cells were also stained with TRITC (Tetramethyl Rhodamine Iso-Thiocyanate) Phalloidin and DAPI (4′,6-diamidino-2-phenylindole) to facilitate imaging of the actin cytoskeleton and nucleus respectively. Z-stack imaging was done using a Fluoview FV100 confocal laser scanning microscope system (Olympus) at a magnification of 60x (oil immersion) with a step size of 0.5 µm.
All image analysis was completed using ImageJ software71. The z-stacks of the images taken were combined as maximum intensity projections and these combined images were used for all image analysis. Cell area and overall actin fluorescence intensity were measured using the actin stained images. Cell area was measured by thresholding the images to remove background fluorescence and then using the “wand tool” to select the region of interest around each cell. Where two cells were touching, the region of interest was drawn manually with the “freehand tool”. Anisotropy of the actin fibrils was determined using a ImageJ plugin, known as FibrilTool72. The vinculin stained images were used to identify distinct focal adhesion sites across the entire cell. Identification of distinct focal adhesion sites was enabled by means of a previously published semi-automatic protocol73. Focal adhesion area was normalised to cell area. The focal adhesion ROIs thresholded in this process were then used to determine the intensity of the αvβ3 staining at focal adhesion sites. All fluorescent intensities were measured using the integrated density of each cell/ROI with their corresponding background integrated density subtracted.
Real Time PCR
Relative gene expression was studied by quantitative Real Time Polymerase Chain Reaction (qRT-PCR). The genes of interest included Cox-2, Rankl, and Opg, with Rpl13A used as a reference gene (Supplementary Table 2). RNA was isolated using Qiagen RNeasy kits as per manufacturer’s instructions. RNA purity and yield were assessed using a spectrophotometer (DS-11 FX, DeNovix), with 260/280 ratios of >1.9 for all samples. 250–500 ng of RNA was then transcribed into cDNA using Qiagen Quantinova reverse transcription kits and thermal cycler (5PRIMEG/O2, Prime). qRT-PCR was carried out with a Qiagen Quantinova SYBR Green PCR kit and a StepOne Plus PCR machine (Applied Biosciences). Analysis of the results was done using the Pfaffl method74.
Statistical analysis
Data is presented as mean ± standard deviation. Statistical significance was determined by means of unpaired two-tailed Student’s t-tests. All statistical analyses were performed using GraphPad Prism version 6 (Windows, GraphPad Software, La Jolla California USA, www.graphpad.com) and p-value of 0.05.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
McNamara, L. In Comprehensive Biomaterials (eds Ducheyne, P., Healy, K. E., Hutmacher, D. W., Grainger, D. W. & Kirkpatrick, C. J.) 169–186, https://doi.org/10.1016/B978-0-08-055294-1.00068-4 (Elsevier, 2011).
Lee, K. L. et al. The primary cilium functions as a mechanical and calcium signaling nexus. Cilia 4, 7 (2015).
Lyons, J. S. et al. Microtubules tune mechanotransduction through NOX2 and TRPV4 to decrease sclerostin abundance in osteocytes. Sci. Signal. 10 (2017).
McNamara, L. M., Majeska, R. J., Weinbaum, S., Friedrich, V. & Schaffler, M. B. Attachment of osteocyte cell processes to the bone matrix. Anat Rec 292, 355–363 (2009).
Lanyon, L. E. Osteocytes, strain detection, bone modeling and remodeling. Calcif. Tissue Int. 53 Suppl 1, S102-6; discussionS106-7 (1993).
Bonewald, L. F. The amazing osteocyte. J. Bone Miner. Res. 26, 229–238 (2011).
Han, Y., You, X., Xing, W., Zhang, Z. & Zou, W. Paracrine and endocrine actions of bone—the functions of secretory proteins from osteoblasts, osteocytes, and osteoclasts. Bone Res. 6, 16 (2018).
Li, X. et al. Sclerostin Binds to LRP5/6 and Antagonizes Canonical Wnt Signaling. J. Biol. Chem. 280, 19883–19887 (2005).
Nakashima, T. et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 17, 1231 (2011).
Schaffler, M. B., Cheung, W.-Y., Majeska, R. & Kennedy, O. Osteocytes: Master Orchestrators of Bone. Calcif. Tissue Int. 94, 5–24 (2014).
Hughes, D. E., Salter, D. M., Dedhar, S. & Simpson, R. Integrin expression in human bone. J. Bone Miner. Res. 8, 527–533 (1993).
Zaidel-Bar, R., Cohen, M., Addadi, L. & Geiger, B. Hierarchical assembly of cell–matrix adhesion complexes. Biochem. Soc. Trans. 32, 416 LP–420 (2004).
Mitra, S. K., Hanson, D. A. & Schlaepfer, D. D. Focal adhesion kinase: in command and control of cell motility. Nat. Rev. Mol. Cell Biol. 6, 56 (2005).
Lee, D.-Y. et al. Oscillatory Flow-induced Proliferation of Osteoblast-like Cells Is Mediated by αvβ3 and β1 Integrins through Synergistic Interactions of Focal Adhesion Kinase and Shc with Phosphatidylinositol 3-Kinase and the Akt/mTOR/p70S6K Pathway. J. Biol. Chem. 285, 30–42 (2010).
Haugh, M. G., Vaughan, T. J. & McNamara, L. M. The role of integrin alpha(V)beta(3) in osteocyte mechanotransduction. J Mech Behav Biomed Mater 42, 67–75 (2015).
Litzenberger, J. B., Kim, J. B., Tummala, P. & Jacobs, C. R. Beta1 integrins mediate mechanosensitive signaling pathways in osteocytes. Calcif Tissue Int 86, 325–332 (2010).
Thi, M. M., Suadicani, S. O., Schaffler, M. B., Weinbaum, S. & Spray, D. C. Mechanosensory responses of osteocytes to physiological forces occur along processes and not cell body and require alphaVbeta3 integrin. Proc Natl Acad Sci USA 110, 21012–21017 (2013).
le Duc, Q. et al. Vinculin potentiates E-cadherin mechanosensing and is recruited to actin-anchored sites within adherens junctions in a myosin II–dependent manner. J. Cell Biol. 189, 1107–1115 (2003).
Yao, M. et al. The mechanical response of talin. Nat. Commun. 7, 11966 (2016).
Le, S. et al. Mechanotransmission and Mechanosensing of Human alpha-Actinin 1. Cell Rep. 21, 2714–2723 (2017).
von Wichert, G. et al. RPTP-α acts as a transducer of mechanical force on αvβ3-integrin–cytoskeleton linkages. J. Cell Biol. 161, 143 LP–153 (2003).
Cabahug‐Zuckerman, P. et al. Potential role for a specialized β3 integrin‐based structure on osteocyte processes in bone mechanosensation. J. Orthop. Res. 36, 642–652 (2017).
You, L.-D., Weinbaum, S., Cowin, S. C. & Schaffler, M. B. Ultrastructure of the osteocyte process and its pericellular matrix. Anat. Rec. Part A Discov. Mol. Cell. Evol. Biol. 278A, 505–513 (2004).
You, L., Cowin, S. C., Schaffler, M. B. & Weinbaum, S. A model for strain amplification in the actin cytoskeleton of osteocytes due to fluid drag on pericellular matrix. J. Biomech. 34, 1375–1386 (2001).
Wang, Y., McNamara, L. M., Schaffler, M. B. & Weinbaum, S. A model for the role of integrins in flow induced mechanotransduction in osteocytes. Proc Natl Acad Sci USA 104, 15941–15946 (2007).
McNamara, L. M. Perspective on post-menopausal osteoporosis: establishing an interdisciplinary understanding of the sequence of events from the molecular level to whole bone fractures. J R Soc Interface 7, 353–372 (2010).
Krum, S. A. et al. Estrogen protects bone by inducing Fas ligand in osteoblasts to regulate osteoclast survival. EMBO J. 27, 535–545 (2008).
Bakker, A. D. et al. Additive effects of estrogen and mechanical stress on nitric oxide and prostaglandin E2 production by bone cells from osteoporotic donors. Osteoporos. Int. 16, 983–989 (2005).
Zaman, G. et al. Osteocytes Use Estrogen Receptor α to Respond to Strain but Their ERα Content Is Regulated by Estrogen. J. Bone Miner. Res. 21, 1297–1306 (2009).
Castillo, A. B., Triplett, J. W., Pavalko, F. M. & Turner, C. H. Estrogen receptor-β regulates mechanical signaling in primary osteoblasts. Am. J. Physiol. - Endocrinol. Metab. 306, E937–E944 (2014).
Jia, J., Zhou, H., Zeng, X. & Feng, S. Estrogen stimulates osteoprotegerin expression via the suppression of miR-145 expression in MG-63 cells. Mol. Med. Rep. 15, 1539–1546 (2017).
Yeh, C.-R. et al. Estrogen Augments Shear Stress–Induced Signaling and Gene Expression in Osteoblast-like Cells via Estrogen Receptor–Mediated Expression of β(1)-Integrin. J. Bone Miner. Res. 25, 627–639 (2010).
Galea, G. L. et al. Estrogen Receptor α Mediates Proliferation of Osteoblastic Cells Stimulated by Estrogen and Mechanical Strain, but Their Acute Down-regulation of the Wnt Antagonist Sost Is Mediated by Estrogen Receptor β. J. Biol. Chem. 288, 9035–9048 (2013).
Bord, S., Ireland, D. C., Beavan, S. R. & Compston, J. E. The effects of estrogen on osteoprotegerin, RANKL, and estrogen receptor expression in human osteoblasts. Bone 32, 136–141 (2003).
Marathe, N., Rangaswami, H., Zhuang, S., Boss, G. R. & Pilz, R. B. Pro-survival Effects of 17β-Estradiol on Osteocytes Are Mediated by Nitric Oxide/cGMP via Differential Actions of cGMP-dependent Protein Kinases I and II. J. Biol. Chem. 287, 978–988 (2012).
Plotkin, L. I., Aguirre, J. I., Kousteni, S., Manolagas, S. C. & Bellido, T. Bisphosphonates and Estrogens Inhibit Osteocyte Apoptosis via Distinct Molecular Mechanisms Downstream of Extracellular Signal-regulated Kinase Activation. J. Biol. Chem. 280, 7317–7325 (2005).
Ren, J. & Wu, J. H. 17β-Estradiol Rapidly Activates Calcium Release from Intracellular Stores via the GPR30 Pathway and MAPK Phosphorylation in Osteocyte-Like MLO-Y4 Cells. Calcif. Tissue Int. 90, 411–419 (2012).
Ren, J., Wang, X.-H., Wang, G.-C. & Wu, J.-H. 17β Estradiol regulation of connexin 43-based gap junction and mechanosensitivity through classical estrogen receptor pathway in osteocyte-like MLO-Y4 cells. Bone 53, 587–596 (2013).
Deepak, V., Kayastha, P. & McNamara, L. M. Estrogen deficiency attenuates fluid flow–induced [Ca2+]i oscillations and mechanoresponsiveness of MLO-Y4 osteocytes. FASEB J., https://doi.org/10.1096/fj.201601280R (2017).
Sterck, J. G. H., Klein-Nulend, J., Lips, P. & Burger, E. H. Response of normal and osteoporotic human bone cells to mechanical stress in vitro. Am. J. Physiol. - Endocrinol. Metab. 274, E1113 LP–E1120 (1998).
Brennan, M. A. et al. Estrogen Withdrawal from Osteoblasts and Osteocytes Causes Increased Mineralization and Apoptosis. Horm. Metab. Res. 46, 537–545 (2014).
Voisin, M. & McNamara, L. M. Differential β 3 and β 1 Integrin Expression in Bone Marrow and Cortical Bone of Estrogen Deficient Rats. Anat. Rec. 298, 1548–1559 (2015).
Verbruggen, S. W. W., Mc Garrigle, M. J. J., Haugh, M. G. G., Voisin, M. C. C. & McNamara, L. M. M. Altered Mechanical Environment of Bone Cells in an Animal Model of Short- and Long-Term Osteoporosis. Biophys. J. 108, 1587–1598 (2015).
Brennan, M. A., Gleeson, J. P., O’Brien, F. J. & McNamara, L. M. Effects of ageing, prolonged estrogen deficiency and zoledronate on bone tissue mineral distribution. J. Mech. Behav. Biomed. Mater. 29, 161–170 (2014).
Miyauchi, A. et al. AlphaVbeta3 integrin ligands enhance volume-sensitive calcium influx in mechanically stretched osteocytes. J Bone Min. Metab 24, 498–504 (2006).
Vaughan, T. J., Mullen, C. A., Verbruggen, S. W. & McNamara, L. M. Bone cell mechanosensation of fluid flow stimulation: a fluid–structure interaction model characterising the role integrin attachments and primary cilia. Biomech. Model. Mechanobiol. 14, 703–718 (2015).
Clarke, B. Normal Bone Anatomy and Physiology. Clin. J. Am. Soc. Nephrol. 3, S131 LP–S139 (2008).
Boyce, B. F. & Xing, L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Arch. Biochem. Biophys. 473, 139–146 (2008).
Yasuda, H. et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. 95, 3597 LP–3602 (1998).
Asagiri, M. & Takayanagi, H. The molecular understanding of osteoclast differentiation. Bone 40, 251–264 (2007).
Takayanagi, H. The Role of NFAT in Osteoclast Formation. Ann. N. Y. Acad. Sci. 1116, 227–237 (2007).
Emerton, K. B. et al. Osteocyte apoptosis and control of bone resorption following ovariectomy in mice. Bone 46, 577–583 (2010).
Cabahug-Zuckerman, P. et al. Osteocyte Apoptosis Caused by Hindlimb Unloading is Required to Trigger Osteocyte RANKL Production and Subsequent Resorption of Cortical and Trabecular Bone in Mice Femurs. J. Bone Miner. Res. 31, 1356–1365 (2016).
Forwood, M. R. Inducible cyclo-oxygenase (COX-2) mediates the induction of bone formation by mechanical loading in vivo. J. Bone Miner. Res. 11, 1688–1693 (1996).
Brennan, O. et al. Temporal changes in bone composition, architecture, and strength following estrogen deficiency in osteoporosis. Calcif Tissue Int 91, 440–449 (2012).
Castel, S. et al. Alpha v integrin antagonists induce the disassembly of focal contacts in melanoma cells. Eur. J. Cell Biol. 79, 502–512 (2000).
Reid, P. E., Brown, N. J. & Holen, I. Breast cancer cells stimulate osteoprotegerin (OPG) production by endothelial cells through direct cell contact. Mol. Cancer 8, 49 (2009).
Litzenberger, J. B., Tang, W. J., Castillo, A. B. & Jacobs, C. R. Deletion of β1 Integrins from Cortical Osteocytes Reduces Load-Induced Bone Formation. Cell. Mol. Bioeng. 2, 416–424 (2009).
Santos, A., Bakker, A. D., Zandieh-Doulabi, B., de Blieck-Hogervorst, J. M. A. & Klein-Nulend, J. Early activation of the β-catenin pathway in osteocytes is mediated by nitric oxide, phosphatidyl inositol-3 kinase/Akt, and focal adhesion kinase. Biochem. Biophys. Res. Commun. 391, 364–369 (2010).
Sanchez, A. M. et al. Estrogen Receptor-α Promotes Breast Cancer Cell Motility and Invasion via Focal Adhesion Kinase and N-WASP. Mol. Endocrinol. 24, 2114–2125 (2010).
Sanchez, A. M. et al. Estrogen receptor-α promotes endothelial cell motility through focal adhesion kinase. MHR Basic Sci. Reprod. Med. 17, 219–226 (2011).
Alford, A. I., Jacobs, C. R. & Donahue, H. J. Oscillating fluid flow regulates gap junction communication in osteocytic MLO-Y4 cells by an ERK1/2 MAP kinase-dependent mechanism small star, filled. Bone 33, 64–70 (2003).
Cheng, B. et al. Expression of functional gap junctions and regulation by fluid flow in osteocyte-like MLO-Y4 cells. J. Bone Miner. Res. 16, 249–259 (2001).
Vaughan, T. J., Haugh, M. G. & McNamara, L. M. A fluid-structure interaction model to characterize bone cell stimulation in parallel-plate flow chamber systems. J. R. Soc. Interface 10, 20120900 (2013).
Sowers, M. R. et al. Estradiol Rates of Change in Relation to the Final Menstrual Period in a Population-Based Cohort of Women. J. Clin. Endocrinol. Metab. 93, 3847–3852 (2008).
Smeester, B., et al The relationship of bone-tumor induced spinal cord astrocyte activation and aromatase expression to mechanical hyperalgesia and cold hypersensitivity in intact female and ovariectomized mice. Neuroscience 324 (2016).
Han, Y., Cowin, S. C., Schaffler, M. B. & Weinbaum, S. Mechanotransduction and strain amplification in osteocyte cell processes. Proc. Natl. Acad. Sci. USA 101, 16689–16694 (2004).
Verbruggen, S. W., Vaughan, T. J. & McNamara, L. M. Strain amplification in bone mechanobiology: a computational investigation of the in vivo mechanics of osteocytes. J. R. Soc. Interface 9, 2735–44 (2012).
Weinbaum, S., Cowin, S. C. & Zeng, Y. A model for the excitation of osteocytes by mechanical loading-induced bone fluid shear stresses. J. Biomech. 27, 339–360 (1994).
Li, J., Rose, E., Frances, D., Sun, Y. & You, L. Effect of oscillating fluid flow stimulation on osteocyte mRNA expression. J. Biomech. 45, 247–251 (2012).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671 (2012).
Boudaoud, A. et al. FibrilTool, an ImageJ plug-in to quantify fibrillar structures in raw microscopy images. Nat. Protoc. 9, 457 (2014).
Horzum, U., Ozdil, B. & Pesen-Okvur, D. Step-by-step quantitative analysis of focal adhesions. MethodsX 1, 56–59 (2014).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 29, e45–e45 (2001).
Acknowledgements
This publication has emanated from research conducted with the financial support of Science Foundation Ireland (SFI) and is co-funded under the European Regional Development Fund under Grant Number 13/RC/2073 and Grant Number 14/IA/2884. The authors acknowledge funding from European Research Council (ERC) Starting Grant (336882) and Science Foundation Ireland (SFI) Grants 13/ERC/L2864 and 12/RC/2278. The MLO-Y4 cell line used in this study was received as a kind gift from Professor Lynda Bonewald (School of Dentistry, University of Missouri, Kansas City, MO, U.S.A.). The authors acknowledge the facilities and scientific and technical assistance of the Centre for Microscopy & Imaging at the National University of Ireland Galway (www.imaging.nuigalway.ie). The authors acknowledge the facilities and scientific and technical assistance of the Genomics and Screening Core at the National University of Ireland Galway, a facility that is funded by NUIG and the Irish Government’s Programme for Research in Third Level Institutions, Cycles 4 and 5, National Development Plan 2007–2013.
Author information
Authors and Affiliations
Contributions
I.G. designed and performed experiments, interpreted the data, and wrote the manuscript; D.A.H. designed experiments, interpreted the data, and wrote the manuscript; and L.M.M. designed experiments, interpreted the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Geoghegan, I.P., Hoey, D.A. & McNamara, L.M. Estrogen deficiency impairs integrin αvβ3-mediated mechanosensation by osteocytes and alters osteoclastogenic paracrine signalling. Sci Rep 9, 4654 (2019). https://doi.org/10.1038/s41598-019-41095-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-41095-3
This article is cited by
-
Estrogen withdrawal alters cytoskeletal and primary ciliary dynamics resulting in increased Hedgehog and osteoclastogenic paracrine signalling in osteocytes
Scientific Reports (2021)
-
Mechanotransduction via the coordinated actions of integrins, PI3K signaling and Connexin hemichannels
Bone Research (2021)
-
Osteocytes and Estrogen Deficiency
Current Osteoporosis Reports (2021)
-
Scl-Ab reverts pro-osteoclastogenic signalling and resorption in estrogen deficient osteocytes
BMC Molecular and Cell Biology (2020)
-
A FAK/HDAC5 signaling axis controls osteocyte mechanotransduction
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.