Abstract
Core β-1,2-xylose and α-1,3-fucose are antigenic motifs on schistosome N-glycans, as well as prominent IgE targets on some plant and insect glycoproteins. To map the association of schistosome infection with responses to these motifs, we assessed plasma IgE and IgG reactivity using microarray technology among Ugandans from rural Schistosoma mansoni (Sm)-endemic islands (n = 209), and from proximate urban communities with lower Sm exposure (n = 62). IgE and IgG responses to core β-1,2-xylose and α-1,3-fucose modified N-glycans were higher in rural versus urban participants. Among rural participants, IgE and IgG to core β-1,2-xylose were positively associated with Sm infection and concentration peaks coincided with the infection intensity peak in early adolescence. Responses to core α-1,3-fucose were elevated regardless of Sm infection status and peaked before the infection peak. Among urban participants, Sm infection intensity was predominantly light and positively associated with responses to both motifs. Principal component and hierarchical cluster analysis reduced the data to a set of variables that captured core β-1,2-xylose- and α-1,3-fucose-specific responses, and confirmed associations with Sm and the rural environment. Responses to core β-1,2-xylose and α-1,3-fucose have distinctive relationships with Sm infection and intensity that should further be explored for associations with protective immunity, and cross-reactivity with other exposures.
Similar content being viewed by others
Introduction
Schistosomiasis is second only to malaria as a parasitic cause of human morbidity, with over 230 million infections globally, the majority of which occur in tropical and subtropical sub-Saharan Africa1,2,3. Despite important strides in coverage of anthelminthic treatment, reductions in infection prevalence have only been modest4,5,6, and the long struggle for a vaccine breakthrough continues7. The host immunological response to Schistosoma infection is shaped to a significant extent by schistosome surface-exposed and secreted glycans and glycoproteins. For example, anti-glycan antibody responses dominate the host humoral response to schistosome larvae and eggs8,9,10 and Schistosoma soluble egg antigen (SEA)-mediated Th2-polarisation profoundly relies on glycosylation11,12. In a mouse model for periovular granuloma formation, periodate treatment of SEA-coated beads inhibited their granulomogenic activity13, further demonstrating the functional relevance of glycan-specific responses in Schistosoma-mediated immunity and pathology. A better understanding of the human immune response to the Schistosoma glycome may be beneficial to the current drive towards identification of better Schistosoma diagnostic markers and potent vaccine candidates14,15,16,17,18.
Current insights into the Schistosoma glycome, the most characterised among parasites, have been particularly aided by mass spectrometry-based (MS) studies19,20,21. Analysis of asparagine (N)-linked glycans expressed by schistosomes reveals two standout, non-mammalian substitutions22,23 on the trimannosyl-chitobiose core (Man3GlcNAc2, conserved in all eukaryotes): an α-1,3-fucose (α3Fuc) linked to the asparagine-linked N-acetylglucosamine (GlcNAc) of the chitobiose component and a β-1,2-xylose (β2Xyl) linked to the β-mannose of the trimannosyl component24 (Fig. 1). These substitutions are also found on nematode glycans from Haemonchus contortus and Caenorhabditis elegans25,26,27,28, and on invertebrate29,30 and plant glycans31,32,33, but have so far not been detected on glycans from other helminths prevalent in the tropics19. Detailed MS studies have neither detected core β2Xyl nor core α3Fuc modified N-glycans in adult schistosome worms but both are present in miracidia and eggs, while cercariae express core β2Xyl but no α3Fuc on the core GlcNAc19. Other common alterations to the schistosome Man3GlcNAc2 core include addition of antennae composed of GalNAcβ1-4GlcNAc (LacdiNAc, LDN), GalNAcβ1-4(Fucα1-3)GlcNAc (fucosylated LacdiNAc, LDN-F) and Galβ1-4(Fucα1-3)GlcNAc (Lewis X, LeX) units. These antennary modifications are expressed in schistosomes (at all developmental stages, albeit with varying surface expression patterns)34 but are rare in mammals35, and occur variably in other helminth species19.
Non-mammalian carbohydrate substitutions on the N-glycan core. Non-mammalian monosaccharide substitutions are denoted by blue brackets. ±implies that motifs in brackets are present or absent in different species. Figure drawn using GlycoWorkbench software, version 2.1 (European Carbohydrates Database Project).
Core β2Xyl and α3Fuc modified schistosome egg N-glycoproteins induce potent Th2-type cellular responses36. In plants, core β2Xyl and α3Fuc may be the most common N-glycan epitopes targeted by human IgE37,38. It is plausible that N-glycan core substitutions play a major role in the glycan-dependent host response to chronic schistosomiasis. For example, most N-glycans on the SEA-derived glycoprotein omega-1 carry core α3Fuc motifs in combination with terminal LeX units39. Omega-1 drives both immunoregulatory40 and Th2 responses41, the latter in a glycan-dependent manner12. Kappa-5, another major component of the Th2-polarising SEA42, expresses glycans modified with both core β2Xyl and core α3Fuc43. Whether protective immunity against Schistosoma infection and reinfection (long associated with host IgE responses44,45) can be credited to these epitopes will require further investigations in animal and human studies.
The advent of glycan microarray technology enabled serum/plasma profiling of antibodies raised to a wide repertoire of N-glycan variants during schistosome infections. This technology has been employed in a small number of human studies. Recently, in Ghana, sera from a few S. haematobium infected schoolchildren showed elevated IgE responses to core β2Xyl modified N-glycans on a synthetic glycan microarray46, and in sera from a small cohort of Schistosoma mansoni (Sm)-infected children and adults near Lake Albert, Uganda, IgG1-4 subclass responses to core β2Xyl and α3Fuc motifs were examined using the same array47. Two other human studies employing shotgun microarrays constructed of complex native schistosome N-glycans showed strong anti-glycan IgG and IgM responses against a wider range of N-glycans during schistosome infections48,49. A better understanding of population-level immune responses to Schistosoma glycans is important for research and clinical applications, and requires larger, well-defined immuno-epidemiological studies in endemic settings.
Fishing villages in the Lake Victoria islands of Koome, Uganda, have a high prevalence of Sm50,51,52,53, and have been surveyed as part of a portfolio of studies on helminth infections and allergy-related outcomes in Uganda. This setting provided a unique opportunity, within the context of a well-characterised large study50, to correlate epidemiological trends pertaining to Sm infection (and intensity) with microarray-detected plasma IgE and IgG responses to N-glycans with and without core α-1,3-fucosylation and/or β-1,2-xylosylation. Plasma from residents of nearby mainland urban communities with lower Sm exposure enabled us to make rural-urban comparisons of anti-glycan antibody responses.
Methods
Study design and population
Individuals included in the current investigation were randomly selected using a Stata program (StataCorp, College Station, USA) from participants of two cross-sectional surveys in rural and urban Uganda, who had a sufficient volume of stored plasma. The rural survey was the outcome survey (year three, September 2015–August 2016) of the Lake Victoria Island Intervention Study on Worms and Allergy-related diseases (LaVIISWA; ISRCTN47196031)50, a cluster-randomised trial of community-based standard versus intensive anthelminthic intervention in 26 Sm-endemic fishing villages of Koome islands (Lake Victoria, Uganda). The trial description50,53 and survey results after three years of intervention52 have been published: briefly, standard intervention included annual, community-based, mass drug administration of praziquantel; intensive intervention included quarterly praziquantel. The urban survey (September 2016–September 2017) was conducted in the 24 sub-wards of Entebbe municipality, an area with lower helminth exposure, located on the northern shores of Lake Victoria (approximately 35 km from Koome). It was designed to collect data from an urban setting for comparison with the Sm-endemic rural survey.
In both surveys, intestinal helminth infections were assessed using the Kato-Katz (KK) method54 on a single stool sample (prepared on two slides, read by different technicians). The remaining sample was stored at −80 °C and later investigated for Sm, Necator americanus and Strongyloides stercoralis infections using multiplex real-time PCR55,56. Mid-stream urine was also assessed for Sm circulating cathodic antigen (CCA) using a point-of-care test (Rapid Medical Diagnostics, Pretoria, South Africa). Schistosoma haematobium is not present in the surveyed areas57. Blood samples were processed to obtain plasma for immunological measurements, including N-glycan-specific IgE and IgG by microarray (detailed below) and Schistosoma egg [SEA]- and adult worm [SWA] antigen-specific IgE, IgG4 and IgG by ELISA (Supplementary Material).
The research ethics committees of the Uganda Virus Research Institute and the London School of Hygiene and Tropical Medicine, and the Uganda National Council for Science and Technology approved this work. All methods were performed in accordance with guidelines and regulations of these committees. Informed consent was obtained from all participants and/or their legal guardians and assent from children aged ≥8 years.
Microarray detection of N-glycan-specific IgE and IgG
Immunoglobulin E and G responses to 135 chemically synthesised glycans with and without core α-1,3-fucosylation and, or, β-1,2-xylosylation (Supplementary Fig. S1) were assessed using a non-commercial microarray. Fluorescently-labeled bovine serum albumin (BSA) was included as an array printing control. Microarray construction procedures have been described in detail elsewhere48,58. The glycan antibody binding assay was adapted from existing procedures17,46,49,59, as follows: Nexterion H N-hydroxysuccinimide-coated microarray slides (Schott AG, Mainz, Germany) (pre-blocked with 50 mM ethanolamine in 50 mM sodium borate buffer pH 9.0, and stored at −20 °C) were thawed at room temperature (RT) and covered with silicone gaskets to create seven wells with printed microarrays per slide. Each microarray was incubated with 300 μl of a 1:30 plasma dilution in 1% BSA - 0.01% Tween20 for one hour at RT while shaking. After sequential washes with PBS-0.05% Tween20 and PBS, the slides were incubated for 30 minutes at RT in the dark with PromoFluor 647-labelled anti-human IgE (diluted 1/150 in PBS-0.01% Tween20) and Cy3-labelled anti-human IgG (diluted 1/1000 in PBS-0.01% Tween20), while shaking. After a final wash with PBS-0.05% Tween20, PBS and deionised water, sequentially, the slides were dried and kept in the dark until scanning. The slides were scanned for fluorescence at a 10μm resolution with a G2565BA scanner (Agilent Technologies, CA, USA) using 633 nm and 532 nm lasers for detection of reactivity to glycan-specific IgE and IgG, respectively.
Data analysis
Using GenePix Pro 7.0 software (Molecular Devices, CA, USA), a spot-finding algorithm was used to align and re-size fluorescence spots in the microarray images, without setting a composite pixel intensity threshold. Data on median fluorescence intensity (MFI) for each spot and the local background were then exported to Microsoft Excel software, where background MFI subtraction was done for each glycan structure, averaged over four spots. Further processing of IgG and IgE MFIs in Excel was done as described by Oyelaran et al.60 and Amoah et al.46, respectively, to yield log2-transformed values.
Graphical representations of antibody responses and further data analyses were done using Stata 13.1 (College Station, Texas, USA), R (R foundation for Statistical Computing, Vienna, Austria) via the RStudio interface (version 1.1.383, RStudio, Inc. Boston, USA) and GraphPad Prism (version 6.0e, Fay Avenue, La Jolla, CA, USA). Schistosoma mansoni infection and the rural-urban environment were the main exposures of interest: we compared anti-glycan antibody responses between Sm infected and uninfected participants separately in the rural and urban survey, and thereafter between rural and urban participants. Initial analyses considered each anti-glycan antibody response independently, while further analyses combined antibody responses to reduce the dimensionality of the outcome data, as detailed below.
Rural-urban differences in Sm prevalence and Schistosoma-specific antibodies were assessed using survey design-based logistic and linear regression, respectively. Most log2-transformed anti-glycan IgE responses maintained a skewed distribution. Therefore, Mann-Whitney tests were used to assess differences in individual glycan structure-specific antibody responses between Sm infected and uninfected participants and between rural and urban participants. Most log2-transformed anti-glycan IgG responses were normally distributed and were assessed using unpaired t tests. The Kruskal-Wallis (IgE responses) and one-way ANOVA test (IgG responses) were also conducted to assess differences along the infection intensity gradient. Since many of the anti-glycan antibody responses were correlated, the above tests were conducted within a Monte Carlo simulation approach based on 1000 permutations, to generate empirical p-values corrected for multiple testing.
Given the large number of outcomes, two data reduction techniques were used to investigate associations between exposures and outcomes. First, principal component analysis (PCA) was run in Stata to transform groups of correlated anti-glycan responses into fewer, uncorrelated artificial variables (principal components, PCs), which were then compared by 1) survey setting, and 2) Sm infection and intensity status using survey design-based linear regression. Second, unsupervised hierarchical clustering analysis (HCA, complete linkage using Euclidean distance) was conducted in R to further identify homogeneous sets of N-glycan-specific responses. The resultant IgE and IgG clusters were then assessed for associations with survey setting and Sm infection using the global test61,62,63 executed in R with the Globaltest package (version 5.33.0).
Results
Characteristics of the rural and urban survey participants included in this analysis are presented in Table 1. Rural participants were, on average, older [median age (IQR) 22 (5, 37)] than urban participants [median age (IQR) 11 (5, 18)] (p < 0.001). A significantly higher percentage of rural, compared to urban participants, were infected with Sm (KK, p = 0.002; PCR, p < 0.001; CCA, p = 0.015). Furthermore, median levels of total IgE (p < 0.001) and SEA- and SWA-specific IgE (p < 0.001), IgG4 (p = 0.001) and IgG (p = 0.002 and p < 0.001, respectively) were higher among rural compared to urban participants.
We recently reported that community-based intensive versus standard anthelminthic intervention in the rural survey reduced Sm infection intensity but had no effect on the overall Sm prevalence (measured using the urine CCA test)52. The current analysis found no evidence of an effect of intensive versus standard treatment on total IgE, SEA- or SWA-specific antibodies, or on antibody reactivity to any of the N-glycans on the microarray. Therefore, data from the rural survey were not stratified by trial treatment arm in the further analyses presented herein.
Associations between S. mansoni infection and IgE and IgG responses to individual core β-1,2-xylosylated and core α-1,3-fucosylated N-glycans
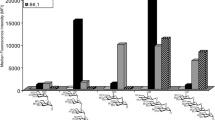
In the rural survey, IgE and IgG responses to the β2Xyl modified Man3GlcNAc2 core (G34) were significantly higher among Sm infected (KK and/or PCR, and CCA positive), compared to uninfected individuals, and were positively associated with Sm infection intensity (KK) [Fig. 2a–f] and SWA- and SEA-specific IgE and IgG (Supplementary Table S1). Observations were similar for the N-glycan core carrying both β2Xyl and α3Fuc (G37). However, IgE and IgG responses to the N-glycan core carrying α3Fuc only (G73) were similar between Sm infected and uninfected rural individuals (Fig. 2a–f), but positively associated with SWA- and SEA-specific IgE and IgG (Table S1).
Associations between S. mansoni infection and IgE and IgG reactivity to N-glycans carrying non-mammalian core modifications. Plasma from S. mansoni infected and uninfected rural and urban individuals were assessed for IgE and IgG reactivity to N-glycan structural variants with and without α-1,3-fucosylation and β-1,2-xylosylation, on a microarray platform. Box-and-whisker plots show background-subtracted and log2-transformed median fluorescence intensities (MFI) representing IgE (a–c,g,h) and IgG (d–f,i,j) reactivity to the Man3GlcNAc2 core structure (G42) and to α3Fuc- and/or β2Xyl-carrying Man3GlcNAc2 core structures (G34, G73 and G37). The plots show a horizontal line denoting the median, a box indicating the interquartile range (IQR), and whiskers drawn using the Tukey method (1.5 times IQR). Outliers (greater than 1.5 times IQR away from the median) are plotted as individual points. Mann-Whitney (IgE responses) and unpaired t test (IgG responses) were conducted within the framework of a Monte Carlo simulation algorithm based on 1000 permutations (in order to adjust for multiple testing), to assess differences between infected and uninfected individuals. The Kruskal-Wallis (IgE responses) and one-way ANOVA test (IgG responses) were also conducted using the permutation approach to assess differences along the infection intensity gradient (b and e) in the rural survey. #Infection prevalence and intensity was relatively low in the urban survey so analysis by Sm intensity is not shown for the urban setting. *p < 0.05; **p < 0.01; ***p < 0.001. Sm: S. mansoni infection determined by detection of eggs in a single stool sample by Kato-Katz and/or PCR (rural infected n = 84, uninfected n = 113; urban infected n = 6, uninfected n = 42); CCA: S. mansoni infection determined by a positive urine circulating cathodic antigen (CCA) result (rural infected n = 118, uninfected n = 81; urban infected n = 21, uninfected n = 37).
In the urban survey, IgE and IgG reactivity to core β2Xyl and/or core α3Fuc modified glycans was also higher in Sm infected (KK and/or PCR) compared to uninfected individuals, although differences were not statistically significant (Fig. 2g–j). However, IgE reactivity to core α3Fuc and core α3Fuc + core β2Xyl modified glycans was significantly positively associated with SEA- and SWA-specific IgE (Table S1).
In the rural survey, Sm infection prevalence (and intensity) and median levels of Schistosoma-specific antibodies (except SEA-IgE) and β-1,2-xylosylated glycan (G34)-specific IgE and IgG were highest among 10-14-year old individuals (Fig. 3). However, IgE and IgG reactivity to glycans carrying either core α3Fuc (G73) or both core β2Xyl and core α3Fuc (G37) peaked earlier (in the 5-9-year age group), akin to SEA-specific IgE. Age-stratified antibody reactivity patterns were less clear in the urban survey.
Age-stratified prevalence of S. mansoni infection and median IgE and IgG reactivity to SWA, SEA and α-1,3-fucosylated and β-1,2-xylosylated N-glycans. (a,e) Rural and urban prevalence and intensity of S. mansoni infection, by age group. (b,f) Median IgG, IgG4 and IgE reactivity to SWA and SEA, by age group, among rural and urban participants, respectively. (c,g) Median IgE reactivity to the Man3GlcNAc2 core and to α3Fuc- and/or β2Xyl modified Man3GlcNAc2 core structures, by age group, among rural and urban participants, respectively. Plotted results are from all participants, irrespective of Sm infection status. (d,h) Median IgG reactivity to the Man3GlcNAc2 core and to α3Fuc- and/or β2Xyl-modified Man3GlcNAc2 core structures, by age group, among rural and urban participants, respectively. Plotted results are from all participants, irrespective of Sm infection status. CCA: S. mansoni infection determined by a positive urine circulating cathodic antigen (CCA) result; KK: S. mansoni infection determined by detection of eggs in a single stool sample by Kato-Katz (KK); epg: Eggs per gram of stool; SWA: Schistosoma adult worm antigen; SEA: Schistosoma egg antigen.
Immunoglobulin E and G responses to other N-glycan structural variants with core β2Xyl or both core β2Xyl and core α3Fuc48 were also higher in Sm infected versus uninfected individuals (Fig. S2). IgE and IgG reactivity to non-xylosylated and non-fucosylated glycans was not associated with Sm infection (data not shown), except for those glycans with antennae constructed of LDN-F (G90) and LeX (G89) units (Fig. S3).
Infection with other helminths, malaria or HIV was not associated with IgE or IgG reactivity to any glycans on the microarray (data not shown).
Principal component analysis of anti-glycan antibody responses
Antibody responses to individual core modified N-glycans were strongly correlated. Principal component analysis (PCA) was conducted to summarise these responses, and to evaluate to what extent the resultant principal components (PCs) were associated with Sm infection.
Scatterplots of PC1 and PC2 loadings are shown in Fig. 4. In the rural survey, the first two IgE and IgG PCs each accounted for 37% of the total variance in the data (IgE: PC1 28.2%, PC2 8.8%; IgG: PC1 27.7%, PC2 9.7%). Principal component 1 was characterized by responses to core β2Xyl and/or α3Fuc modified glycans while PC2 was characterized by responses to non-xylosylated and non-fucosylated glycans (Fig. 4, panel a and b). Scores for IgE PC1, but not PC2, were higher among Sm infected (KK or PCR) compared to uninfected individuals (crude p = 0.028, age- and sex-adjusted p = 0.167). Similarly, IgG PC1 scores were higher among Sm infected (KK or PCR) compared to uninfected individuals (crude p = 0.009, adjusted p = 0.027). There were no differences in PC scores between CCA+ and CCA− individuals.
Principal component analysis of anti-glycan antibody responses. Scatterplots of first (PC1) and second factor (PC2) loadings derived from principal component analysis of IgE and IgG responses to 135 synthetic N-glycans. Box-and-whisker plots show comparison of PC1 and PC2 scores between S. mansoni infected and uninfected individuals. The plots show a horizontal line denoting the median, a box indicating the interquartile range (IQR), and whiskers drawn using the Tukey method (1.5 times IQR). Outliers (greater than 1.5 times IQR away from the median) are plotted as individual points. Panels a and b show IgE and IgG profiles, respectively, among rural participants. Panels c and d show IgE and IgG profiles, respectively, among urban participants. Associations between factor loading scores and S. mansoni infection and intensity were assessed by linear regression analysis in Stata 13.1. Crude and age- and sex-adjusted p values are shown for significant associations. All analyses were adjusted for survey design using the ‘svy’ command in Stata. PC1: Principal Component 1; PC2: Principal Component 2; Sm: S. mansoni infection determined by detection of eggs in a single stool sample by Kato-Katz and/or PCR (rural infected n = 84, uninfected n = 113; urban infected n = 6, uninfected n = 42); CCA: S. mansoni infection determined by a positive urine circulating cathodic antigen (CCA) result (rural infected n = 118, uninfected n = 81; urban infected n = 21, uninfected n = 37).
In the urban survey, the first two IgE and IgG PCs accounted for 31% and 35% of the total variance, respectively (IgE: PC1 19.4%, PC2 11.5%; IgG: PC1 24.2%, PC2 10.6%). Interestingly, most IgE responses to glycans carrying core β2Xyl without α3Fuc clustered with non-xylosylated and non-fucosylated glycans in PC2 while responses to glycans carrying both core β2Xyl and α3Fuc and those carrying core α3Fuc without β2Xyl clustered together in PC1 (Fig. 4, panel c). Akin to the rural survey, scores for IgE and IgG PC1 were higher among Sm infected compared to uninfected urban individuals.
Scores for PC1 were positively associated with SWA- and SEA-specific IgE and IgG in both surveys, while PC2 scores were inversely associated with the same Schistosoma-specific antibodies (Table S1).
In addition to PCA, we conducted HCA to further identify groups of anti-glycan IgE and IgG responses that might be jointly elicited in Sm infected versus uninfected individuals. Figure S4 shows clusters of IgE and IgG responses in the rural and urban survey, and the dominant core substitutions on the glycans in these clusters. Generally, antibody clusters comprising core β2Xyl modified glycans were positively associated with Sm infection and intensity in both surveys (Table S2).
Rural-urban comparisons of anti-glycan antibody responses
Immunoglobulin E responses to individual core β2Xyl and/or α3Fuc modified glycans were higher among rural compared to urban participants, as exemplified in Fig. 5a. Principal component analysis of data combined from both surveys yielded distinct groups of anti-glycan responses (Fig. 5b,f): PC1 was characterized by responses to core β2Xyl and/or α3Fuc modified glycans while PC2 was characterized by responses to non-xylosylated and non-fucosylated glycans. Scores for IgE PC1 (Fig. 5c), but not PC2 (Fig. 5d), were higher among rural compared to urban individuals (p = 0.002). Differences in IgG PC1 scores were not statistically significant. However, IgG PC2 scores were lower among rural compared to urban individuals (p = 0.013).
Rural-urban comparisons of anti-glycan antibody responses. (a,e) Box-and-whisker plots showing background-subtracted and log2-transformed median fluorescence intensities (MFI) representing IgE and IgG reactivity to individual α3Fuc- and/or β2Xyl-carrying Man3GlcNAc2 core structures in rural versus urban participants. The plots show a horizontal line denoting the median, a box indicating the interquartile range (IQR), and whiskers drawn using the Tukey method (1.5 times IQR). Outliers (greater than 1.5 times IQR away from the median) are plotted as individual points. Mann-Whitney (IgE responses) and unpaired t test (IgG responses) were conducted within the framework of a Monte Carlo simulation algorithm based on 1000 permutations, to assess differences between rural and urban individuals. (b,f) First and second principal component loadings of N-glycan-specific IgE and IgG responses among all participants, irrespective of survey setting. (c,d,g,h) Box-and-whisker plots showing comparison of PC1 and PC2 scores between rural and urban individuals. Associations between PC scores and survey setting were assessed by linear regression in Stata 13.1. Crude and age- and sex-adjusted p values are shown for significant associations. All analyses were adjusted for survey design using the ‘svy’ command in Stata. *p < 0.05; **p < 0.01; ***p < 0.001. PC1: Principal Component 1; PC2: Principal Component 2.
Further assessment by HCA showed that clusters that comprised IgE responses to core β2Xyl and/or α3Fuc modified glycans (IgE-C1, IgE-C2 and IgE-C4; Fig. S5) were positively associated with the rural setting (Table S4), while IgE-C3 (characterised by very low responses, raised against non-xylosylated and non-fucosylated glycans) was positively associated with the urban setting. Immunoglobulin G response clusters were generally similar between rural and urban settings, except for IgG-C7 which comprised responses to non-xylosylated and non-fucosylated glycans and was positively associated with the urban setting.
Discussion
By studying rural Sm-endemic Ugandan fishing communities and a proximate urban community, we have dissected antibody responses to core β2Xyl and α3Fuc modified N-glycans. Antibody responses to the core modified glycans were higher in the rural communities compared to a proximate urban community. In the urban community, IgE and IgG to both core β2Xyl and core α3Fuc were positively associated with Sm infection. In the rural communities, IgE and IgG to core β2Xyl were strongly positively associated with Sm infection while reactivity to core α3Fuc was elevated in both Sm infected and uninfected individuals. In the rural communities the concentration of antibodies to core α3Fuc modified N-glycans peaked ahead of the peak of Sm infection intensity, while the peak of antibodies to N-glycans with only core β2Xyl coincided with it.
The positive association between current Sm infection and IgE and IgG reactivity to N-glycans carrying only core α3Fuc in the urban, but not the rural communities, might reflect universal exposure to infection, and persistence of light infection despite treatment, in the rural setting. Core α3Fuc is abundant on N-glycans from Sm eggs but is not expressed by cercarial and adult worm N-glycans19,20,24. It is plausible that responses to core α3Fuc persist after active infection in high Sm exposure rural settings: in mice, eggs and hepatic granulomas persist long after clearance of worms64. Another explanation for elevated responses to α3Fuc in the rural communities, regardless of Sm infection status, is cross-reactivity. Core α-1,3-fucosylation and β-1,2-xylosylation are also present on certain plant and insect glycoproteins29,30,65, hence similar core α3Fuc responses in both Sm infected and uninfected individuals may also be explained by an exposure other than schistosomes, more prevalent in the rural than the urban setting, that carries core α3Fuc. The observation that antibodies to core β2Xyl were significantly higher among Sm infected individuals in both urban and rural settings implies a dominant role for core β2Xyl (compared to core α3Fuc) in Sm-specific humoral immunity, shown here for the first time. It also appears that responses only to core β2Xyl are more responsive to change in Sm exposure: core β2Xyl is abundant on cercarial N-glycans despite being absent in adult worms.
The prominent contribution of core β2Xyl and α3Fuc to cross-reactivity between schistosomes and other environmental exposures such as pollen, hymenoptera venom and vegetable foods22,37,38 is a caveat against the use of core modified glycans in schistosome diagnostic tests. Cross-reactivity with other helminth infections might also occur, but only a few other helminth species25,26,27,28, none of which are prevalent in humans in our survey settings, have so far been demonstrated to express glycans with core β2Xyl and α3Fuc motifs. More extensive glycomic studies of other helminths in our survey settings (S. stercoralis, hookworm, T. trichiura, A. lumbricoides, M. perstans) are warranted. However, we did not find any significant associations between these infections and IgE or IgG reactivity to core modified glycans.
Our observations that IgE and IgG reactivity to N-glycans modified with antennae carrying LDNF and LeX units were associated with Sm infection in both surveys are consistent with previous studies in animal models and in humans49,66. No associations were observed with responses to glycans carrying unsubstituted LDN units.
Principal component analysis indicated strong correlations between antibody responses to β-1,2-xylosylated glycans and responses to α-1,3-fucosylated glycans in both surveys. Core β2Xyl and α3Fuc epitopes can be found on similar Sm antigens, where they may be expressed on the same glycoproteins and glycans (such as those expressed by SEA)19, inducing analogous immune responses36. Non-xylosylated and non-fucosylated glycans with antennae constructed of LDN, LDN-F or LeX units may be expressed on the same Sm antigens as glycans with core β2Xyl and α3Fuc motifs. Furthermore, these terminal antennary substitutions can occur on the same glycans as core β2Xyl and α3Fuc19,39,43. However, PCA showed that responses to non-core-substituted glycans with LDN, LDN-F or LeX units did not cluster with core β2Xyl/α3Fuc substituted glycans. Temporal changes in expression of glycans on Sm antigens have been reported20,34; it is possible that these two groups of glycans are expressed at varying magnitudes during Sm antigen maturation. Positive associations between Sm infection and the first principal component (representing responses to core β2Xyl and α3Fuc) reflect the important role of these core substitutions in the glycan-dependent host response to Sm. To further evaluate their contribution to the host immune response to Sm, it will be important to compare their antibody reactivity with that of other highly antigenic terminal motifs absent from glycans on the array used in this study, such as multi-fucosylated LDN motifs59. It is important to note that while IgG is abundantly detected to many schistosome glycans, and is triggered by Sm infection, only to core β2Xyl and α3Fuc modified glycans is IgE abundantly detected46,47,67.
Notably in the urban survey, PCA showed that IgE responses to core β2Xyl modified glycans clustered with responses to non-xylosylated and non-fucosylated glycans (Fig. 4c), and IgG responses to core β2Xyl and α3Fuc separated out less distinctly (Fig. 4d) than in the rural survey. The observed β2Xyl clustering patterns may be attributed to the greater intensity of repeated exposure to schistosome cercariae (core β2Xyl) and sustained egg deposition (core β2Xyl and α3Fuc) among rural compared to urban participants. In other words, rural-urban differences in antibody responses to core modified glycans may be indicative of differences in the intensity of Sm infection and/or degree of exposure between the two settings. However, this study did not have sufficient power to assess statistical interactions between the rural and the urban setting. Rural-urban differences in antibody responses to core modified glycans may also be explained by exposures other than schistosomes (mentioned above), perhaps more prevalent in the rural than the urban setting; however, this is unlikely as we observed strong associations between Sm infection and reactivity to core modified N-glycans, particularly to those carrying core β2Xyl. It is also noteworthy that urban survey participants were significantly younger than rural participants; however, this disparity did not seem to influence the observed rural-urban differences in anti-glycan responses, as observed from test statistics before and after adjusting for age.
One of the key challenges in schistosomiasis vaccine development is the risk of allergic (IgE) sensitisation to candidate vaccine antigens68. Glycans are attractive vaccine candidates because they are generally considered to be benign as allergenic determinants69,70. There are a few known exceptions, such as the galactose-α-1,3-galactose (α-1,3-gal) epitope (found in non-primate mammalian proteins, and shown to elicit severe allergy)71, so assessment for any associations between IgE to antigenic Sm glycans and allergy-related phenomena are important. The case for consideration of core modified Sm glycans as Schistosoma vaccine candidates will also need definite proof for an association between reactivity to core modified glycans and protection from Sm infection/re-infection. Our data suggests that a protective role, if any, is more plausible for core β2Xyl than core α3Fuc: in the rural survey, antibody responses to core α3Fuc (G73 and G37, Fig. 3c,d) peaked in childhood, prior to the Sm infection peak in early adolescence, while responses to core β2Xyl (G34) coincided with the Sm infection peak (preceding the more ‘protected’ period in adulthood). However, concrete evidence is required from further population and mechanistic studies exploring the role of Sm N-glycans in protective immunity. For example, it may be important to assess antibodies to these core modifications (and other antigenic terminal motifs) in re-infection study cohorts evaluating the immunological characteristics of individuals who are Sm-resistant following anthelminthic treatment.
In conclusion, we provide an immuno-epidemiological description of IgE and IgG responses to N-glycans in rural and urban Uganda, highlighting the significance of core β2Xyl and core α3Fuc to the glycan-dependent host immune response during chronic schistosomiasis. Moreover, our data imply that IgE and IgG responses to core β2Xyl and α3Fuc modified N-glycans have distinctive relationships with Sm infection and intensity, which may reflect their different contributions towards protective immunity against Sm that need to be further explored using mechanistic animal and human studies.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Colley, D. G., Bustinduy, A. L., Secor, W. E. & King, C. H. Human schistosomiasis. The Lancet 383, 2253–2264, https://doi.org/10.1016/S0140-6736(13)61949-2 (2014).
Toor, J. et al. Are We on Our Way to Achieving the 2020 Goals for Schistosomiasis Morbidity Control Using Current World Health Organization Guidelines? Clin Infect Dis 66, S245–S252, https://doi.org/10.1093/cid/ciy001 (2018).
Global Health Estimates 2015: Disease burden by Cause, Age, Sex, by Country and by Region, 2000–2015. Geneva, World Health Organization (2016).
Secor, W. E. & Montgomery, S. P. Something old, something new: is praziquantel enough for schistosomiasis control? Future Med Chem 7, 681–684, https://doi.org/10.4155/fmc.15.9 (2015).
King, C. H. The evolving schistosomiasis agenda 2007–2017-Why we are moving beyond morbidity control toward elimination of transmission. PLoS Negl Trop Dis 11, e0005517, https://doi.org/10.1371/journal.pntd.0005517 (2017).
Sokolow, S. H. et al. Global Assessment of Schistosomiasis Control Over the Past Century Shows Targeting the Snail Intermediate Host Works Best. PLoS Negl Trop Dis 10, e0004794, https://doi.org/10.1371/journal.pntd.0004794 (2016).
Tebeje, B. M., Harvie, M., You, H., Loukas, A. & McManus, D. P. Schistosomiasis vaccines: where do we stand? Parasites & Vectors 9, 528, https://doi.org/10.1186/s13071-016-1799-4 (2016).
Omer-Ali, P., Magee, A. I., Kelly, C. & Simpson, A. J. A major role for carbohydrate epitopes preferentially recognized by chronically infected mice in the determination of Schistosoma mansoni schistosomulum surface antigenicity. Journal of immunology (Baltimore, Md.: 1950) 137, 3601–3607 (1986).
Eberl, M. et al. Antibodies to glycans dominate the host response to schistosome larvae and eggs: is their role protective or subversive? J Infect Dis 183, 1238–1247, https://doi.org/10.1086/319691 (2001).
Kariuki, T. M., Farah, I. O., Wilson, R. A. & Coulson, P. S. Antibodies elicited by the secretions from schistosome cercariae and eggs are predominantly against glycan epitopes. Parasite immunology 30, 554–562, https://doi.org/10.1111/j.1365-3024.2008.01054.x (2008).
Okano, M., Satoskar, A. R., Nishizaki, K., Abe, M. & Harn, D. A. Jr. Induction of Th2 responses and IgE is largely due to carbohydrates functioning as adjuvants on Schistosoma mansoni egg antigens. Journal of immunology (Baltimore, Md.: 1950) 163, 6712–6717 (1999).
Everts, B. et al. Schistosome-derived omega-1 drives Th2 polarization by suppressing protein synthesis following internalization by the mannose receptor. The Journal of experimental medicine 209, 1753–1767, s1751, https://doi.org/10.1084/jem.20111381 (2012).
van de Vijver, K. K. et al. Glycans of Schistosoma mansoni and keyhole limpet haemocyanin induce hepatic granulomas in vivo. Int J Parasitol 34, 951–961, https://doi.org/10.1016/j.ijpara.2004.04.009 (2004).
Elliott, A. M. et al. Ethical and scientific considerations on the establishment of a controlled human infection model for schistosomiasis in Uganda: report of a stakeholders’ meeting held in Entebbe, Uganda. [version 1; referees: 2 approved] AAS Open Research, https://doi.org/10.12688/aasopenres.12841.1 (2018).
PATH. Schistosomiasis: The Business Case for New Diagnostics. Seattle: PATH (2016).
Utzinger, J., Becker, S. L., van Lieshout, L., van Dam, G. J. & Knopp, S. New diagnostic tools in schistosomiasis. Clinical Microbiology and Infection 21, 529–542, https://doi.org/10.1016/j.cmi.2015.03.014 (2015).
van Diepen, A. et al. Development of a Schistosoma mansoni shotgun O-glycan microarray and application to the discovery of new antigenic schistosome glycan motifs. Int J Parasitol 45, 465–475, https://doi.org/10.1016/j.ijpara.2015.02.008 (2015).
Nyame, A. K., Kawar, Z. S. & Cummings, R. D. Antigenic glycans in parasitic infections: implications for vaccines and diagnostics. Archives of biochemistry and biophysics 426, 182–200, https://doi.org/10.1016/j.abb.2004.04.004 (2004).
Hokke, C. H. & van Diepen, A. Helminth glycomics – glycan repertoires and host-parasite interactions. Molecular and Biochemical Parasitology 215, 47–57, https://doi.org/10.1016/j.molbiopara.2016.12.001 (2017).
Smit, C. H. et al. Glycomic Analysis of Life Stages of the Human Parasite Schistosoma mansoni Reveals Developmental Expression Profiles of Functional and Antigenic Glycan Motifs. Molecular & cellular proteomics: MCP 14, 1750–1769, https://doi.org/10.1074/mcp.M115.048280 (2015).
Haslam, S. M., Morris, H. R. & Dell, A. Mass spectrometric strategies: providing structural clues for helminth glycoproteins. Trends in parasitology 17, 231–235 (2001).
Altmann, F. The role of protein glycosylation in allergy. Int Arch Allergy Immunol 142, 99–115, https://doi.org/10.1159/000096114 (2007).
Commins, S. P. Carbohydrates as allergens. Current allergy and asthma reports 15, 492, https://doi.org/10.1007/s11882-014-0492-y (2015).
Khoo, K.-H., Chatterjee, D., Caulfield, J. P., Morris, H. R. & Dell, A. Structural mapping of the glycans from the egg glycoproteins of Schistosoma mansoni and Schistosoma japonicum: identification of novel core structures and terminal sequences. Glycobiology 7, 663–677, https://doi.org/10.1093/glycob/7.5.663 (1997).
van Die, I. et al. Core alpha1– > 3-fucose is a common modification of N-glycans in parasitic helminths and constitutes an important epitope for IgE from Haemonchus contortus infected sheep. FEBS letters 463, 189–193 (1999).
Yan, S. et al. Core Richness of N-Glycans of Caenorhabditis elegans: A Case Study on Chemical and Enzymatic Release. Analytical chemistry 90, 928–935, https://doi.org/10.1021/acs.analchem.7b03898 (2018).
Haslam, S. M., Coles, G. C., Reason, A. J., Morris, H. R. & Dell, A. The novel core fucosylation of Haemonchus contortus N-glycans is stage specific. Molecular and Biochemical Parasitology 93, 143–147, https://doi.org/10.1016/S0166-6851(98)00020-6 (1998).
Haslam, S. M. et al. Haemonchus contortus glycoproteins contain N-linked oligosaccharides with novel highly fucosylated core structures. The Journal of biological chemistry 271, 30561–30570 (1996).
Aalberse, R. C., Koshte, V. & Clemens, J. G. J. Immunoglobulin E antibodies that crossreact with vegetable foods, pollen, and Hymenoptera venom. Journal of Allergy and Clinical Immunology 68, 356–364, https://doi.org/10.1016/0091-6749(81)90133-0 (1981).
Fabini, G., Freilinger, A., Altmann, F. & Wilson, I. B. H. Identification of Core α1,3-Fucosylated Glycans and Cloning of the Requisite Fucosyltransferase cDNA from Drosophila melanogaster: Potential Basis of the Neural Anti-Horseradish Peroxidase Epitope. Journal of Biological Chemistry 276, 28058–28067, https://doi.org/10.1074/jbc.M100573200 (2001).
Fötisch, K., Altmann, F., Haustein, D. & Vieths, S. Involvement of Carbohydrate Epitopes in the IgE Response of Celery–Allergic Patients. International Archives of Allergy and Immunology 120, 30–42 (1999).
Fournet, B. et al. Primary structure of an N-glycosidic carbohydrate unit derived from Sophora japonica lectin. European journal of biochemistry 166, 321–324 (1987).
Ramirez-Soto, D. & Poretz, R. D. The (1—3)-linked alpha-L-fucosyl group of the N-glycans of the Wistaria floribunda lectins is recognized by a rabbit anti-serum. Carbohydrate research 213, 27–36 (1991).
Smit, C. H. et al. Surface expression patterns of defined glycan antigens change during Schistosoma mansoni cercarial transformation and development of schistosomula. Glycobiology 25, 1465–1479, https://doi.org/10.1093/glycob/cwv066 (2015).
Wuhrer, M., Koeleman, C. A., Deelder, A. M. & Hokke, C. H. Repeats of LacdiNAc and fucosylated LacdiNAc on N-glycans of the human parasite Schistosoma mansoni. The FEBS journal 273, 347–361, https://doi.org/10.1111/j.1742-4658.2005.05068.x (2006).
Faveeuw, C. et al. Schistosome N-glycans containing core alpha 3-fucose and core beta 2-xylose epitopes are strong inducers of Th2 responses in mice. European journal of immunology 33, 1271–1281, https://doi.org/10.1002/eji.200323717 (2003).
Wilson, I. B. & Altmann, F. Structural analysis of N-glycans from allergenic grass, ragweed and tree pollens: corealpha1,3-linked fucose and xylose present in all pollens examined. Glycoconj J 15, 1055–1070 (1998).
Wilson, I. B. H., Harthill, J. E., Mullin, N. P., Ashford, D. A. & Altmann, F. Core α1,3-fucose is a key part of the epitope recognized by antibodies reacting against plant N-linked oligosaccharides and is present in a wide variety of plant extracts. Glycobiology 8, 651–661, https://doi.org/10.1093/glycob/8.7.651 (1998).
Meevissen, M. H. et al. Structural characterization of glycans on omega-1, a major Schistosoma mansoni egg glycoprotein that drives Th2 responses. Journal of proteome research 9, 2630–2642, https://doi.org/10.1021/pr100081c (2010).
Zaccone, P. et al. The S. mansoni glycoprotein omega-1 induces Foxp3 expression in NOD mouse CD4(+) T cells. Eur. J. Immunol. 41, 2709–2718, https://doi.org/10.1002/eji.201141429 (2011).
Everts, B. et al. Omega-1, a glycoprotein secreted by Schistosoma mansoni eggs, drives Th2 responses. The Journal of experimental medicine 206, 1673–1680, https://doi.org/10.1084/jem.20082460 (2009).
Schramm, G. et al. Molecular characterisation of kappa-5, a major antigenic glycoprotein from Schistosoma mansoni eggs. Mol Biochem Parasitol 166, 4–14, https://doi.org/10.1016/j.molbiopara.2009.02.003 (2009).
Meevissen, M. H. et al. Targeted glycoproteomic analysis reveals that kappa-5 is a major, uniquely glycosylated component of Schistosoma mansoni egg antigens. Molecular & cellular proteomics: MCP 10, M110 005710, https://doi.org/10.1074/mcp.M110.005710 (2011).
Dunne, D. W., Butterworth, A. E., Fulford, A. J., Ouma, J. H. & Sturrock, R. F. Human IgE responses to Schistosoma mansoni and resistance to reinfection. Memorias do Instituto Oswaldo Cruz 87(Suppl 4), 99–103 (1992).
Demeure, C. E. et al. Resistance to Schistosoma mansoni in Humans: Influence of the IgE/IgG4 Balance and IgG2 in Immunity to Reinfection after Chemotherapy. The Journal of Infectious Diseases 168, 1000–1008 (1993).
Amoah, A. S. et al. Identification of dominant anti-glycan IgE responses in school children by glycan microarray. The Journal of allergy and clinical immunology 141, 1130–1133, https://doi.org/10.1016/j.jaci.2017.09.040 (2018).
Yang, Y. Y., van Diepen, A., Brzezicka, K., Reichardt, N. C. & Hokke, C. H. Glycan microarray-assisted identification of IgG subclass targets in schistosomiasis. Frontiers in Immunology, https://doi.org/10.3389/fimmu.2018.02331 (2018).
Brzezicka, K. et al. Synthesis and microarray-assisted binding studies of core xylose and fucose containing N-glycans. ACS Chem Biol 10, 1290–1302, https://doi.org/10.1021/cb501023u (2015).
van Diepen, A. et al. Differential anti-glycan antibody responses in Schistosoma mansoni-infected children and adults studied by shotgun glycan microarray. PLoS Negl Trop Dis 6, e1922, https://doi.org/10.1371/journal.pntd.0001922 (2012).
Nampijja, M. et al. The Lake Victoria island intervention study on worms and allergy-related diseases (LaVIISWA): study protocol for a randomised controlled trial. Trials 16, 187, https://doi.org/10.1186/s13063-015-0702-5 (2015).
Nkurunungi, G. et al. Schistosoma mansoni-specific immune responses and allergy in Uganda. Parasite immunology 40, https://doi.org/10.1111/pim.12506 (2018).
Sanya, R. E. et al. The Impact of Intensive Versus Standard Anthelminthic Treatment on Allergy-related Outcomes, Helminth Infection Intensity, and Helminth-related Morbidity in Lake Victoria Fishing Communities, Uganda: Results From the LaVIISWA Cluster-randomized Trial. Clinical Infectious Diseases, ciy761–ciy761, https://doi.org/10.1093/cid/ciy761 (2018).
Webb, E. L. et al. Helminths are positively associated with atopy and wheeze in Ugandan fishing communities: results from a cross-sectional survey. Allergy 71, 1156–1169, https://doi.org/10.1111/all.12867 (2016).
Katz, N., Chaves, A. & Pellegrino, J. A simple device for quantitative stool thick-smear technique in Schistosomiasis mansoni. Rev Inst Med Trop Sao Paulo 14, 397–400 (1972).
Verweij, J. J. et al. Simultaneous detection and quantification of Ancylostoma duodenale, Necator americanus, and Oesophagostomum bifurcum in fecal samples using multiplex real-time PCR. The American journal of tropical medicine and hygiene 77, 685–690 (2007).
Verweij, J. J. et al. Molecular diagnosis of Strongyloides stercoralis in faecal samples using real-time PCR. Trans R Soc Trop Med Hyg 103, 342–346, https://doi.org/10.1016/j.trstmh.2008.12.001 (2009).
Emmanuel, I. O. A. & Ekkehard, D. Epidemiology, of bilharzias (schistosomiasis) in Uganda from 1902 until 2005. African Health Sciences 8, 239–243 (2008).
Echeverria, B. et al. Chemoenzymatic Synthesis of N-glycan Positional Isomers and Evidence for Branch Selective Binding by Monoclonal Antibodies and Human C-type Lectin Receptors. ACS Chemical Biology 13, 2269–2279, https://doi.org/10.1021/acschembio.8b00431 (2018).
Yang, Y. Y. et al. Specific anti-glycan antibodies are sustained during and after parasite clearance in Schistosoma japonicum-infected rhesus macaques. PLoS Negl Trop Dis 11, e0005339, https://doi.org/10.1371/journal.pntd.0005339 (2017).
Oyelaran, O., McShane, L. M., Dodd, L. & Gildersleeve, J. C. Profiling human serum antibodies with a carbohydrate antigen microarray. Journal of proteome research 8, 4301–4310, https://doi.org/10.1021/pr900515y (2009).
Goeman, J. J. & Oosting, J. Globaltest R package, version 5.33.0. (2016).
Goeman, J. J., van de Geer, S. A., de Kort, F. & van Houwelingen, H. C. A global test for groups of genes: testing association with a clinical outcome. Bioinformatics 20, 93–99 (2004).
Goeman, J. J., Van De Geer, S. A. & Van Houwelingen, H. C. Testing against a high dimensional alternative. Journal of the Royal Statistical Society: Series B 68, 477–493 (2006).
Cheever, A. W., Macedonia, J. G., Deb, S., Cheever, E. A. & Mosimann, J. E. Persistence of eggs and hepatic fibrosis after treatment of Schistosoma mansoni-infected mice. The American journal of tropical medicine and hygiene 46, 752–758 (1992).
Bardor, M. et al. Immunoreactivity in mammals of two typical plant glyco-epitopes, core alpha(1,3)-fucose and core xylose. Glycobiology 13, 427–434, https://doi.org/10.1093/glycob/cwg024 (2003).
Luyai, A. E. et al. Differential expression of anti-glycan antibodies in schistosome-infected humans, rhesus monkeys and mice. Glycobiology 24, 602–618, https://doi.org/10.1093/glycob/cwu029 (2014).
van Diepen, A. et al. Parasite glycans and antibody-mediated immune responses in Schistosoma infection. Parasitology 139, 1219–1230, https://doi.org/10.1017/s0031182012000273 (2012).
Diemert, D. J. et al. Generalized urticaria induced by the Na-ASP-2 hookworm vaccine: implications for the development of vaccines against helminths. The Journal of allergy and clinical immunology 130, 169–176.e166, https://doi.org/10.1016/j.jaci.2012.04.027 (2012).
Mari, A. et al. Evaluation by double-blind placebo-controlled oral challenge of the clinical relevance of IgE antibodies against plant glycans. Allergy 63, 891–896, https://doi.org/10.1111/j.1398-9995.2008.01703.x (2008).
Hemmer, W. In Anticarbohydrate Antibodies: From Molecular Basis to Clinical Application (eds Paul, K. & Sven, M.-L.) 181–202 (Springer Vienna, 2012).
Commins, S. P. et al. Delayed anaphylaxis, angioedema, or urticaria after consumption of red meat in patients with IgE antibodies specific for galactose-α-1,3-galactose. Journal of Allergy and Clinical Immunology 123, 426–433.e422, https://doi.org/10.1016/j.jaci.2008.10.052 (2009).
Acknowledgements
We thank Koome sub-county and Entebbe municipality community members for participating in the rural survey (LaVIISWA) and in the urban survey, respectively. We are grateful for important contributions from all LaVIISWA and urban survey staff. We also thank Ms Grace Nabakooza and Mr Moses Egesa for technical assistance in the implementation of HCA and the global test in R. The LaVIISWA study (herein the ‘rural survey’) and the urban survey were funded by the Wellcome Trust, grant 095778 awarded to A.M.E. G.N. is supported by a PhD fellowship from the African Partnership for Chronic Disease Research (APCDR) and received further support for glycan microarray experiments from the Royal Society of Tropical Medicine and Hygiene (grant GR000904). G.N. is an honorary fellow, and R.E.S a PhD fellow, of the Makerere University – Uganda Virus Research Institute Centre of Excellence for Infection and Immunity Research and Training (MUII-plus). MUII-plus is funded under the DELTAS Africa Initiative. The DELTAS Africa Initiative is an independent funding scheme of the African Academy of Sciences (AAS), Alliance for Accelerating Excellence in Science in Africa (AESA) and supported by the New Partnership for Africa’s Development Planning and Coordinating Agency (NEPAD Agency) with funding from the Wellcome Trust (grant 107743) and the UK Government.
Author information
Authors and Affiliations
Contributions
G.N., C.H.H., M.Y., A.M.E., R.v.R. and A.v.D. contributed to the conception and experimental design of the study. A.M.E., R.E.S. and M.N. led the field and clinic procedures. S.S. and N.C.R. constructed the synthetic microarrays. G.N. conducted the microarray antibody binding experiments. G.N., J.N., J.K. and I.N. participated in establishing and conducting all other immunological and parasitological experiments. G.N. analysed the data with important contributions from E.L.W. and A.v.D. G.N. wrote the manuscript, with all authors contributing to the interpretation of the results, and revision and approval of the final manuscript. G.N. is the guarantor of the article.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nkurunungi, G., van Diepen, A., Nassuuna, J. et al. Microarray assessment of N-glycan-specific IgE and IgG profiles associated with Schistosoma mansoni infection in rural and urban Uganda. Sci Rep 9, 3522 (2019). https://doi.org/10.1038/s41598-019-40009-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-40009-7
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.