Abstract
Small artery occlusion (SAO) is responsible for 31.3% of all ischemic strokes in China. However, reports regarding the recurrence rate of SAO in China are rare. We aimed to assess the recurrence rate and factors associated with SAO in China. All consecutive patients with SAO hospitalized at Tianjin Huanhu Hospital from 2005 to 2014 were recruited. We assessed the association between stroke subtype, severity, and disease history with recurrence at 3, 12, and 36 months of onset using multivariate logistic regression analysis. A total of 2,524 SAO patients were included in this study, including 1696 (67.2%) men and 828 (32.8%) women. The recurrence rates were 3.1% at 3 months, 12.7% at 12 months, and 36.5% at 36 months. Compared with women, men had a higher risk of recurrence at 3 months after SAO (P = 0.003). Old age and severity of stroke were also associated with a higher risk of recurrence (P < 0.05). Patients with an elevated C-reactive protein had a higher risk of recurrence at 12 months (P = 0.003). On the other hand, the risk of recurrence at 12 months was 39% lower in patients who consumed alcohol than in those who did not (P = 0.037). Hypertension, atrial fibrillation, and obesity were independent risk factors of recurrence at 36 months. These findings suggest that modification of risk factors in patients with SAO, particularly men, is essential for reducing the rate of recurrence and the overall burden of stroke in China.
Similar content being viewed by others
Introduction
Stroke has become the second most common cause of death and the leading cause of disability worldwide1. In China, 21.6% of deaths among men and 20.8% of deaths among women are attributed to stroke2. Moreover, stroke has become the leading cause of death in rural areas and the third most common cause of death in urban areas in China3. While the incidence of stroke has declined in developed countries over the past several decades4,5, it has increased in developing countries, particularly in China6,7,8. Furthermore, the total incidence of stroke is expected to increase considerably over the next two decades9.
Stroke is a complex disease caused by multiple potential etiologies. It is important to identify the underlying causes and the subtype of stroke in order to determine the most appropriate treatment and to accurately predict patient outcomes10. Small artery occlusion (SAO) is a distinct type of ischemic stroke according to the Trial of ORG10172 in Acute Stroke Treatment (TOAST) classification. The age-standardized incidence rate of SAO has been reported to be 25.8 per 100,000 persons (95% confidence interval, 21.5, 30.9) in Europe11. SAO accounts for approximately 20% of all ischemic strokes in Europe12, but up to 31.3% of cases in China13.
Previous studies have demonstrated that disability, mortality, and recurrence rates after ischemic stroke are associated with stroke subtype11,14. However, there is limited evidence on the recurrence rates of SAO in China. Thus, it is crucial to assess the recurrence rate and the relevant patient factors associated with SAO in order to manage risk and improve secondary prevention, particularly since SAO is associated with longer survival and lower severity compared with other stroke subtypes15.
In this study, we identified the recurrence rates and relevant risk factors at 3, 12, and 36 months after stroke among patients with SAO at a single center in China.
Results
Description of the demographical and clinical features among SAO patients
A total of 11,330 patients with acute ischemic stroke were recruited during the study period. After excluding 8679 patients with non-SAO strokes and 127 without TOAST classification data, the final analysis included 2524 patients with SAO. Follow-up data for the assessment of recurrence was available for 2497 SAO patients (98.9%) at 3 months, 2326 (95.4%) at 12 months, and 1636 (86%) at 36 months (Fig. 1).
Flow chat of patients’ selection. A total of 11,330 patients with acute ischemic stroke were recruited during the study period. After excluding 8679 patients with non-SAO strokes and 127 without TOAST classification data, the final analysis included 2524 patients with SAO. Follow-up data for the assessment of recurrence was available for 2497 SAO patients (98.9%) at 3 months, 2326 (95.4%) at 12 months, and 1636 (86%) at 36 months.
Men comprised 67.2% (n = 1,696) of the SAO cohort while women accounted for 32.8% (n = 828), with an average age of 62.71 years. Most patients were between 50 and 64 years (55.9%), and mild stroke was the most common (82.7%). The prevalence of hypertension, diabetes mellitus, atrial fibrillation (AF), artery stenosis, obesity, current smoking, and alcohol consumption was 75.9%, 32.4%, 2.3%, 13.9%, 11.5%, 37.8%, and 18.8%, respectively. Moreover, the average levels of fasting plasma glucose, total cholesterol, triglycerides, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, high-sensitivity C-reactive protein (Hs-CRP), and homocysteine (HCY) were 6.44 mmol/L, 4.92 mmol/L, 1.70 mmol/L, 1.06 mmol/L, 3.00 mmol/L, 6.23 mmol/L, and 15.89 mmol/L, respectively (Table 1).
Risk factors for recurrence on univariate analysis
The overall 3-month recurrence rate was 3.1%, with 3.8% in men and 1.7% in women. The corresponding recurrence rates at 12 and 36 months after SAO were 12.7%, 12.4%, and 13.2%; and 36.5%, 35.2%, and 34.4%, respectively. Recurrence was correlated with older age (P = 0.036 for 3 months and < 0.001 for 12 months and 36 months) and stroke severity (P = 0.008 for 3 months, P < 0.001 for 12 months and 36 months). Moreover, sex was associated with recurrence at 3 months after stroke; diabetes, current smoking, alcohol consumption, and Hs-CRP levels at admission were associated with recurrence at 12 months; and hypertension, diabetes, AF, obesity, current smoking, and Hs-CRP levels at admission were associated with recurrence at 36 months after SAO (all P < 0.05; Tables 2 and 3).
Determinants of recurrence on multivariate analysis
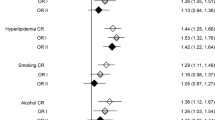
Compared to women, men were more likely to experience a recurrence at 3 months after SAO (relative risk [95% confidence interval], 2.50 [1.38 to 4.53; P = 0.003]). Old age was a common risk factor for recurrence at 3, 12, and 36 months after SAO. Patients ≥80 years had a 2.07 and 1.12-fold increased risk of recurrence than those <50 years at 3 and 12 months, respectively (P < 0.05). Meanwhile, there was a 77% higher risk of recurrence at 36 months after SAO in patients between 70 and 79 years (P = 0.007). In comparison to patients with mild stroke, there was an increased risk of recurrence at 12 months in those with moderate stroke, and at 36 months in those with moderate and severe strokes. Moreover, the risk of recurrence at 12 months after SAO was 39% lower in patients who drank alcohol compared to those who did not (P = 0.037). The risk of recurrence increased in accordance with CRP level elevation (P = 0.003). Hypertension, AF, and obesity were independent risk factors of recurrence at 36 months after SAO (relative risk [95% confidence interval], 1.38 [1.04, 1.84; P = 0.027], 3.09 [1.28, 7.47; P = 0.012], and 1.62 [1.13, 2.31; P = 0.008], respectively; Table 4).
Discussion
In the present study, we assessed the recurrence rates and their associated factors among patients with SAO at 3, 12, and 36 months after stroke. To our knowledge, this is the first study to report the recurrence rates and determinants of SAO in China. The recurrence rates were 3.1% at 3 months, 12.7% at 12 months, and 36.5% at 36 months after SAO. Male sex and older age (≥80 years) were independent risk factors for recurrence at 3 months after SAO. Older age (≥80 years), moderate stroke, and elevated CRP level were independent risk factors of recurrence at 12 months after SAO, but alcohol drinking was negatively associated with recurrence. Older age (70–79 years), moderate/severe stroke, hypertension, AF, and obesity were determinants of recurrence at 36 months after SAO. The Oxford Vascular Study had shown that the risk of recurrence varied between stroke subtypes and the 3-month rate for SAO was 6.2%14.
The outcomes and recommended therapeutic interventions differ according to the stroke subtype14,16. Although SAO is typically associated with more favorable outcomes, the expected long-term survival following SAO can increase the risk of adverse events, particularly recurrence.
Previous studies have demonstrated poor stroke outcomes among elderly patients17,18,19,20,21. However, reports of outcomes among patients with SAO have been limited and controversial. The previous studies demonstrated favorable short-term outcomes among SAO patients22,23, but the unfavorable long-term outcomes among elderly SAO patients22,24. Moreover, young patients with SAO have low 5-year mortality rates25. In this study, the short and long-term risk of recurrence was higher among elderly patients.
Several studies have reported that the National Institutes of Health Stroke Scale (NIHSS) score was highly related to stroke outcome18,26,27. Ischemic stroke subtypes according to the TOAST criteria were correlated with short- and long-term recurrence after stroke14,28. Age, NIHSS score on admission, and female sex were independent risk factors for poor outcome (defined as a modified Rankin Scale [mRS] score >2 or death at 3-month follow-up)29. A higher rate of recurrence was observed in men with SAO than in women30. In the present study, we found that stroke severity according to the NIHSS score on admission was significantly associated with long-term recurrence (12 and 36 months). However, we found a higher risk of recurrence at 3 months in male patients. Moreover, hypertension, AF, and obesity increased the risk of recurrence in patients at 36 months. Poor control of risk factors (including hypertension, AF, and obesity) may contribute to recurrence in SAO patients, particularly in men.
CRP is currently used for evaluating pathological inflammation and has been extensively studied in relation to the progression of atherosclerosis31. The level of Hs-CRP at admission predicts future recurrence in patients with acute ischemic stroke32,33. Moreover, a recent study revealed that higher CRP levels were associated with subclinical markers of cerebral small vessel disease and neurodegeneration34. In the present study, we found that elevated levels of Hs-CRP increased the risk of recurrence of SAO at 12 months. Several mechanisms may explain this finding. First, elevated CRP levels are involved in vessel occlusion, altered cerebral autoregulation, increased vascular permeability, and are a marker of arteriolosclerosis35. Second, elevated CRP is associated with large-vessel atherosclerosis, which affects the microvascular endothelium of cerebral small vessels by reducing cerebral blood flow and producing inflammatory mediators and free radicals36. Finally, CRP is associated with endothelial cell activation, which plays a key role in small- and large-vessel disease37.
Previously reports on the effects of alcohol consumption on outcome after stroke have been controversial. Excessive alcohol consumption showed no association with functional outcome after stroke38,39. The North East Melbourne Stroke Incidence Study found that pre-stroke alcohol consumption did not independently predict disability at 2 years after stroke on multivariate analysis40. The Copenhagen Stroke Study demonstrated that daily alcohol consumption was not an independent predictor of good outcomes on multivariate analysis41. A population-based study indicated that light-to-moderate alcohol consumption was not associated with reduced stroke risk42, while heavier consumption increased the risk of stroke or death42,43. In contrast to these previous studies, we found that alcohol consumption was associated with a low risk of recurrence at 12 months after SAO. Differences in the study participants (general population or patients with stroke), alcohol consumption style (categorized by qualitative or quantitative factors), and study endpoints (risk of developing stroke or prognosis after stroke) may partly explain this disparity. Moreover, differences between the life styles of Western and Eastern populations may have contributed to inversion of effects, especially in terms of alcohol consumption. Some studies have reported that excessive alcohol consumption increases the relative risk of stroke, while light consumption may be protective against ischemic stroke44,45,46.
There are several limitations to this study. First, this study was conducted at a single hospital and thus represents a limited population. Moreover, Tianjin Huanhu Hospital is a specialized neurology and neurosurgery hospital in Tianjin, China. The patients treated here are usually in a more critical condition than those at other hospitals and this may result in selective bias. Additionally, we assessed the association of alcohol consumption with the risk of recurrence after SAO using the qualitative categorization (no drinking versus current drinking) those ever alcohol drinkers was involved in no drinking group. Moreover, there was no quantitative analysis of alcohol consumption per day or week. Further quantitative evaluation of the impact of alcohol consumption on the prognosis after SAO is urgently needed. Finally, this study may have excluded some patients with severe stroke and those who died before hospital admission, which could have resulted in an unfair assessment.
In conclusion, we observed a lower rate of recurrence at 3 months after SAO, but a 4- and 12-fold increase in risk at 12 and 36 months, respectively. Age, sex, stroke severity, hypertension, AF, obesity, and elevated CRP level were associated with an increased risk of recurrence, while alcohol consumption decreased this risk. Poor management and uncontrolled risk factors might contribute to the high risk of recurrence among SAO patients. These findings suggest that improving risk factor control in patients with SAO, particularly among male patients, is critical to reducing the recurrence rate and alleviating the overall stroke burden in China.
Methods
All patients in this study were recruited from the stroke unit at Tianjin Huanhu Hospital, China between May 2005 and September 2014.The procedure used to establish the stroke registry has been described previously47,48,49. In brief, all consecutive patients with acute ischemic stroke were entered into the registry system, and detailed information, including clinical features and outcomes, were collected.
The ethics committee of Tianjin Huanhu Hospital approved the study, and written informed consent was obtained from all patients or their next-of-kin.
Stroke patients were identified according to the World Health Organization’s criteria, and all cases of stroke were confirmed using neuroimaging50. All acute ischemic stroke patients were categorized as either of five etiological subtypes according to their medication history, location of ischemic zone, and vessels status (TOAST classification). First, computed tomography (CT) or magnetic resonance imaging (MRI) was performed to ascertain the location and diameter of the ischemic focus in all patients. Patients with penetrating artery or lacunar infarctions, and a focus diameter of <1.5 cm on CT or MRI were classified SAO51. All patients subsequently underwent carotid ultrasonography for exclusion of large artery stenosis, and echocardiography for exclusion of cardiac emboli. Some patients with abnormal blood flow velocities, multiple carotid plaques, or severe artery stenosis underwent additional CT or magnetic resonance angiography.
Stroke severity was categorized into three groups according to NIHSS scores: mild (NIHSS score: ≤7), moderate (NIHSS score: 8–16), and severe (NIHSS score: ≥17)52. Patients were considered to have been using oral anticoagulants if they were receiving oral anticoagulants prior to stroke onset. Stroke risk factors, such as previous medical history (including hypertension, diabetes mellitus, AF, hyperlipidemia, and obesity) and life style (including smoking and alcohol consumption) were recorded on admission. The presence of hypertension, diabetes mellitus, and hyperlipidemia were defined according to self-reporting of disease history or use of antihypertensives, antidiabetics, or lipid-lowering medication, respectively. AF was defined as a history of AF, confirmed through at least one electrocardiogram, or the presence of arrhythmia during hospitalization. Obesity was defined as a body mass index ≥30 kg/m2. The NIHSS score and Barthel Index were evaluated on admission and at discharge. The mRS score was assessed on admission; at discharge; and at 3, 12, and 36 months after stroke. Moreover, TC, (TG), HDL-C, LDL-C, FPG, Hs-CRP, and HCY levels on admission were recorded.
Recurrence was defined as all new-onset vascular events, including stroke, myocardial infarction, and venous thrombosis after 1 month of stroke onset. Follow-up was conducted according to a predetermined procedure: trained neurologists assessed patients in the outpatient department at 3, 12, 24, and 36 months after stroke. All patients were followed-up by face-to-face interview, but those reexamined at the local hospital were followed-up by telephone.
The metric variables are presented as means with standard deviations for age as well as TC, TG, HDL-C, LDL-C, Hs-CRP, and HCY levels. NIHSS score, Barthel Index, and mRS score are presented as medians with ranges. Differences in metric variables between groups were compared using Student’s t-test or the Mann-Whitney U test. Categorized variables, including sex, stroke severity, stroke risk factors, and recurrence rate during follow-up, are presented as number of cases (percentages). Differences in risk factors were compared between groups using the Chi-squared test. Multivariate analysis of determinants of recurrence (as a dichotomous variable: yes or no) was assessed using logistic regression after adjusting for covariables, which included age (<75 years, ≥75 years), sex (male, female), stroke severity (mild, moderate, severe), medical history (as a dichotomous variable: yes or no), risk factors (as a dichotomous variable: yes or no), and laboratory measurements (as a continuous variable). The results are presented as adjusted relative risk with 95% confidence intervals. All statistical analyses were performed using SPSS version 19.0 (SPSS Inc., Chicago, IL), and a two-tailed P value < 0.05 indicated statistical significance.
References
Bonita, R. et al. The global stroke initiative. Lancet Neurol. 3, 391–393 (2004).
National Center for Cardiovascular Diseases, China: Report on cardiovascular diseases in China 2014. Beijing: Encyclopedia of China Publishing House (2015).
The Ministry of Health of the People’s Republic of China: China health statistics yearbook 2011. Beijing: China Union Medical University Press (2011).
Redon J, et al. Stroke mortality and trends from 1990 to 2006 in 39 countries from Europe and Central Asia: implications for control of high blood pressure. Eur Heart J. 32, 1424–1431 (2011).
Lloyd-Jones, D. M. et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: the American Heart Association’s strategic Impact Goal through 2020 and beyond. Circulation. 121, 586–613 (2010).
Wang, J. et al. Increasing stroke incidence and prevalence of risk factors in a low-income Chinese population. Neurology. 84, 374–381 (2015).
Zhao, D. et al. Epidemiological transition of stroke in China: twenty-one-year observational study from the Sino-MONICA-Beijing Project. Stroke. 39, 1668–1674 (2008).
Jiang, B. et al. Incidence and trends of stroke and its subtypes in China: results from three large cities. Stroke. 37, 63–68 (2006).
Meairs, S. et al. Stroke research priorities for the next decade–A representative view of the European scientific community. Cerebrovasc Dis. 22, 75–82 (2006).
Chen, P. H. et al. Classifying ischemic stroke, from TOAST to CISS. CNS Neurosci Ther. 18, 452–456 (2012).
Kolominsky-Rabas, P. L., Weber, M., Gefeller, O., Neundoerfer, B. & Heuschmann, P. U. Epidemiology of ischemic stroke subtypes according to TOAST criteria: incidence, recurrence, and long-term survival in ischemic stroke subtypes: a population-based study. Stroke. 32, 2735–2740 (2001).
Small Artery Occlusive Disease. Chapter. In book: Primer on Cerebrovascular diseases, 303–307 (1997).
Zhou, H., Wang, Y. J., Wang, S. X. & Zhao, X. Q. TOAST subtyping of acute ischemic stroke. Zhonghua Nei Ke Za Zhi. 43, 495–498 (2004).
Lovett, J. K., Coull, A. J. & Rothwell, P. M. Early risk of recurrence by subtype of ischemic stroke in population-based incidence studies. Neurology. 62, 569–573 (2004).
Grau, A. J. et al. Risk factors, outcome, and treatment in subtypes of ischemic stroke: the German stroke data bank. Stroke. 32, 2559–2566 (2001).
Marnane, M. et al. Stroke subtype classification to mechanism-specific and undetermined categories by TOAST, A-S-C-O, and causative classification system: direct comparison in the North Dublin population stroke study. Stroke. 41, 1579–1586 (2010).
Deng, Y. X. et al. Age Differences in Clinical Characteristics, Health care, and Outcomes after Ischemic Stroke in China. CNS Neuroscience & Therapeutics. 18, 819–26 (2012).
Soares, I., Abecasis, P. & Ferro, J. M. Outcome of first-ever acute ischemic stroke in the elderly. Arch Gerontol Geriat. 53, e81–e87 (2011).
Fonarow, G. C. et al. Get With the Guidelines-Stroke Steering Committee and Investigators. Age-related differences in characteristics, performance measures, treatment trends, and outcomes in patients with ischemic stroke. Circulation. 121, 879–91 (2010).
Saposnik, G. et al. Registry of the Canadian Stroke Network for the Stroke Outcome Research Canada (SORCan) Working Group. Variables associated with 7-day, 30-day, and 1-year fatality after ischemic stroke. Stroke. 39, 2318–24 (2008).
Palnum, K. D. et al. Older patients with acute stroke in Denmark: quality of care and shortterm mortality. A nationwide follow-up study. Age Ageing. 37, 90–5 (2008).
Bamford, J., Sandercock, P., Dennis, M., Burn, J. & Warlow, C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet. 337, 1521–6 (1991).
Shin, D., Lee, P. & Bang, O. Mechanisms of recurrence in subtypes of ischemic stroke: a hospital-based follow-up study. Arch Neurol. 62, 1232–7 (2005).
Eriksson, S. E. & Olsson, J. E. Survival and recurrent strokes in patients with different subtypes of stroke: a fourteen-year follow-up study. Cerebrovasc Dis. 12, 171–80 (2001).
Putaala, J. et al. Causes of death and predictors of 5-year mortality in young adults after first-ever ischemic stroke: the Helsinki Young Stroke Registry. Stroke. 40, 2698–703 (2009).
Denti, L. et al. The poor outcome of ischemic stroke in very old people: A cohort study of its determinants. J Am Geriatr Soc. 58, 12–17 (2010).
Gur, A. Y. et al. Stroke in the very elderly: characteristics and outcome in patients aged ≥85 years with a first-ever ischemic stroke. Neuroepidemiology. 39, 57–62 (2012).
Schulz, U. G. & Rothwell, P. M. Differences in vascular risk factors between etiological subtypes of ischemic stroke: importance of population-based studies. Stroke. 34, 2050–2059 (2003).
Li, S. et al. Risk factors for poor outcome and mortality at 3 months after the ischemic stroke in patients with atrial fibrillation. J Stroke Cerebrovasc Dis. 22, e419–425 (2013).
Qiao, Q. et al. Sex differences in outcomes and associated factors among stroke patients with small artery occlusion in China. Biol Sex Differ. 9(1), 35 (2018).
Calabro, P., Golia, E. & Yeh, E. T. Role of C-reactive protein in acute myocardial infarction and stroke: possible therapeutic approaches. Curr Pharm Biotechnol. 13(1), 4–16 (2012).
Zhang, Y. B. et al. Association of circulating high-sensitivity C-reactive protein with late recurrence after ischemic stroke. Neuroreport 28(10), 598–603 (2017).
Kitagawa, K. et al. Reduction in High-Sensitivity C-Reactive Protein Levels in Patients with Ischemic Stroke by Statin Treatment: Hs-CRP Sub-Study in J-STARS. J Atheroscler Thromb 24(10), 1039–1047 (2017).
Hilal, S. et al. C-Reactive Protein, Plasma Amyloid-β Levels, and Their Interaction With Magnetic ResonanceImaging Markers. Stroke. 49, 2692–2698 (2018).
Pantoni, L. & Garcia, J. H. Pathogenesis of leukoaraiosis: a review. Stroke. 28, 652–659 (1997).
van Dijk, E. J. et al. C-reactive protein and cerebral small-vessel disease: the Rotterdam Scan Study. Circulation. 112, 900–905 (2005).
Khreiss, T., Jozsef, L., Potempa, L. A. & Filep, J. G. Conformational rearrangement in C-reactive protein is required for proinflammatory actions on human endothelial cells. Circulation. 109, 2016–2022 (2004).
Mukamal, K. J. et al. Alcohol use and risk of ischemic stroke among older adults: the Cardiovascular Health Study. Stroke. 36, 1830–1834 (2005).
Klára, F. et al. Prestroke Alcohol Consumption and Smoking Are Not Associated with Stroke Severity, Disability at Discharge, and Case Fatality. Journal of Stroke and Cerebrovascular Diseases 23(1), e31–e37 (2014).
Karoliina, A. et al. Long-Term Mortality After First-Ever and Recurrent Stroke in Young Adults. Stroke 45(9), 2670–2676 (2014).
Jørgensen, H. S. et al. What determines good recovery in patients with the most severe strokes? The Copenhagen. Stroke Study. Stroke 30, 2008–2012 (1999).
Sturm, W. et al. Determinants of handicap after stroke: The North East Melbourne. Stroke Incidence Study [NEMESIS]. Stroke 35, 715–720 (2004).
Jones, S. B. et al. Midlife Alcohol Consumption and the Risk of Stroke in the Atherosclerosis Risk in Communities Study. Stroke. 46(11), 3124–30 (2015).
Patra, J. et al. Alcohol consumption and the risk of morbidity and mortality for different stroke types–a systematic review and meta-analysis. BMC Public Health. 10, 258 (2010).
Mukamal, K. J. et al. Alcohol and Risk for Ischemic Stroke in Men: The Role of Drinking Patterns and Usual Beverage. Ann Intern Med 142(1), 11–9 (2005 Jan).
Reynolds Kristi, L. et al. Alcohol Consumption and Risk of Stroke. JAMA 289(5), 579 (2003).
Zhou, G. et al. Sex differences in outcomes after stroke among patients with low total cholesterol levels: a large hospital-based prospective study. Biol Sex Differ. 7, 62 (2016).
Xing, Y. et al. Low density lipoprotein cholesterol and the outcome of acute ischemic stroke: results of a large hospital-based study. Eur Neurol. 76, 195–201 (2016).
Zhao, W. et al. Low total cholesterol level is the independent predictor of poor outcomes in patients with acute ischemic stroke: a hospital-based prospective study. BMC Neurol. 16, 36 (2016).
Stroke, Recommendations on stroke prevention, diagnosis, and therapy: Report of the WHO task force on stroke and other cerebrovascular disorders. Stroke. 20, 1407–1431 (1989).
Adams, H. P. Jr et al. Marsh EE 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 24(1), 35–41 (1993).
Kim, J. S., Lee, K. B., Roh, H., Ahn, M. Y. & Hwang, H. W. Gender differences in the functional recovery after acute stroke. J Clin Neurol. 6, 183–188 (2010).
Author information
Authors and Affiliations
Contributions
Y.Z. and Y.G. were involved in conception and design, and critical review for this article. Y.G. were involved in data analysis for this article. Y.Z. was involved in manuscript drafting. Y.Z., Y.G., S.L., M.Z., M.W., W.S. and H.W. were involved in data collection, case diagnosis and confirmation for this article. All authors have reviewed and approved publication the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Y., Guan, Y., Zhang, Y. et al. Recurrence Rate and Relevant Associated Factors of Stroke among Patients with Small Artery Occlusion in Northern China. Sci Rep 9, 2834 (2019). https://doi.org/10.1038/s41598-019-39207-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39207-0
This article is cited by
-
Association of α-HBDH levels with the severity and recurrence after acute ischemic stroke
European Journal of Medical Research (2024)
-
Cumulative risk of stroke recurrence over the last 10 years: a systematic review and meta-analysis
Neurological Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.