Abstract
Two influenza B virus lineages, B/Victoria and B/Yamagata, are co-circulating in human population. While the two lineages are serologically distinct and TIV only contain one lineage. It is important to investigate the epidemiological and evolutionary dynamics of two influenza B virus lineages in Beijing after the free influenza vaccine policy from 2007. Here, we collected the nasopharyngeal swabs of 12657 outpatients of influenza-like illness and subtyped by real-time RT-PCR during 2011–2017. The HA and NA genes of influenza B were fully sequenced. The prevalence is the highest in the 6–17 years old group among people infected with influenza B. Yamagata-lineage virus evolved to two inter-clade from 2011–2014 to 2014–2017. The amino acids substitutions of HA1 region were R279K in strains of 2011–2014 and L173Q, M252V in strains of 2014–2017. Substitutions L58P, I146V were observed in HA1 region of Victoria-lineage virus in 2011–2012 and I117V, N129D were showed in 2015–2017. Phylogenetic analysis of NA showed Yamagata-Victoria inter-lineage reassortant occurred in 2013–2014. Influenza B mainly infect the school-aged children in Beijing and the free influenza vaccine inoculation does not seem to block school-age children from infection with influenza B. The antigen characteristics of circulating influenza B were different to the recommended vaccine strains. We concluded that the Victoria-lineage vaccine strain should been changed and the free influenza vaccine should be revalued.
Similar content being viewed by others
Introduction
Influenza virus poses a serious threat to public health by causing the annual epidemics and occasional pandemics. Influenza viruses are divided into influenza A, B, C and D virus based on the antigenic specificity of the nucleoprotein and matrix protein and influenza A and B virus co-circulated globally in a typical seasonal pattern1,2,3. Unlike influenza A which has a broad host range and caused pandemics by antigenic shift, influenza B usually causes local epidemic with no natural animal host (other than seals) and a slower mutation rate4,5,6. However, influenza B still contributes about one third of the global influenza disease burden. Many recent reports are indicated that influenza B is associated with serious illness, such as acute myocardial infarction and so on7. More seriously, influenza B caused 0.058% deaths for all-cause death in Southern China, which was higher than influenza A8. Despite the significance of influenza B to human health, their epidemiological characteristics and antigenic dynamics are far less studied compared to influenza A.
Influenza B was first isolated in 1940, designated as influenza B/Lee/40 and then was classified into two distinct lineages by the genetically relationship of the HA gene, represented by the prototype viruses B/Victoria/2/87 (Victoria-lineage) and B/Yamagata/16/88 (Yamagata-lineage) since 19839,10,11. The Yamagata- and Victoria-lineage have been co-circulating globally and often alternating in regional dominance. Two lineages undergo a slower antigenic variation with genetic evolution of the HA and NA genes including nucleotide mutations, and reassortment12,13.
The primary measure to prevent influenza virus infection is vaccination. Trivalent vaccine (TIV) has been produced containing influenza A/H1N1 and A/H3N2 antigens and a single influenza B antigen since 1977, despite some studies said low level of cross-protection provided by immunization with vaccine containing antigen from a single influenza B lineage14,15. Since 2007, the municipal government of Beijing provided free influenza vaccination for adult residents more than 60 years old and 6–17 years old elementary and high school students (Beijing influenza vaccination program for middle school in 2007, http://zfxxgk.beijing.gov.cn/110088/qt33/2015–10/20/content_625915.shtml)16,17. However, the overall estimate of influenza vaccine effectiveness (VE) was 46.9% for the 2013–2014 season and 5.0% for the 2014–2015 season18. After this policy of free vaccine was implemented in Beijing, there was no continuous monitoring of the restrictions on influenza B infection and the influence to the pathogenic characteristics of influenza B. So, the continuous surveillance for influenza B in Beijing reflected the relationship between influenza vaccination rate and immune protective effect which imperative and has economic significance. The aim of this retrospective surveillance during the 2011–2017 seasons in Beijing was to assess the epidemiological and phylogenetic characteristics of influenza B from outpatients with influenza-like illness (ILI), and analyze the mismatch ratio between the circulating and vaccine strains.
Results
Pathogen spectrum of influenza cases from October 2011 to September 2017
Total 12657 outpatients in the surveillance seasons from October 2011 to September 2017 were included in this study. Among them 446 (3.52%, from 0 to 7.8% per year) were positive for influenza A/H1N1; 1085 (8.57%, from 2.1% to 16.9% per year) were positive for influenza A/H3N2; 395 (3.12%, from 0 to 8.8% per year) were positive for influenza B/Yamagata-lineage; 246 (1.94%, from 0 to 7.2% per year) were positive for influenza B/Victoria-lineage. 3 (0.02%) were co-infected with two influenza viruses (Fig. 1). In total, 29.47% of influenza cases during the six years were infected by influenza B, which was higher than influenza A/H1N1, but lower than influenza A/H3N2. Notably, the positive percentage of influenza Yamagata-lineage in 2014–2015 was 7.2% that was higher than the other three influenza virus subtypes.
The percentage of influenza virus by real-time RT-PCR in surveillance seasons from 2011 to 2017. The y-axis represents positive percentage of influenza virus in assembled ILI outpatients every month. The x-axis shows month from October 2011 to September 2017. The period from October to September next year is defined as one influenza seasons. Blue boxes represent A/H1N1 virus, green boxes represent A/H3N2 virus, red boxes represent B/Yamagata-lineage, yellow boxes represent B/Victoria-lineage.
Age distribution of influenza B
The ILI cases were divided to four age groups (≤5 years old, 6–17 years old, 18–59 years old, ≥60 years old) in order to analyze the age distribution of influenza B. The positive rates of influenza B in four age groups were 17.24%, 4.99%, 4.82% and 6.71% (Table S1). Comparing with subtypes of influenza A, the prevalence of influenza B, especially for Yamagata-lineage, was higher in age group ≤5 years old (χ2 test, p = 0.000). Although the prevalence of influenza B was lower than influenza A/H3N2, it was higher than influenza A/H1N1 in all age groups excepting ≤5 years old group. For all outpatients caused by influenza B, the positive percentage of Yamagata-lineage was the highest in ≤5 years old group. While the prevalence of Victoria-lineage virus was higher in age group 6–17 years old group than the other age groups (Fig. 2). Therefore, influenza B infection people concentrated in preschool children and school-aged children.
The age distribution of Yamagata- and Victoria-lineage virus. The y-axis represents the proportion of Victoria-lineage (a) and Yamagata-lineage (b) in assembled ILI outpatients caused by influenza virus every month. The x-axis shows month from October 2011 to September 2017. The period from October to September next year is defined as one influenza seasons. The age group ≤5 years old was shown by a yellow lines; the age group 6–17 years old was shown by a green lines; the age group 18–59 years old was shown by a blue lines and the age group ≥60 years old was shown by a violet lines.
Matching ratio of influenza B between seasonal viruses and vaccine strains
During 2011–2017 surveillance seasons, Yamagata- and Victoria-lineage were infected synchronously or one lineage was in the ascendancy. But one lineage was recommended vaccine strain (Table S2). In 2011–2012 season, Victoria-lineage infection was predominated, but 34.68% of the circulating influenza B belonged to the opposite lineage to the northern hemisphere influenza B vaccine strains (B/Brisbane/60/2008). Subsequently, Yamagata-lineage was in dominant in 2012–2015 seasons and was consistent with recommended vaccine strain. Then Victoria-lineage was predominated in 2015–2016 season, however, the Yamagata-lineage (B/Phuket/3073/2013) was still recommended as the vaccine strain during this time span. Moreover, 26.67% of the circulating influenza B belonged to the opposite lineage to recommended vaccine strain in 2016–2017 season. On average, 31.20% of circling influenza B was mismatched to recommended influenza B vaccine strain during the consecutive six seasons (Fig. 3).
Distribution and recommended vaccine strain of influenza B in Beijing, 2011–2017. White boxes represent Yamagata-lineage; black boxes represent Victoria-lineage. Asterisks indicate the World Health Organization–recommended influenza B vaccine strain of that year. Data from October 2012 to September 2013 had only 1 case infected by influenza B virus.
Phylogenetic tree of the HA and NA of influenza B
The complete sequence of HA and NA gene were obtained from 83/110 (79.09%) of selected samples. Partial sequence of HA or NA sequence was excluded. A total of 83 full-length HA and 83 full-length NA sequences and 60 published sequences in GISAID and GenBank databases were analyzed for phylogenetic classification.
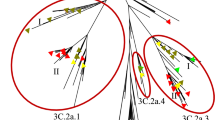
Among 83 HA sequences, 22 (26.51%) of influenza B strains belonged to Victoria-lineage and 61 (73.49%) virus belonged to Yamagata-lineage based on the phylogenetic analysis of the HA sequence. Influenza B has been separated previously into three major antigenically distinct clades (clade 1, the B/Brisbane/60/2008 clade, clade 2, the B/Massachusetts/2/2012 clade, and clade 3, the B/Wisconsin/1/2010 clade), based on phylogenetic analysis of HA and NA genes19. Phylogenetic analysis showed that all HA sequences of Yamagata-lineage belonged to clade 3 (Fig. 4). Comparing to vaccine strain B/Wisconsin/1/2010, a series of amino acid changes were observed in the HA1 region of all strains: N116K, K299E and E313K. In detail, Yamagata-lineage evolved to two inter-clade from 2011–2014 seasons to 2014–2017 seasons. The Yamagata-lineage in 2011–2014 surveillance seasons shared special amino acids substitutions at R278K (9/33, 27.27%) (Tables 1, 2), whereas the Yamagata-lineage in 2014–2017 surveillance seasons shared amino acids substitutions at L172Q (34/34, 100%), K211R (8/34, 23.53%), M251V (26/34, 76.47%), which were related to B/Phuket/3073/2013 strain.
Phylogenetic tree of the HA gene sequences of influenza B viruses. HA sequences of influenza B viruses were compared with WHO recommended candidate vaccine (▲) and reference strains. The phylogeny was reconstructed using Neighbor-Joining (NG) method. Bootstrap values (>70%) and amino acid substitutions are mapped to key branches. Yamagata-lineage (blue), Victoria-lineage (green) are indicated.
Phylogenetic analysis showed that all HA of Victoria-lineage were classified to clade 1. A portion of Victoria-lineage from 2011–2012 season was related to strain B/Brisbane/60/2008 and classified to clade 1B which had amino acids substitutions at L58P (8/8, 100%). All Victoria-lineage virus in 2011–2012 surveillance season shared amino acids substitutions at I146V (21/21), whereas Victoria-lineage virus in 2015–2017 surveillance seasons shared amino acids substitutions at I117V (23/29, 79.31%), N129D (29/29, 100%). They classified to clade 1 A within different inter-clades compared with vaccine strain B/Brisbane/60/2008.
Phylogenetic analysis of NA and HA showed Yamagata-Victoria inter-lineage reassortment occurred in 2013–2014 season (Fig. 5). HA of Yamagata-lineage in 2013–2014 season and NA of Victoria-lineage in 2011–2012 season formed new strains. The NA of Yamagata-lineage fall in clade 3. The Yamagata-lineage was the prevail lineage in 2014–2015 season and amino acids substitutions I45V, A62T, L73P of NA protein showed in most strains comparing to strain B/Wisconsin/1/2010 (Tables 1, 2). NA of Victoria-lineage classified into clade 1 and part of strains in 2011–2012 season developed an inter-clade different to 2015–2017 seasons. Comparing to strain B/Brisbane/60/2008, S41P, P42S substitutions in NA protein were showed in Victoria-lineage of 2011–2012 season, then strains of 2015–2017 seasons showed special amino acids substitutions I45T, I120V, D384G in NA protein.
Phylogenetic tree of the NA gene sequences of influenza B viruses. NA sequences of influenza B viruses were compared with WHO recommended candidate vaccine (▲) and reference strains. The phylogeny was reconstructed using Neighbor-Joining (NG) method. Bootstrap values (>70%) and amino acid substitutions are mapped to key branches. Yamagata-lineage (blue), Victoria-lineage (green) are indicated.
Antigenic analysis of HA1 region of Yamagata- and Victoria-lineage comparing to vaccine strain
Some receptor binding or in the antibody recognition in HA1 regions were observed in the 120 loop (position 116–137), the 150 loop (position 141–150), the 160 loop (162–167) and the helix 190 (194–202)20,21. Aligned HA1 of Yamagata-lineage to reference influenza B vaccine strain (B/Massachusetts/2/2012), only N116K substitutions were found in 2013–2015 seasons was on the 120 loop. Aligning HA of Victoria-lineage virus in HA1 region is different between 2011–2012 and 2015–2017 surveillance seasons when Victoria-lineage was predominant. The N129S and I146V substitutions were specially identified in 120 loop and 150 loop of HA1 region of Victoria-lineage in 2011–2012 surveillance season. Whereas, I117V and N129D substitutions were characteristically showed in 120 loop of HA1 region of Victoria-lineage in 2015–2017 surveillance seasons (Table 3).
Discussion
In this study we described the epidemiology and phylodynamics of influenza B viruses during 2011–2017 surveillance seasons in Chaoyang District in Beijing. Epidemiological results of surveillance outpatients of ILI showed influenza B virus is more likely to infect people under 17 years old, who were preschool and school-aged children. The finding is consistent to the surveillance data of influenza B virus during 2009–2014 seasons in Shanghai and results from several community-based cohort studies that found that children were more frequently infected with B viruses than adults22. Influenza B contributed to a higher proportion (41.9%) among children and young teenagers and were associated with severe disease especially myocardial injury in children, which is observed in almost 70% of fatal cases23,24,25. Despite elementary and high school students can inoculate free influenza vaccines in Beijing, a recent report showed that the average coverage rate of TIV among students of the 43 schools in Beijing was 47.6% in 2014–2015 and children who received both 2013–2014 and 2014–2015 vaccinations had vaccine effectiveness of 29%26. VE against influenza B was higher among vaccinated individuals with no previous vaccination history was 75% compared with vaccinated individuals with a frequent vaccination history was 48% from 8 seasons27. Repeated previous vaccinations over multiple seasons had dose-dependent negative impacts on VE against both influenza A and B virus28. By this token, repeated vaccinations may low the VE of influenza vaccine. Contradictorily, the free influenza vaccine police may not the best protection to prevent infection of influenza B to school children. Further studies to confirm this finding are necessary.
Phylogenetic analysis of nucleotide and amino acids of influenza B virus is important in the assessment of antigenic evolvement. The evolution of the influenza B virus is driven by mutations and under positive selective pressure on the membrane-distal domain of HA1 in specific domains such as four epitopes, including loop-120, loop-150, loop-160 and helix 19029. Characterizing amino acid substitutions that occur along the trunk of Yamagata- and Victoria-lineage gene phylogenies, identifies changes that become fixed in the virus population across seasons. As expected, substitution N116K was key mutation in 2013–2015 seasons and caused Yamagata-lineage to be absolute prevalence. This mutation previously described to circulate in Italian in 2011–201530. We also observed the 120-loop and 150-loop of HA1 of Victoria-lineage showed different mutations in 2011–2012 season and 2015–2017 seasons. The mutations of the antigenic site of HA1 make Victoria-lineage to be the absolute epidemic strain of these periods. Interestingly, we observed Yamagata/Victoria inter-lineage reassortment with HA of Yamagata-lineage and NA of Victoria-lineage, which was dominant in 2013–2014 seasons. The reassortment was reported in other researches and caused Yamagata-lineage to be dominated strain31. In conclusion, one or multiple mutation of four epitopes and subtypes reassortment may cause preponderant strain.
At the moment, TIV is still the effective measure to prevent infections and severe illnesses caused by influenza virus in the world32,33. However, the protective effect of vaccination is very weak34. The overall influenza VE for school children during the influenza season of 2016–2017 was 20.6% and estimated to 32% among risk groups and was 11% among the elderly35. The 3-seasons adjusted VE in preventing hospitalization as determined in a case-control study was 52% for influenza A and 28% for influenza B36. The reasons may consist three items for low protective rate. Firstly, TIV only include one lineage of influenza B and disparities from antigenic mismatches between the predominant circulating influenza B lineage in a given year and that year’s seasonal influenza trivalent vaccine. Our data showed 31.20% of infected by influenza B virus was mismatched to recommended influenza B vaccine strain. Recent surveillance data from the United States and Europe between 2001 and 2011 showed that 46–58% of mismatching between the vaccine and the predominant circulating lineage of influenza B virus18. Vaccine mismatch in 2015–2016 influenza season in Beijing was associated with an increased number of influenza school outbreaks26. Secondly, pathogenic diversity generated by shift and drift prevent vaccine strains from producing protective antibodies against epidemic strains4,21. The constant genetic and antigenic changes of influenza virus render them the ability to evade host immune system, thus limiting the vaccine effectiveness. Thirdly, cross-lineage serum antibodies were not detected on ferrets who were challenged with Victoria-lineage or Yamagata-lineage virus, though a significantly lower level of heterologous challenge virus in the respiratory tract was observed37. Hemagglutination-inhibition antibody titers indicate antibody responses against HA, which is not cross-reactive, and do not protect against mismatching influenza strains38. So, the need for development of quadrivalent influenza vaccines (QIV) containing Yamagata-lineage and Victoria-lineage antigens or engineered synthetic vaccine to encode two novel and broadly cross-protective monoclonal antibodies targeting influenza A and B are imperative33,39,40. The first QIV (a LAIV formulation) entered the market in 2012, and several other QIV formulations based on inactivated vaccine formulations, such as split and subunit formulations, have been licensed. The VE of QIV showed an immunogenicity and safety profile of the vaccine comparable with the two licensed trivalent vaccines containing the same strains41.
In conclusion, influenza virus that circulated in Chaoyang District in Beijing during the influenza seasons 2011–2017 evolved multiple antigenic epitope mutations, reassortment and belonged to different inter-clade comparing to recommended vaccine of influenza B. The epidemic strain of influenza B was the Victoria lineage mismatching to the vaccine strain Yamagata lineage. Though the free influenza vaccine for school-age children in Beijing, pre-school and primary school students still was in high infection by influenza B virus. The reasons may explained the phenomenon including antigenic variation, mismatching strains and repeated vaccination. So, the introduction of the QIV and broad-immunity vaccine are needed and the free vaccine policy in Beijing need to be revalued.
Materials and Methods
Ethics statement
The study was approved by the Ethics Review Committee of Chaoyang District Center for Disease Prevention and Control. All patients signed informed consent before the samples were collected. If the participants were children, their guardians were informed the consent.
Sample collection
According to Influenza Surveillance Program for Beijing, the enrolment criteria for surveillance cases included: (a) outpatients were as ILI cases defined as fever with armpit temperature ≥38 °C accompanied by one of the general symptoms (i.e., cough, pharyngalgia or nasal congestion; (b) the clinical symptoms appeared at last 3 days; (c) patients have no antiviral treatment42. Forty nasopharyngeal or throat swabs were collected from the enrolled patients by 2 sentinel hospitals including China-Japan Friendship Hospital and Beijing Chao-Yang Hospital every week. Specimens were kept in 3 mL virus transport medium at 4–8 °C and sent to the network laboratory of Chaoyang District Center for Disease Prevention and Control within 24 h. The epidemiological information about the patients was input into the Chinese influenza Surveillance Net.
Subtypes identification of influenza virus
Viral nucleic acids of all samples were extracted using QIAamp Viral RNA Mini Kit (Qiagen, Germany) according to the manufacturer’s instructions within 24 h after receiving the specimens and typed by real-time RT-PCR according to Technical Guidelines for National Influenza Surveillance by Chinese National Influenza Centre (http://www.chinaivdc.cn/cnic/fascc/201802/t20180202_158592.htm). Specimens were tested for influenza A and B viruses, then the positive samples were subtyped into A (H1N1) pdm09, H3N2, Victoria-lineage and Yamagata-lineage. Nasopharyngeal swabs which were positive for influenza viruses were cultured in Madin–Darby canine kidney cells for 4 to 5 days. The hemagglutination assay (HA) of cell culture was performed using 1% guinea pig erythrocyte. The culture medium with HA titer ≥1:8 was identified by hemagglutination inhibition method for subtypes of influenza virus.
Sequencing and phylogenetic analyses of HA and NA of influenza B
Three (range 0–4) viral isolates identified as influenza B per month during 2011–2017 were randomly selected for sequencing. Viral nucleic acids were extracted from cell culture and the complete fragment of hemagglutinin (HA) gene and neuraminidase (NA) gene were sequenced. Specific primers used for target HA and NA gene amplification by RT-PCR were designed (Table S4), referencing to the HA and NA gene of strain B/Yamagata/16/88. The RT-PCR was performed using the One Step RT-PCR Kit (Qiagen, Germany) according to the handbook. The PCR products were analyzed using QIAxcel capillary electrophoresis (Qiagen, Germany) and sequenced by ABI 3730 DNA sequencer (PE-Applied Biosystems, Foster City, CA).
Multiple sequence alignment was performed using Clustal W (version 1.83). Genome sequences included in this study were submitted in the Global Initiative on Sharing Avian Influenza Data (GISAID)/GISAID accession numbers: EPI1153147-EPI1153378 (Table S4). A total of 83 HA and 83 NA sequences derived ILI outpatients from 2011 to 2017 were analyzed and 60 strains were downloaded from NCBI and GISAID using as reference strains in this study. The phylogenetic trees were constructed using MEGA software (version 6.06) applying the neighbor-joining method with 1000 bootstrap replicates.
Statistical analysis
Data was analyzed using SPSS (Version 18.0; SPSS Inc., Chicago, IL, USA) and graphs drawing were used with GraphPad Prism software (Version 5.01; GraphPad, La Jolla, CA, USA). Difference between groups was evaluated using Pearson χ2 test or Fisher exact test.
References
A revision of the system of nomenclature for influenza viruses: a WHO memorandum. Bull World Health Organ 58, 585–591 (1980).
Dowdle, W. R., Galphin, J. C., Coleman, M. T. & Schild, G. C. A simple double immunodiffusion test for typing influenza viruses. Bull World Health Organ 51, 213–215 (1974).
Ferguson, L. et al. Influenza D Virus Infection in Feral Swine Populations, United States. Emerg Infect Dis 24, 1020–1028 (2018).
Bedford, T. et al. Global circulation patterns of seasonal influenza viruses vary with antigenic drift. Nature 523, 217–220 (2015).
Chen, J. M. et al. Exploration of the emergence of the Victoria lineage of influenza B virus. Arch Virol 152, 415–422 (2007).
Bodewes, R. et al. Recurring influenza B virus infections in seals. Emerg Infect Dis 19, 511–512 (2013).
Kwong, J. C. et al. Acute Myocardial Infarction after Laboratory-Confirmed Influenza Infection. N Engl J Med 378, 345–353 (2018).
Wang, H. et al. Influenza associated mortality in Southern China, 2010–2012. Vaccine 32, 973–978 (2014).
Francis, T. Jr. A New Type of Virus from Epidemic Influenza. Science 92, 405–408 (1940).
Rota, P. A. et al. Cocirculation of two distinct evolutionary lineages of influenza type B virus since 1983. Virology 175, 59–68 (1990).
Hay, A. J., Gregory, V., Douglas, A. R. & Lin, Y. P. The evolution of human influenza viruses. Philos Trans R Soc Lond B Biol Sci 356, 1861–1870 (2001).
Langat, P. et al. Genome-wide evolutionary dynamics of influenza B viruses on a global scale. PLoS Pathog 13, e1006749 (2017).
Nakagawa, N., Kubota, R., Maeda, A. & Okuno, Y. Influenza B virus victoria group with a new glycosylation site was epidemic in Japan in the 2002–2003 season. J Clin Microbiol 42, 3295–3297 (2004).
Ambrose, C. S. & Levin, M. J. The rationale for quadrivalent influenza vaccines. Hum Vaccin Immunother 8, 81–88 (2012).
Weir, J. P. & Gruber, M. F. An overview of the regulation of influenza vaccines in the United States. Influenza Other Respir Viruses 10, 354–360 (2016).
Yang, J. et al. Seasonal influenza vaccination in China: Landscape of diverse regional reimbursement policy, and budget impact analysis. Vaccine 34, 5724–5735 (2016).
Lv, M. et al. The free vaccination policy of influenza in Beijing, China: The vaccine coverage and its associated factors. Vaccine 34, 2135–2140 (2016).
Qin, Y. et al. Influenza vaccine effectiveness in preventing hospitalization among Beijing residents in China, 2013–15. Vaccine 34, 2329–2333 (2016).
Chen, R. & Holmes, E. C. The evolutionary dynamics of human influenza B virus. J Mol Evol 66, 655–663 (2008).
Wang, Q., Cheng, F., Lu, M., Tian, X. & Ma, J. Crystal structure of unliganded influenza B virus hemagglutinin. J Virol 82, 3011–3020 (2008).
Ni, F., Kondrashkina, E. & Wang, Q. Structural basis for the divergent evolution of influenza B virus hemagglutinin. Virology 446, 112–122 (2013).
Zhao, B. et al. Epidemiological study of influenza B in Shanghai during the 2009-2014 seasons: implications for influenza vaccination strategy. Clin Microbiol Infect 21, 694–700 (2015).
Chan, P. K. et al. Influenza B lineage circulation and hospitalization rates in a subtropical city, Hong Kong, 2000–2010. Clin Infect Dis 56, 677–684 (2013).
Paddock, C. D. et al. Myocardial injury and bacterial pneumonia contribute to the pathogenesis of fatal influenza B virus infection. J Infect Dis 205, 895–905 (2012).
Gentile, A. et al. Influenza virus: 16 years’ experience of clinical epidemiologic patterns and associated infection factors in hospitalized children in Argentina. PLoS One 13, e0195135 (2018).
Zhang, L. et al. Influenza Vaccine Effectiveness in Preventing Influenza Illness Among Children During School-based Outbreaks in the 2014–2015 Season in Beijing, China. Pediatr Infect Dis J 36, e69–e75 (2017).
Cheng, A. C. et al. Repeated Vaccination Does Not Appear to Impact Upon Influenza Vaccine Effectiveness Against Hospitalization With Confirmed Influenza. Clin Infect Dis 64, 1564–1572 (2017).
Saito, N. et al. Dose-Dependent Negative Effects of Prior Multiple Vaccinations against Influenza A and Influenza B among School Children: A Study of Kamigoto Island in Japan during the 2011/12, 2012/13 and 2013/14 Influenza Seasons. Clin Infect Dis (2018).
Shen, J., Kirk, B. D., Ma, J. & Wang, Q. Diversifying selective pressure on influenza B virus hemagglutinin. J Med Virol 81, 114–124 (2009).
Tramuto, F. et al. The Molecular Epidemiology and Evolutionary Dynamics of Influenza B Virus in Two Italian Regions during 2010-2015: The Experience of Sicily and Liguria. Int J Mol Sci 17, 549 (2016).
Oong, X. Y. et al. Whole-Genome Phylogenetic Analysis of Influenza B/Phuket/3073/2013-Like Viruses and Unique Reassortants Detected in Malaysia between 2012 and 2014. PLoS One 12, e0170610 (2017).
Jamotte, A., Chong, C. F., Manton, A., Macabeo, B. & Toumi, M. Impact of quadrivalent influenza vaccine on public health and influenza-related costs in Australia. BMC Public Health 16, 630 (2016).
Soema, P. C., Kompier, R., Amorij, J. P. & Kersten, G. F. Current and next generation influenza vaccines: Formulation and production strategies. Eur J Pharm Biopharm 94, 251–263 (2015).
Demicheli, V., Jefferson, T., Ferroni, E., Rivetti, A. & Di Pietrantonj, C. Vaccines for preventing influenza in healthy adults. Cochrane Database Syst Rev 2, CD001269 (2018).
Souty, C. et al. Early estimates of 2014/15 seasonal influenza vaccine effectiveness in preventing influenza-like illness in general practice using the screening method in France. Hum Vaccin Immunother 11, 1621–1625 (2015).
Sugaya, N. et al. Three-season effectiveness of inactivated influenza vaccine in preventing influenza illness and hospitalization in children in Japan, 2013-2016. Vaccine 36, 1063–1071 (2018).
Kiseleva, I. et al. Cross-Protective Efficacy of Monovalent Live Influenza B Vaccines against Genetically Different Lineages of B/Victoria and B/Yamagata in Ferrets. Biomed Res Int 2018, 9695628 (2018).
Laurie, K. L. et al. Evidence for Viral Interference and Cross-reactive Protective Immunity Between Influenza B Virus Lineages. J Infect Dis (2018).
Belshe, R. B. The need for quadrivalent vaccine against seasonal influenza. Vaccine 28(Suppl 4), D45–53 (2010).
Elliott, S. T. C. et al. DMAb inoculation of synthetic cross reactive antibodies protects against lethal influenza A and B infections. NPJ Vaccines 2, 18 (2017).
Graaf, H. & Faust, S. N. Fluarix quadrivalent vaccine for influenza. Expert Rev Vaccines 14, 1055–1063 (2015).
Fang, Q. et al. Molecular epidemiology and evolution of influenza A and B viruses during winter 2013–2014 in Beijing, China. Arch Virol 160, 1083–1095 (2015).
Acknowledgements
The authors would like to thank two Sentinel hospitals that collected clinical specimens used in these studies, including the influenza-like illness (ILI) surveillance system of CNIC. The authors gratefully acknowledge Yong Zhang, Ke-qiang Huang, Wen-bing Chen.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Ling-li Sun, Shu-ming Li. Performed the experiments: Na Lei, Hai-bin Wang, Yu-song Zhang, Yi Zhong, Yuan-jie Wang. Analyzed the data: Na Lei, Qiang Sun, Lei Yang. Contributed reagents/materials/analysis tools: Hai-bin Wang, Jian-hong Zhao, Ling-li Sun, Shu-ming Li, Yue-long Shu. Wrote the paper: Na Lei. Revised the paper: Ling-li Sun, Shu-ming Li. Li-yong Huang, Jian-xin Ma contributed to sample collection and data collection.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lei, N., Wang, Hb., Zhang, Ys. et al. Molecular evolution of influenza B virus during 2011–2017 in Chaoyang, Beijing, suggesting the free influenza vaccine policy. Sci Rep 9, 2432 (2019). https://doi.org/10.1038/s41598-018-38105-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-38105-1
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.