Abstract
Converging evidence supports the involvement of pro-inflammatory pathways and the gut microbiome in major depressive disorder (MDD). Pre-clinical and clinical studies suggest that decreasing pro-inflammatory signaling may provide clinical benefit in MDD. In this study, we used the chronic unpredictable stress (CUS) paradigm to assess whether mice lacking the pro-inflammatory caspase 1, interferon gamma-receptor, and nitric oxide synthase (Casp1, Ifngr, Nos2)−/− present altered depressive- and anxiety-like behaviour at baseline and in response to CUS. In comparison to wild-type (wt) mice, (Casp1, Ifngr, Nos2)−/− mice displayed decreased depressive- and anxiety-like behaviour, and increased hedonic-like behaviour and locomotor activity at baseline, and resistance to developing anhedonic-like behaviour and a heightened emotional state following stress. Plasma levels of ACTH and CORT did not differ between the triple knockout and wt mice following stress. The faecal microbiome of (Casp1, Ifngr, Nos2)−/− mice differed from that of wt mice at baseline and displayed reduced changes in response to chronic stress. Our results demonstrate that simultaneous deficit in multiple pro-inflammatory pathways has antidepressant-like effects at baseline, and confers resilience to stress-induced anhedonic-like behaviour. Moreover, accompanying changes in the gut microbiome composition suggest that CASP1, IFNGR and NOS2 play a role in maintaining microbiome homeostasis.
Similar content being viewed by others
Introduction
Increasing evidence implicates neuroinflammatory pathways in the development, treatment response, and remission of MDD1,2. Dysregulation of three major inflammatory systems is evident in those patients: (a) increased oxidative stress by means of nitric oxide (NO) overproduction, driven by NOS2 (NO synthase 2 or inducible NOS)3,4, (b) low-grade chronic pro-inflammatory status driven by CASP1 overproduction5,6, and (c) INFG over production driven by Th1 lymphocytes7,8,9.
Each of these pathways has been investigated in isolation as an antidepressant-like strategy for MDD10,11,12. However, the complex nature of MDD pathophysiology and the potential interplay between pathways suggests that multi-targeted pharmacotherapeutic approaches might provide more significant benefit than isolated pathway approaches. To investigate the effects of multiple pro-inflammatory mediator deficiencies at baseline and post-stress behaviour, we generated a triple knockout mouse model lacking Casp1, Ifngr and Nos2 (Casp1, Ifngr, Nos2)−/−, and assessed them in a pre-clinical paradigm of stress-induced depressive-like behaviour.
Immune regulation contributes to the maintenance of gut microbiome homeostasis13. Therefore, changes in inflammatory states are likely to give rise to shifts in gut microbiota composition, potentially contributing to MDD development through a variety of pathways10,14,15. Also, emerging studies implicate the gut microbiota in the regulation of stress responses16,17, while depressed patients display abnormal faecal microbiota composition associated with heightened inflammatory signalling5,7,18,19,20,21.
We therefore further assessed whether: 1) the (Casp1, Ifngr, Nos2)−/− model is associated with an altered basal gut microbiota composition compared with wt, and 2) (Casp1, Ifngr, Nos2)−/− mice are protected from stress-induced shifts in gut microbiome composition, due to deficits on mediating inflammatory pathways.
Results
Casp1, Ifngr and Nos2 deficiency decreases depressive-like and anxiety-like behaviour
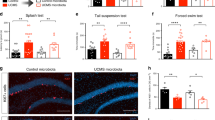
Total floating time in the forced swim test was lower in (Casp1, Ifngr, Nos2)−/− mice compared to wt mice (F1,34 = 14.618, P = 0.001) (Fig. 1a and Supplementary Table 1). Swimming and climbing behaviours were increased in (Casp1, Ifngr, Nos2)−/− mice compared to wt mice (respectively F1,34 = 25.256, P = 0.001, Fig. 1b and F1,34 = 5.929, P = 0.020, Fig. 1c). Similarly, preference for a 1% sucrose solution in the sucrose preference test was increased in the (Casp1, Ifngr, Nos2)−/− genotype compared to wt mice (F1,34 = 23.331, P < 0.001) (Fig. 1d). Moreover, (Casp1, Ifngr, Nos2)−/− mice spent more time in the open arms of the elevated plus maze compared to wt mice (F1,34 = 15.480, P < 0.001) (Fig. 2a) and displayed an increased open/closed arms entries ratio (F1,34 = 17.302, P < 0.001); in contrast, no differences were observed in the total time spent in the centre of the open field arena between (Casp1, Ifngr, Nos2)−/− and wt mice (F1,34 = 0.200, P = 0.658) (Fig. 2b) and in the ratio centre/total distance in the open field test (F1,34 = 3.330, P = 0.077). In the open field test, the (Casp1, Ifngr, Nos2)−/− genotype was associated with a higher number of faecal boli (F1,34 = 4.128, P = 0.050) (Fig. 2c).
Casp1, Ifngr and Nos2 deficiency decreases depressive-like behaviour. (a) (Casp1, Ifngr, Nos2)−/− mice displayed decreased floating time in the forced swim test in comparison to wild-type (wt) mice while displaying (b) increased swimming and (c) climbing behaviours. Moreover, (d) (Casp1, Ifngr, Nos2)−/− mice displayed an increased preference for a 1% sucrose solution. Data are presented as means ± s.e.m. Genotype effect *P < 0.05; **P < 0.01; ***P < 0.001; stress effect +P < 0.05; ++P < 0.01; +++P < 0.001; genotype*stress effect #P < 0.05; ##P < 0.01; ###P < 0.001. wt = wild-type; BL = baseline; CUS = after unpredictable chronic stress paradigm; wt n = 16; (Casp1, Ifngr, Nos2)−/− n = 20.
Casp1, Ifngr and Nos2 deficiency decreases anxiety-like behaviour. (a) (Casp1, Ifngr, Nos2)−/− mice displayed increased time spent in the open arms of the elevated plus maze but (b) similar time in the centre area of the open field test. The (Casp1, Ifngr, Nos2)−/− genotype had a main effect on (c) the number of defecations during the open field test, which was driven by the increased number of faecal boli in (Casp1, Ifngr, Nos2)−/− mice following CUS, while this parameter remained unchanged in wt mice. Data are presented as means ± s.e.m. Genotype effect *P < 0.05; **P < 0.01; ***P < 0.001; stress effect +P < 0.05; ++P < 0.01; +++P < 0.001; genotype*stress effect #P < 0.05; ##P < 0.01; ###P < 0.001; wt = wild-type; BL = baseline; CUS = chronic unpredictable stress paradigm; wt n = 16; (Casp1, Ifngr, Nos2)−/− n = 20.
Casp1, Ifngr and Nos2 deficiency affects the response to chronic unpredictable stress (CUS)
Following 28 days of CUS, the preference for a 1% sucrose solution in the sucrose preference test varied as a function of genotype (F1,34 = 17.485, P < 0.001) (Fig. 1d). Wt mice showed a ~74% decrease in sucrose preference compared to baseline (F1,34 = 57.25 P < 0.001), while (Casp1, Ifngr, Nos2)−/− mice showed a decrease of ~16% (F1,34 = 4.78, P = 0.036), reaching an after-stress sucrose preference of 74.2%.
In the open field test, a significant stress-genotype interaction was observed for total distance travelled (F1,34 = 11.091, P = 0.002) (Fig. 3a). This result was driven by the 17.5% decrease in locomotor activity in (Casp1, Ifngr, Nos2)−/− mice following stress (F1,34 = 24.68, P < 0.001), while the distance travelled by wt mice was unchanged (F1,34 = 0, P = 0.981) compared to baseline. Similarly, a significant stress-genotype interaction was observed for the average moving velocity in the open field test (F1,34 = 11.154, P = 0.002) (Fig. 3b). While the average velocity recorded for wt mice was unchanged (F1,34 = 0, P = 0.979) following CUS, (Casp1, Ifngr, Nos2)−/− mice displayed a statistically significant 17.6% reduction (F1,34 = 24.80, P < 0.001). A significant stress-genotype interaction was recorded for the number of faecal boli in the open field test (F1,34 = 14.285, P < 0.001) (Fig. 2c). While this measure did not differ significantly between genotypes at baseline (F1,34 = 2.11, P = 0.155), and was unchanged in wt mice following chronic stress (F1,34 = 0.58, P = 0.453), it was increased in (Casp1, Ifngr, Nos2)−/− mice following stress (68.4% increase, F1,34 = 23.23, P < 0.001).
Casp1, Ifngr and Nos2 deficiency increases locomotor activity. (a) (Casp1, Ifngr, Nos2)−/− mice displayed increased locomotor activity and (b) average moving velocity in the open field test. Moreover, (c) (Casp1, Ifngr, Nos2)−/− genotype had a main effect on the total number of entries in any of the arms of the elevated plus maze, irrespective of them being open or closed. All those parameters showed a stress-genotype interaction, with a common trend of being decreased as a result of Casp1, Ifngr and Nos2 deficiency following chronic unpredictable stress (CUS). Data are presented as means ± s.e.m. Genotype effect *P < 0.05; **P < 0.01; ***P < 0.001; stress effect +P < 0.05; ++P < 0.01; +++P < 0.001; genotype*stress effect #P < 0.05; ##P < 0.01; ###P < 0.001; wt = wild-type mice; BL = baseline; wt n = 16; (Casp1, Ifngr, Nos2)−/− n = 20.
Casp1, Ifngr and Nos2 deficiency increases locomotor activity
Casp1, Nos2 and Ifngr deficiency was associated with increased locomotor activity (F1,34 = 58.883, P < 0.001; Fig. 3a) and average moving velocity (F1,34 = 58.777, P < 0.001; Fig. 3b) in the open field test in comparison to wt mice. Furthermore, the number of centre visits but not the total time (Fig. 2b) spent in the centre was increased in (Casp1, Ifngr, Nos2)−/− mice (respectively F1,34 = 35.424, P < 0.001 and F1,34 = 0.200, P = 0.658) in the open field test. Moreover, (Casp1, Ifngr, Nos2)−/− mice displayed increased number of entries in the closed arms of the elevated plus maze (F1,34 = 64.426, P < 0.001) and in the total number of entries in any of the arms, irrespective of whether they were open or closed (F1,34 = 20.348, P < 0.001) (Fig. 3c).
Casp1, Ifngr and Nos2 deficiency does not affect ACTH and CORT plasma levels following stress
At the endpoint of the experiment, Casp1, Ifngr and Nos2 deficiency was not associated with altered plasma levels of ACTH (t2,25 = 0.1465, P = 0.8847) (Fig. 4a) or CORT (t2,28 = 0.3851, P = 0.7031) (Fig. 4b) when compared to wt mice.
Casp1, Ifngr and Nos2 deficiency does not affect the levels of adrenocorticotropic hormone and corticosterone following chronic unpredictable stress. (a) (Casp1, Ifngr, Nos2)−/− mice displayed similar levels of adrenocorticotropic hormone (ACTH), and (b) corticosterone (CORT) compared to wild-type (wt) mice at euthanasia. Data are presented as means ± s.e.m. n.s. = not significant. ACTH wt n = 14, (Casp1, Ifngr, Nos2)−/− n = 13; CORT wt n = 12, (Casp1, Ifngr, Nos2)−/− n = 18.
CUS affects anxiety- and depressive-like behaviour
28-day CUS exposure exacerbated depressive-like behaviour, as measured by increased floating (F1,34 = 4.299, P = 0.046; Fig. 1a) and decreased climbing behaviours in the forced swim test (F1,34 = 6.545, P = 0.015; Fig. 1c). Moreover, CUS induced anhedonia, as measured by a decreased preference for a 1% sucrose solution in the sucrose preference test (F1,34 = 50.384, P < 0.001; Fig. 1d). Furthermore, CUS increased anxiety-like behaviour, as it decreased the time spent in the open arms of the elevated plus maze (F1,34 = 10.423, P = 0.003; Fig. 2a), decreased the open/closed arms entries ratio (F1,34 = 15.595, P < 0.001) and decreased the time spent in the centre section of the open field test arena (F1,34 = 12.583, P < 0.001; Fig. 2b). However, no differences were observed in the centre distance/total distance ratio in the open field test (F1,34 = 2.442, P = 0.127).
Casp1, Ifngr and Nos2 deficiency affects gut microbiome composition
To determine whether Casp1, Ifngr and Nos2 deficiency influenced the gut microbiota composition, faecal samples were analysed by 16S rRNA sequencing (Supplementary Fig. 2). Alpha diversity analysis indicated that the faecal microbial community of (Casp1, Ifngr, Nos2)−/− mice had significantly increased richness (number of bacterial taxa observed), evenness and diversity when compared to wt (P < 0.0001) (Supplementary Fig. 3). Besides, the faecal microbiota composition of wt and (Casp1, Ifngr, Nos2)−/− mice differed significantly, as determined by permutational ANOVA (PERMANOVA) analysis (P(perm) = 0.0001, Pseudo-F = 38.22, 9950 permutations).
Comparison of genus level relative abundances between the groups using LEfSe analysis indicated that the relative abundance of several genera within the Lachnospiraceae family including Anaerofustis, Marvinbryantia, and Roseburia, as well as Ruminicococcus and Ruminiclostridium, Streptococcus, Dialister, and Veillonella were significantly higher in (Casp1, Ifngr, Nos2)−/− mice (Fig. 5). Significantly higher relative abundances were also observed for other genera, including Bacteroides, Odoribacter, Prevotellaceae UCG-001, and Alistipes in (Casp1, Ifngr, Nos2)−/− mice. The relative abundance of S24-7 (a member of the order Bacteroidales) was significantly lower in (Casp1, Ifngr, Nos2)−/− mice. Also, the relative abundance of Bifidobacterium, Allobaculum, Turicibacter, and the mucin-degrading bacterium Akkermansia were significantly lower in (Casp1, Ifngr, Nos2)−/− mice.
Taxonomic cladogram based on LEfSe analysis of bacterial taxa identified by 16S rRNA sequencing of faecal samples. Taxa that are enriched in wt and (Casp1, Ifngr, Nos2)−/− faecal samples are indicated in green and red, respectively. Differential taxa were determined based on an LDA threshold score of >3 and a statistical significance level of 0.05.
Casp1, Ifngr and Nos2 deficiency affects the change in gut microbiota composition in response to chronic stress
Assessment of the faecal microbial community at baseline and after 28 days of CUS treatment showed no significant changes in measures of richness, evenness and diversity in (Casp1, Ifngr, Nos2)−/− mice following stress (P > 0.05) (Supplementary Fig. 4). Significant increases in microbial evenness were observed for wt mice following stress (P = 0.004), but not for (Casp1, Ifngr, Nos2)−/− mice, when compared to their respective baselines. Microbial richness or diversity did not differ in either genotype when subject to CUS (P > 0.005). However, stochastic changes for microbial richness, which include significant increases in the wt control group and a significant decrease in the (Casp1, Ifngr, Nos2)−/− control group were observed. PERMANOVA analysis indicated a significant interaction between genotype, time and treatment on the faecal microbiota composition (P(perm) = 0.017, Pseudo-F = 2.978, 9939 permutations).
To determine the influence of stress on faecal microbiota, pairwise comparisons between pre- and post-treatment samples were performed for all groups. CUS for 28 days significantly altered the faecal microbiota of wt mice (P(perm) = 0.0012, t = 3.213, 9931 permutations). Significant compositional changes were also observed in control (Casp1, Ifngr, Nos2)−/− (P(perm) = 0.004, t = 3.049, 9929 permutations) and stressed (Casp1, Ifngr, Nos2)−/− mice (P(perm) = 0.010, t = 1.739, 9915 permutations). Bacterial taxa that were altered in wt or (Casp1, Ifngr, Nos2)−/− mice as a result of CUS were identified by CAP analysis. Based on CAP biplots, bacterial taxa in the opposing quadrants separating the pre- and post-chronic stress groups for the wt (Fig. 6A) or (Casp1, Ifngr, Nos2)−/− mice (Fig. 6B) were identified for further statistical analysis. The bacterial taxa Lactobacillus and S24-7 were found to contribute substantially to the compositional differences observed in wt mice following stress, while the taxa Acetatifactor, Odoribacter, Turicibacter, Ruminococcus1, Faecalibaculum, Gastranaerophilales, and Ruminococcaceae UCG-010 were found to contribute to the differences in the (Casp1, Ifngr, Nos2)−/− genotype following stress exposure. While the taxon Bifidobacterium was not identified by CAP analysis, further comparisons were also performed for this taxon due to its documented contribution to the stress response22,23.
Canonical analysis of principal coordinates (CAP) biplot constrained by time and treatment. The biplot was determined based on a Spearman’s correlation score of 0.4. Only bacterial taxa in the two opposing quadrants separating the CUS (chronic unpredictable stress) group pre- and post-treatment are indicated.
Pairwise comparison of the pre- and post-treatment relative abundances of bacterial taxa confirmed a significant reduction in the relative abundance of Lactobacillus in wt mice following stress (FDR P = 0.018), but not in (Casp1, Ifngr, Nos2)−/− mice (Fig. 7A). An increased relative abundance of Bifidobacterium (FDR P = 0.028) (Fig. 7B), Faecalibaculum (FDR P = 0.029) (Fig. 7C), Ruminococcus1 (FDR P = 0.029) (Fig. 7D) and Gastranaerophilales (FDR P = 0.036) (Fig. 7E) were observed in (Casp1, Ifngr, Nos2)−/− mice subjected to CUS, and not in the control groups. Significant decreases in the relative abundance of S24-7 were observed in wt CUS mice (FDR P = 0.018) but not in (Casp1, Ifngr, Nos2)−/− CUS mice, although increased relative abundance was observed in (Casp1, Ifngr, Nos2)−/− control mice (FDR P = 0.049) (Fig. 7F). Ruminococcaceae UCG-010 (FDR P = 0.029) and Turicibacter (FDR P = 0.029) relative abundances significantly increased in the (Casp1, Ifngr, Nos2)−/− CUS group and the control groups, independent of genotype [(Casp1, Ifngr, Nos2)−/− (FDR P = 0.048) and wt (FDR P = 0.018)].
Relative abundance of selected bacterial taxa that were identified to be differential due to chronic stress. Identification of bacterial taxa was based on the CAP biplots. Pairwise comparison between time-points was performed for each group using the Wilcoxon test based on a statistical significance level of 0.05. CUS, chronic unpredictable stress.
Discussion
Increasing evidence suggests a dysregulation of neuroinflammatory pathways and a potential alteration of gut microbiome equilibrium in MDD. We aimed to determine whether genetic deletion of Casp1, Nos2 and Ifngr affects depression- and anxiety-like behaviours in mice, either in the absence of stress or in response to CUS. Furthermore, we also determined whether faecal microbiota composition was changed as a result of altered genotype, either in isolation or when combined with chronic stress.
Mice deficient in Casp1, Ifngr and Nos2 displayed decreased depressive-like behaviour at baseline, as measured by decreased floating time in the forced swim test, and increased hedonic-like behaviour, as measured by increased sucrose preference. Moreover, (Casp1, Ifngr, Nos2)−/− mice displayed decreased levels of anxiety-like behaviour in the elevated plus maze, as they spent more time in the open arms of the maze compared to wt mice. These mice also exhibited increased locomotor activity and moving velocity.
After 28 days of CUS exposure, we observed a different stress response in (Casp1, Ifngr, Nos2)−/− mice. CUS increased floating and decreased swimming and struggling time, as reported previously24,25. While CUS decreased sucrose preference in both wt and (Casp1, Ifngr, Nos2)−/− genotypes, the decrease observed in wt mice was greater and fell below the anhedonic threshold of 65% preference, while the decrease in (Casp1, Ifngr, Nos2)−/− mice did not, suggesting that (Casp1, Ifngr, Nos2)−/− mice are protected from developing anhedonic-like behaviour following stress. In accordance with this finding, inhibition of hippocampal NOS2 was shown to prevent the decrease in sucrose preference following CUS26. Similarly, NOS2 inhibition results in antidepressant-like effects in mice while preventing the exacerbation of depressive-like behaviour11. Accordingly, it was reported that Ifng deficiency affects basal emotionality while blunting some of the behavioural and biochemical responses to chronic stressors12. Casp1−/− mice have decreased basal anxiety and depressive-like behaviour and decreased exacerbation of those behaviours following stress exposure27. Simultaneously inhibiting Casp1, Ifngr and Nos2 may result in a complex neuro-behavioural phenotype, different from individual gene knockouts, which decreases anxiety- and depressive-like behaviours at baseline, while preventing the exacerbation of anhedonic- but not of depressive- or anxiety-like behaviours following chronic stress exposure.
(Casp1, Ifngr, Nos2)−/− mice, but not wt mice, displayed a higher number of faecal pellets excreted during the open field test following stress exposure. Similar effects have been reported in germ-free mice and rats exposed to stress28,29. It has been hypothesised that such increases result from stress-induced colonic hypermotility and hyperalgesia28,29. This finding is in contrast to a previous report that wt, but not Casp1−/− mice, displayed an increased number of defecations in the open field test following chronic restraint stress, which was hypothesized to reflect stress resilience in Casp1−/− mice27. These apparently contrasting findings might be due to the different stress paradigms used in the present study and/or to the different gut microbiome composition of the (Casp1, Ifngr, Nos2)−/− genotype. Although studying a causal correlation between faecal output and gut microbiome composition was beyond the scope of this study, it cannot be excluded that changes in faecal output might be mediated by the observed changes in gut bacteria composition between genotypes.
There are many potential explanations for the increased locomotor activity displayed by (Casp1, Ifngr, Nos2)−/− mice. Pre-clinical studies have identified several systems that modulate spontaneous locomotor activity such as the dopaminergic system, the glutamatergic system, and the caspase 1 system27,30,31,32,33. Hence, it could be hypothesized that (Casp1, Ifngr, Nos2)−/− mice have increased levels of movement-related neurotransmitters, such as dopamine or glutamate. In order to test this hypothesis, future studies might interrogate monoamines levels in brain regions that are relevant to locomotion (e.g., substantia nigra and striatum) and MDD (e.g., prefrontal cortex and hippocampus) in the transgenic model used in this study. If (Casp1, Ifngr, Nos2)−/− mice indeed have increased levels of dopamine and/or glutamate, they might prove useful as pre-clinical models of Parkinson’s disease. In this disease, the main neurodegenerative features include inflammation-mediated loss of dopaminergic neurons in areas involved with movement and locomotor activity, such as the substantia nigra and the striatum34. Unsurprisingly, it has been shown that the CASP1 and CASP3 inhibitor minocycline prevents nigrostriatal dopaminergic neurodegeneration in a mouse model of Parkinson’s disease35.
Faecal microbiota analysis indicated significant compositional differences between (Casp1, Ifngr, Nos2)−/− and wt mice. Notably, rather than affecting the gut microbiota as a whole, the deletion of Casp1, Ifngr and Nos2 genes result in specific and reproducible changes in the relative abundance of discrete bacterial taxa. Previous studies have shown the effect of Casp1−/− gene knockout on gut microbiota composition36,37. These studies indicated an overrepresentation of Prevotellaceae in Casp1−/− mice, which were recapitulated in the (Casp1, Ifngr, Nos2)−/− genotype in this study. However, in contrast to our findings, the Casp1−/− mice described by Brinkman and colleagues exhibited decreased Lachnospiraceae relative abundance, while another study showed that Ifng and Ifngr-deficient mice had decreased abundance of Bacteroides/Prevotella or bifidobacteria37,38. The discrepancy in taxa relative abundances could be due to the effect on gut microbiome composition of multiple, as opposed to single, gene deletions. Previous studies have shown that deletion of genes involved in immune function and exposure to opportunistic pathogens can influence commensal microbiota composition39.
Exposure to CUS for 28 days resulted in a significant decrease in the relative abundance of Lactobacillus in wt mice, compared to baseline levels, consistent with trends demonstrated in other CUS studies40 and pre-clinical models of social stress41,42. Accordingly, decreased Lactobacillus levels have been reported in MDD patients43. Interestingly, Lactobacillus levels did not decrease in (Casp1, Ifngr, Nos2)−/− mice after CUS, suggesting that their genetic makeup may provide resistance towards Lactobacillus shifts and that this trait might be involved in their observed resilience to developing anhedonic-like behaviour. Lactobacillus has been suggested to have beneficial effects such as reducing levels of TNFA and IFNG in human intestinal epithelial cells44, and those cytokines are associated with MDD45 and pre-clinical MDD models20. Indeed, monocolonisation of germ-free mice with Lactobacillus plantarum PS128 improves anxiety-like behaviour and increases striatal serotonin and dopamine levels20, while chronic treatment with Lactobacillus rhamnosus reduces corticosterone levels in a vagal-dependent manner46.
The relative abundance of Bifidobacterium, Faecalibaculum and Ruminococcus significantly increased in (Casp1, Ifngr, Nos2)−/− mice subjected to CUS treatment but not in wt CUS mice or those in the control treatment groups. Our findings are consistent with our previous study, which showed that mice subjected to chronic restraint stress had reduced levels of Bifidobacterium27. Increased Bifidobacterium levels are associated with resilience to chronic social defeat stress, while Bifidobacterium supplementation increases resilience status in previously susceptible mice23. Monocolonisation of germ-free mice with Bifidobacterium infantis reverses the exaggerated HPA stress response observed in these mice, potentially by preventing the elevation of stress hormones such as ACTH and CORT22.
Ruminococcus and Bifidobacterium, which were increased in (Casp1, Ifngr, Nos2)−/− mice following stress, are both able to produce short chain fatty acids (SCFA)47. Similarly, Faecalibaculum was also increased and can produce lactic acid, a precursor of the SCFA butyrate48. Given that SCFA may play a role in reducing stress responses by reducing intestinal epithelial barrier permeability17, it cannot be excluded that the increase of those taxa in (Casp1, Ifngr, Nos2)−/− mice following stress might be involved in their resilience to developing anhedonic-like behaviour.
Limitations of the Study
The mouse model used in this study simultaneously lacks three genes, making the assignment of observed effects to particular proteins difficult. The rationale to generate this model stemmed from the evidence that each single knockout of these genes displays altered responses to stress or antidepressant-like phenotypes10,11,12; therefore, we hypothesized that by combining these knockouts, the effects might be additive and result in a greater antidepressant-like phenotype. In order to bypass this limitation, it might prove valuable to compare the present results with single KO mouse models for each of the genes investigated, while generating combinations of double KOs to ascertain if they have greater antidepressant-like behaviour than the respective individual KOs. Another limitation of this study is that, although the circulating levels of adrenocorticotropic hormone and corticosterone were measured following CUS, such measurements were not performed at the beginning of the stress regime. Further studies should determine whether (Casp1, Ifngr, Nos2)−/− mice present altered levels of ACTH and/or CORT at baseline. Lastly, the changes in gut microbiome observed between genotypes and stress groups are correlative nature, rather than causal.
Conclusions
In conclusion, we investigated the effects of simultaneous Casp1, Ifngr and Nos2 gene deletion in a CUS model of stress-induced depressive-like behaviour. (Casp1, Ifngr, Nos2)−/− mice displayed decreased levels of depressive- and anxiety-like behaviour while exhibiting increased locomotor activity and moving velocity. Following chronic stress exposure, (Casp1, Ifngr, Nos2)−/− mice present an attenuated anhedonic-like behaviour compared to wt mice while presenting no alterations in ACTH and CORT plasma levels compared to wt at the time of euthanasia. Correlative faecal microbiota changes were observed as a function of genotype, stress exposure, and genotype-stress interaction. Future studies are needed to investigate the clinical potential of inhibiting these pro-inflammatory signalling pathways in MDD.
Methods
All procedures were approved by the South Australian Health and Medical Research Institute (SAHMRI) Ethics committee and are in accordance with the Australian Code for the Care and Use of Animals for Scientific Purposes (8th edition, 2013). Male C57BL/6 J mice (wild-type, wt, n = 26) aged 60 days were obtained from the SAHMRI Bioresources Facility (Adelaide, Australia). Age-matched (Casp1, Ifngr, Nos2)−/− mice (n = 30) with C57BL/6 J background were generated by back-crossing mice with individual gene deletions49,50,51.
Chronic unpredictable stress
The CUS protocol consisted of 28 days exposure to randomly scheduled, low and mild intensity social and environmental stressors, applied each day during the light phase of the light cycle. Briefly, the stressors were: (a) restraint, (b) removal of bedding and nesting material, (c) soiled bedding, (d) 45° cage tilting, (e) predator stress, (f) forced swim test, (g) fasting, (h) social stress, (i) light cycle disruption and (j) light cycle reversal (for detailed procedures refer to Supplementary Table 2 and Supplementary Methods).
Behavioural testing
Mice were submitted to the open field, elevated plus maze, forced swim and sucrose preference tests at baseline and following CUS. All tests were video-recorded and analysed using Ethovision XT 10 software (Noldus, Wageningen, Holland) for behaviour recognition and scoring. See Supplementary Fig. 1 for a timeline of experimental procedures.
Adrenocorticotropic hormone (ACTH) and corticosterone (CORT) measurements
Plasma ACTH and plasma CORT level were measured respectively by a competitive inhibition ELISA kit (Cloud-Clone Corp., Wuhan, Hubei, China) and a competitive immunoassay ELISA kit (Enzo Life Sciences, Farmingdale, New York, USA) following the manufacturers’ instructions.
16S rRNA gene amplicon sequencing and bioinformatics analysis
Fresh faecal pellets were collected between 10–11 am with sterile toothpicks on experimental day 41 during weighing procedures and placed into sterile 1.5 mL Eppendorf tubes and stored at −80 °C. DNA was extracted from faecal samples using the DNeasy PowerSoil HTP 96 kit (Qiagen, Hilden, Germany) optimized for the Biomek 4000 Automation Workstation (Beckman Coulter Inc., Lane Cove, NSW, Australia). Barcoded amplicons of the V4 hypervariable region of the bacterial 16S rRNA gene were generated from the faecal DNA extracts based on the Illumina Miseq. 16S Metagenomic Sequencing Library Preparation protocol with modifications, as described previously52, and sequencing was performed on an Illumina Miseq platform. A detailed description of library preparation and sequencing are provided in the Supplementary Methods.
Bioinformatics analysis of paired-end sequences was performed using Quantitative Insights Into Microbial Ecology (QIIME, v1.9.1)53 based on a previously described bioinformatics pipeline54. Paired forward and reverse barcoded reads were quality filtered and merged using the Paired-End read merger (PEAR v0.9.10)55. Chimera filtering (USEARCH, v6.1)56 and open reference operational taxonomic unit (OTU) assignment was performed against the SILVA reference database (release 128) that were clustered at 97% identity. Only sequences with >80% identity to the reference sequence were used for OTU assignment step. All samples had spurious OTUs removed and were subsampled to the lowest sequence depth at 7,354 sequence reads, providing an average Good’s coverage score of 97.13% (±0.7%).
Statistical analysis
Power analysis was performed based on the effect size seen in a previous pilot study investigating the effects of simultaneous Casp1, Ifngr and Nos2 deficiency on total floating time in the forced swim test (our primary outcome measure). Statistical analyses of the behavioural tests were performed using the Statistical Package for the Social Sciences version 23.0 for Windows (SPSS, Chicago, Illinois) using a general linear model for repeated measures (repeated measures ANOVA). The effects of genotype, stress, treatment and their interaction were explored and the significance set at P ≤ 0.05. Statistically significant stress-genotype interactions were further assessed as described previously57. ELISA data were analysed by two-tailed unpaired student T-test. Alpha diversity scores of observed species, Simpson (1-D) and Shannon were calculated using QIIME53. Beta-diversity analyses were performed using the PRIMER software (v6, PRIMER-E Ltd, Plymouth, UK). Comparison of microbiota composition between groups was performed on weighted Unifrac distances using a permutational multivariate analysis of variance (PERMANOVA) test with 9,999 permutations. LEfSe (linear discriminant analysis effect size) analysis58 was used for the comparison of bacterial taxa relative abundances between genotypes, based on a Linear Discriminant Analysis (LDA) threshold score of 3. Specific taxa that contributed to group differences in the wt or (Casp1, Ifngr and Nos2)−/− group following stress were identified using a canonical analysis of principal coordinates (CAP) plot generated based on the factors treatment (control, CUS) and time (baseline, 28 days). Within-group paired comparisons of the relative abundances of specific taxa were further assessed using the Wilcoxon test with a false discovery rate (FDR) correction for multiple testing. The significance level was set at 0.05 for all statistical tests.
References
Leonard, B. & Maes, M. Mechanistic explanations how cell-mediated immune activation, inflammation and oxidative and nitrosative stress pathways and their sequels and concomitants play a role in the pathophysiology of unipolar depression. Neurosci Biobehav Rev 36, 764–785, https://doi.org/10.1016/j.neubiorev.2011.12.005 (2012).
Licinio, J. & Wong, M. L. The role of inflammatory mediators in the biology of major depression: central nervous system cytokines modulate the biological substrate of depressive symptoms, regulate stress-responsive systems, and contribute to neurotoxicity and neuroprotection. Mol Psychiatry 4, 317–327 (1999).
Black, C. N., Bot, M., Scheffer, P. G., Cuijpers, P. & Penninx, B. W. Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 51, 164–175, https://doi.org/10.1016/j.psyneuen.2014.09.025 (2015).
Kudlow, P., Cha, D. S., Carvalho, A. F. & McIntyre, R. S. Nitric Oxide and Major Depressive Disorder: Pathophysiology and Treatment Implications. Curr Mol Med 16, 206–215 (2016).
Alcocer-Gomez, E. et al. NLRP3 inflammasome is activated in mononuclear blood cells from patients with major depressive disorder. Brain Behav Immun 36, 111–117, https://doi.org/10.1016/j.bbi.2013.10.017 (2014).
Kaufmann, F. N. et al. NLRP3 inflammasome-driven pathways in depression: Clinical and preclinical findings. Brain Behav Immun 64, 367–383, https://doi.org/10.1016/j.bbi.2017.03.002 (2017).
Maes, M. et al. Increased neopterin and interferon-gamma secretion and lower availability of L-tryptophan in major depression: further evidence for an immune response. Psychiatry Res 54, 143–160 (1994).
Myint, A. M., Leonard, B. E., Steinbusch, H. W. & Kim, Y. K. Th1, Th2, and Th3 cytokine alterations in major depression. J Affect Disord 88, 167–173, https://doi.org/10.1016/j.jad.2005.07.008 (2005).
Inserra, A., Mastronardi, C. A., Rogers, G., Licinio, J. & Wong, M. L. Neuroimmunomodulation in Major Depressive Disorder: Focus on Caspase 1, Inducible Nitric Oxide Synthase, and Interferon-Gamma. Mol Neurobiol, https://doi.org/10.1007/s12035-018-1359-3 (2018).
Wong, M. L. et al. Inflammasome signaling affects anxiety- and depressive-like behavior and gut microbiome composition. Mol Psychiatry 21, 797–805, https://doi.org/10.1038/mp.2016.46 (2016).
Montezuma, K. et al. Inhibition of iNOS induces antidepressant-like effects in mice: pharmacological and genetic evidence. Neuropharmacology 62, 485–491, https://doi.org/10.1016/j.neuropharm.2011.09.004 (2012).
Litteljohn, D. et al. Interferon-gamma deficiency modifies the effects of a chronic stressor in mice: Implications for psychological pathology. Brain Behav Immun 24, 462–473, https://doi.org/10.1016/j.bbi.2009.12.001 (2010).
Peterson, C. T., Sharma, V., Elmen, L. & Peterson, S. N. Immune homeostasis, dysbiosis and therapeutic modulation of the gut microbiota. Clin Exp Immunol 179, 363–377, https://doi.org/10.1111/cei.12474 (2015).
Wang, Y. & Kasper, L. H. The role of microbiome in central nervous system disorders. Brain Behav Immun 38, 1–12, https://doi.org/10.1016/j.bbi.2013.12.015 (2014).
Rogers, G. B. et al. From gut dysbiosis to altered brain function and mental illness: mechanisms and pathways. Mol Psychiatry 21, 738–748, https://doi.org/10.1038/mp.2016.50 (2016).
Sudo, N. & Microbiome, H. P. A. axis and production of endocrine hormones in the gut. Adv Exp Med Biol 817, 177–194, https://doi.org/10.1007/978-1-4939-0897-4_8 (2014).
Carabotti, M., Scirocco, A., Maselli, M. A. & Severi, C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Ann Gastroenterol 28, 203–209 (2015).
Zheng, P. et al. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Mol Psychiatry 21, 786–796, https://doi.org/10.1038/mp.2016.44 (2016).
Chen, J. J. et al. Sex differences in gut microbiota in patients with major depressive disorder. Neuropsychiatr Dis Treat 14, 647–655, https://doi.org/10.2147/NDT.S159322 (2018).
Liu, Y. N. et al. TNFalpha mediates stress-induced depression by upregulating indoleamine 2,3-dioxygenase in a mouse model of unpredictable chronic mild stress. Eur Cytokine Netw 26, 15–25, https://doi.org/10.1684/ecn.2015.0362 (2015).
Inserra, A., Rogers, G. B., Licinio, J. & Wong, M. L. The Microbiota-Inflammasome Hypothesis of Major Depression. Bioessays 40, e1800027, https://doi.org/10.1002/bies.201800027 (2018).
Sudo, N. et al. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J Physiol 558, 263–275, https://doi.org/10.1113/jphysiol.2004.063388 (2004).
Yang, C. et al. Bifidobacterium in the gut microbiota confer resilience to chronic social defeat stress in mice. Sci Rep 7, 45942, https://doi.org/10.1038/srep45942 (2017).
Zhu, S., Shi, R., Wang, J., Wang, J. F. & Li, X. M. Unpredictable chronic mild stress not chronic restraint stress induces depressive behaviours in mice. Neuroreport 25, 1151–1155, https://doi.org/10.1097/WNR.0000000000000243 (2014).
Zhu, S. et al. Unpredictable chronic mild stress induces anxiety and depression-like behaviors and inactivates AMP-activated protein kinase in mice. Brain Res 1576, 81–90, https://doi.org/10.1016/j.brainres.2014.06.002 (2014).
Wang, D., An, S. C. & Zhang, X. Prevention of chronic stress-induced depression-like behavior by inducible nitric oxide inhibitor. Neurosci Lett 433, 59–64, https://doi.org/10.1016/j.neulet.2007.12.041 (2008).
Wong, M. L. et al. Inflammasome signaling affects anxiety- and depressive-like behavior and gut microbiome composition. Mol Psychiatry, https://doi.org/10.1038/mp.2016.46 (2016).
De Palma, G. et al. Microbiota and host determinants of behavioural phenotype in maternally separated mice. Nat Commun 6, 7735, https://doi.org/10.1038/ncomms8735 (2015).
Bradesi, S. et al. Repeated exposure to water avoidance stress in rats: a new model for sustained visceral hyperalgesia. Am J Physiol Gastrointest Liver Physiol 289, G42–53, https://doi.org/10.1152/ajpgi.00500.2004 (2005).
Gainetdinov, R. R., Mohn, A. R., Bohn, L. M. & Caron, M. G. Glutamatergic modulation of hyperactivity in mice lacking the dopamine transporter. Proc Natl Acad Sci USA 98, 11047–11054, https://doi.org/10.1073/pnas.191353298 (2001).
Burns, L. H., Everitt, B. J., Kelley, A. E. & Robbins, T. W. Glutamate-dopamine interactions in the ventral striatum: role in locomotor activity and responding with conditioned reinforcement. Psychopharmacology (Berl) 115, 516–528 (1994).
Swanson, C. J. & Kalivas, P. W. Regulation of locomotor activity by metabotropic glutamate receptors in the nucleus accumbens and ventral tegmental area. J Pharmacol Exp Ther 292, 406–414 (2000).
Karlsson, R. M., Tanaka, K., Heilig, M. & Holmes, A. Loss of glial glutamate and aspartate transporter (excitatory amino acid transporter 1) causes locomotor hyperactivity and exaggerated responses to psychotomimetics: rescue by haloperidol and metabotropic glutamate 2/3 agonist. Biol Psychiatry 64, 810–814, https://doi.org/10.1016/j.biopsych.2008.05.001 (2008).
McGeer, P. L. & McGeer, E. G. Inflammation and neurodegeneration in Parkinson’s disease. Parkinsonism Relat Disord 10(Suppl 1), S3–7, https://doi.org/10.1016/j.parkreldis.2004.01.005 (2004).
Du, Y. et al. Minocycline prevents nigrostriatal dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease. Proc Natl Acad Sci USA 98, 14669–14674, https://doi.org/10.1073/pnas.251341998 (2001).
Elinav, E. et al. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 145, 745–757, https://doi.org/10.1016/j.cell.2011.04.022 (2011).
Brinkman, B. M. et al. Caspase deficiency alters the murine gut microbiome. Cell Death Dis 2, e220, https://doi.org/10.1038/cddis.2011.101 (2011).
Thoene-Reineke, C. et al. Composition of intestinal microbiota in immune-deficient mice kept in three different housing conditions. PLoS One 9, e113406, https://doi.org/10.1371/journal.pone.0113406 (2014).
Galvez, E. J. C., Iljazovic, A., Gronow, A., Flavell, R. & Strowig, T. Shaping of Intestinal Microbiota in Nlrp6- and Rag2-Deficient Mice Depends on Community Structure. Cell Rep 21, 3914–3926, https://doi.org/10.1016/j.celrep.2017.12.027 (2017).
Marin, I. A. et al. Microbiota alteration is associated with the development of stress-induced despair behavior. Sci Rep 7, 43859, https://doi.org/10.1038/srep43859 (2017).
Bailey, M. T. et al. Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor-induced immunomodulation. Brain Behav Immun 25, 397–407, https://doi.org/10.1016/j.bbi.2010.10.023 (2011).
Galley, J. D. et al. Exposure to a social stressor disrupts the community structure of the colonic mucosa-associated microbiota. BMC Microbiol 14, 189, https://doi.org/10.1186/1471-2180-14-189 (2014).
Aizawa, E. et al. Possible association of Bifidobacterium and Lactobacillus in the gut microbiota of patients with major depressive disorder. J Affect Disord 202, 254–257, https://doi.org/10.1016/j.jad.2016.05.038 (2016).
Resta-Lenert, S. & Barrett, K. E. Probiotics and commensals reverse TNF-alpha- and IFN-gamma-induced dysfunction in human intestinal epithelial cells. Gastroenterology 130, 731–746, https://doi.org/10.1053/j.gastro.2005.12.015 (2006).
Mikova, O., Yakimova, R., Bosmans, E., Kenis, G. & Maes, M. Increased serum tumor necrosis factor alpha concentrations in major depression and multiple sclerosis. Eur Neuropsychopharmacol 11, 203–208 (2001).
Bravo, J. A. et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc Natl Acad Sci USA 108, 16050–16055, https://doi.org/10.1073/pnas.1102999108 (2011).
LeBlanc, J. G. et al. Beneficial effects on host energy metabolism of short-chain fatty acids and vitamins produced by commensal and probiotic bacteria. Microb Cell Fact 16, 79, https://doi.org/10.1186/s12934-017-0691-z (2017).
Lim, S., Chang, D. H., Ahn, S. & Kim, B. C. Whole genome sequencing of “Faecalibaculum rodentium” ALO17, isolated from C57BL/6J laboratory mouse feces. Gut Pathog 8, 3, https://doi.org/10.1186/s13099-016-0087-3 (2016).
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000–2003 (1995).
Laubach, V. E., Shesely, E. G., Smithies, O. & Sherman, P. A. Mice lacking inducible nitric oxide synthase are not resistant to lipopolysaccharide-induced death. Proc Natl Acad Sci USA 92, 10688–10692 (1995).
Huang, S. et al. Immune response in mice that lack the interferon-gamma receptor. Science 259, 1742–1745 (1993).
Choo, J. M., Leong, L. E. & Rogers, G. B. Sample storage conditions significantly influence faecal microbiome profiles. Sci Rep 5, 16350, https://doi.org/10.1038/srep16350 (2015).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7, 335–336, https://doi.org/10.1038/nmeth.f.303 (2010).
Jervis-Bardy, J. et al. Deriving accurate microbiota profiles from human samples with low bacterial content through post-sequencing processing of Illumina MiSeq data. Microbiome 3, 19, https://doi.org/10.1186/s40168-015-0083-8 (2015).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620, https://doi.org/10.1093/bioinformatics/btt593 (2014).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461, https://doi.org/10.1093/bioinformatics/btq461 (2010).
Kinnear, P. R. & Gray, C. D. PASW statistics 17 made simple (replaces SPSS statistics 17). 1st edn, (Psychology Press, 2010).
Segata, N. et al. Metagenomic biomarker discovery and explanation. Genome Biol 12, R60, https://doi.org/10.1186/gb-2011-12-6-r60 (2011).
Acknowledgements
This work was supported by institutional funding from the Australian National University (Canberra, ACT, Australia), and the South Australian Health and Medical Research Institute (Adelaide, Australia), and a Flinders University Faculty of Medicine and Health Sciences Grant (JL and M-LW). Institutional funding from the Australian National University supported the generation of the (Casp1, Ifngr, Nos2)−/− mouse model.
Author information
Authors and Affiliations
Contributions
M.-L.W. and J.L. conceived the experiments. G.B.R., M.-L.W. and J.L. designed the experiments and revised the manuscript. A.I. and J.C. performed the experiments, performed the data analysis and wrote the manuscript draft. M.D.L. supervised the experiments and revised the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Inserra, A., Choo, J.M., Lewis, M.D. et al. Mice lacking Casp1, Ifngr and Nos2 genes exhibit altered depressive- and anxiety-like behaviour, and gut microbiome composition. Sci Rep 9, 6456 (2019). https://doi.org/10.1038/s41598-018-38055-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-38055-8
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.