Abstract
Caribbean lionfish (Pterois spp.) are considered the most heavily impacting invasive marine vertebrate ever recorded. However, current management is largely inadequate, relying on opportunistic culling by recreational SCUBA divers. Culling efficiency could be greatly improved by exploiting natural aggregations, but to date this behaviour has only been recorded anecdotally, and the drivers are unknown. We found aggregations to be common in situ, but detected no conspecific attraction through visual or olfactory cues in laboratory experiments. Aggregating individuals were on average larger, but showed no further differences in morphology or life history. However, using visual assessments and 3D modelling we show lionfish prefer broad-scale, but avoid fine-scale, habitat complexity. We therefore suggest that lionfish aggregations are coincidental based on individuals’ mutual attraction to similar reef structure to maximise hunting efficiency. Using this knowledge, artificial aggregation devices might be developed to concentrate lionfish densities and thus improve culling efficiency.
Similar content being viewed by others
Introduction
Since their introduction in the early 1980s1, lionfish (Pterois volitans and P. miles) have spread throughout the western Atlantic with devastating impacts on the region’s coral reefs2. Successful eradications of marine invasive species are rare and generally involve species with a small invaded range3,4. Complete eradication of lionfish is considered unlikely5, as they are now well established with a large geographical distribution6 and have the potential for long distance dispersal7. Rather than eradication, management should therefore be focused on population reduction through culling8.
Lionfish culling is typically performed by recreational SCUBA divers and snorkelers using hand nets and pole spears8. This can be opportunistic or as part of organised events involving competing teams of divers and fishermen9. On shallow reefs, culling has been successful at reducing lionfish densities8,10 and allowing native species to recover11. However, whilst culling is largely restricted to depths <30 m12, lionfish are known to have invaded deeper mesophotic and sub-mesophotic habitats to at least 304 m12,13. Technical diving provides a partial solution, although cost, expertise and safety concerns will limit the extent of its use14, and lionfish traps to collect lionfish from deeper water are still being tested15.
Groups of lionfish have been observed throughout the invaded range10,16,17 and it has been noted that aggregating individuals often appear larger than solitary individuals16,17. By exploiting this natural behaviour and artificially stimulating aggregations, culling or trapping efficiency could be improved. Mass trapping of invasive and pest species has been successful in both aquatic and terrestrial ecosystems18,19,20 but requires knowledge of the species’ aggregation drivers to inform the design of effective lures to trap them. Although previous work has focused on many aspects of the lionfish invasion21, nothing is known about the drivers of lionfish aggregations, and quantitative data on their prevalence is lacking.
One possible driver of lionfish aggregations is social attraction, which may be for cooperative hunting (as observed in P. miles in its native range22), spawning23, minimising the chance of predation24 or pooling experiences to solve problems25. Social attraction in fish can be initiated by visual26, olfactory27, acoustic26 or a combination of cues28. Zebrafish will even aggregate with robotic fish that mimic their shape and movement29. Another possible aggregation driver is habitat preference. Fish may seek out specific habitat because it provides a source of prey, good hunting conditions or shelter from predators30. Marine habitat complexity can be measured via a range of metrics (e.g31,32) and is known to be a strong predictor of fish abundance33,34. Although lionfish have been anecdotally reported to be found in more complex habitat10,16,17, this has not been determined quantitatively.
Here, we provide the first quantitative assessment of lionfish aggregations and their drivers, specifically exploring: (i) morphology and life history traits of aggregating and solitary individuals via in situ population surveys and ex situ dissections, (ii) social attraction using laboratory binary choice tests focusing on visual and olfactory cues, and (iii) habitat preference and the role of habitat complexity using 3D modelling and visual scores.
Results
Aggregating lionfish are larger and heavier, but otherwise similar to solitary individuals
A total of 283 lionfish were caught and dissected for this study, comprising 178 solitary and 105 aggregating individuals. We recorded 33 aggregations of varying sizes from 2 to 7 individuals, with a mean (±1 SE) of 3 (±0.34) and a median of 2. Lionfish were collected from depths of 3.0–18.4 m, with similar mean depths (±1 SE) for solitary lionfish (10.7 ± 0.28 m) and aggregations (10.6 ± 0.52 m).
Lionfish found in aggregations were significantly larger than solitary individuals (p < 0.001) and were also heavier for their size (p < 0.001). Mean total lengths (±1 SE) were 22.2 cm (±0.55) for aggregating lionfish and 18.4 cm (±0.54) for solitary lionfish. However, there was no difference in sex ratio (p > 0.05), the gonadosomatic indices of males and females (p > 0.05 for both) or percentage body fat (p > 0.05) between solitary and aggregating individuals.
Lionfish are not attracted to conspecific visual or olfactory cues
The proportion of trials where lionfish entered the stimulus zone first did not differ significantly from chance levels for any of the stimuli tested (p > 0.05 for all stimuli; Table 1). Lionfish in the visual, visual + olfactory and artificial model treatments showed no significant preference for either stimulus or control (p > 0.05 for each; Table 1). Lionfish in the olfactory treatment showed a significant preference for the control, or avoidance of the stimulus, with a mean (±1 SE) time difference of −229 (±62) seconds.
Lionfish prefer broad-scale, rather than fine-scale, habitat complexity
Habitat assessment scores found that aggregating lionfish inhabited areas with significantly larger refuge sizes (p < 0.001), higher percentage live cover (p < 0.05) and a greater variety of growth forms (p < 0.05) than the areas inhabited by solitary lionfish (Fig. 1). There were no significant differences for rugosity or substratum height (p > 0.05 for both).
We also used 3D modelling to test for lionfish preferences towards habitat complexity. As there were no significant differences in the three complexity metrics (linear rugosity, vector dispersion and fractal dimension) between reef areas harbouring solitary lionfish and aggregations, these data sets were pooled to represent complexity with lionfish (lionfish quadrats) and compared with randomly selected areas of reef without lionfish (background quadrats).
Although there was no significant difference in linear rugosity between lionfish quadrats and background quadrats (p > 0.05; Table 2), lionfish were found in areas with significantly lower vector dispersion values (mean ± 1 SE; 0.144 ± 0.009 compared with 0.239 ± 0.007), indicating an association with low complexity at fine spatial scales (p < 0.001; Table 2). This is supported by assessment of fractal dimension across five spatial scale ranges (Table 2; Fig. 2). Lionfish quadrats were significantly less complex than background quadrats at the finest spatial scale (1–5 cm; p < 0.001). Conversely, lionfish quadrats were significantly more complex than background quadrats at the broadest spatial scales (30–60 cm and 60–120 cm; p < 0.05 for both). At intermediate spatial scales (5–15 cm and 15–30 cm) there was no significant difference. These results demonstrate an association with low complexity at fine spatial scales but high complexity at broad spatial scales.
Fractal dimension at varying spatial scales. Fractal dimension values were calculated at five spatial scales: 1–5 cm, 5–15 cm, 15–30 cm, 30–60 cm and 60–120 cm. Means (±1 SE) are plotted at the midpoint of each spatial scale range for both lionfish quadrats (orange; n = 30) and background quadrats (grey; n = 44). * indicates p < 0.05.
Discussion
Prior to this study, lionfish aggregation behaviour had only been observed anecdotally and little was known about its drivers. Through a combination of approaches and both in situ and ex situ data collection we have made a substantial contribution to this aspect of invasive lionfish research.
We found that aggregating individuals were larger than solitary individuals. Although larger female fish are often more fecund35, we found no difference in reproductive maturity between solitary and aggregating lionfish. This suggests that the aggregations observed during our study period were not for the purpose of spawning, unlike in other reef fish species36,37. Alternatively, aggregating lionfish may be larger because of higher growth rates, which may occur through cooperative hunting (which has been demonstrated to improve hunting success38) or because lionfish choose areas that maximise their individual hunting success. Cooperative hunting has been observed in another lionfish species, Dendrochirus zebra, in the laboratory38 and in P. miles in its native range22. However, in Mexico lionfish were more likely to be seen hunting when solitary than when in aggregations39. Although we found no difference in percentage body fat between aggregating and solitary individuals, we did find a difference in the value of Fulton’s condition factor, with aggregating individuals being heavier for their size. This difference may result from the benefits of cooperative hunting, or because aggregations form in areas where hunting success is higher.
Many fish that are found in groups exhibit social attraction40,41 but lionfish showed no preference for any of the cues tested here, both in initial reaction or overall preference. This is not unique; a lack of response to visual cues has been observed in other aggregating fish27. The lack of response might have been caused by differing personalities between individuals, resulting in differing motivation to aggregate. Several fish species show personality (e.g42,43) and personalities can influence social attraction43. Although we did not control for personality differences, the lionfish we caught may have been the boldest individuals44, thus minimising personality differences between individuals.
Lionfish did show a significant response in the olfactory treatment, however, their preference was for the control. This may have been due to the presence of waste products (e.g. ammonia45) in the olfactory cue, which are known to stimulate avoidance behaviour in other fish species46. Based on this result we suggest that future studies should use alternative methods of introducing olfactory cues, such as a lionfish behind an opaque, but water permeable, barrier.
Sound has a well-established role in reef fish behaviour47,48 and lionfish are known to produce sound49, therefore the use of acoustic cues for social attraction is possible. Acoustic cues were not standardised in our experiment therefore we cannot deduce whether lionfish use acoustic cues for social attraction. However, our findings do suggest that aggregating behaviour in invasive lionfish is not driven by visual or olfactory cues between conspecifics.
Previous studies provide mixed results on the association of lionfish with habitat complexity. Lionfish have been anecdotally reported from more complex habitat10,16,17 and shown to be associated with areas of greater hard coral cover and overhanging structures50. However, other studies have reported no relationship between habitat complexity and lionfish density or biomass10,51,52. By using more rigorous methods and quantifying multiple metrics of reef structure, we found a clear link between lionfish, aggregations and habitat complexity.
Firstly, our data show aggregations were associated with larger refuge sizes, higher live cover and a greater variety of growth forms. Preference of lionfish for these habitats may increase food availability; larger refuges can be associated with greater fish abundance53, increased coral cover may provide preferred habitat for larval settlement of prey species54 and a greater variety of growth forms can be associated with increased species richness31.
Lionfish habitat preference varied across different spatial scales. At finer scales (1–5 cm for fractal dimension and 1 cm for vector dispersion) lionfish were associated with lower complexity. Predatory fish may hunt less effectively in habitats that are complex at fine scales55, therefore lionfish may avoid high complexity at this scale to maximise hunting success. If this is indeed the case, lionfish hunting success may be increased because of reef flattening which is occurring throughout the Caribbean56. At broader scales (30–60 cm and 60–120 cm for fractal dimension) lionfish were associated with higher complexity that is often created by large crevices and overhangs. The erosion of broad-scale complexity as part of reef flattening may be less important to lionfish as artificial reefs such as shipwrecks and concrete structures can provide this type of complexity. This may explain why lionfish were found at higher density on artificial, rather than natural, reefs in the Gulf of Mexico57. Our study highlights the importance of considering spatial scale in assessments of species associations with habitat complexity. By creating “complexity signatures” using fractal dimension (Fig. 2) a much more detailed picture can be gained of how a species associates with both the quality and quantity of complexity32.
Caves and overhangs provide low light environments that may increase hunting success, given that lionfish prey capture rates have been shown to be higher on overcast than clear days58. The physical structure of caves and overhangs may also benefit lionfish hunting; lionfish have been observed herding prey towards vertical and concave surfaces before striking58.The inverse relationship between substratum height and availability of prey refugia59 suggests that these habitats often lack refuges for prey species. However, these habitats may provide shelter for certain prey and mutualist species. For example, overhangs provide shelter for Gramma loreto60, a fish that has been found in lionfish stomachs61,62 but that is also a potential lionfish cleaner16.
Based on these findings, we propose that aggregations of invasive lionfish are coincidental and form when multiple lionfish are attracted to the same area of reef, rather than through visual or olfactory attraction to conspecifics. This attraction to broad-scale over fine-scale complexity is likely to be driven by a search for maximised hunting efficiency, characterised by reduced refugia for prey items, leading multiple lionfish to congregate on areas of reef where feeding is easiest.
This knowledge could be used to significantly improve the efficiency of culling efforts. Our results can be used to inform the design of artificial aggregation devices or to improve existing lionfish traps that already involve some complexity15, but which could benefit from incorporating broad-scale crevices and minimising fine-scale refugia. Artificial aggregation devices and traps could in turn extend the depth limit of culling efforts to exploit the significant populations found on mesophotic reefs12,13, which are believed to include the most reproductively active individuals12. Only through improved understanding of the behaviour and ecology of this widespread invader will managers have the upper hand. By expanding and improving culling efforts using the findings of this study, a long-term solution will be one step closer.
Methods
Study sites
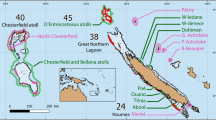
All data were collected from reefs in the mainland Tela Bay, Honduras (Fig. 3; Supplementary Table 1) between June and August 2017 using open-circuit SCUBA. Lionfish were culled for dissection at six sites on the offshore reef system of Banco Capiro at depths between 10–18 m (see Bodmer et al.63 for a general site description). Lionfish were collected alive for use in laboratory social attraction experiments from four sites on the shallow nearshore reef system of La Ensenada at depths <10 m. A recent genetic study suggests invasive lionfish in Honduras are likely to all be P. volitans64, and so we assumed this to be the case. However, we did not identify them to species level and so will refer to them simply as lionfish hereafter.
Map of our study sites in Honduras. Population structure data and habitat assessment scores were recorded from all 10 sites. Triangles mark the four sites on the La Ensenada reef system where lionfish were collected live for laboratory social attraction experiments. Circles mark the six sites on the Banco Capiro reef system where lionfish were culled and 3D modelling was carried out. The location of Tela bay, Honduras within central America is shown on the inset map.
In situ population structure surveys and ex situ dissections
Roving diver surveys were conducted, and any lionfish encountered deeper than 10 m were culled using pole spears before being transferred into a ZooKeeper container (ZooKeeper LLC, USA) for transport back to the laboratory. Approximate size, supra-ocular tentacle type and distinguishing features (e.g. unusual patterning) were noted in situ to enable later identification of individuals. Depth of capture and, if aggregating, the number of individuals within the aggregation were also recorded. An aggregation was defined here as two or more individual lionfish being found within approximately four body lengths of one another24.
On return to the laboratory, culled lionfish were refrigerated and dissected within 12 hours. Total length was recorded to the nearest mm from the tip of the snout to the longest point of the tail. Percentage body fat, which was used as a measure of feeding success, was calculated as fat weight (to the nearest 0.1 g) divided by body weight (to the nearest g), then multiplied by 100. Fat weight was obtained by removing all fat deposits from within the gut cavity and weighing these using an electronic balance. Fulton’s condition factor, which was also used as a measure of feeding success, was calculated as body weight divided by total length cubed65,66, with higher values indicating that fish were heavier for their size. Macroscopic analysis of the gonads was used to determine sex67. Lionfish reproduce throughout the year68, which enabled us to measure reproductive maturity during our study. Reproductive maturity of males and females was measured using the gonadosomatic index68, which was calculated as gonad weight (to the nearest 0.1 g) divided by body weight and then multiplied by 100. Dissection methods followed Green et al.67.
Laboratory visual and olfactory attraction experiments
Lionfish capture and experimental setup
A total of 58 lionfish were captured alive using hand nets and transferred to large plastic dry-bags, before being slowly brought to the surface. Lionfish were transported inside the dry-bags to the laboratory by 15-minute boat ride. On arrival at the laboratory lionfish were immediately transferred to the holding tank. Live collections were restricted to <10 m to avoid barotrauma injuries. Lionfish ranged in total length from 4.7–29.5 cm with a mean (±1 SE) of 17.0 (±0.88) cm. Each lionfish was used once to explore one of (i) visual cues (n = 15), (ii) olfactory cues (n = 13), (iii) visual + olfactory cues (n = 18) or (iv) artificial model cues (n = 12). Post-trial, some lionfish were used as stimulus fish for up to two trials. Stimulus fish were never re-used as test fish.
The holding and trial tanks measured 245 × 56 × 24 cm (length x width x depth) and were filled with unfiltered natural seawater to a depth of 16 cm. Water in the tanks was maintained at ambient temperature (29 °C). Water changes were conducted at 100% every 24 hours in the holding tank, and 100% between each trial in the trial tank using freshly collected seawater (which was assumed to be constant in aeration and quality). No lionfish were present in the tanks during water changes. Tanks were naturally lit by large windows and thus followed the ambient light:dark cycle. All experiments were conducted during daylight hours between 6:00 am and 6:00 pm. During the trials two artificial lights (10 watt LED lamps) were used above the trial tank to account for shading by the building structure and ensure standardisation of lighting between all trials.
Lionfish were kept in the holding tank for an acclimatisation period of 24 hours during which they were starved to ensure similar hunger levels between individuals69. A maximum of six lionfish were maintained in the holding tank at any time. The holding tank was fitted with a Tetra whisper power filter (Spectrum Brands Inc., USA) and Aqua Culture air pump (Walmart Stores Inc., USA) to ensure the water remained clean and aerated.
We used one trial tank that was divided into two preference zones of 90.5 cm (one at either end of the tank) and a central zone of 64 cm, by placing tape across the top of the tank (Fig. 4). The preference zones encompassed a 15.5 cm diameter stimulus container plus approximately 4.5 body lengths distance from the stimulus container, based on the average total length of 17.0 cm for lionfish in this study. This distance encompassed the four body length distance used to define fish aggregations24 to ensure that, if present, aggregation behaviour was detected. The tank was surrounded by opaque white plastic to prevent interference from external stimuli70.
Tank set-up for laboratory social attraction experiments. The tank was divided into two preference zones (stimulus and control) and a central zone. Dotted lines mark boundaries between zones, which were marked by placing tape across the top of the tank. A GoPro Hero5 camera (GoPro Inc., USA) was mounted on the ceiling 1 m above the centre of the tank. Diagram is not drawn to scale. Lionfish photo ©iStock.com/GlobalP.
Experimental design
Stimuli were added to one end of the trial tank based on the following: (i) live lionfish and water from the trial tank added to a submerged transparent plastic container (visual cue), (ii) water from the holding tank added to a submerged transparent plastic container with 30 evenly spaced 4 mm diameter holes (olfactory cue), (iii) live lionfish and water from the trial tank added to a submerged transparent plastic container with 30 evenly spaced 4 mm diameter holes (visual + olfactory cue), and (iv) 3D printed lionfish model (Fig. 5) and water from the trial tank added to a submerged transparent plastic container (artificial model cue). A control was placed at the opposite end of the tank, comprising water from the trial tank added to a submerged transparent plastic container without holes (visual and artificial model controls) or with 30 evenly spaced 4 mm diameter holes (olfactory and visual + olfactory controls). The end of the tank containing the stimulus/control was randomised between trials and the order of trials was randomised between days.
Artificial model lionfish used in the laboratory social attraction experiments. The model was moulded from aquarium grade silicone using a custom-made mould created photogrammetrically using video footage of a live lionfish. The model measures 15 cm total length. Underwater the pectoral fins spread out to give a more realistic appearance (inset photograph).
A randomly chosen lionfish was placed in a 30 cm diameter mesh acclimatisation cylinder in the centre of the tank. After a ten minute acclimatisation period29 (which allowed the lionfish to acclimatise to the water, stimuli and artificial lighting), the cylinder was lifted and the lionfish was filmed for a trial period of ten minutes29 using a GoPro Hero5 camera (GoPro Inc., USA) mounted 1 m above the centre of the tank. Subsequently, the videos were analysed and the position of the lionfish (stimulus, control or central zone) recorded every second for the duration of the trial, as well as which preference zone (stimulus or control) was entered first. Position was determined as the zone that the tip of the lionfish’s snout was in. Overall preference was calculated as time spent in the stimulus zone minus time spent in the control zone.
Once lionfish had been used in a trial they were humanely culled by cervical transection, followed by pithing to destroy the brain tissue71. These lionfish were subsequently dissected as described earlier, except that fat weight was not recorded.
Habitat preference and habitat complexity
Whenever lionfish were encountered on the reef, a 1 × 1 m quadrat was placed over the reef centred on the position of the individual lionfish or the centre of the aggregation. Habitat assessment scores were then performed following Gratwicke and Speight31, whereby five categories of structure were estimated visually on a five-point scale. Maximum substratum height, refuge size and variety of growth forms were recorded, along with average live cover (percentage cover of sessile organisms such as live corals, macroalgae and sponges) and rugosity scores for each quadrat. Over the course of the study 153 quadrats were assessed. Only one data point for complexity was taken per aggregation.
In addition, 30 large quadrats (2 × 2 m) were placed on reef areas where lionfish were encountered (23 solitary and 7 aggregations). These quadrats were filmed in a lawnmower pattern, approximately 0.5 m above the benthos, using a GoPro Hero3 camera (GoPro Inc., USA). Areas of reef without lionfish present were also sampled to give a background average for the reef system. Transects were laid at randomly chosen directions from each mooring line and quadrats were placed at regular intervals along each transect to give a total of 44 background quadrats. The resulting footage was converted into stills and used to render 3D models in Agisoft Photoscan (Agisoft LLC, Russia), before being analysed for habitat complexity using Rhinoceros 3D (Robert McNeel and Associates, USA), following the method of Young et al.32. Habitat complexity was measured using three metrics: linear rugosity (2 cm resolution), vector dispersion (1 cm resolution) and fractal dimension. Fractal dimension was measured at five spatial resolutions (1–5 cm, 5–15 cm, 15–30 cm, 30–60 cm and 60–120 cm) to explore patterns between lionfish habitat preference and habitat complexity at varying spatial scales.
Statistical analyses
All tests were two-tailed with an a-priori significance level of 0.05 and were conducted in R version 3.4.2 (R Core Team, Austria). Prior to analysis, datasets were checked for normality using a Shapiro-Wilk test, and groups were checked for homogeneity of variance using Fisher’s F test. When the assumption of normality was violated, transformation was attempted. When data transformation did not normalise the data, a non-parametric test was conducted on the original data.
For the social attraction experiments, initial preference was tested using binomial tests and overall preference was tested using one-sample t-tests or sign tests. For tests between two groups (lionfish quadrats and background quadrats or solitary lionfish and aggregations), two-sample t-tests were used for continuous, normally distributed data, whilst two-proportions Z-tests were used for proportions. For continuous data that violated the normality assumptions, and for ordinal data, Mann-Whitney U-tests were conducted. Data that violated both normality and homogeneity of variance assumptions were analysed using a ranked Welch’s t-test72,73.
Ethics statement
Lionfish culling was carried out in accordance with the American Veterinary Medical Association Guidelines for the Euthanasia of Animals71. All experimental protocols were approved by the University of Oxford Animal Welfare and Ethical Review Body. No permits were required to collect lionfish, however, a research permit to allow this work to be conducted in Honduras was obtained from the Instituto de Conservacion Forestal Honduras (permit number: DE-MP-081-2017).
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Schofield, P. J. Geographic extent and chronology of the invasion of non-native lionfish (Pterois volitans (Linnaeus 1758) and P. miles (Bennett 1828)) in the western north Atlantic and Caribbean Sea. Aquat. Invasions 4, 473–479 (2009).
Albins, M. A. & Hixon, M. A. Worst case scenario: potential long-term effects of invasive predatory lionfish (Pterois volitans) on Atlantic and Caribbean coral-reef communities. Environ. Biol. Fishes 96, 1151–1157 (2013).
Anderson, L. W. J. California’s reaction to Caulerpa taxifolia: A model for invasive species rapid response. Biol. Invasions 7, 1003–1016 (2005).
Culver, C. S. & Kuris, A. M. The apparent eradication of a locally established introduced marine pest. Biol. Invasions 2, 245–253 (2000).
Barbour, A. B., Montgomery, M. L., Adamson, A. A., Diaz-Ferguson, E. & Silliman, B. R. Mangrove use by the invasive lionfish Pterois volitans. Mar. Ecol. Prog. Ser. 401, 291–294 (2010).
Schofield, P. J. Update on geographic spread of invasive lionfishes (Pterois volitans Linnaeus, 1758 and P. miles Bennett, 1828) in the Western North Atlantic Ocean, Caribbean Sea and Gulf of Mexico. Aquat. Invasions 5, S117–S122 (2010).
Cote, I. M., Green, S. J. & Hixon, M. A. Predatory fish invaders: Insights from Indo-Pacific lionfish in the western Atlantic and Caribbean. Biol. Conserv. 164, 50–61 (2013).
Green, S. J., Underwood, E. B. & Akins, J. L. Mobilizing volunteers to sustain local suppression of a global marine invasion. Conserv. Lett. 10, 726–735 (2017).
Malpica-Cruz, L., Chaves, L. C. T. & Cote, I. M. Managing marine invasive species through public participation: Lionfish derbies as a case study. Mar. Pol. 74, 158–164 (2016).
de Leon, R. et al. Effectiveness of lionfish removal efforts in the southern Caribbean. Endanger. Species Res. 22, 175–182 (2013).
Green, S. J. et al. Linking removal targets to the ecological effects of invaders: a predictive model and field test. Ecol. Appl. 24, 1311–1322 (2014).
Andradi-Brown, D. A. et al. Depth-dependent effects of culling-do mesophotic lionfish populations undermine current management? R. Soc. Open Sci. 4, 15, https://doi.org/10.1098/rsos.170027 (2017).
Gress, E. et al. Lionfish (Pterois spp.) invade the upper-bathyal zone in the western Atlantic. PeerJ 5, 15, https://doi.org/10.7717/peerj.3683 (2017).
Andradi-Brown, D. A. et al. Large-scale invasion of western Atlantic mesophotic reefs by lionfish potentially undermines culling-based management. Biol. Invasions 19, 939–954 (2017).
Gittings, S. R. et al. Going deep for lionfish: designs for two new traps for capturing lionfish in deep water. 9 (U.S. Department of Commerce, National Oceanic and Atmospheric Administration, Office of National Marine Sanctuaries, Silver Spring, MD, 2017).
Garcia-Rivas, M. D. et al. What are the characteristics of lionfish and other fishes that influence their association in diurnal refuges? Mar. Biol. Res. 13, 899–908 (2017).
Jud, Z. R. & Layman, C. A. Site fidelity and movement patterns of invasive lionfish, Pterois spp., in a Florida estuary. J. Exp. Mar. Biol. Ecol. 414, 69–74 (2012).
Alpizar, D. et al. Pheromone mass trapping of the west Indian sugarcane weevil and the American palm weevil (Coleoptera: Curculionidae) in palmito palm. Fla. Entomol. 85, 426–430 (2002).
Brown, M., Roth, J., Smith, B. & Boscarino, B. The light at the end of the funnel?: Using light-based traps for the detection and collection of a nearshore aquatic, invasive invertebrate, Hemimysis anomala. J. Gt. Lakes Res. 43, 717–727 (2017).
Muller, B. J. & Schwarzkopf, L. Success of capture of toads improved by manipulating acoustic characteristics of lures. Pest Manag. Sci. 73, 2372–2378 (2017).
Cote, I. M. & Smith, N. S. The lionfish Pterois sp invasion: Has the worst-case scenario come to pass? J. Fish Biol. 92, 660–689 (2018).
McTee, S. A. & Grubich, J. R. Native densities, distribution, and diurnal activity of Red Sea lionfishes (Scorpaenidae). Mar. Ecol. Prog. Ser. 508, 223–232 (2014).
Domeier, M. L. & Colin, P. L. Tropical reef fish spawning aggregations: Defined and reviewed. Bull. Mar. Sci. 60, 698–726 (1997).
Magurran, A. E. & Pitcher, T. J. Provenance, shoal size and the sociobiology of predator-evasion behaviour in minnow shoals. Proc. R. Soc. B Biol. Sci. 229, 439–465 (1987).
Webster, M. M., Whalen, A. & Laland, K. N. Fish pool their experience to solve problems collectively. Nat. Ecol. Evol. 1, 5 (2017).
Lecchini, D., Shima, J., Banaigs, B. & Galzin, R. Larval sensory abilities and mechanisms of habitat selection of a coral reef fish during settlement. Oecologia 143, 326–334 (2005).
Ward, A. J. W., Axford, S. & Krause, J. Mixed-species shoaling in fish: the sensory mechanisms and costs of shoal choice. Behav. Ecol. Sociobiol. 52, 182–187 (2002).
Johnston, N. K. & Dixson, D. L. Anemonefishes rely on visual and chemical cues to correctly identify conspecifics. Coral Reefs 36, 903–912 (2017).
Kim, C., Ruberto, T., Phamduy, P. & Porfiri, M. Closed-loop control of zebrafish behaviour in three dimensions using a robotic stimulus. Sci. Rep. 8, 15, https://doi.org/10.1038/s41598-017-19083-2 (2018).
Bohnsack, J. A. Are high-densities of fishes at artificial reefs the result of habitat limitation or behavioural preference? Bull. Mar. Sci. 44, 631–645 (1989).
Gratwicke, B. & Speight, M. R. The relationship between fish species richness, abundance and habitat complexity in a range of shallow tropical marine habitats. J. Fish Biol. 66, 650–667 (2005).
Young, G. C., Dey, S., Rogers, A. D. & Exton, D. Cost and time-effective method for multiscale measures of rugosity, fractal dimension, and vector dispersion from coral reef 3D models. PLoS ONE 12, 18, https://doi.org/10.1371/journal.pone.0175341 (2017).
Darling, E. S. et al. Relationships between structural complexity, coral traits, and reef fish assemblages. Coral Reefs 36, 561–575 (2017).
Gonzalez-Rivero, M. et al. Linking fishes to multiple metrics of coral reef structural complexity using three-dimensional technology. Sci. Rep. 7, 15, https://doi.org/10.1038/s41598-017-14272-5 (2017).
Hixon, M. A., Johnson, D. W. & Sogard, S. M. BOFFFFs: on the importance of conserving old-growth age structure in fishery populations. ICES J. Mar. Sci. 71, 2171–2185 (2014).
Erisman, B. E., Buckhorn, M. L. & Hastings, P. A. Spawning patterns in the leopard grouper, Mycteroperca rosacea, in comparison with other aggregating groupers. Mar. Biol. 151, 1849–1861 (2007).
Farmer, N. A. et al. Timing and locations of reef fish spawning off the southeastern United States. PLoS ONE 12, 35, https://doi.org/10.1371/journal.pone.0172968 (2017).
Lonnstedt, O. M., Ferrari, M. C. O. & Chivers, D. P. Lionfish predators use flared fin displays to initiate cooperative hunting. Biol. Lett. 10, 4 (2014).
Garcia-Rivas, M. D. et al. Age-dependent strategies related to lionfish activities in the Mexican Caribbean. Environ. Biol. Fishes 101, 563–578 (2018).
Hiermes, M., Mehlis, M., Rick, I. P. & Bakker, T. C. M. Habitat-dependent olfactory discrimination in three-spined sticklebacks (Gasterosteus aculeatus). Anim. Cogn. 18, 839–846 (2015).
Ward, A. J. W. & Mehner, T. Multimodal mixed messages: the use of multiple cues allows greater accuracy in social recognition and predator detection decisions in the mosquitofish. Gambusia holbrooki. Behav. Ecol. 21, 1315–1320 (2010).
Houslay, T. M., Vierbuchen, M., Grimmer, A. J., Young, A. J. & Wilson, A. J. Testing the stability of behavioural coping style across stress contexts in the Trinidadian guppy. Funct. Ecol. 32, 424–438 (2018).
Jolles, J. W. et al. The role of social attraction and its link with boldness in the collective movements of three-spined sticklebacks. Anim. Behav. 99, 147–153 (2015).
Cote, I. M. et al. What Doesn’t Kill You Makes You Wary? Effect of Repeated Culling on the Behaviour of an Invasive Predator. PLoS ONE 9, 6, https://doi.org/10.1371/journal.pone.0094248 (2014).
Ip, Y. K. & Chew, S. F. Ammonia production, excretion, toxicity, and defense in fish: a review. Front. Physiol. 1, 20 (2010).
Richardson, J., Williams, E. K. & Hickey, C. W. Avoidance behaviour of freshwater fish and shrimp exposed to ammonia and low dissolved oxygen separately and in combination. N. Z. J. Mar. Freshw. Res. 35, 625–633 (2001).
Simpson, S. D., Meekan, M., Montgomery, J., McCauley, R. & Jeffs, A. Homeward sound. Science 308, 221–221 (2005).
van Oosterom, L., Montgomery, J. C., Jeffs, A. G. & Radford, C. A. Evidence for contact calls in fish: conspecific vocalisations and ambient soundscape influence group cohesion in a nocturnal species. Sci. Rep. 6, 8, https://doi.org/10.1038/srep19098 (2016).
Beattie, M., Nowacek, D. P., Bogdanoff, A. K., Akins, L. & Morris, J. A. The roar of the lionfishes Pterois volitans and Pterois miles. J. Fish Biol. 90, 2488–2495 (2017).
Biggs, C. R. & Olden, J. D. Multi-scale habitat occupancy of invasive lionfish (Pterois volitans) in coral reef environments of Roatan, Honduras. Aquat. Invasions 6, 347–353 (2011).
Anton, A., Simpson, M. S. & Vu, I. Environmental and Biotic Correlates to Lionfish Invasion Success in Bahamian Coral Reefs. PLoS ONE 9, 10, https://doi.org/10.1371/journal.pone.0106229 (2014).
Valdivia, A., Bruno, J. F., Cox, C. E., Hackerott, S. & Green, S. J. Re-examining the relationship between invasive lionfish and native grouper in the Caribbean. PeerJ 2, 16, https://doi.org/10.7717/peerj.348 (2014).
Friedlander, A. M. & Parrish, J. D. Habitat characteristics affecting fish assemblages on a Hawaiian coral reef. J. Exp. Mar. Biol. Ecol. 224, 1–30 (1998).
Jones, G. P., McCormick, M. I., Srinivasan, M. & Eagle, J. V. Coral decline threatens fish biodiversity in marine reserves. Proc. Natl. Acad. Sci. USA 101 (2004).
Gotceitas, V. & Colgan, P. Predator foraging success and habitat complexity- quantitative test of the threshold hypothesis. Oecologia 80, 158–166 (1989).
Alvarez-Filip, L., Dulvy, N. K., Gill, J. A., Cote, I. M. & Watkinson, A. R. Flattening of Caribbean coral reefs: region-wide declines in architectural complexity. Proc. R. Soc. B 276, 3019–3025 (2009).
Dahl, K. A. & Patterson, W. F. Habitat-Specific Density and Diet of Rapidly Expanding Invasive Red Lionfish, Pterois volitans, Populations in the Northern Gulf of Mexico. PLoS ONE 9, 13, https://doi.org/10.1371/journal.pone.0105852 (2014).
Cote, I. M. & Maljkovic, A. Predation rates of Indo-Pacific lionfish on Bahamian coral reefs. Mar. Ecol. Prog. Ser. 404, 219–225 (2010).
Bejarano, S., Lohr, K., Hamilton, S. & Manfrino, C. Relationships of invasive lionfish with topographic complexity, groupers, and native prey fishes in Little Cayman. Mar. Biol. 162, 253–266 (2015).
Ingeman, K. E. Lionfish cause increased mortality rates and drive local extirpation of native prey. Mar. Ecol. Prog. Ser. 558, 235–245 (2016).
Harms-Tuohy, C. A., Schizas, N. V. & Appeldoorn, R. S. Use of DNA metabarcoding for stomach content analysis in the invasive lionfish Pterois volitans in Puerto Rico. Mar. Ecol. Prog. Ser. 558, 181–191 (2016).
Valdez-Moreno, M., Quintal-Lizama, C., Gomez-Lozano, R. & Garcia-Rivas, M. D. Monitoring an alien invasion: DNA barcoding and the identification of lionfish and their prey on coral reefs of the Mexican Caribbean. Plos One 7, 8, https://doi.org/10.1371/journal.pone.0036636 (2012).
Bodmer, M. D. V., Rogers, A. D., Speight, M. R., Lubbock, N. & Exton, D. A. Using an isolated population boom to explore barriers to recovery in the keystone Caribbean coral reef herbivore Diadema antillarum. Coral Reefs 34, 1011–1021 (2015).
Betancur-R, R. et al. Reconstructing the lionfish invasion: insights into Greater Caribbean biogeography. J. Biogeogr. 38, 1281–1293 (2011).
Ricker, W. E. Computation and interpretation of biological statistics of fish populations. Vol. 191 1–382 (Bulletin of the Fisheries Research Board of Canada, 1987).
Villumsen, K. R., Koppang, E. O., Christensen, D. & Bojesen, A. M. Alternatives to mineral oil adjuvants in vaccines against Aeromonas salmonicida subsp salmonicida in rainbow trout offer reductions in adverse effects. Sci. Rep. 7, 15, https://doi.org/10.1038/s41598-017-06324-7 (2017).
Green, S. J., Akins, J. L. & James, A. Lionfish dissection: Techniques and applications. 24 (NOAA Technical Memorandum NOS NCCOS 139, 2012).
Gardner, P. G., Frazer, T. K., Jacoby, C. A. & Yanong, R. P. E. Reproductive biology of invasive lionfish (Pterois spp.). Front. Mar. Sci. 2 (2015).
Webster, M. M., Adams, E. L. & Laland, K. N. Diet-specific chemical cues influence association preferences and prey patch use in a shoaling fish. Anim. Behav. 76, 17–23 (2008).
Gomez-Laplaza, L. M., Caicoya, A. L. & Gerlai, R. Quantity discrimination in angelfish (Pterophyllum scalare) is maintained after a 30-s retention interval in the large but not in the small number range. Anim. Cogn. 20, 829–840 (2017).
Leary, S. L. et al. AVMA guidelines for the euthanasia of animals: 2013 edition. (American Veterinary Medical Association, USA, 2013).
Ruxton, G. D. The unequal variance t-test is an underused alternative to Student’s t-test and the Mann-Whitney U test. Behav. Ecol. 17, 688–690 (2006).
Zimmerman, D. W. & Zumbo, B. D. Rank transformations and the power of the Student t test and Welch t′ test for non-normal populations with unequal variances. Can. J. Exp. Psychol.-Rev. Can. Psychol. Exp. 47, 523–539 (1993).
Acknowledgements
We thank all the Operation Wallacea volunteers and staff who assisted with data collection. We also thank Steve Dey and colleagues at ThinkSee3D Ltd. (UK) for producing the 3D printed lionfish model. This work was financially supported by the Natural Environment Research Council (Grant number: NE/L002612/1) and Operation Wallacea.
Author information
Authors and Affiliations
Contributions
C.L.H., D.A.E. and A.D.R. designed the population structure and habitat complexity aspect of the study, the data for which were collected by C.L.H., G.R.K. and H.W. C.L.H., J.C.Q., H.C., D.A.E. and A.D.R. designed the social attraction experiments, which were performed by C.L.H. M.D.V.B. provided fieldwork support. C.L.H. analysed the data and wrote the manuscript. C.L.H. and D.A.E. produced the figures. All authors reviewed drafts of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hunt, C.L., Kelly, G.R., Windmill, H. et al. Aggregating behaviour in invasive Caribbean lionfish is driven by habitat complexity. Sci Rep 9, 783 (2019). https://doi.org/10.1038/s41598-018-37459-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-37459-w
This article is cited by
-
The bioeconomic paradox of market-based invasive species harvest: a case study of the commercial lionfish fishery
Biological Invasions (2023)
-
The ecological importance of habitat complexity to the Caribbean coral reef herbivore Diadema antillarum: three lines of evidence
Scientific Reports (2021)
-
Microhabitats of sharknose goby (Elacatinus evelynae) cleaning stations and their links with cleaning behaviour
Coral Reefs (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.