Abstract
Generally, lattice distortions play a key role in determining the electronic ground states of materials. Although it is well known that trigonal distortions are generic to most two dimensional transition metal dichalcogenides, the impact of this structural distortion on the electronic structure and topological properties has not been understood conclusively. Here, by using a combination of polarization dependent X-ray absorption spectroscopy (XAS), X-ray photoelectron spectroscopy (XPS) and atomic multiplet cluster calculations, we have investigated the electronic structure of titanium dichalcogenides TiX2 (X = S, Se, Te), where the magnitude of the trigonal distortion increase monotonically from S to Se and Te. Our results reveal the presence of an anomalously large crystal field splitting. This unusual kind of crystal field splitting is likely responsible for the unconventional electronic structure of TiX2 compounds and ultimately controls the degree of the electronic phase protection. Our findings also indicate the drawback of the distorted crystal field picture in explaining the observed electronic ground state and emphasize the key importance of trigonal symmetry, metal-ligand hybridization and electron-electron correlations in defining the electronic structures at the Fermi energy.
Similar content being viewed by others
Introduction
The realization of numerous exotic electronic phases of graphene1,2,3 and the relentless tendency to miniaturization of silicon-based electronics4 have ignited exhaustive research in a wide range of two dimensional (2D) layered materials. As a result, 2D transition metal dichalcogenides have emerged as the promising platform with intriguing topological and electronic ground states and high potential for applicability in the field of microelectronics5,6, nanophotonics7,8, optoelectronics9,10 and photovoltaics11,12,13 to name a few.
A generic feature of this dichalcogenide family is the presence of structural distortions which play a critical role in defining the electronic ground state and invariably the topological properties of these systems. Specifically, the lattice deformations can strongly alter the interatomic interaction strength and thereby are responsible for various novel electronic phases including charge density wave in VSe214,15, NbSe216 and TaSe217,18, superconductivity in FeSe1−xTex19, insulating ground states in ReSe220, Weyl semi-metallic phase in MoTe221 and many more. On the other hand, recent results on topological phases (TPs) have demonstrated a remarkable degree of protection against perturbations as long as the key symmetries remain intact22. Experimentally, surprisingly very little is know about the TP stability subjected to lattice distortions associated with trigonal symmetry as trigonal distortions could be used to either enter a topological phase, or exit a topological phase with a trigonal distortion22,23,24,25. Naturally, a way to microscopically probe and understand the effect of lattice deformations, induced covalency, and electron-electron correlations along with their impact on the electronic structure are essential for deterministic control of the rich physical phases of these TMDs compounds.
Recently, titanium dichalcogenides TiX2 (here X = S, Se and Te) have attracted significant attention of the community as the exemplary TMD systems26,27,28,29,30. While the usual crystallographic form of TiX2 is the layered CdI2 type31, these systems also possess a distinct trigonal distortion from an ideal octahedral crystal environment31,32. Previously, detailed structural investigation established that the magnitude of the distortion varies monotonically from nearly octahedral in TiS231 to highly distorted in TiTe232. In addition, large body of work on transition metal compounds suggest that these generic trigonal distortions may have strong influence in modifying the energy level and electronic structure of the chalcogenides. The problem is vividly illustrated by the case of TiSe2 which undergoes the transition into a chiral charge density wave (CDW) state33,34 and further into a conventional CDW resulting in dramatic renormalization of electronic and structural properties. The fundamental challenge here is to separate the many-body effects associated with excitonic condensate35,36,37 from the Jahn-Teller like instability from the strong-electron phonon coupling38,39. In addition, recent extensive theoretical work based on LDA + U40 along with the detailed ARPES41 study hinted on the importance of lattice distortions coupled with strong electron-electron correlations to explain the ground state properties of TiSe2. Based on those findings and given the obvious importance of these the whole TiX2 family, there have been several experimental and theoretical investigations to understand the impact of trigonal distortion on the electronic structure in these systems42,43,44. However, in absence of systematic spectroscopic investigations corroborated with theoretical calculations the consequence of the trigonal distortion on the electronic structure of the TiX2 systems has not convincingly been reported thus far. Here, we have addressed this issue by investigating the whole family of TiX2 single crystals by means of polarization dependent X-ray absorption spectroscopy in conjunction with the multiplet cluster calculations. Our results unambiguously demonstrate the failure of the standard ionic configuration and distorted crystal field picture in predicting electronic ground states and thereby reveal the key importance of metal-ligand hybridization between titanium and chalcogen ions and electronic correlations in defining their electronic properties.
Results and Discussion
As shown in Fig. 1(a), all members of the TiX2 family crystallize into trigonal CdI2 type layered structure with space group P3m131. The crystal structure consists of repeated tri-layers (Fig. 1(a)) along crystallographic z direction; each tri-layer in these systems contains a titanium layer sandwiched between two layers of chalcogenides. Although the interactions between titanium and chalcogenides are strong within a tri-layer, the chalcogen bonding between two tri-layers is weak and dominated by the weak van der Waals type interaction. Each tri-layer further experiences an elongated trigonal distortion where all six Ti-X bond length remains constant but X-Ti-X bond angle deviates from an ideal 90° in such a manner that the crystallographic lattice parameter along z direction increases in length. As illustrated in Fig. 1(b) the strength of elongated trigonal distortion increases monotonically from TiS2 to TiTe2 with three different X-Ti-X bond angles. The magnitude of such elongated trigonal distortion can be estimated from c/a ratio where c and a represent the corresponding unit cell parameter along z and x direction, respectively; For an ideal octahedral environment, the c/a ratio is close to 1.633 whereas the c/a ratio for TiS2, TiSe2 and TiTe2 are found to be 1.726, 1.732 and 1.808, respectively. This type of structural distortion can also be alternatively explained in terms of the distortion angle Θ between the diagonals of the pq, qr, rp plane as schematically shown in Fig. 1(c). For a regular octahedron Θ = 60° and becomes lesser and greater than 60° for elongated and compressive trigonal distortions, respectively.
(a) Schematic crystal structure of the TiS2, TiSe2, and TiTe2 compounds showing nearly octahedral environment of each TiX2 unit. (b) Distortion of the ideal octahedral environment across the chalcogenide series. It is to be noted that the distortion does not change the bond length but alters the bond angle. (c) Schematic representation of the trigonal distortions.
Chemical quality, and the absence of chalcogen vacancies critical for most dichalcogenides are verified by X-ray photo electron spectroscopy (XPS) measurements carried out on freshly cleaved TiX2 single crystals to rule out the presence of any anion vacancies. The basic process associated with the XPS technique is shown schematically in Fig. 2(a). Generally, the chemical shift associated with each core level provides important information about the charge state of the different elements of the system under investigation. Figure 2(b) displays typical Ti 2p core level spectra for three different TiX2 systems (Core level XPS spectra of Chalcogenides are shown in Supporting Information). Each Ti 2p core-level spectrum shown in Fig. 2(b) is composed of two intense spin-orbit split doublet (marked as A and B) with a spin-orbit splitting strength close to 5.8 eV. Feature A appearing at a binding energy range between 454 eV to 459 eV represents Ti 2p3/2 like states whereas feature B arises mainly from Ti 2p1/2 like states. Interestingly, a systematic shift in peak position was found from higher to lower binding energy in Ti 2p core level spectra from TiS2 to TiTe2, respectively. These shifts in the binding energy were previously attributed to the reduction in ionic contribution in Ti-X chemical bond formations45. In this work, each spectrum was decomposed using a Gaussian-Lorentz line profile and a Shirley type background function and can be accounted within a single Gaussian-Lorentz line profile. The absence of the multiple peak structure in Ti 2p core level spectra clearly implies the presence single valence Ti, and rules out any signature of anion vacancies in our TiX2 samples.
(a) Schematic representations of the X-ray photoelectron (top) and X-ray absorption (bottom) spectroscopy process. (b) X-ray photoelectron spectra of Ti 2p core level. Each spectrum was decomposed with Lorentzian function convoluted with Gaussian functions. (c) Experimental XAS spectra (open circle) of three different TiX2 systems were compared with atomic multiplet calculations (shaded area).
To investigate the evolutions in electronic structure across the TiX2 series, XAS measurements were carried out on Ti L2,3 edge at beamline 4.0.2 of the Advanced Light Source, at Lawrence Berkeley National Laboratory. In a typical Ti L3,2 XAS process (see bottom of Fig. 2(a)), electrons are excited from Ti 2p core level (from 2p3/2 and 2p1/2) to the Ti 3d conduction band; each L3,2 XAS spectrum splits into two edges (L2 and L3) due to the spin-orbit coupling of Ti 2p states. Experimentally obtained Ti L2,3 XAS spectra for three different TiX2 systems are shown in Fig. 2(c) as open circles. As seen both L3 and L2 peaks of each spectrum exhibit a double hump feature arising primarily from nearly octahedral crystal field effects which splits the fivefold degenerate 3d orbitals of Ti into doubly degenerate eg and triply degenerate t2g orbitals. For a quantitative understanding, theoretical atomic multiplet cluster calculations were performed within a TiX6 cluster with Oh point group symmetry and Ti4 + ionic configuration46. The obtained calculated spectra are shown as shaded line in Fig. 2(c) confirm that only one types of charge state Ti4 + is present in our samples. In good agreement with the XPS spectra, we observed a gradual shift in the XAS spectra from TiS2 to TiTe2. This monotonic spectral shift suggests a systematic decrease in (Udd − Upd) from TiS2 to TiTe2 where Udd symbolizes the onsite coulomb interaction strength and Upd defines the core hole interaction potential. This result indicates that in case of Te, the strongest interatomic p − d interaction strength arises from the markedly increased metal-ligand hybridization. The summary of the relative energy position of the occupied 2p states and unoccupied 3d states of titanium obtained from XPS and XAS measurements is given schematically in Fig. 3.
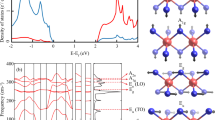
Despite high XAS sensitivity to charge state and local symmetry, it is still very challenging to capture the effect of trigonal distortions from non-polarized XAS measurement. To further explore the impact of trigonal distortion, detailed polarization dependent XAS measurements were carried out. Polarization dependent XAS process can probe local orbital character depending on their relative orientation with respect to crystallographic axes; for example it can be sensitive to the sub-band splitting of t2g states which may emerge from local lattice distortions. The process associated with the polarization dependent XAS (in-plane vs. out-of-plane) are schematically shown in Fig. 4(a). Since our samples are aligned along their natural [111] direction (z axis in Fig. 1(a) is along [111] direction), it is expected that out-of-plane polarization will be sensitive to the orbitals oriented along [111] direction whereas the in-plane polarization will probe those states which are oriented perpendicular to the [111] directions. Both out-of-plane (H) and in-plane (V) polarization dependent spectra are shown in Fig. 4(b). As immediately seen, a clear difference between the in- and out-of-plane XAS spectra is present which in turn implies the presence of distinct d-orbital anisotropy. The corresponding XLD signal or the difference between the in- and out-of-plane XAS spectrum is shown as the shaded area in Fig. 4(b) for all three samples.
(a) Experimental setup of the in-plane and out-of-plane polarization dependent XAS measurements. (b) Ti L3,2 edge XAS/XLD spectra of three different TiX2 compounds at room temperature. (c) Conventional theoretical model of an elongated trigonal distortion with t2g sub-band splitting. (d) Experimentally observed Ti 3d sub-band splitting in TiX2. (e) PDOS of 3\({d}_{{z}^{2}}\) and 3dxy/3\({d}_{{x}^{2}-{y}^{2}}\) obtained from previous theoretical calculations49 on TiS2 system where x, y, z represent global co-ordinates axis as shown schematically in Fig. 1(a).
To make a connection between the XLD results and orbital occupation we discuss electronic structure of the TiX2 compounds. In the purely ionic limit, the electronic structure of these systems comprises of fully occupied p states of chalcogen atom (S or Se or Te) forming valence bands and unoccupied conduction bands with the predominant Ti 3d character. Due to the effect of octahedral crystal field, five-fold degenerate 3d orbitals of Ti split into a doubly degenerate higher energy eg and a triply degenerate lower energy t2g states32. In addition, as shown schematically in Fig. 4(c) due to elongated trigonal distortion47 t2g states further split into a higher energy singlet a1g and lower energy doublet \({e}_{g}^{\pi }\) states. The wave function of a1g and \({e}_{g}^{\pi }\) states is a linear combination of dxy, dyz and dxz orbitals and can be expressed as as \(|{a}_{1g}\rangle =\frac{1}{\sqrt{3}}(|xy\rangle +|yz\rangle +|xz\rangle )\) and \(|{e}_{g\pm }^{\pi }\rangle =\pm \,\frac{1}{\sqrt{3}}(|xy\rangle +{e}^{\mp 2i\pi /3}|yz\rangle +{e}^{\pm 2i\pi /3}|xz\rangle )\)48. The description of the mixed a1g and \({e}_{g}^{\pi }\) states becomes simplified when one assumes z axis along the [111] direction as shown in Fig. 1(a). The wave function of the a1g state has a similar shape along [111] direction as the \({d}_{{z}^{2}}\) orbital along [001]; \({e}_{g}^{\pi }\) states are perpendicular to the [111] directions and oriented in xy plane (see the shape of the a1g and \({e}_{g}^{\pi }\) orbital Fig. 4(a)).
Based on this picture, one would naively expect that in-plane polarization will largely probe \({e}_{g}^{\pi }\) states which are at lower energy compared to the higher energy a1g state that are more sensitive to out-of-plane polarization. Contrary to this expectation, the experimental spectra for all three systems are in complete variation with the conventional picture. More specifically, in the case of t2g states labeled by feature I and III in Fig. 2(c), the out-of-plane polarization (shown by solid line in Fig. 4(b)) exhibits a lower energy peak position compared to the in-plane polarization (shown by dotted line). This implies the stabilization of a1g as the lowest occupied orbital contrary to the expected \({e}_{g}^{\pi }\) state. This is completely opposite to what one would infer from the standard crystals field splitting arguments. The crystal field inversion picture is schematically shown in Fig. 4(d) (Quantitative Sub-band splitting of each TiX2 compound has been shown schematically in Supporting Information). These findings clearly suggest that purely ionic picture which is normally very efficient in capturing the excitation spectra of Ti4 + derived states in oxide systems is inadequate in explaining the origin of XLD signal in dichalcogenides. Several factors might be responsible for such discrepancy in the description of the XAS spectra. Because of the markedly enlarged p-orbitals in S, Se and especially Te, unlike oxides, covalency may play a dominant role in transition metal dichalcogenides. The synergistic effect of high metal-ligand hybridization and significant ligand field can strongly affect the local electronic description of these systems.
Adding more to the surprising finding, the crystal field splitting gap (10 Dq) between t2g and eg is found to be strongly dependent on the direction of polarization of the incident light. In particular, out-of-plane polarization (solid line in Fig. 4(b)) shows a higher crystal field gap compared to in-plane polarizations (dotted line in Fig. 4(b)). This is unexpected since for the trigonal distortion, the eg states should not experience any sub-band splitting. However, the energy position of the eg state is found to depend strongly on the types of polarization. All these discrepancies clearly establish that conventional crystal field picture fails to explain the observed features in these system contrary to the Ti4+ based oxide materials.
To resolve this inconsistency, the XAS spectra were compared with calculated projected partial density of states (PDOS) of 3\({d}_{{z}^{2}}\) and 3dxy states of TiS245,49,50,51 as shown in Fig. 4(e). Interestingly, the calculated PDOS of 3\({d}_{{z}^{2}}\) of titanium exhibit a double hump structure analogous to the experiments when the systems were probed with out-of-plane polarization and PDOS of the 3dxy/3\({d}_{{x}^{2}-{y}^{2}}\) states (these two states are degenerate) also shows a double hump feature similar to in-plane polarization. Moreover, the energy gap between the doublet features is greater in the case of 3\({d}_{{z}^{2}}\) PDOS as compared to 3dxy, and 3\({d}_{{x}^{2}-{y}^{2}}\) PDOS which is remarkably similar to the experimentally observed trend. The calculated PDOS is qualitatively consistent with experimental observations and explains the appearance of the all features and their energy position as observed from experiments. The key feature of the theoretical calculation is the inclusion of strong metal-chalcogen hybridization which is responsible for the observed crystal field inversion and allows to capture the details of electronic structure within the band-like description. This result implies that indeed covalency and metal-ligand hybridization are critical for the electronic properties of the TiX2 family of materials.
In conclusion, detailed polarization dependent XAS measurements were carried out to probe and microscopically understand the effect of trigonal lattice distortion on the electronic structure of the TiX2 family. All systems were characterized to rule out the presence of anion vacancies in these compounds. XLD spectra demonstrate the failure of conventional crystal field arguments in explaining the observed experimental features implying the crucial importance of covalency/metal-ligand hybridization in defining the electronic structure. Orbital projected DOS successfully reproduced the spectral features observed in our experiments. The excellent agreement between theory and the XAS spectra suggests the importance of the band-like description including trigonal lattice deformations, electron-electron correlations and metal-ligand covalency to elucidate the electronic structure of transition metal dichalcogenides.
Methods
XPS measurements
Ti 2p, S 2p, Se 3d and Te 3d core level XPS measurements were carried out in a lab-based Thermo Scientific X-ray Photoelectron Spectrometer furnished with a monochromatic Al Kα photon source and a hemi-spherical analyser with total energy resolution close to 0.45 eV. The base pressure of the main chamber was below 2 × 10−8 mbar during XPS measurements. All three TiX2 compounds were mechanically exfoliated just before XPS measurements to avoid any surface contamination. The bulk sensitivity of the measurements was increased by collecting the ejected photoelectrons in a surface normal geometry. Photon energy of the source was calibrated using C 1 s core level spectra with a characteristic peak at around 284.6 eV binding energy. Each core level spectrum was decomposed in a casaXPS software using Gaussian-Lorentz type line profile.
XAS /XLD measurements
XAS/XLD measurements on three TiX2 compounds were carried out at Ti L3,2 edge at beamline 4.0.2 of the Advanced Light Source (ALS), at Lawrence Berkeley National Laboratory, USA. To avoid surface contamination, each single crystal was exfoliated mechanically in nitrogen atmosphere just before carrying out XAS measurements. No further surface treatments were performed as like vacuum heating or sputtering to clean the surface as these techniques could produce chalcogen vacancy in our systems. Total electron yield (TEY) detection technique were used during the XAS/XLD measurements.
References
Castro Neto, A. H., Guinea, F., Peres, N. M. R., Novoselov, K. S. & Geim, A. K. The electronic properties of graphene. Rev. Mod. Phys. 81, 109 (2009).
Allen, M. J., Tung, V. C. & Kaner, R. B. Honeycomb Carbon: A Review of Graphene. Chem. Rev. 110, 132–145 (2010).
Kumar, S. & Chatterjee, K. Comprehensive Review on the Use of Graphene-Based Substrates for Regenerative Medicine and Biomedical Devices. ACS Appl. Mater. Interfaces 8, 16431–26457 (2016).
Frazier, A. B., Warrington, R. O. & Friedrich, C. The Miniaturization Technologies: Past, Present, and Future. IEEE Trans. Ind. Appl. 42, 423–430 (1995).
Gong, C. et al. Band alignment of two-dimensional transition metal dichalcogenides: Application in tunnel field effect transistors. Appl. Phys. Lett. 103, 053513 (2013).
Podzorov, V., Gershenson, M. E., Kloc, C., Zeis, R. & Bucher, E. High-mobility field-effect transistors based on transition metal dichalcogenides. Appl. Phys. Lett. 84, 3301–3303 (2004).
Xia, F., Wang, H., Xiao, D., Dubey, M. & Ramasubramaniam, A. Two-dimensional material nanophotonics. Nat. Photonics 8, 899–907 (2014).
Mak, K. F. & Shan, J. Photonics and optoelectronics of 2D semiconductor transition metal dichalcogenides. Nat. Photonics 10, 216–226 (2016).
Wang, Q. H., Zadeh, K. K., Kis, A., Coleman, J. N. & Strano, M. S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 7, 699–712 (2012).
Baugher, B. W. H., Churchill, H. O. H., Yang, Y. & Jarillo-Herrero, P. Optoelectronic devices based on electrically tunable p-n diodes in a monolayer dichalcogenide. Nat. Nanotechnol 9, 262–267 (2014).
Bernardi, M., Palummo, M. & Grossman, J. C. Extraordinary Sunlight Absorption and One Nanometer Thick Photovoltaics Using Two-Dimensional Monolayer Materials. Nano Lett. 13, 3664–3670 (2013).
Britnell, L. et al. Strong Light-Matter Interactions in Heterostructures of Atomically Thin Films. Science 340, 13111314 (2013).
Wi, S. et al. Enhancement of Photovoltaic Response in Multilayer MoS2 Induced by Plasma Doping. ACS Nano 8, 5270–5281 (2014).
Tsutsumi, K. X-ray-diffraction study of the periodic lattice distortion associated with a charge-density wave in 1T-VSe2. Phys. Rev. B 26, 5756 (1982).
Woolley, A. M. & Wexler, G. Band structures and Fermi surfaces for 1T-TaS2, 1T-TaSe2 and 1T-VSe2. J. Phys. C 10, 2601–2616 (1977).
Xi, X. et al. Strongly enhanced charge-density-wave order in monolayer NbSe2. Nat. Nanotechnol 10, 765–769 (2015).
Zhu, P. et al. Dynamic separation of electron excitation and lattice heating during the photoinduced melting of the periodic lattice distortion in 2H-TaSe2. Appl. Phys. Lett. 103, 071914 (2013).
Dai, J. et al. Microscopic evidence for strong periodic lattice distortion in two-dimensional charge-density wave systems. Phys. Rev. B 89, 165140 (2014).
Ingle, K. E. et al. Importance of structural distortions in enhancement of transition temperature in FeSe1−xTex superconductors. Supercond. Sci. Technol 28, 015015 (2015).
Zhong, H.-X., Gao, S., Shi, J.-J. & Yang, L. Quasiparticle band gaps, excitonic effects, and anisotropic optical properties of the monolayer distorted 1 T diamond-chain structures ReS2 and ReSe2. Phys. Rev. B 92, 115438 (2015).
Deng, K. et al. Experimental observation of topological Fermi arcs in type-II Weyl semimetal MoTe2. Nat. Phys 12, 1105–1110 (2016).
Bahramy, M. S. et al. Ubiquitous formation of bulk Dirac cones and topological surface states from a single orbital manifold in transition-metal dichalcogenides. Nat. Mater. 17, 21–28 (2018).
Regg, A., Mitra, C., Demkov, A. A. & Fiete, G. A. Lattice distortion effects on topological phases in (LaNiO3)2/(LaAlO3)N heterostructures grown along the [111] direction. Phys. Rev. B 88, 115146 (2013).
Kargarian, M., Wen, J. & Fiete, G. A. Competing exotic topological insulator phases in transition-metal oxides on the pyrochlore lattice with distortion. Phys. Rev. B 83, 165112 (2011).
Yang, B.-J. & Kim, Y. B. Topological insulators and metal-insulator transition in the pyrochlore iridates. Phys. Rev. B 82, 085111 (2010).
Chen, P. et al. Charge density wave transition in single-layer titanium diselenide. Nat Commun. 6, 8943 (2015)
Gu, Y., Katsura, Y., Yoshino, T., Takagi, H. & Taniguchi, K. Rechargeable magnesium-ion battery based on a TiSe2-cathode with d-p orbital hybridized electronic structure. Sci. Rep 5, 12486 (2015).
Sun, X., Bonnick, P. & Nazar, L. F. Layered TiS2 Positive Electrode for Mg Batteries. ACS Energy Lett 1, 297–301 (2016).
Lv, R. et al. Transition Metal Dichalcogenides and Beyond: Synthesis, Properties, and Applications of Single- and Few-Layer Nanosheets. Acc. Chem. Res. 48, 56–64 (2015).
Whittingham, M. S. Chalcogenide battery. US Patent No. 4,009,052 (1977).
Chianelli, R. R., Scanlon, J. C. & Thompson, A. H. Structure refinement of stoichiometric TiS2. Mater. Res. Bull. 10, 1379–1382 (1975).
Arnaud, Y. & Chevreton, M. Etude comparative des composs TiX2 (X = S, Se, Te). Structures de TiTe2 et TiSeTe. J. Solid State Chem. 39, 230–239 (1981).
Ishioka, J. et al. Chiral Charge-Density Waves. Phys. Rev. Lett. 105, 176401 (2010).
Castellan, J.-P. et al. Chiral Phase Transition in Charge Ordered 1T-TiSe2. Phys. Rev. Lett. 110, 196404 (2013).
Cercellier, H. et al. Evidence for an Excitonic Insulator Phase in 1T-TiSe2. Phys. Rev. Lett. 99, 146403 (2007).
Wilson, J. A. Modelling the contrasting semimetallic characters of TiS2 and TiSe2. Physica status solidi (b) 86, 11–36 (1978).
Wilson, J. A. Concerning the semimetallic characters of TiS2 and TiSe2. Solid State Commun. 22, 551–553 (1977).
Hughes, H. P. Structural distortion in TiSe2 and related materials-a possible Jahn-Teller effect? J. Phys. C: Solid State Physics 10, L319–L323 (1977).
Rossnagel, K., Kipp, L. & Skibowski, M. Charge-density-wave phase transition in 1T-TiSe2 Excitonic insulator versus band-type Jahn-Teller mechanism. Phys. Rev. B 65, 235101 (2002).
Bianco, R., Calandra, M. & Mauri, F. Electronic and vibrational properties of TiSe2 in the charge-density-wave phase from first principles. Phys. Rev. B 92, 094107 (2015).
Rohwer, T. et al. Collapse of long-range charge order tracked by time-resolved photoemission at high momenta. Nature 471, 490–493 (2011).
Wu, Z. Y., Ouvrard, G., Moreau, P. & Natoli, C. R. Interpretation of preedge features in the Ti and S K-edge x-ray-absorption near-edge spectra in the layered disulfides TiS2 and TaS2. Phys. Rev. B 55, 9508 (1997).
Wan, C. et al. Flexible n-type thermoelectric materials by organic intercalation of layered transition metal dichalcogenide TiS2. Nat. Mater. 14, 622–627 (2015).
Bullett, D. W. Electronic band structure and bonding in transition metal layered dichalcogenides by atomic orbital methods. J. Phys. C: Solid State Phys. 11, 4501–4514 (1978).
Shkvarin, A. S. et al. Electronic structure of titanium dichalcogenides TiX2 (X = S, Se, Te). Journal of Expt. and Theo. Phys 114, 150–156 (2012).
Stavitski, E. & de Groot, F. M. F. The CTM4XAS program for EELS and XAS spectral shape analysis of transition metal L edges. Micron 41, 687–694 (2010).
Wilson, J. A. & Yoffe, A. D. The transition metal dichalcogenides discussion and interpretation of the observed optical, electrical and structural properties. Adv. Phys. 18, 193–334 (1969).
Khomskii, D. I. Transition Metal Compounds (Cambridge University Press).
Simunek, A., Sipr, O., Bocharov, S., Heumann, D. & Drager, G. Unoccupied electron states of TiS2 studied by means of polarized x-ray absorption. Phys. Rev. B 56, 12232 (1997).
Clerc, D. G. & Poshusta, R. D. Periodic Hartree-Fock Study of TiS2. J. Phys. Chem. 100, 15735 (1996).
Fang, C. M., de Groot, R. A. & Haas, C. Bulk and surface electronic structure of 1T-TiS2 and 1T-TiSe2. Phys. Rev. B 56, 4455 (1997).
Acknowledgements
J.C., Y.C. and B.P. are supported by the Gordon and Betty Moore Foundation EPiQS Initiative through Grant No. GBMF4534. This work is partially supported by the National Natural Science Foundation of China (Grant No. 11874058). Y.C. is supported by the Pioneer Hundred Talents Program of the Chinese Academy of Sciences and the Ningbo 3315 Innovation Team. X.L. and F.W. are supported by the US Department of Energy (DOE) under Grant No. DOE DE-SC 00012375 for synchrotron work and bulk crystal characterization. This research used resources of the Advanced Light Source, which is a Department of Energy Office of Science User Facility under Contract No. DE- AC0205CH11231.
Author information
Authors and Affiliations
Contributions
B.P., Y.C. and J.C. have initiated the project. X.L., F.W. and M.K. has performed crystal characterization. B.P. has performed X.P.S. measurements. Y.C., X.L., A.T.N.D., P.S. and E.A. has carried out the XAS measurements. All authors have discussed the results of the studies. B.P., Y.C., X.L. and J.C. co-wrote the paper. All authors have commented on the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pal, B., Cao, Y., Liu, X. et al. Anomalous orbital structure in two-dimensional titanium dichalcogenides. Sci Rep 9, 1896 (2019). https://doi.org/10.1038/s41598-018-37248-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-37248-5
This article is cited by
-
Density-of-states similarity descriptor for unsupervised learning from materials data
Scientific Data (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.