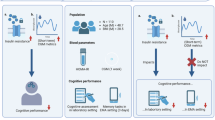
Abstract
This study cross-sectionally examines in the elderly population: (a) the association of type 2 diabetes with executive function (EF); (b) the effect of BMI on both type 2 diabetes and EF; (c) the association between glycaemia control and EF in type 2 diabetes. 6823 older individuals with overweight/obesity and metabolic syndrome participating in the PREDIMED-PLUS study, were assessed with a battery of cognitive tests and a medical interview. ANOVA showed a significantly worse performance on EF in type 2 diabetes vs. non-diabetic individuals. Two complementary models were displayed: (1) in the whole sample, the presence of type 2 diabetes, depressive symptoms and BMI had a direct negative effect on EF, while apnoea had an indirect negative effect; (2) in the diabetes subsample, higher illness duration was associated with worse performance in EF. Participants with type 2 diabetes and HbA1c<53 mmol/mol displayed better cognitive performance when compared to those with HbA1c≥53 mmol/mol. Our results provide a controlled comprehensive model that integrates relevant neuropsychological and physical variables in type 2 diabetes. The model suggests that, to improve treatment adherence and quality of life once diabetes has been diagnosed, cognitive decline prevention strategies need to be implemented while monitoring depressive symptoms, BMI and glycaemia control.
Similar content being viewed by others
Introduction
Type 2 diabetes is caused by a combination of resistance to insulin action and an inadequate compensatory insulin secretory response1. This is one of the most frequent diseases among the elderly individuals, with studies reporting a prevalence ranging from 19% to 33% in general population2,3. With global population ageing at significant pace and given the high prevalence of type 2 diabetes among the elderly, the negative effects of this chronic metabolic disease on health and cognitive functioning are of notable clinical interest.
With this regards, the main cognitive domains that have been related to type 2 diabetes are the following: attention, memory, processing speed and executive function (EF; i.e. working memory, cognitive flexibility, inhibitory control, etc.), as well as global cognitive functioning. When examining cognitive impairment in older individuals (>60 years), results confirmed that patients with type 2 diabetes in comparison with those without type 2 diabetes displayed higher impairment in global cognitive function, working memory and cognitive flexibility4,5. Similarly, when exploring other cohorts of elderly population (>65) with normal cognition, it was confirmed that type 2 diabetes participants obtained lower scores than non-diabetic patients in some cognitive domains such as attention, psychomotor function, information-processing speed and executive function, but they did not differ in memory6,7.
Different prospective studies have analyzed the association between the presence of type 2 diabetes and the risk of cognitive dysfunction. In older adults (42–89 years), results showed that women with type 2 diabetes had an increased risk of major cognitive decline in verbal fluency after 4 years when compared with non-diabetic women8. Similarly, other longitudinal studies concluded that older women (>65) with type 2 diabetes experienced an accelerated cognitive decline in general functioning and other specific domains, including visual and working memory as well as verbal fluency5,9,10. In addition, research in old population exploring the duration of type 2 diabetes point towards an association between the duration of this metabolic disease and a poorer performance in verbal memory and concept formation11 as well as in general cognition10. A longer duration of type 2 diabetes together with an earlier onset, has also been reported to increase the risk of mild cognitive impairment development12.
The increasing number of individuals suffering from type 2 diabetes among older people during the last few decades might be due to inappropriate dietary habits and a sedentary lifestyle, which lead to overweight and obesity. An elevated BMI may cause insulin resistance and it is common in adults with type 2 diabetes13. Some findings show that both high BMI and obesity are associated with a worsened performance in measures associated with EF, working memory, short-term memory and verbal fluency14,15,16.
Finally, type 2 diabetes has been associated with cognitive dysfunction regardless of ethnicity and cultural factors17. Nevertheless, other aspects seem to be relevant when examining the negative effects of type 2 diabetes on cognitive performance. In this line, different studies report that gender8, low glycaemia control18,19 as well as the presence of depressive symptoms20 are associated with worse cognitive performance in type 2 diabetes individuals. It has also to be note that sleep disorders can have an impact on cognition, especially in older age population at higher risk of cognitive decline21.
In light of previous studies and in order to specify the association between the presence of type 2 diabetes and cognitive decline in older individuals (above 55 years of age) with overweight/obesity and metabolic syndrome, the objectives of this study are: (a) to explore the associations of type 2 diabetes with cognitive performance (i.e.: EF); (b) to evaluate the effect of BMI on type 2 diabetes and cognitive performance when controlling for other confounding variables (e.g.: depressive symptoms, sleep disorders, education, age and sex); (c) to explore the association between glycaemia control and cognitive function performance in individuals with type 2 diabetes.
Methods
This is a cross-sectional analysis on baseline data within the frame of the PREDIMED-Plus study, a 6-year multicenter, randomized, parallel-group, primary prevention clinical trial conducted in Spain to assess the effect of an intensive weight loss intervention program based on an energy-restricted traditional Mediterranean diet, physical activity promotion and behavioural support (intervention group), compared to a usual care intervention and energy-unrestricted Mediterranean diet recommendations (control group). A more detailed description of the PREDIMED-plus study and the study protocol is available at http://predimedplus.com/wp-content/uploads/2016/07/Protocolo_PREDIMED_PLUS_eng_23112016_adl.pdf. This study was registered at the International Standard Randomized Controlled Trial (ISRCT; http://www.isrctn.com/ISRCTN89898870) with number 89898870. All participants were recruited and randomized from October 2013 to December 2017 across 23 centres from different universities, hospitals and research institutes of Spain. Each of these centres recruited participants from several Primary Care Health Facilities belonging to the National Health System. The eligible participants were community-dwelling adults (aged between 55 and 75 years in case of men; and between 60 and 75 years in women) with overweight/obesity (BMI between 27 and 40 kg/m2), who met at least three components of the metabolic syndrome according to the updated harmonized criteria of the International Diabetes Federation and the American Heart Association and National Heart, Lung and Blood Institute22. Specific PREDIMED-plus exclusion criteria can be found in the website http://predimedplus.com/en. Participants included in the current analysis provided data on cognitive performance and depressive symptoms, as well as detailed specification of BMI, physical conditions and sociodemographic data. Additionally, glucose and HbA1c concentrations were measured at the same time point of the mood and cognitive assessment. All participants provided written informed consent, and the study protocol and procedures were approved according to the ethical standards of the Declaration of Helsinki by the Research Ethics Committees from all the participating institutions (Supplementary Info 1).
Exclusion criteria for the present analysis were: (1) reported dementia (2) head trauma, learning disability or intellectual disabilities; (3) presence of depression or a lifetime mental disorder according to DSM-5. Finally, all the included participants presented glucose levels within the normal range during the neuropsychological assessment.
Instruments/measures
All participants completed a self-report questionnaire for exploring depressive symptoms as well as a set of neuropsychological tests for assessing different cognitive domains that included attention, short term memory, verbal fluency, cognitive flexibility, visuospatial skills and working memory. Sociodemographic information was also collected by means of a self-reported questionnaire and a hetero-administered clinical interview assessing medical conditions (including current presence of type 2 diabetes and years of duration since its onset, presence of apnoea, etc.).
Clinical and neuropsychological assessment
-The Beck Depression Inventory–II (BDI-II)23 is a 21-item self-report measure for assessing the severity of depressive symptoms in adults and adolescents (ages from 13 to 80 years). The BDI-II reflects the diagnostic criteria for Major Depressive Disorder listed in the Diagnostic and Statistical Manual of Mental Disorders (4th ed.)24. Scores for each item range from 0 to 3; the total score is the sum of all responses. The Cronbach’s alpha in our sample shows a good internal consistency (α = 0.883).
The Trail Making Test (TMT)25 consists of 25 circles spread out over two sheets of paper (parts A and B). In the part A, participants are requested to connect consecutive numbers (1–2–3–4-…) in the correct order by drawing a line. In the part B, they have to connect consecutive numbers and letters in an alternating numeric and alphabetic sequence (1-A, 2-B, 3-C-…). Each part is scored according to the time spent to complete the task (lower score mean better performance). TMT is frequently used to assess executive function. More specifically, TMT-A is used for measuring attention, psychomotor speed and visuospatial skills; while, TMT-B is used for measuring divided attention, cognitive flexibility and interference.
The Verbal Fluency Test26 assess verbal ability and executive control. Participants need to retrieve words of their language by accessing their mental lexicon, but also focus on the task and avoid repetition that involves executive control processes. In the phonemic fluency task, participants are requested to produce as many words as possible that start with the letter P during 60 seconds. Participants were asked to avoid producing names of people or places and repetitions of the same word with a different suffix. Secondly, in the semantic fluency task, participants have to name as many different animals as they can without repeating them during 60 seconds. The total raw score for each of the tasks corresponds to the number of words the participant achieves to say.
The Digit Span (DS) test of the Wechsler Adult Intelligence Scale-III (Spanish version)27 is compound of two different subtests: DS Forward, considered as a test of attention and short-term memory capacity; and DS Backward recall, considered as a test of working memory capacity. The DS Forward recall requires participants to repeat orally a series of random single digits in the same order they heard them; the sequence of digits varies in length from three to nine. Contrary, in the DS Backward recall participants are asked to repeat a series of random single digits in reverse order; the sequence varies from two to eight. The performance on the DS can be reported by a direct score of the backward performance (ranging from 1 to 16) and the forward performance (ranging from 1 to 14) and, by the span of immediate recall (backward span range from 1 to 8 and forward span range from 1 to 7).
Anthropometric and biochemical measurements
Weight and height were measured with light clothing and no shoes with calibrated scales and a wall-mounted stadiometer, respectively. BMI was calculated as weight in kilograms divided by the square of height in meters. Serum glucose levels were measured using standard enzymatic methods. HbA1c was measured by standard routine methods and glycaemia control was specified (HbA1c<53 mmol/mol [7%])1.
Statistical analysis
Analyses were carried out with Stata15 for Windows. We used the study baseline database generated in August 2017. The comparisons of the cognitive measures between participants with and without type 2 diabetes was done with ANOVA procedures adjusted for the individuals’ sex, age, years of school attendance and presence of sleep apnoea syndrome. Cohen’s-d coefficients measured the effect size for the mean comparisons (low effect size was considered low for |d|> 0.2, moderate for |d|> 0.5 and large for |d|> 0.8)28. The Finner’s method, a procedure included into the Familywise error rate stepwise procedures which offers a more powerful test than the classical Bonferroni correction29, was used to control Type-I error due to multiple comparisons. The same method was used to explore the association between glycaemia control (HbA1c<53 mmol/mol [7%]) and cognitive function performance in the subsample of participants who presented type 2 diabetes.
The underlying mechanisms between BMI, type 2 diabetes and cognitive measures were examined with path-analysis modelling, a straightforward extension of multiple regression techniques that can be used for both confirmatory and exploratory modelling. The aim of this procedure is to estimate the magnitude and significance of hypothesized connections/relationships into a set of variables, and so they allow to theory testing and theory development30. The Path analysis used in this study was implemented through Structural Equation Modelling (SEM) with exploratory aims, with the advantage (compared to classical regression models) to allow the inclusion of multiple relationships among a set of variables, including mediational associations. The maximum-likelihood estimation method of parameter estimation was used and goodness-of-fit was evaluated using standard statistical measures31: the root mean square error of approximation (RMSEA), Bentler’s Comparative Fit Index (CFI), the Tucker-Lewis Index (TLI), and the standardized root mean square residual (SRMR). Adequate model fit was considered if the following criteria were met: RMSEA< 0.08, TLI> 0.9, CFI> 0.9 and SRMR< 0.1; the global predictive capacity of the model was measured by the coefficient of determination. Separate models were obtained for the whole sample (n = 6,825) and for the type 2 diabetes sub-sample (n = 1,860).
Results
The initial sample was constituted of 6865 participants. According to the exclusion criteria of this study, for the present analyses forty nine participants were excluded for presenting hypo or hyperglycaemia during the cognitive assessment (≤60 mmol/l, hypoglycaemia; ≥150 mmol/l, hyperglycaemia). The final sample for the present analysis consisted of 6823 participants (mean age: 64.95 years; 48.6% females) distributed as follow: 4964 with no type 2 diabetes and 1859 with reported type 2 diabetes. At the moment of the assessment most of the patients with type 2 diabetes had presented this affection for more than 5 years (55.4%), some of them during 1 to 5 years (32.2%) and a 10.4% of them were only diagnosed during the last year (see the Supplementary Figure S1 for the flowchart graphic of the sample which is available as online supporting material).
Table 1 includes the descriptive data for the categorical variables (top of the table) and quantitative measures (bottom part of the table), for the whole sample and stratified by the sub-samples defined by the presence-absence of type 2 diabetes. No differences between groups were found for the origin or the civil status. However, the subsample who presented type 2 diabetes included a higher proportion of men, non-employed, with lower level studies, older age, a higher prevalence of sleep apnoea and depression symptomatology, higher BMI and higher concentrations of HbA1c and glucose.
Comparison of cognitive measures between participants with and without type 2 diabetes
Table 2 includes the results obtained in the ANOVA procedures adjusted for sex, age, years of school attendance and presence of apnoea. Some significant results emerged comparing patients with and without type 2 diabetes: the presence of type 2 diabetes was associated with lower scores in the phonological and semantic verbal fluency, and to higher scores in the trail making test.
Table 3 includes the ANOVA procedures exploring the relationship between glycaemia control (HbA1c<53 mmol/mol [7%] versus HbA1c≥53 mmol/mol [7%]) and cognitive function performance in the subsample of participants who presented type 2 diabetes. Statistical differences emerged for the measures of phonological fluency and digit backward, achieving better functioning (higher means) the participants into the HbA1c<53 mmol/mol (7%) group.
SEM examining the association between BMI, type 2 diabetes and cognitive functioning
Figure 1 shows the standardized coefficients of the SEM obtained for the whole sample (first panel, n = 6,823) and for the subsample with type 2 diabetes (n = 1,859). Both models included a latent variable (labelled cognitive) which was defined by the cognitive scores analyzed in this study (as high the score in this variable the better the neuropsychological functioning). Goodness-of-fit was obtained for both models, as well as very good global predictive capacity. Supplementary Info S2 and S3 contain the complete results of the path-analyses, including the parameters estimating the direct, indirect and total effects. contain the complete results of the path-analyses, including the parameters estimating the direct, indirect and total effects.
In the whole sample, the presence of type 2 diabetes was predictive of worse cognitive functioning (direct negative effect); while the BMI directly contributed on the cognitive performance (as high the BMI as poor the cognitive performance). In the subsample with type 2 diabetes, the higher duration of this metabolic condition was predictive of a lower cognitive functioning (direct effect), while BMI did not contributed to (with direct or indirect effects) on the cognitive levels.
Regarding the remaining variables included in the SEM, for both models better cognitive functioning was directly related to male sex, younger age and higher education. The apnoea achieved indirect effects on the cognitive performance only in the whole sample, through one path-way: the presence of apnoea was directly related to type 2 diabetes, which was also a predictor of poor cognitive execution. No direct effect between the presence of apnoea and cognitive performance emerged in any of the SEM.
As a synthesis of the results of the study, Figure S2 (Supplementary Material) includes the scheme of the main direct and indirect effects on the cognitive construct analyzed in the study.
Discussion
The present study examined the effect of type 2 diabetes on cognitive performance specifically with EF tasks, in a large cohort of 6823 patients above 55 years of age. Additionally, the role that BMI, apnoea, age, sex, depressive symptoms and education play in these effects was also explored or controlled when appropriate. Mainly, results display worse cognitive functioning when participants present type 2 diabetes, with a larger effect on those who have a longer duration of this metabolic condition. Additionally, some other variables are also found to directly or indirectly worsen cognitive function among this population, namely BMI, sex, depressive symptoms and education.
Within the first analysis when comparing patients presenting type 2 diabetes with individuals without type 2 diabetes and controlling for other co-related variables that could interfere with the cognitive assessment (i.e.: sex, age, education and apnoea), results show a significantly worse cognitive performance on EF when type 2 diabetes is present. Thus, the presence of type 2 diabetes in patients above 55 years of age seems to have a clear impact on cognitive function which would not be explained by the education, age, sex or the presence of apnoea. In the same line, previous studies have also reported that type 2 diabetes has a negative impact on cognitive function and physical health4,5,6,7,8,9,10,32,33; taken together, our results add some extra evidence to previous findings and point towards the need to target these neurocognitive domains once type 2 diabetes has been diagnosed and its treatment protocol implemented. In an attempt to further explore the underlying mechanisms of the reported association between type 2 diabetes and cognitive performance, the present study sought to step further, by exploring the association of glycated haemoglobin with the neuropsychological measures in the subsample with type 2 diabetes. Previous studies have found that individuals with a weighted mean HbA1c less than 53 mmol/mol (7%) performed significantly better on tests of motor speed and psychomotor efficiency than those subjects whose time weighted mean HbAlc was greater34. In our study, individuals with type 2 diabetes and HbA1c<53 mmol/mol (7%) displayed better cognitive function when compared to those with HbA1c≥53 mmol/mol (7%), specifically on verbal fluency and working memory which highlights the importance of maintain good glycaemia control in this population which is already at risk of cognitive decline.
Additionally, we sought to assess the associations of the presence of type 2 diabetes and its duration, cognitive performance and BMI in a model that also took into account other variables reported to be associated with cognitive performance (i.e.: sex, mood, education and apnoea). As a result of this analysis, two complementary different models were obtained. These two models provide relevant information about the interaction and contribution of the described variables to the cognitive performance of population suffering from type 2 diabetes, which can help improving the current understanding and treatment of this affection. Firstly, it is important to note that, as expected, type 2 diabetes and a higher duration of this metabolic condition was associated with worse cognitive performance. Consequently, it seems highly important to implement cognitive stimulation protocols within the treatment of type 2 diabetes that could help to reduce the progression of the cognitive decline once type 2 diabetes has been diagnosed. Secondly, a high BMI has previously been linked to specific cognitive impairments in different sample populations, especially in extreme weight conditions14,15,16,35. Our findings with the whole sample model give extra evidence on the negative impact of higher BMI on cognitive performance with this older age sample population. Additionally, higher BMI also negatively affects the participants’ mood in our sample. In turn, the lower mood in our sample is linked to poorer cognitive performance, which has also been reported in another study conducted with clinically depressed individuals36. Thus, health programs should give special emphasis in promoting weight loss within these individuals as well as strategies to monitor and improve mood when needed. On the contrary, in our older age sample with overweight/obesity, BMI differences are not significant enough to be robustly linked to the presence or duration of type 2 diabetes. However, BMI is described as a risk factor for the occurrence of type 2 diabetes in older age population37. Thus, the results observed regarding the influence of BMI on type 2 diabetes may most certainly only be circumscribed to our sample.
With regards to the presence of sleep disturbances/disorders and cognitive decline21, in our sample sleep apnoea (which unsurprisingly presents an association with higher BMI) is only linked to cognitive decline through its role in type 2 diabetes. Finally, it is also relevant that as reported in older adult populations a better cognitive function is directly related to males, younger age and higher education38,39.
The present study should be considered under some limitations. Firstly, the cross-sectional design does not imply causality. Also, given that our study participants are senior adults with a metabolic syndrome the present findings cannot be extrapolated to other population groups. However, it is one of the strengths of this study that it is conducted with a large sample of men and women and analyses are adjusted for potential confounding factors. Thus, although the results cannot be generalizable to other populations, they are representative of individuals over 55 years of age who suffer from a metabolic syndrome. Future studies should further explore the associations reported regarding the presence of type 2 diabetes, cognitive decline and BMI in different sample populations. Cognitive dysfunction is of particular importance because of its impact on quality of life. Future studies should test if an early intervention for improving cognitive deficits as well as depressive symptoms would help to reduce de cognitive decline and to improve the type 2 diabetes treatment adherences as well as self-care and quality of life.
To conclude, in this cross-sectional assessment type 2 diabetes (including illness duration), high BMI and lower mood was linked to poorer cognitive function in older individuals with overweight/obesity and metabolic syndrome. A good glycaemia control in type 2 diabetes was associated with a better cognitive performance. Our results provide a controlled comprehensive model that integrates different neuropsychological and physical variables that are relevant in type 2 diabetes. The model also suggests that, to improve treatment adherence and quality of life once type 2 diabetes has been diagnosed, cognitive decline prevention strategies need to be implemented while closely monitoring depressive symptoms, BMI and glycaemia control. However, the etiopathogenic factors associated with the reported associations are still unclear.
References
American Diabetes Association. 2. Classification and diagnosis of diabetes. Diabetes Care 38, S8–S16 (2015).
Kirkman, M. S. et al. Diabetes in older adults. Diabetes Care 35, 2650–2664 (2012).
Federation International Diabetes. IDF Diabetes Atlas. (International Diabetes Federation, 2017).
Halling, A. & Berglund, J. Association of diagnosis of ischaemic heart disease, diabetes mellitus and heart failure with cognitive function in the elderly population. Eur J Gen Pr. 12, 114–119 (2006).
Gregg, E. W. Is Diabetes Associated With Cognitive Impairment and Cognitive Decline Among Older Women? Arch. Intern. Med. 160, 174 (2000).
Nandipati, S., Luo, X., Schimming, C., Grossman, H. T. & Sano, M. Cognition in non-demented diabetic older adults. Curr. Aging Sci. 5, 131–5 (2012).
Qiu, C. et al. Diabetes, markers of brain pathology and cognitive function: The Age, Gene/Environment Susceptibility-Reykjavik Study. Ann. Neurol. 75, 138–146 (2014).
Kanaya, A. M., Barrett-Connor, E., Gildengorin, G. & Yaffe, K. Change in cognitive function by glucose tolerance status in older adults: A 4-year prospective study of the Rancho Bernardo Study cohort. Arch. Intern. Med. 164, 1327–1333 (2004).
Logroscino, G., Kang, J. H. & Grodstein, F. Prospective study of type 2 diabetes and cognitive decline in women aged 70-81 years. BMJ 328, 548 (2004).
Grodstein, F., Chen, J., Wilson, R. S. & Manson, J. E. Type 2 diabetes and cognitive function in community-dwelling elderly women. Diabetes Care 24, 1060–5 (2001).
Elias, P. K. et al. NIDDM and blood pressure as risk factors for poor cognitive performance. The Framingham Study. Diabetes Care 20, 1388–1395 (1997).
Roberts, R. O. et al. Association of duration and severity of diabetes mellitus with mild cognitive impairment. Arch. Neurol. 65, 1066–1073 (2008).
The GBD 2015 Obesity Collaborators. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 377, 13–27 (2017).
Gunstad, J., Lhotsky, A., Wendell, C. R., Ferrucci, L. & Zonderman, A. B. Longitudinal examination of obesity and cognitive function: Results from the baltimore longitudinal study of aging. Neuroepidemiology 34, 222–229 (2010).
Wu, X., Nussbaum, M. A. & Madigan, M. L. Executive Function and Measures of Fall Risk Among People With Obesity. Percept. Mot. Skills 122, 825–839 (2016).
Elias, M. F., Elias, P. K., Sullivan, L. M., Wolf, P. A. & D’Agostino, R. B. Obesity, diabetes and cognitive deficit: The Framingham Heart Study. Neurobiol. Aging 26 (2005).
Zhao, Q. et al. . Diabetes is Associated with Worse Executive Function in Both Eastern and Western Populations: Shanghai Aging Study and Mayo Clinic Study of Aging. J. Alzheimer’s Dis. 47, 167–176 (2015).
Launer, L. J. et al. Effects of intensive glucose lowering on brain structure and function in people with type 2 diabetes (ACCORD MIND): A randomised open-label substudy. Lancet Neurol. 10, 969–977 (2011).
Cukierman-Yaffe, T. et al. Relationship between baseline glycemic control and cognitive function in individuals with type 2 diabetes and other cardiovascular rIsk factors the action to control cardiovascular risk in diabetes-memory in diabetes (ACCORD-MIND) trial. Diabetes Care 32, 221–226 (2009).
Zhang, D. A., Lam, V., Chu, V. & Li, M. Type 2 Diabetes with Comorbid Depression in Relation to Cognitive Impairment: an Opportunity for Prevention? Molecular Neurobiology 55, 1–5 (2017).
Sindi, S. et al. Sleep disturbances and later cognitive status: A multi-centre study. Sleep Med. https://doi.org/10.1016/j.sleep.2017.11.1149 (2018).
Alberti, K. et al. Harmonizing the Metabolic Syndrome A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention for the Study of Obesity. Circulation 120, 1640–1645 (2009).
Sanz, J., Perdigón, A. & Luis Vázquez Valverde, C. The Spanish adaptation of Beck’s Depression Inventory-II (BDI-II): 2. Psychometric properties in the general population. Clínica y Salud 14, 249–280 (2003).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, (DSM IV) (1994).
Periáñez, J. A. et al. Trail Making Test in traumatic brain injury, schizophrenia, and normal ageing: Sample comparisons and normative data. Arch. Clin. Neuropsychol. 22, 433–447 (2007).
Benton, A., Hamsher, K. deS. & Sivan, A. Multilingual Aphasia Examination. (Psychological Assessment Resources, Inc, 1994).
TEA, S. A. WAIS-III: Escala de inteligencia de Wechsler para Adultos. Tercera versión [WAIS-III: Wechsler adult Intelligence scale. Third version]. (TEA, 1999).
Kelley, K. & Preacher, K. J. On effect size. Psychol. Methods 17, 137–152 (2012).
Finner, H. On a monotonicity problem in step-down multiple test procedures. J. Am. Stat. Assoc. 88, 920–923 (1993).
von Oertzen, T. Power equivalence in structural equation modelling. Br. J. Math. Stat. Psychol. 63, 257–272 (2010).
Barrett, P. Structural equation modelling: Adjudging model fit. Pers. Individ. Dif. 42, 815–824 (2007).
Kumari, M. & Marmot, M. Diabetes and cognitive function in a middle-aged cohort: Findings from the Whitehall II study. Neurology 65, 1597–1603 (2005).
Ebady, S. A., Arami, M. A. & Shafigh, M. H. Investigation on the relationship between diabetes mellitus type 2 and cognitive impairment. Diabetes Res. Clin. Pract. 82, 305–309 (2008).
Genuth, S. et al. Long-term effect of diabetes and its treatment on cognitive function. N. Engl. J. Med. 356, 1842–1852 (2007).
Solas, M., Milagro, F. I., Ramírez, M. J. & Martínez, J. A. Inflammation and gut-brain axis link obesity to cognitive dysfunction: plausible pharmacological interventions. Current Opinion in Pharmacology 37, 87–92 (2017).
Baune, B. T. et al. Cognition in depression: Can we THINC-it better? J. Affect. Disord. 225, 559–562 (2018).
Ye, M. et al. Changes in body mass index and incidence of diabetes: A longitudinal study of Alberta’s Tomorrow Project Cohort. Prev. Med. (Baltim). 106, 157–163 (2018).
Burke, S. L. et al. Sex differences in the development of mild cognitive impairment and probable Alzheimer’s disease as predicted by the hippocampal volume or white matter hyperintensities. J. Women Aging 1–25 https://doi.org/10.1080/08952841.2018.1419476(2018).
Huntley, J. et al. Online assessment of risk factors for dementia and cognitive function in healthy adults. International Journal of Geriatric Psychiatry 33, e286–e293 (2017).
Acknowledgements
We thank the PREDIMED-PLUS Biobank Network, as a part of the National Biobank Platform of the Carlos III Health Institute (ISCIII) for storing and managing the PREDIMED-PLUS biological samples. Contract Juan Rodés from the Instituto de Salud Carlos III to Olga Castañer (JR17/00022) is duly acknowledged. This project is funded by Instituto de Salud Carlos III (ISCIII), the Spanish Government Official Agency for funding biomedical research - with competitive grants for the periods 2014–2016, 2015–2017, 2017–2019 and 2018–2020, through the Fondo de Investigación para la Salud (FIS), which is co-funded by the European Regional Development Fund [grants: PI13/00673, PI13/00492, PI13/00272, PI13/01123, PI13/00462, PI13/00233, PI13/02184, PI13/00728 PI13/01090 PI13/01056, PI14/01722, PI14/00636, PI14/00618, PI14/00696, PI14/01206, PI14/01919, PI14/00853, PI14/01374, PI16/00473, PI16/00662, PI16/01873, PI16/01094, PI16/00501, PI16/00533, PI16/00381, PI16/00366, PI16/01522, PI16/01120, PI17/00764, PI17/01183, PI17/00855, PI17/01347, PI17/00525, PI17/01827, PI17/00532, PI17/00215, PI17/01441, PI17/00508, PI17/01732, PI17/00926] and the European Research Council [Advanced Research Grant 2014–2019; agreement #340918]. Additional grants: Acciones Especiales from ISCIIII, Consejería-Salud, Junta -Andalucía [PI0458/2013, PS0358/2016], Recercaixa-grant 2013 [2013ACUP00194], a SEMERGEN grant, International Nut & Dried Fruit Council – FESNAD No. 201302. Fundación Patrimonio Comunal Olivarero is providing the necessary amounts of olive oil. Nuts were initially and only partially provided by Pistachios Growers and Almond Board of California, for the pilot study. This research was also partially funded by EU-H2020 Grant (Eat2beNICE/H2020-SFS-2016–2; Ref 728018), PI14/00290 y PI17/1167(Co-funded by European Regional Development Fund, ERDF, a way to build Europe), PERIS (Generalitat de Catalunya, SLT006/17/00077), Grant PROMETEO/2017/017 (Generalitat Valenciana) and Grant FEA/SEA 2017 for Primary Care Research. We thank CERCA Programme/Generalitat de Catalunya for institutional support. ML-M is supported by a predoctoral Grant of the Ministerio de Educación, Cultura y Deporte (FPU15/02911). None of these funding sources plays any role in the design, collection, analysis, or interpretation of the data or in the decision to submit manuscripts for publication.
Author information
Authors and Affiliations
Contributions
The authors’ responsibilities were as follows: F.F.-A., R.d.T., J.S.-S., D.C., M.A.M.-G., J.V., D.R., J.A.M., J.L.-M. and X.P. designed the study; M.M.-G., N.M.-B., E.T., A.C.-R., J.W., R.E., A.B.-C., A.A.-G, J.A.T., F.J.T., L.S.-M., V.M., J.L., C.V., X.P., J.V., L.D., J.J.G., P.M., E.R., P.B.-C., R.B., M.B., O.C., M.G.-H., A.M.Y., I.A., .A.G.-R., M.R.-C. and A.D.-L. conducted the study and collected the data; M.M.-G., N.M.-B., E.T., A.C.-R., J.W., R.E., A.B.-C., A.A.-G., J.A.T., F.J.T., L.S.-M., V.M., J.L., C.V., X.P., J.V., L.D., J.J.G., P.M., E.R., P.B.-C., R.B., M.B., O.C., M.G.-H., A.M.Y., I.A., A.G.-R., M.R.-C. and A.D.-L. provided essential materials; R.G analyzed the data; N.M.-B., M.L.-M., R.G., S.J.-M., F.F.-A. and R.d.T.: wrote the manuscript; N.M.-B., M.L.-M., R.G., A.C.-R., S.J.-M., F.F.-A., R.d.T. and J.S.-S. had primary responsibility for the final content; and all authors participated in data interpretation, provided critical review and commentary on the draft of the manuscript, and read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mallorquí-Bagué, N., Lozano-Madrid, M., Toledo, E. et al. Type 2 diabetes and cognitive impairment in an older population with overweight or obesity and metabolic syndrome: baseline cross-sectional analysis of the PREDIMED-plus study. Sci Rep 8, 16128 (2018). https://doi.org/10.1038/s41598-018-33843-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33843-8
Keywords
This article is cited by
-
Correlation between fat-to-muscle mass ratio and cognitive impairment in elderly patients with type 2 diabetes mellitus: a cross-sectional study
BMC Geriatrics (2024)
-
Dysglycemia, gender, and cognitive performance in older persons living with mild cognitive impairment: findings from a cross-sectional, population-based study
Aging Clinical and Experimental Research (2024)
-
Association between type 2 diabetes and depressive symptoms after a 1-year follow-up in an older adult Mediterranean population
Journal of Endocrinological Investigation (2024)
-
Associations of metabolic syndrome, its severity with cognitive impairment among hemodialysis patients
Diabetology & Metabolic Syndrome (2023)
-
Fingolimod Alleviates Cognitive Deficit in Type 2 Diabetes by Promoting Microglial M2 Polarization via the pSTAT3-jmjd3 Axis
Molecular Neurobiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.