Abstract
Hybridization of parasites can generate new genotypes with high virulence. The fungal amphibian parasite Batrachochytrium dendrobatidis (Bd) hybridizes in Brazil’s Atlantic Forest, a biodiversity hotspot where amphibian declines have been linked to Bd, but the virulence of hybrid genotypes in native hosts has never been tested. We compared the virulence (measured as host mortality and infection burden) of hybrid Bd genotypes to the parental lineages, the putatively hypovirulent lineage Bd-Brazil and the hypervirulent Global Pandemic Lineage (Bd-GPL), in a panel of native Brazilian hosts. In Brachycephalus ephippium, the hybrid exceeded the virulence (host mortality) of both parents, suggesting that novelty arising from hybridization of Bd is a conservation concern. In Ischnocnema parva, host mortality in the hybrid treatment was intermediate between the parent treatments, suggesting that this species is more vulnerable to the aggressive phenotypes associated with Bd-GPL. Dendropsophus minutus showed low overall mortality, but infection burdens were higher in frogs treated with hybrid and Bd-GPL genotypes than with Bd-Brazil genotypes. Our experiment suggests that Bd hybrids have the potential to increase disease risk in native hosts. Continued surveillance is needed to track potential spread of hybrid genotypes and detect future genomic shifts in this dynamic disease system.
Similar content being viewed by others
Introduction
The host-parasite dynamic is a classic example of an evolutionary arms race; hosts face pressure to evolve defenses against parasites, while parasites face pressure to overcome host defenses1,2. Compared to vertebrate hosts, parasites have the advantages of short generation times and large population sizes, which may promote rapid rates of evolution3. In many cases, vertebrate hosts overcome parasite pressure through sexual recombination, which allows for the forming of new and potentially beneficial gene combinations4,5,6. However, many parasites also possess dynamic genomes, further increasing their evolutionary potential7,8. Pathogen recombination through hybridization occurs when previously isolated lineages come into contact, a process which has intensified in our increasingly globalized world7,9,10,11. As a result, hybridization of parasites is an emerging concern for biodiversity conservation9,12,13.
Traits of hybrid parasites may differ in various ways from the parental lineages10,14. One possible outcome of hybridization is ‘hybrid vigor’ in which phenotypes exceed the ranges of the parent populations through mechanisms such as additive, complementary, or dominance gene interactions14,15,16,17. Disease progression can change rapidly to the detriment of hosts if fitness components such as parasite fecundity, infectivity, transmission, virulence, reproductive rate, ability to exploit novel host resources, or ability to evade host immunity are enhanced through hybridization13,18. For example, the mobility of a hybrid cucumber mosaic virus (genus Cucumovirus) was enhanced by a protein from the congeneric tomato aspermy virus19 and the viability and replicability of a hybrid avian influenza virus were enhanced by a protein segment from a human influenza virus20. Similarly, hybrids of the fungal insect pathogen Beauveria expressed the unique insecticidal functions of both parents, increasing the biocontrol efficiency against moth and beetle crop pests21,22. In addition, enhanced physiological traits such as tolerance of heat or host toxicity could allow for expansion of the geographic ranges or host specificities of parasites16. For instance, in the fungal plant pathogen Phytophthora, hybrids colonized an expanded array of hosts compared to the parent fungi10,23.
Alternatively, hybridization may produce phenotypes that are intermediate between the parental populations, a form of ‘hybrid breakdown’ that results from incompatibilities between interacting genes17. For instance, the proportion of deaths of guinea pigs infected with E. coli-Shigella hybrids was intermediate between those of guinea pigs infected with the parental bacteria24. Similarly, the average time until death in mice infected with a hybrid pseudorabies virus (genus Varicellovirus) was intermediate between those of mice infected with the parental viruses25. First generation crosses often show hybrid vigor, whereas later generation crosses often show hybrid breakdown because of disruption of co-adapted gene complexes26.
The fungal parasite Batrachochytrium dendrobatidis (Bd) causes the potentially lethal amphibian disease chytridiomycosis27. During its complex evolutionary history, Bd diverged into multiple genetic lineages which primarily reproduce clonally28. Epizootic outbreaks of chytridiomycosis are predominantly associated with spread of the most recently derived lineage of Bd, termed the Global Panzootic Lineage (Bd-GPL). These outbreaks occurred in the Andes29, Brazil30, Central America31, the Caribbean32, California33, Spain34, Cameroon35, New Zealand36, and eastern Australia37, mostly in the 1970s–2000s. In contrast to Bd-GPL, Bd lineages of Brazilian38, Chinese39, Japanese40, Korean41, Swiss, and South African32 origin diverged early in the evolutionary history of Bd and have not been directly linked to host population declines. Early diverging Bd lineages show high genetic structure, indicating long-term presence of Bd in select geographic areas28,39,40,41,42. Moreover, low incidence, prevalence, host burden, and host mortality associated with some ancestral lineages32,39,40,41,43,44, despite environmental suitability for Bd43,45, has led to the hypothesis that older Bd lineages are locally hypovirulent toward native hosts as an outcome of long-term co-evolution41,42,43,46. However, controlled studies have not been undertaken to determine the relative virulence of global and putatively enzootic Bd genotypes across panels of local hosts.
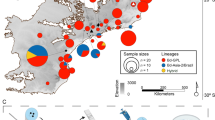
The Atlantic Forest of southeastern Brazil is a major amphibian biodiversity hotspot47, despite high levels of deforestation and forest fragmentation48,49, and is the only known region in which divergent lineages of Bd coexist and hybridize38,42. The enzootic Brazilian lineage of Bd (Bd-Brazil) occurs in the southern Atlantic Forest, where it appears to have evolved with a relatively small group of endemic host species against a backdrop of specific microclimatic conditions42. In contrast, Bd-GPL appears to be effective at dispersing across the microclimatically diverse landscape of this region and infects a wide array of native and introduced amphibian hosts42, despite estimates that it was introduced to the Atlantic Forest only within the last few centuries50. Hybrid genotypes between Bd-Brazil and Bd-GPL were recently detected in a mountain range within the geographic range of Bd-Brazil38,42. Amphibian declines and extirpations in the Atlantic Forest have been linked to Bd30, but it is unknown whether outbreaks resulted from the introduction of Bd-GPL, an increase in virulence of Bd-Brazil, the emergence of GPL-Brazil hybrids, or some combination of these factors30. Thus, testing the relative virulence of these genotypes is a research priority, especially considering that the section of the Atlantic Forest in which amphibian declines and extirpations were most strongly linked to Bd overlaps with the hybrid zone30.
Our aim was to compare the virulence (ability to cause harm to the host) of hybrid Bd genotypes to the parental lineages Bd-Brazil and Bd-GPL, in native Brazilian hosts. In keeping with the hypothesis that Bd-Brazil shared a longer co-evolutionary history with Brazilian frog species, we predicted that Bd-Brazil would have lower virulence in native Brazilian frog species than Bd-GPL and hybrid genotypes. The relative virulence of Bd-GPL and hybrid genotypes were more difficult to predict because the influence of genetic recombination on Bd virulence factors has never been tested, so we proposed two possible hypotheses. First, hybrid genotypes could have intermediate virulence compared to the parental lineages. Second, hybrid genotypes could have higher virulence than both parental lineages. Under controlled laboratory conditions, we inoculated two relatively Bd-intolerant direct-developing frog species (Brachycephalus ephippium and Ischnocnema parva) and one more tolerant aquatic-breeding frog species (Dendropsophus minutus) with Bd isolates representing Bd-Brazil, Bd-GPL, and hybrids. We measured relative virulence by comparing mortality rates and infection burdens among treatments and considered the implications of our results for the evolutionary future of this ecologically important parasite and its diverse amphibian hosts.
Results
Brachycephalus ephippium and I. parva (host species with direct development)
The ranked mortality by treatment (from highest to lowest) for B. ephippium was: hybrid, Bd-GPL, and Bd-Brazil. (χ2 = 33.938; d.f. = 3; p < 0.0001; Fig. 1a). Mortality in B. ephippium exposed to Bd-Brazil was similar to the control (p = 0.100; Fig. 1a). Mortality was first observed in frogs exposed to Bd-Brazil on day 35 and 2/9 frogs (22%) died by the end of the experiment on day 40 (Fig. 1a). In contrast, frogs exposed to Bd-GPL (p = 0.002) and the hybrid (p < 0.0001) had higher mortality rates than in the control, and the mortality rate was higher in the hybrid treatment than in the Bd-GPL treatment (p = 0.0008). Mortality was first observed in frogs exposed to the hybrid on day 19 and 9/9 frogs (100%) died by day 34 (Fig. 1a). Mortality was first observed in frogs exposed to Bd-GPL on day 26 and 6/9 frogs (67%) died by the end of the experiment (Fig. 1a).
The ranked mortality by treatment for I. parva was: Bd-GPL, hybrid, and Bd-Brazil (χ2 = 36.723; d.f. = 3; p < 0.0001; Fig. 1b). Mortality in I. parva exposed to Bd-Brazil was similar to the control (p = 0.071; Fig. 1b). Mortality was first observed in frogs treated with Bd-Brazil on day 52 and 2/5 frogs (40%) died by the end of the experiment on day 58 (Fig. 1b). In contrast, frogs exposed to Bd-GPL (p < 0.0001) and the hybrid (p = 0.0003) had higher mortality rates than in the control, and the mortality rate was higher in the Bd-GPL treatment than in the hybrid treatment (p < 0.0001). Mortality was first observed in frogs exposed to Bd-GPL on day 18 and 6/6 frogs (100%) died by day 27 (Fig. 1b). Mortality was first observed in frogs exposed to the hybrid on day 31 and 6/7 frogs (86%) died by the end of the experiment (Fig. 1b). All direct-developing individuals that were not experimentally inoculated survived during both experiments.
Infection loads at the time of mortality were greater in frogs exposed to the hybrid than to Bd-GPL (F [22,3] = 92.349; r2 = 0.926; p < 0.0001) but the magnitude of this effect was greater for B. ephippium than for I. parva (t = −2.89; p = 0.0084). For B. ephippium, frogs exposed to Bd-GPL had an average infection load (±SD) of 3,762 (±2,115) zoospore genome equivalents (g.e.) at the time of mortality (range = 1,116–6,555 g.e.), whereas frogs exposed to the hybrid had an average infection load of 12,441 (±6,091) g.e. at the time of mortality (range = 4,260–22,876 g.e.; Fig. 2). In contrast, infection loads at the time of mortality for I. parva were at least one order of magnitude higher than for B. ephippium and were more similar between Bd-GPL (mean ± SD = 213,961 ± 85,611 g.e.; range = 123,681–321,625 g.e.) and the hybrid (221,062 ± 109,100 g.e.; range = 2,303–409,665 g.e.; Fig. 2).
Dendropsophus minutus (host species with aquatic larval development)
We did not detect differences in mortality of D. minutus among infection treatments or between infection and control treatments (χ2 = 8.297; d.f. = 9, p = 0.504). Mortality was first observed in frogs exposed to Bd-Brazil on day 19 and 12/30 frogs (40%) died by the end of the experiment on day 60 (CLFT 041: 4/10 [40%], CLFT 142: 5/10 [50%], CLFT 150: 3/10 [30%]). Mortality was first observed in frogs exposed to Bd-GPL on day 10 and 14/30 frogs (47%) died by the end of the experiment (CLFT 073: 4/10 [40%], CLFT 131: 5/10 [50%], CLFT 137: 5/10 [50%]). Mortality was first observed in frogs inoculated with hybrid genotypes on day 14 and 13/30 frogs (43%) died by the end of the experiment (CLFT 024.2: 3/10 [30%], CLFT 160: 4/10 [40%], CLFT 039: 6/10 [60%]). Mortality was first observed in control frogs on day 22 and 3/20 frogs (15%) died by the end of the experiment.
We detected differences in infection loads on day 60 within and among treatments (p < 0.001; Fig. 3). Average infection loads on frogs exposed to hybrids and Bd-GPL were highly variable within treatments (hybrids: 7,356 g.e.; 8,679 g.e.; 248,663 g.e.; Bd-GPL: 9,893 g.e.; 49,316 g.e.; 141,879 g.e.; Fig. 3). Average infection loads within the Bd-Brazil treatment were less variable and were generally lower than in frogs treated with hybrids and Bd-GPL (Bd-Brazil: 3,437 g.e.; 17,614 g.e.; 26,169 g.e.; Fig. 3). Thirteen of 20 control frogs carried natural Bd infections. Average infection loads on control frogs were lower than those on treatment frogs, likely reflecting what they were carrying in the field (Fig. 3). Passage rate was not a significant predictor of Bd infection loads, independent of Bd lineage (F = 0.228, p = 0.634).
Average infection burdens 60 days post-inoculation (±SE) for Dendropsophus minutus inoculated with Bd-Brazil (green bars), Bd-GPL (red bars), and hybrid genotypes (yellow bars); each bar represents a different Bd genotype. Gray bar depicts natural infection burdens of the control group after 60 days incubation.
Discussion
Amphibian declines and extirpations throughout the Atlantic Forest have been attributed to Bd, and the zone of hybridization between Bd-Brazil and Bd-GPL is within the region with the clearest signature of disease-related mortality, underscoring the need to test the virulence of hybrid genotypes in native hosts30,42. In our direct-developing host species, mortality rates of frogs exposed to a hybrid genotype were higher (B. ephippium) or intermediate (I. parva) compared to Bd-Brazil and Bd-GPL. This finding indicates that hybrid virulence is context specific; in certain scenarios these genotypes may produce disease outcomes that far exceed parental ranges, while in others these genotypes may elicit intermediate disease outcomes. Since it is well-established that Bd has a dynamic genome and is now nearly globally distributed, this finding highlights the importance of global surveillance to detect future genomic shifts in this disease system that could lead to new outbreaks of chytridiomycosis, especially because a non-Brazilian host (Lithobates sylvaticus) also exhibited increased mortality in response to a Bd hybrid relative to the parental genotypes in a preliminary trial51. In addition, in other chytrid fungi, sexual reproduction produces a thick-walled resting stage that is capable of tolerating desiccation and high levels of heat and salinity52. Resting spores can also be produced by asexual reproduction, as in the chytrid Rhizophydium brooksianum53, which is in the same taxonomic order (Rhizophydiales) as Bd. No definitive evidence of a resting stage in Bd has been observed to date54,55, but this would have important implications for the geographic and temporal scales at which transmission of Bd could occur.
The relative virulence of hybrid genotypes and Bd-GPL appeared to depend on host factors, highlighting the context dependency of disease outcomes that is a hallmark of this disease system56,57. Compared to I. parva, B. ephippium died with relatively low Bd burdens, suggesting a defense tradeoff in which B. ephippium invested more resources in resistance defenses (minimizing parasite burden) at the expense of tolerance defenses (minimizing harm caused by parasites, such as mortality effects in this study)58. Theoretically, resistance strategies are expected to reduce pathogen fitness, eliciting a strong antagonistic host-pathogen co-evolution59,60. The effectiveness of an immune defense strategy that is heavily influenced by host-pathogen co-evolution should be positively correlated with the length of time that the host has been exposed to the parasite. In accordance with this expectation, B. ephippium exhibited the longest incubation periods and highest survival rate (variables indicating the most developed resistance defenses) when exposed to Bd-Brazil, the lineage with which this species has probably co-existed in the wild for the longest time period. It follows that incubation periods and survival were intermediate when B. ephippium was exposed to Bd-GPL, the lineage with which this species has probably co-existed in the wild for a time period intermediate between Bd-Brazil and hybrid genotypes. Lastly, this species exhibited the shortest incubation periods and lowest survival rate, as well as the highest infection burdens at the time of death (variables indicating the least developed resistance defenses), when exposed to hybrid genotypes, the group with which this species has probably co-existed in the wild for the shortest time period, or not at all.
These findings indicate that B. ephippium may be particularly vulnerable to infections by hybrid genotypes in nature, primarily due to the relative novelty of these genotypes. The vulnerability of B. ephippium to novel pathogens may be linked to its association with patchy, high-altitude habitats, which may have limited its exposure to pathogens throughout its evolutionary history or lowered its immunogenetic diversity61,62,63. In the case of Bd, anthropogenic habitat alteration may tip the host-pathogen balance further in favor of the pathogen, as deforestation may promote the evolutionary isolation of B. ephippium while at least some relatively novel genotypes of Bd appear highly competent at dispersing across even the most fragmented landscapes in the Atlantic Forest, such as our B. ephippium collection site at Jundiái42. Previous studies indicate that F1 hybrids often show hybrid vigor because of outbreeding enhancement, whereas F2 hybrids might express hybrid breakdown as a result of recombination26. Considering that all living Bd hybrid isolates are first-generation crosses (TYJ pers. comm.)64, we are unable to test whether our results for B. ephippium represent an instance of hybrid vigor that could weaken through subsequent crosses16. However, an F2 backcrossed to Bd-Brazil was observed in the field and its survival provides some indication that parasitemia of Bd hybrids may not be limited to the F1 generation64.
In contrast to B. ephippium, I. parva died with relatively high Bd burdens, suggesting the reverse defense tradeoff in which I. parva invested more resources in tolerance defenses at the expense of resistance defenses. Tolerance strategies are not expected to negatively affect the success of pathogen populations, so we would expect relatively weak co-evolutionary pressures between parasites and hosts that invest primarily in this type of defense strategy59,60. This may help to explain why the responses of I. parva to our panel of Bd treatments were not correlated with the relative length of time to which this species was probably exposed to each lineage in the wild. Rather, it is possible that this species was more responsive to the expression of hybrid phenotypes that were intermediate between the hypervirulent Bd-GPL and the hypovirulent Bd-Brazil, resulting in intermediate incubation periods and mortality in frogs exposed to the hybrid compared to Bd-GPL and Bd-Brazil. Our findings suggest that I. parva is particularly vulnerable to pathogens with aggressive phenotypes, such as Bd-GPL, regardless of the extent to which this species has shared an evolutionary history with co-occurring pathogens. Thus, an important avenue for future study is to determine the genetic or physiological factors that make Bd-GPL particularly damaging to hosts and the ecological backdrop against which these genotypes emerged. Immunological comparisons of disease progression among Bd genotypes and host species are necessary to verify the mechanisms we have proposed to explain the host-specific patterns in our data. Nevertheless, our results offer convincing evidence that both Bd-GPL and hybrid genotypes are virulent in the Atlantic Forest.
Compared to hybrid genotypes and Bd-GPL, frogs treated with Bd-Brazil had the lowest ranked mortality in both direct-developing host species and the lowest infection burdens in D. minutus, consistent with the hypothesis that Bd-Brazil has shared a long co-evolutionary history with endemic Brazilian frogs and is hypovirulent in endemic hosts28,38,42,50. A recent study also found that the Bd-Brazil genotype CLFT 001 (isolated from the Atlantic Forest) exhibited lower in vitro growth performance than the Bd-GPL genotype CJB5-2 and the Bd-Brazil genotype UM 142 (unknown geographic origin)65, another indication that the relative threat of endemic Atlantic Forest Bd genotypes is low. In contrast, the only other study that tested the relative virulence of Bd-Brazil and Bd-GPL reported that 50% of hosts died when exposed to Bd-Brazil and that the virulence of Bd-Brazil (one genotype) fell within the range of virulence shown by Bd-GPL (three genotypes)66. This conflicting study used the Bd-Brazil genotype UM 142, different Bd-GPL genotypes (from the eastern U.S. and Panama), and a North American host species. Our conflicting results may thus reflect host-independent variability in virulence within Bd lineages or suggest that Bd-Brazil has higher virulence toward non-native hosts. Evidence suggests that humans are facilitating the spread of Bd in South America67 and globally38. For instance, the Bd-Brazil genotype UM 142 was isolated from a bullfrog (Lithobates catesbeianus) collected from a U.S. amphibian market. We reiterate the concern raised by Becker et al.66 that lineages endemic to one region may lead to declines of naïve host populations in other regions.
Terrestrial, direct-developing frog species have typically been considered less vulnerable to chytridiomycosis than species with more aquatic life histories that may experience high levels of exposure to aquatic Bd zoospores68,69,70. However, both of our terrestrial study species acquired heavy Bd infections and experienced mortality from chytridiomycosis under laboratory conditions mimicking the microhabitat of direct-developing frogs in nature, consistent with laboratory data for other Brazilian direct-developing species63. In the wild, Brazilian direct-developers had high Bd infection loads71,72, but low infection prevalence63, the latter of which could be underestimated if frogs die quickly from infections or if sick frogs remain stationary in hidden refugia63,73. Even if low prevalence of Bd is currently facilitating population persistence of direct-developing host species in Brazil, this host-pathogen balance is precarious in an era of global change61. For example, in Puerto Rico, Bd dynamics in the direct-developing species Eleutherodactylus coqui shifted from enzootic to epizootic when extreme drought conditions associated with global climate change forced frogs to congregate in humid refugia, increasing transmission and reinfection rates73,74. Moreover, reduced levels of recruitment stemming from increased Bd-related mortality of juvenile E. coqui has led to recent, low-level population declines75,76, similar to other examples of negative population effects from Bd in the absence of drastic epizootic events77,78,79. Future research should investigate the potential effects of Bd on the population persistence of Brazil’s diverse direct-developing amphibian fauna, especially considering that the narrow geographic ranges of many Brazilian direct-developers leave them vulnerable to other natural and anthropogenic stressors and that Bd caused population declines of a direct-developing Arthroleptis in Cameroon35 and likely the extinction of several direct-developing species in the Atlantic Forest (see Supplementary Table S2).
In contrast to our direct-developing species, mortality rates of D. minutus were low across treatments, suggesting that this species is relatively tolerant to Bd regardless of variation in the genetic attributes of the fungus. This finding is consistent with previous studies of D. minutus in the laboratory63,80 and in the wild81. Patterns of infection loads in D. minutus by lineage matched mortality rates in our direct-developing species, with the lowest infection loads in frogs exposed to Bd-Brazil and higher infection loads in frogs exposed to Bd-GPL and hybrid genotypes, corroborating the evidence from our direct-developing species that Bd-Brazil is less virulent in native hosts. Infection loads were also less variable among genotypes within Bd-Brazil than within Bd-GPL and hybrid genotypes, which could reflect consistency in host immune responses stemming from long-term exposure to Bd-Brazil.
Our study shows that hybridization can be associated with high levels of virulence in Bd. Hybrid Bd genotypes emerged relatively recently in Brazil, at some point after the introduction of Bd-GPL to Brazil in the last few centuries41. The known distribution of hybrid genotypes is small (an isolated mountain range in the Atlantic Forest), which could suggest that it is functioning primarily as a parasite with intermediate rather than extreme fitness relative to parental populations and could be outcompeted by quick-dispersing Bd-GPL. However, it is unknown whether the current geographic distribution of hybrid genotypes is a product of their short evolutionary history (i.e., they might currently be spreading), their specific microhabitat requirements, or gaps in field sampling. Nevertheless, their coexistence with Bd-GPL suggests that they could be adept competitors in Brazilian landscapes. Applying newly developed, non-invasive techniques that can discriminate among Bd genotypes on skin swabs would help to paint a more complete picture of the spatial distribution of Bd lineages in Brazil as well as coinfection dynamics82. The same techniques could also be used with contemporary and retrospective sampling of preserved specimens to increase our understanding of which genotypes and lineages are associated with amphibian mortality in Brazil, track the spread of Bd-GPL through time, and determine whether the geographic distributions of hybrid genotypes and Bd-Brazil have changed over time82. Another useful avenue for future study is to determine how coexistence and hybridization of Bd genotypes in Brazil could influence host population recoveries through adaptive responses, which have been documented in other regions in the decades following mass declines83,84.
Our results do not point definitively to a single genetic culprit of Bd-related amphibian declines in Brazil. Bd-GPL is likely to have played a large role given its high level of virulence in some hosts and widespread geographic distribution42, but the hybrid zone overlaps the region with significant evidence of disease-related mortality30, and our results indicate that some host species are especially vulnerable to new genotypes, suggesting a possible role of hybrids in declines. Taking all these factors into account, a plausible scenario for disease-linked amphibian declines in Brazil is that the impacts of the introduction of Bd-GPL were exacerbated by the emergence of hybrids, possibly by overloading host immune systems with an even more genetically and spatiotemporally diverse assemblage of pathogen strains. As much as evolutionary novelty can aid species adaptation in an era of rapid environmental change, our findings underscore that this plasticity can also be advantageous for parasites, with serious consequences for the persistence of host populations13,16.
Materials and Methods
Study species
We selected three experimental host species with varying life histories and levels of tolerance (i.e., ability to minimize harm caused by parasites, such as mortality) to Bd. Dendropsophus minutus (Hylidae: Dendropsophinae) is a habitat generalist tree frog with indirect development and a close association with water bodies throughout its life history. The geographic range of this species covers most of tropical South America85,86, and it exhibits relatively high survival rates when challenged with Bd-GPL under laboratory conditions63,80. Ischnocnema parva and Brachycephalus ephippium (Brachycephalidae) are direct-developing leaf litter frogs that occur in southeastern Brazil85 and exhibit relatively low survival rates when challenged with Bd-GPL under laboratory conditions63. All three species are listed as Least Concern by the International Union for the Conservation of Nature87. In a vulnerability assessment of amphibians of the Brazilian Atlantic Forest, all three species were considered to have large geographic ranges and high local abundances but D. minutus and I. parva were classified as having wide habitat specificities, whereas B. ephippium was classified as having a narrow habitat specificity due to its association with high-altitude habitats47.
We collected adults of our study species in the municipality of Jundiaí, near Serra do Japi (B. ephippium and D. minutus) and in the municipality of São Luiz do Paraitinga, adjacent to Parque Estadual da Serra do Mar, Núcleo Santa Virgínia (I. parva), São Paulo state, Brazil. Both collection sites are located to the north of the Bd hybrid zone identified by Jenkinson et al.41. Some D. minutus tested positive for Bd at the time of collection but we elected not to treat infections to avoid any negative treatment-associated side effects and instead controlled for natural infections in our statistical analyses88. We only used I. parva and B. ephippium that tested negative for Bd in the field. For the experiments, frogs were randomly assigned to treatments and housed individually in plastic containers with sterile moist sphagnum (all species) and a sterile leaf for cover (B. ephippium and I. parva). Frogs were fed pinhead crickets ad libitum during the experiments.
Bd isolates
Bd isolates were obtained from infected tadpoles sampled throughout the Atlantic Forest of Brazil from 2011 to 201542. Tadpoles were screened in the field with a 10X hand lens for signs of chytrid infection by assessing the level of oral tissue dekeratinization89. Animals with signs of Bd infection were euthanized and oral tissues excised. Infected tissues were prepared for pathogen isolation on 1% tryptone agar with 0.2 mg.mL−1 penicillin-G and 0.4 mg.mL−1 streptomycin sulfate. Isolates of Bd were maintained on 1% tryptone agar at 21–23 °C until sufficient growth had occurred for DNA extraction. Isolates were genotyped to determine Bd lineage/group (Bd-Brazil, Bd-GPL, or hybrid) following the procedures described in Jenkinson et al.41. Cultures were maintained at 4 °C at Universidade Estadual de Campinas, UNICAMP, Brazil, and passaged every 4 mo.
Challenge experiment
We cultured three hybrid, three Bd-Brazil, and four Bd-GPL genotypes (see Supplementary Table S1) in Petri dishes containing 1% tryptone agar at 19 °C for 7 d. The three hybrid genotypes represent all living hybrid isolates, and analyses of whole genome sequences indicate that all are F1 hybrids (TYJ pers. comm.)64. The Bd-Brazil and Bd-GPL genotypes were selected haphazardly. To inoculate frogs with Bd (day zero), we filled each Petri dish with 5 ml of distilled water for 30 minutes and scraped the substrate with a sterile scalpel to facilitate zoospore release. We then transferred the liquid contents of each dish to a sterile beaker, sampled 1 ml of the solution to quantify the zoospore concentration with a hemocytometer, and diluted the solution with distilled water to obtain the desired zoospore concentration for experimental inoculations.
We inoculated D. minutus with hybrid genotypes CLFT 024.2, CLFT 039, and CLFT 160; Bd-Brazil genotypes CLFT 041, CLFT 142, and CLFT 150; and Bd-GPL genotypes CLFT 073, CLFT 131, and CLFT 137. We inoculated 10 frogs with each genotype. Each frog was inoculated individually in a Petri dish containing 1.8 × 106 zoospores in 2.5 ml of distilled water for 45 minutes. We exposed 20 additional individuals to the same volume of distilled water as Bd-unexposed controls.
We inoculated B. ephippium and I. parva with hybrid genotype CLFT 160 (B. ephippium: n = 9; I. parva: n = 7), Bd-Brazil genotype CLFT 150 (B. ephippium: n = 9; I. parva: n = 5), and Bd-GPL genotype CLFT 156 (B. ephippium: n = 9; I. parva: n = 6). Due to technical difficulties with preparing Bd-GPL for this component of the experiment, we were unable to use one of the three Bd-GPL genotypes that were tested with D. minutus. We inoculated each frog individually in a Petri dish containing 3.375 × 106 zoospores in 1.5 ml of distilled water for 45 minutes. We exposed additional individuals to the same volume of distilled water as controls (B. ephippium: n = 8; I. parva: n = 6).
Survival was monitored daily, dead animals were noted, and dying animals were euthanized with an overdose of the anesthetic MS-222 if they showed lack of righting response, which is a typical sign of advanced stages of chytridiomycosis90. Dead animals were swabbed immediately following the protocol described by Hyatt et al.91. The experiment concluded on day 60 (D. minutus), 58 (I. parva), or 40 (B. ephippium; length of experiment truncated because this species can become stressed after long periods in captivity), at which point all remaining animals were swabbed and euthanized.
We extracted DNA from skin swabs using 50 ml PrepMan Ultra and screened samples for Bd presence and load using Taqman qPCR assays92. For D. minutus, we used Bd genotype-specific standard curves (for each genotype used in the experiment) ranging from 0.1 to 1000 zoospore genome equivalents (g.e.). For B. ephippium and I. parva, we built standard curves (0.1–1000 g.e.) using CLFT 159, a Bd-GPL genotype isolated from a Hylodes frog collected in the Atlantic Forest. We were unable to standardize infection load data for B. ephippium and I. parva due to unforeseen culturing difficulties with one of our genotypes, but we feel confident in using non-standardized infection loads in our analyses for these species because standardization did not influence the overall patterns in our data for D. minutus (see Supplementary Fig. S1).
All experimental protocols were approved by Instituto Chico Mendes de Conservação da Biodiversidade –Instituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis/Brazil (Permits 29964–11, 27745–13, and 57098–1) and the local Animal Care and Use Committee (Comissão de Ética no Uso de Animal – CEUA/UNESP permit #29/2016). Our experiment was carried out in accordance with all ethics guidelines and regulations.
Statistical analyses
We used proportional hazards survival analyses93 to compare mortality rates among frogs exposed to Bd-Brazil, Bd-GPL, and hybrids, independently for each species. For B. ephippium and I. parva, we compared mortality rates among one genotype from each of the three treatments. For D. minutus, we compared mortality rates among nine Bd genotypes within the three treatments, including genotype as a fixed effect.
We used a Generalized Linear Mixed Model (GLMM) to compare infection loads on day 60 among D. minutus exposed to Bd-Brazil, Bd-GPL, and hybrids. In this model, we included the following explanatory variables: genotype, lineage/group, and the interaction between genotype and lineage/group as fixed effects, and infection load at the time of capture in the wild as a random effect. We also performed a GLMM to test for effects of passage rate on Bd loads of D. minutus with Bd lineage/group as a random effect.
For B. ephippium and I. parva, we performed independent General Linear Models (standard least squares) to compare infection loads at the time of mortality between frogs exposed to Bd-GPL and hybrids. For these models, we included log10-transformed infection loads as the response variable and Bd lineage/group (Bd-GPL or hybrid), host species (B. ephippium or I. parva), and the interaction between lineage/group and host species as explanatory variables.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Van Valen, L. A new evolutionary law. Evolutionary Theory 1, 1–30 (1973).
Ewald, P. W. The evolution of virulence: a unifying link between parasitology and ecology. J. Parasitol. 81, 659–669 (1995).
Hamilton, W. D. Sex versus non-sex versus parasite. Oikos 35, 282–290 (1980).
Hamilton, W. D., Axelrod, R. & Tanese, R. Sexual reproduction as an adaptation to resist parasites (a review). Proc. Natl. Acad. Sci. USA 87, 3566–3573 (1990).
Levin, D. A. Pest pressure and recombination systems in plants. Am. Nat. 109, 437–451 (1975).
Jaenike, J. A hypothesis to account for the maintenance of sex within populations. Evol. Theory 94, 191–194 (1978).
Stukenbrock, E. H. The role of hybridization in the evolution and emergence of new fungal plant pathogens. Phytopathology 106, 104–112 (2016).
Stukenbrock, E. H., Christiansen, F. B., Hansen, T. T., Dutheil, J. Y. & Schierup, M. H. Fusion of two divergent fungal individuals led to the recent emergence of a unique widespread pathogen species. Proc. Natl. Acad. Sci. USA 109, 1–6 (2012).
Brasier, C. The rise of the hybrid fungi. Nature 405, 134–135 (2000).
Olson, Å. & Stenlid, J. Pathogenic fungal species hybrids infecting plants. Microbes Infect. 4, 1353–1359 (2002).
Brasier, C. M., Cooke, D. E. & Duncan, J. M. Origin of a new Phytophthora pathogen through interspecific hybridization. Proc. Natl. Acad. Sci. USA 96, 5878–5883 (1999).
Ghosh, P. & Fisher, M. C. Dr. Jekyll and Mrs Hyde: Risky hybrid sex by amphibian-parasitizing chytrids in the Brazilian Atlantic Forests. Mol. Ecol. 25, 2961–2963 (2016).
King, K. C., Stelkens, R. B., Webster, J. P., Smith, D. F. & Brockhurst, M. A. Hybridization in parasites: consequences for adaptive evolution, pathogenesis, and public health in a changing world. Plos Pathog. 11, 1–12 (2015).
Grigg, M. E. et al. Success and virulence in Toxoplasma as the result of sexual recombination between two distinct ancestries. Science 294, 161–165 (2001).
Dittrich-Reed, D. R. & Fitzpatrick, B. M. Transgressive hybrids as hopeful monsters. Evol. Biol. 40, 310–315 (2013).
Pereira, R. J., Barreto, F. S. & Burton, R. S. Ecological novelty by hybridization: experimental evidence for increased thermal tolerance by transgressive segregation in Tigriopus californicus. Evolution (N. Y). 68, 204–215 (2014).
Johansen-Morris, A. D. & Latta, R. G. Fitness consequences of hybridization between ecotypes of Avena barbata: hybrid breakdown, hybrid vigor, and transgressive segregation. Evolution (N. Y). 60, 1585–1595 (2006).
Schelkle, B., Faria, P. J., Johnson, M. B., van Oosterhout, C. & Cable, J. Mixed infections and hybridisation in monogenean parasites. Plos One 7 (2012).
Ding, S. W., Shi, B. J., Li, W. X. & Symons, R. H. An interspecies hybrid RNA virus is significantly more virulent than either parental virus. Proc. Natl. Acad. Sci. USA 93, 7470–7474 (1996).
Li, C. et al. Reassortment between avian H5N1 and human H3N2 influenza viruses creates hybrid viruses with substantial virulence. Proc. Natl. Acad. Sci. 107, 4687–4692 (2010).
Couteaudier, Y., Viaud, M. & Riba, G. Genetic nature, stability, and improved virulence of hybrids from protoplast fusion in Beauveria. Microb. Ecol. 32, 1–10 (1996).
Viaud, M. et al. Molecular analysis of hypervirulent somatic hybrids of the entomopathogenic fungi Beauveria bassiana and Beauveria sulfurescens. Appl. Environ. Microbiol. 64, 88–93 (1998).
Ersek, T., English, J. T. & Schoelz, J. E. Creation of species hybrids of Phytophthora with modified host ranges by zoospore fusion. Phytopathology 85, 1343–1347 (1995).
Falkow, S., Schneider, H., Baron, L. S. & Formal, S. B. Virulence of Escherichia-Shigella genetic hybrids for the guinea pig. J. Bacteriol. 86, 1251–1258 (1963).
Berns, A., van den Ouweland, A., Quint, W., van Oirschot, J. & Gielkens, A. Presence of markers for virulence in the unique short region or repeat region or both of pseudorabies hybrid viruses. J Virol 53, 89–93 (1985).
Edmands, S. Heterosis and outbreeding depression in interpopulation crosses spanning a wide range of divergence. Evolution (N. Y). 53, 1757–1768 (1999).
Longcore, J. E., Pessier, A. P. & Nichols, D. K. Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia 91, 219–227 (1999).
Rosenblum, E. B. et al. Complex history of the amphibian-killing chytrid fungus revealed with genome resequencing data. Proc. Natl. Acad. Sci. USA 110, 9385–90 (2013).
Catenazzi, A., Lehr, E., Rodriguez, L. O. & Vredendurg, V. T. Batrachochytrium dendrobatidis and the collapse of anuran species richness and abundance in the upper Manu National Park, southeastern Peru. Conserv. Biol. 25, 382–391 (2011).
Carvalho, T., Becker, C. G. & Toledo, L. F. Historical amphibian declines and extinctions in Brazil linked to chytridiomycosis. Proc. R. Soc. B Biol. Sci. 284, 20162254 (2017).
Lips, K. R. Mass mortality and population declines of anurans at an upland site in western Panama. Conserv. Biol. 13, 117–125 (1999).
Farrer, R. A. et al. Multiple emergences of genetically diverse amphibian-infecting chytrids include a globalized hypervirulent recombinant lineage. Proc. Natl. Acad. Sci. 108, 18732–18736 (2011).
Vredenburg, V. T., Knapp, R. A., Tunstall, T. S. & Briggs, C. J. Dynamics of an emerging disease drive large-scale amphibian population extinctions. Proc. Natl. Acad. Sci. USA 107, 9689–9694 (2010).
Bosch, J., Mart nez-Solano, I. & Garcia-Paris, M. Evidence of a chytrid fungus infection involved in the decline of the common midwife toad (Alytes obstetricans) in protected areas of central Spain. Biol. Conserv. 97, 331–337 (2001).
Hirschfeld, M. et al. Dramatic declines of montane frogs in a Central African biodiversity hotspot. Plos One 11, e0155129 (2016).
Bell, B. D., Carver, S., Mitchell, N. J. & Pledger, S. The recent decline of a New Zealand endemic: how and why did populations of Archey’s frog Leiopelma archeyi crash over 1996–2001? Biol. Conserv. 120, 189–199 (2004).
Berger, L. et al. Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc. Natl. Acad. Sci. USA 95, 9031–9036 (1998).
Schloegel, L. M. et al. Novel, panzootic and hybrid genotypes of amphibian chytridiomycosis associated with the bullfrog trade. Mol. Ecol. 21, 5162–5177 (2012).
Bai, C., Liu, X., Fisher, M. C., Garner, T. W. J. & Li, Y. Global and endemic Asian lineages of the emerging pathogenic fungus Batrachochytrium dendrobatidis widely infect amphibians in China. Divers. Distrib. 18, 307–318 (2012).
Goka, K. et al. Amphibian chytridiomycosis in Japan: distribution, haplotypes and possible route of entry into Japan. Mol. Ecol. 18, 4757–4774 (2009).
Bataille, A. et al. Genetic evidence for a high diversity and wide distribution of endemic strains of the pathogenic chytrid fungus Batrachochytrium dendrobatidis in wild Asian amphibians. Mol. Ecol. 22, 4196–4209 (2013).
Jenkinson, T. S. et al. Amphibian-killing chytrid in Brazil comprises both locally endemic and globally expanding populations. Mol. Ecol. 25, 2978–2996 (2016).
Swei, A. et al. Is chytridiomycosis an emerging infectious disease in Asia? Plos One 6, e23179 (2011).
Kusrini, M. D., Skerratt, L. F., Garland, S., Berger, L. & Endarwin, W. Chytridiomycosis in frogs of Mount Gede Pangrango, Indonesia. Dis. Aquat. Organ. 82, 187–194 (2008).
Rödder, D., Kielgast, J. & Lötters, S. Future potential distribution of the emerging amphibian chytrid fungus under anthropogenic climate change. Dis. Aquat. Organ. 92, 201–207 (2010).
James, T. Y. et al. Disentangling host, pathogen, and environmental determinants of a recently emerged wildlife disease: lessons from the first 15 years of amphibian chytridiomycosis research. Ecol. Evol. 5, 4079–4097 (2015).
Toledo, L. F., Becker, C. G., Haddad, C. F. B. & Zamudio, K. R. Rarity as an indicator of endangerment in neotropical frogs. Biol. Conserv. 179, 54–62 (2014).
Becker, C. G., Fonseca, C. R., Haddad, C. F. B., Batista, R. F. & Prado, P. I. Habitat split and the global decline of amphibians. Science 318, 1775–1777 (2007).
Ribeiro, M. C., Metzger, J. P., Martensen, A. C., Ponzoni, F. J. & Hirota, M. M. The Brazilian Atlantic Forest: how much is left, and how is the remaining forest distributed? Implications for conservation. Biol. Conserv. 142, 1141–1153 (2009).
Rodriguez, D., Becker, C. G., Pupin, N. C., Haddad, C. F. B. & Zamudio, K. R. Long-term endemism of two highly divergent lineages of the amphibian-killing fungus in the Atlantic Forest of Brazil. Mol. Ecol. 23, 774–787 (2014).
Betancourt-Román, C. M. Differential virulence among panzootic, novel and hybrid lineages of Batrachochytrium dendrobatidis on a single host species, Lithobates sylvaticus. MS Thesis. (University of Michigan, 2014).
Sparrow, F. K. Aquatic Phycomycetes. at, http://biodiversitylibrary.org/page/4685126 (University of Michigan Press, 1960).
Longcore, J. E. Rhizophydium brooksianum sp. nov., a multipored chytrid from soil. Mycologia 96, 162–171 (2004).
Di Rosa, I., Simoncelli, F., Fagotti, A. & Pascolini, R. Ecology: the proximate cause of frog declines? Nature 447, E4–E5 (2007).
Hossack, B. R., Muths, E., Anderson, C. W., Kirshtein, J. D. & Corn, P. S. Distribution limits of Batrachochytrium dendrobatidis: a case study in the Rocky Mountains, USA. J. Wildl. Dis. 45, 1198–1202 (2009).
Gervasi, S., Gondhalekar, C., Olson, D. H. & Blaustein, A. R. Host identity matters in the amphibian-Batrachochytrium dendrobatidis system: fine-scale patterns of variation in responses to a multi-host pathogen. Plos One 8, e54490 (2013).
Daskin, J. H. & Alford, R. A. Context-dependent symbioses and their potential roles in wildlife diseases. Proc. R. Soc. Biol. Sci. 279, 1457–1465 (2012).
Best, A., White, A. & Boots, M. Maintenance of host variation in tolerance to pathogens and parasites. Proc. Natl. Acad. Sci. USA 105, 20786–20791 (2008).
Miller, M. R., White, A. & Boots, M. The evolution of parasites in response to tolerance in their hosts: the good, the bad, and apparent commensalism. Evolution (N. Y). 60, 945–956 (2006).
Rausher, M. D. Co-evolution and plant resistance to natural enemies. Nature 411, 857–864 (2001).
Altizer, S., Harvell, D. & Friedle, E. Rapid evolutionary dynamics and disease threats to biodiversity. Trends Ecol. Evol. 18, 589–596 (2003).
Savage, A. E., Becker, C. G. & Zamudio, K. R. Linking genetic and environmental factors in amphibian disease risk. Evol. Appl. n/a–n/a, https://doi.org/10.1111/eva.12264 (2015).
Mesquita, A. F. C. et al. Low resistance to chytridiomycosis in direct-developing amphibians. Sci. Rep. 7, 16605 (2017).
Jenkinson, T. Population structure and evolutionary ecology of Batrachochytrium dendrobatidis in the Brazilian Atlantic Forest. PhD Thesis (University of Michigan, 2017).
Voyles, J. et al. Diversity in growth patterns among strains of the lethal fungal pathogen Batrachochytrium dendrobatidis across extended thermal optima. Oecologia 184, 363–373 (2017).
Becker, C. G. et al. Variation in phenotype and virulence among enzootic and panzootic amphibian chytrid lineages. Fungal Ecol. 26, 45–50 (2017).
Becker, C. G., Rodriguez, D., Lambertini, C., Toledo, L. F. & Haddad, C. F. B. Historical dynamics of Batrachochytrium dendrobatidis in Amazonia. Ecography (Cop.). 39, 954–960 (2016).
Lips, K. R., Reeve, J. D. & Witters, L. R. Ecological traits predicting amphibian population declines in Central America. Conserv. Biol. 17, 1078–1088 (2003).
Bustamante, M. R., Ron, S. R. & Coloma, L. A. Cambios en la diversidad en siete comunidades de anuros en los Andes de Ecuador. Biotropica 37, 180–189 (2005).
Olson, D. H. et al. Mapping the global emergence of Batrachochytrium dendrobatidis, the amphibian chytrid fungus. Plos One 8, e56802 (2013).
Gründler, M. C. et al. Interaction between breeding habitat and elevation affects prevalence but not infection intensity of Batrachochytrium dendrobatidis in Brazilian anuran assemblages. Dis. Aquat. Organ. 97, 173–184 (2012).
Ruggeri, J., Carvalho-e-silva, S. P., De, James, T. Y. & Toledo, L. F. Amphibian chytrid infection is influenced by rainfall seasonality and water availability. Dis. Aquat. Organ. 127, 107–115 (2018).
Longo, A. V., Burrowes, P. A. & Joglar, R. L. Seasonality of Batrachochytrium dendrobatidis infection in direct-developing frogs suggests a mechanism for persistence. Dis. Aquat. Organ. 92, 253–260 (2010).
Burrowes, P. A., Joglar, R. L. & Green, D. E. Potential causes for amphibian declines in Puerto Rico. Herpetologica 60, 141–154 (2004).
Longo, A. V. & Burrowes, P. A. Persistence with chytridiomycosis does not assure survival of direct-developing frogs. Ecohealth 7, 185–195 (2010).
Langhammer, P. F., Burrowes, P. A., Lips, K. R., Bryant, A. B. & Collins, J. P. Susceptibility to the amphibian chytrid fungus varies with ontogeny in the direct-developing frog. Eleutherodactylus coqui. J. Wildl. Dis. 50, 438–446 (2014).
Phillott, A. D. et al. Chytridiomycosis and seasonal mortality of tropical stream-associated frogs 15 years after introduction of Batrachochytrium dendrobatidis. Conserv. Biol. 27, 1058–1068 (2013).
Pilliod, D. et al. Effects of amphibian chytrid fungus on individual survival probability in wild boreal toads. Conserv. Biol. 24, 1259–1268 (2010).
Valenzuela-Sánchez, A. et al. Cryptic disease-induced mortality may cause host extinction in an apparently stable host–parasite system. Proc. R. Soc. B Biol. Sci. 284, 20171176 (2017).
Becker, C. G. et al. Partitioning the net effect of host diversity on an emerging amphibian pathogen. Proc. R. Soc. B Biol. Sci. 281, 20141796 (2014).
Becker, C. G., Longo, A. V., Haddad, C. F. B. & Zamudio, K. R. Land cover and forest connectivity alter the interactions among host, pathogen and skin microbiome. Proc. R. Soc. B Biol. Sci. 284, 20170582 (2017).
Byrne, A. Q. et al. Unlocking the story in the swab: a new genotyping assay for the amphibian chytrid fungus Batrachochytrium dendrobatidis. Mol. Ecol. Resour. 17, 1283–1292 (2017).
Knapp, R. A. et al. Large-scale recovery of an endangered amphibian despite ongoing exposure to multiple stressors. Proc. Natl. Acad. Sci. USA 113, 11889–11894 (2016).
Scheele, B. C. et al. After the epidemic: Ongoing declines, stabilizations and recoveries in amphibians afflicted by chytridiomycosis. Biol. Conserv. 206, 37–46 (2017).
Haddad, C. et al. Guia dos anfíbios da Mata Atlântica: diversidade e biologia. (Anolis Books, 2013).
Frost, D. R. Amphibian species of the world: an online reference. Version 6.0. American Museum of Natural History, New York, USA. at, http://research.amnh.org/herpetology/amphibia/index.html (2018).
IUCN. IUCN Red List of Threatened Species. Version 2017. 3. at, www.iucnredlist.org (2018).
Pessier, A. P. & Mendelson, J. R. A manual for control of infectious diseases in amphibian survival assurance colonies and reintroduction programs. IUCN/SSC Conservation Breeding Specialist Group, Apple Valley, MN (2010).
Knapp, R. A. & Morgan, J. A. T. Tadpole mouthpart depigmentation as an accurate indicator of chytridiomycosis, an emerging disease of amphibians. Copeia 2006, 188–197 (2006).
Voyles, J. et al. Pathogenesis of chytridiomycosis, a cause of catastrophic amphibian declines. Science 326, 582–585 (2009).
Hyatt, A. D. et al. Diagnostic assays and sampling protocols for the detection of Batrachochytrium dendrobatidis. Dis. Aquat. Organ. 73, 175–192 (2007).
Boyle, D. G., Boyle, D. B., Olsen, V., Morgan, J. & Hyatt, A. Rapid quantitative detection of chytridiomycosis (Batrachochytrium dendrobatidis) in amphibian samples using real-time Taqman PCR assay. Dis. Aquat. Organ. 60, 141–148 (2004).
Cox, D. R. Regression models and life-tables. J. R. Stat. Soc. Ser. B 34, 187–220 (1972).
Acknowledgements
Our collaborative work was funded by grants and fellowships from the São Paulo Research Foundation (FAPESP #2011/51694-7; #2013/50741-7; #2016/25358-3) and National Council for Scientific and Technological Development (CNPq #405285/2013-2; #302518/2013-4; #300980/2014-0; #312895/2014-3; #300896/2016-6). Thanks to Lucas Michelotti and Rebecca Clemons for technical assistance.
Author information
Authors and Affiliations
Contributions
C.G.B. conceived the idea and designed the study. C.G.B., C.L., T.C., L.F.T. and C.F.B.H. conducted field work. C.G.B., C.L., T.C. and T.Y.J. conducted laboratory work. C.G.B. and S.E.G. analyzed the data. S.E.G. wrote the manuscript. All authors critically revised the manuscript and gave final approval for publication.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Greenspan, S.E., Lambertini, C., Carvalho, T. et al. Hybrids of amphibian chytrid show high virulence in native hosts. Sci Rep 8, 9600 (2018). https://doi.org/10.1038/s41598-018-27828-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-27828-w
This article is cited by
-
Body size mediates latitudinal population differences in the response to chytrid fungus infection in two amphibians
Oecologia (2024)
-
Coinfection with chytrid genotypes drives divergent infection dynamics reflecting regional distribution patterns
Communications Biology (2023)
-
Chytrid in the clouds: an alternative passive transport of a lethal pathogen for amphibians
Hydrobiologia (2023)
-
Host–multiparasite interactions in amphibians: a review
Parasites & Vectors (2021)
-
Interspecific hybridization as a driver of fungal evolution and adaptation
Nature Reviews Microbiology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.