Abstract
Modifications to the upper vocal tract involving hyper-elongated tracheae have evolved many times within crown birds, and their evolution has been linked to a ‘size exaggeration’ hypothesis in acoustic signaling and communication, whereby smaller-sized birds can produce louder sounds. A fossil skeleton of a new extinct species of wildfowl (Galliformes: Phasianidae) from the late Miocene of China, preserves an elongated, coiled trachea that represents the oldest fossil record of this vocal modification in birds and the first documentation of its evolution within pheasants. The phylogenetic position of this species within Phasianidae has not been fully resolved, but appears to document a separate independent origination of this vocal modification within Galliformes. The fossil preserves a coiled section of the trachea and other remains supporting a tracheal length longer than the bird’s body. This extinct species likely produced vocalizations with a lower fundamental frequency and reduced harmonics compared to similarly-sized pheasants. The independent evolution of this vocal feature in galliforms living in both open and closed habitats does not appear to be correlated with other factors of biology or its open savanna-like habitat. Features present in the fossil that are typically associated with sexual dimorphism suggest that sexual selection may have resulted in the evolution of both the morphology and vocalization mechanism in this extinct species.
Similar content being viewed by others
Introduction
A key characteristic of nearly all living birds is their ability to vocalize and produce a variety of sounds for acoustic signaling and communication. The majority of these sounds are produced by the avian vocal organ, or syrinx, located at the tracheobronchial junction. The syrinx likely was present in the ancestral crown group bird and evolved by the end of the Cretaceous1. However, fossil syrinxes are rare1,2 compared to more commonly preserved cartilaginous and ossified rings of the trachea in crown birds1,3. This near absence of fossil evidence has made the reconstruction of the evolution of vocal behavior in extinct birds difficult, although phylogenetic and molecular evidence has offered clues4,5,6.
Sounds produced by the syrinx may be modified by the upper vocal tract, including the shape of the oropharyngeal cavity affected by the mouth and tongue6,7, tracheal bullae8,9 and modifications to the diameter of the trachea, expansion of the esophagus while vocalizing5,7,10, and significant tracheal elongation (TE) where the trachea forms permanent coils or loops11,12. Significant elongation of the trachea has likely evolved many times in birds5 and is known to occur in at least 60 species, including species of the extinct giant moas12, Galliformes (Cracidae, Numinidae, and Tetraoninae), Gruiformes (Gruidae), Ciconiiformes (Threskiornithidae), Charadriiformes (Rostratulidae), and songbirds11. There are a number of different ways that TE occurs in birds including the trachea extending into a single loop or multiple loops subcutaneously over the pectoral muscles caudally (even to the caudal region of the body), the lengthened trachea overlaying only the lateral surface of the pectoral muscles, extension of the trachea into the body of the sternum or furcula itself, or looping of the trachea intrathoracically13,14,15.
Many hypotheses have been proposed for the evolution and function of TE in birds including production of lower fundamental frequencies or increased amplitudes (for increased signal propagation in particular habitats), and size exaggeration (i.e., sounding like a larger bodied bird), either to attract a potential mate or startle a predator, and even temperature or CO2 regulation, water retention, and heat conservation11. Among these hypotheses, thermoregulation and vocalization have been thought to be the most likely factors that result in tracheal elongation16. Positive selection for a longer trachea may occur in birds to compensate for limited nasal turbinate sizes, aiding in the reduction of evaporative heat and water loss16. However, this hypothesis does not appear to explain its absence in birds of similar ecologies to those exhibiting TE or dimorphism in the trait within a single species. Size exaggeration is another hypothesis for the evolution of TE in many clades of birds that can explain cases of sexual dimorphism in TE11,17. An elongated trachea allows birds to produce a vocalization similar to that of a larger-bodied bird by manipulating the acoustic output11. Size selection sometimes acts mainly on male individuals and female preferences for low-frequency sounds might explain why males tend to have longer tracheas than females in some clades. The role of TE in vocal tract filtering remains unclear10,11,18, but the filtering is known to occur frequently and be more important than previously thought in bird vocalziation19.
Here, we describe a new late Miocene (~7–11 Ma) fossil of a pheasant from Gansu Province, northwest China that preserves the oldest known instance of TE in a bird, as part of a rare nearly complete skeleton. The new species is placed phylogenetically within a polytomy including Perdix, a few members of Phasianinae, Tetraoninae, and Meleagridinae (Galliformes: Phasianidae). TE is present in many extant galliform birds, including most male cracids (e.g., Ortalis veula), male Tetrao urogallus (Tetraoninae), and both sexes of Guttera11,13 (Numididae). In addition to Tetraoninae, this taxon represents the third record of elongation of the trachea in Phasianidae (besides Tetrao urogallus and Lagopus mutus) and the first within Phasianinae, a diverse group including over 150 extant species of pheasants, peafowl, francolins, and others. In addition, this new species from the Miocene deposits of Gansu Province adds new data to the ecological diversity of this extinct steppe-dwelling avifauna at the edge of the Tibetan Plateau that also includes an extinct long-legged kestrel, ostrich, and Old World vultures20,21,22,23.
Results
Systematic paleontology
AVES Linnaeus, 1758
GALLIFORMES Linnaeus, 1758
PHASIANIDAE Vigors, 1825
Panraogallus hezhengensis gen et sp. nov.
Etymology
The genus name is the pinyin of the Chinese characters meaning ‘coiling’ and the Latin for ‘chicken,’ referring to the preserved elongate trachea in this species. The specific epithet, ‘hezhengensis’ refers to Hezheng area in the Linxia Basin of Gansu Province where abundant fossils, including the holotype, have been collected.
Specimen number
HMV 1876 (HMV, Hezheng Paleozoological Museum, Vertebrate Collection, Gansu Province, China).
Locality and age
Baihua area near Zhuangkeji Township, Guanghe County, Gansu Province, China exposes the Liushu Formation that is late Miocene (7.25–11.1 Ma)24.
Holotype
The holotype specimen of Panraogallus hezhengensis is a nearly complete skeleton (Figs 1–6). It is from a young adult individual as demonstrated by the fusion of all major skeletal elements, a smooth periosteal surface on the bones, and formation of a tarsometatarsal process for spur attachment. Part of the skull, including the premaxillae, maxillae, nasofrontal region, and cranial cervical vertebrae were conspicuously reconstructed in plaster and other materials (grey regions, Fig. 1) prior to acquisition by the HMV; these parts are ignored in the description of the new taxon. The fossil preserves the jaw and caudal portion of the skull. The rest of the postcranial skeleton caudal to the penultimate caudal cervical vertebrae is unmodified original fossil material. The fossil is embedded in reddish clay, identical to that of other avian fossil specimens collected in the Linxia Basin in units attributed to the Liushu Formation23. The occurrence of TE and a bony blunt process for the attachment of a spur on the tarsometatarsus indicate that this individual may have been male (see below for more discussion).
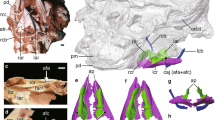
Photo and line drawing of the holotype specimen (HMV 1876). Anatomical abbreviations: co–coracoid, cmc–carpometacarpus, cv–cervical vertebrate, ad-1, first phalanx of alular digit; md-1–first phalanx of major digit, fe–femur, fu–furcula, hh–humeral head, hu–humerus, il–ilium, ped–pedal digits, pr–projection for the spur attachment, py–pygostyle, ra–radius, ri–rib, sca–scapula, sk–skull, st–sternum, ti–tibiotarsus, tmt–tarsometatarsus, tpi–terminal process of the ischium, tr–tracheal rings, tra–trabecula, and ul–ulna.
Photo and line drawing of the thoracic region of the holotype specimen of Panraogallus hezhengensis (HMV 1876). The tracheal loop is indicated by the red line and compared with that in the Helmeted curassow (Pauxi pauxi) (c), Black curassow (Crax alector) (d), Crested guineafowl (Guttera pucherani) (e), and Wood grouse (Tetrao urogallus) (f), which are adapted from ref.48. Anatomical abbreviations: cav–free caudal vertebrae, cc–cnemial crest, cmc–carpometacarpus, dc–deltopectoral crest, fi–fibula, ft–femur trochanteric crest, fu–furcula, hh–humeral head, if–ilioischiadicum foramen, ml?–muscular line, pat–patches, pd–pedal digits, pp–preacetabular process, py–pygostyle, pr–projection for the spur attachment on the tarsometatarsus, ses–sesamoid bone, st–sternum, tr–tracheal ring, tra–trabecula, and trc–tracheal coiling/loop.
Photo and line drawing of the forearm and hind limb of the holotype specimen of Panraogallus hezhengensis (HMV 1876). Anatomical abbreviations: ad-1–first phalanx of the alular digit, co–coracoid, cmc–carpometacarpus, cv–cervical vertebrate, dc–deltopectoral crest, fe–femur, hu–humerus, il–ilium, mc iii–minor metacarpal, ot–ossified tendon, ph–phalanx, py–pygostyle, ra–radius, ri–rib, ti–tibiotarsus, tmt–tarsometatarsus, and ul–ulna.
Photos of the left proximal femur in Panraogallus hezhengensis holotype (HMV 1876): (a) compared to that of Tympanuchus phasianellus, National Museum of Natural History, Smithsonian Institution, USNM 559940 (b), and Lophophorus impejanus USNM 347359 (c) in lateral view. Identification of hind limb muscular scars adopted from ref.63. Anatomical abbreviations: ISF/ IFE (m. iliofemoralis externus, m. ischiofemoralis), ITC (m. iliotrochantericus caudalis), ITCR (m. iliotrochantericus cranialis), and ITM (m. iliotrochantericus medius), OM or PIFE (obturatorius muscle or m. puboischiofemoralis). The white straight arrow (Ant) indicates the cranial direction.
Left hind limb of the holotype of Panraogallus hezhengensis (HMV 1876). Anatomical abbreviations: hpc–hypotarsal crest, is–ischium, pd–pedal digits, pr–projection for the spur attachment on the tarsometatarsus, py–pygostyle, ses–sesamoid bone, st–sternum, ti–tibiotarsus, tmt–tarsometatarsus, and tr–tracheal rings.
Photos of the proximal view of the left tibiotarsus in Panraogallus hezhengensis (HMV 1876) (a); with comparisons to various phasianids, Tragopan temminckii USNM 621027 (b), Lophophorus impejanus USNM 347359 (c), Bonasa bonasia USNM 586135 (d), Dendragapus obscurus USNM 502269 (e), and Tympanuchus phasianellus USNM 559940 (f). Anatomical abbreviations: cra. cne.–cranial cnemial crest, fib–fibula, interart.–interarticular area, med. art. fac.–medial articular facies, pat.cr.–patellar crest, retrop. fos.–retropatellar fossa, and tib. inc–tibia incisure.
Diagnosis
The new specimen differs from Cracidae and Megapodiidae in having a shorter and more robust femur25,26 and lacking an enlarged laterally-directed ridge on the lateral crest of the hypotarsus (for the passage of the m. fibularis longus tendon). Panraogallus hezhengensis also differs from Cracidae in having a strongly proximally-projected cranial cnemial crest27,28. In comparison to Megapodiidae, the Chinese specimen lacks a hooked acromion on the scapula and widely-splayed trochleae on the distal tarsometatarsi25,29. The presence of an enlarged intermetacarpal process is a derived feature present in both new world quail (Odontophoridae) and pheasants (Phasianidae)30. The intermetacarpal process of Panraogallus is similar to Tragopan and other core phasianids, but much larger than that of Odontophoridae25. The absence of a foramen on the caudal mandible additionally distinguishes the new specimen from other phasianids (e.g., turkeys [Meleagris], and grouse [Tetraoninae])25,31.
The new species is the third phasianid in addition to the Western Capercaillie (Tetrao urogallus) and Rock Ptarmigan (Lagopus mutus) that exhibits tracheal elongation: TE is a proposed autapomorphy of each species32. No extant species of Phasianinae has an elongate trachea, diagnosing the fossil as distinct from any living member of that clade. Panraogallus differs significantly from other phasianines (i.e. partridges, francolins, and Old World quails) in its sternal shape (with very deep caudal incisures), and in its significantly larger body size (estimated 2.5 kg). The size of the new specimen differentiates it from other Asian Neogene taxa. It is about twice the size of Shandongornis, which also differs in the absence of spur attachment on the tarsometatarsus, and is much larger than most other known extinct Asian Neogene galliform taxa33,34,35,36. Furthermore, this new species is smaller than extant species of pavonine pheasants (e.g., Pavo and Argusianus), and other large-bodied phasianines. The species has a femur longer than the humerus in contrast to the state present in Cracidae, Numididae, Odontophoridae, and Tetraoninae25. In comparison with extinct members of Phasianidae, Palaeocryptonyx exhibits a second (dorsal) pneumotricipital fossa on the proximal humerus that is absent in the new species37; Palaeortyx is much smaller in size and has relatively shorter tarsometatarsus than Panraogallus38. The new species also differs from Miogallus in the morphology of the acrocoracoid process of the coracoid, which is more medially recurved in the new species34,35,36. Compared to Tologuica, the new species lacks a large tubercle on the dorsal surface of the acrocoracoid process. The femoral condyles appear to be stouter in the new species than the condition in Lophogallus34,35,36. The width of the third metatarsal trochlea in the new species is relatively stouter and shorter than that of Diangallus33. The new specimen has a third pedal digit relatively longer as compared to the tarsometatarsus, in contrast to the shorter condition in Linquornis31. It also differs from extinct species of Syrmaticus in the morphology of the minor metacarpal34,35,36, and from Plioperdix in having a larger intermetacarpal process34,35,36.
Description and comparison
The new specimen represents a medium-sized species of phasianin similar in size to eared pheasants (Crossoptilon), with longer and robust hind limbs, but with relatively shorter forelimbs (see Table 1 for measurements). The specimen is exposed primarily in left lateral view (Fig. 1). The skull is poorly preserved and the premaxillae, nasals, maxillae, and bones of the frontal-orbital region are partially reconstructed with plaster (Fig. 1, highlighted in grey). The basicranium and jaw are original bony material. The elongated postorbital process fuses with the zygomatic process distally, a feature shared with other phasianids27. No foramen is present on the lateral surface of the caudal mandible, as opposed to a large foramen present on the mandible in guans (Penelope), turkeys (Meleagris), and grouse (Tetraoninae)38.
The cervical vertebrae are incompletely preserved, and the cranial part of the series is reconstructed with glue. Two caudal cervical vertebrae are preserved in situ and in articulation (Fig. 1). A lamina acrocostalis38 is present in these vertebrae. Neither a notarium, nor the morphology of the thoracic vertebrae are visible. Three articulated caudal vertebrae display long transverse processes and high spinal processes. The pygostyle is triangular in outline in lateral view, and its caudal tip extends only slightly dorsally, differing from that in turkeys (Meleagris) and guineafowl (Guttera), in which the distal pygostyle strongly projects dorsally. A weak ridge is discernible on the ventral side of the pygostyle in the new specimen (Fig. 2).
Several sternal and thoracic ribs are preserved, overlain by the left hind limb (Fig. 2). The thoracic ribs are mediolaterally thin but craniocaudally wide, with fused elongate and wide uncinate processes. The furcula is partially exposed and the rami appear to meet at the apophysis to form a ‘V-shape’ as in other phasianids, but this region is partially obscured by matrix. The omal ends of the furcula are expanded (Fig. 2). The omal end of the left coracoid is partially exposed in ventrolateral view. The acrocoracoid process is hooked medially, with a shallow impression marked the cranial surface (Fig. 2). The proximal right scapula is only partially visible in medial view, and the scapular acromion extends cranially. The coracoid tubercle is slightly convex.
The cranial margin of the sternum is slightly concave in outline, but less concave than the curvature in most phasianids (e.g., Francolinus). The carinal apex of the sternum is positioned caudally relative to the external sternal rostral spine as in crown galliforms38, and the apex also is caudal to the cranial edge of the sternal incisures as in Lophophorus, Bonasa, and species with an enlarged crop38. The sternal body is similar to turkey and grouse in the dorsoventral width of the keel, which is greater than most other phasianids. Two caudal incisures (the left and right incisura lateralis39) are visible (Fig. 2). The cranial extension of the lateral incisure is quite deep compared to that of Crax or Acryllium, and similar to other phasianids. The width of the lateral trabecular arm of the new specimen is similar to other pheasants, but narrower than that in cracids and megapodes. The medial trabecula is longer and narrower than the lateral, with a shallow groove on the external surface. The craniolateral process of the sternum is not preserved. The mediolateral thickness of the keel varies along its ventral edge (Fig. 2) and is thickest in its cranial portion. The coiled trachea abuts the cranial margin of the sternal keel (Fig. 2). There is no evidence of a crop being preserved in this region of the specimen.
The left humerus is visible in cranial view, and the right one only has part of the humeral head exposed (Fig. 2). The deltopectoral crest deflects dorsally, and a tubercle is present on the cranial surface of the crest (Fig. 3). The second (dorsal) pneumatic fossa (pneumotricipital fossa) on the proximal humerus appears to be shallow or absent. The dorsal condyle of the humerus is rounded and appears to be longer than the ventral condyle. The ulna is robust and is slightly shorter than the length of the humerus. Only the dorsal ramus of the ulnare is exposed, and a concave facet for articulation with the dorsal condyle of the ulna is visible. The left carpometacarpus is preserved in dorsal view. The intermetacarpal process is present with a triangular shape and does not fuse to the minor metacarpal (Fig. 3). However, it slightly overlaps the edge of this metacarpal. By contrast, the intermetacarpal process is only weakly developed in Odontophoridae25. The intermetacarpal process of Panraogallus is shorter than that in Tetraoninae and similar to Tragopan and other phasianids. The extensor sulcus is visible on the distal half of the major metacarpal (Fig. 3). The minor metacarpal is bowed caudally, less than that of Cracidae25, with its apex distal to the intermetacarpal process. The minor metacarpal bears a concave cranial surface with a flange protruded caudally (Fig. 3). The supratrochlear surface is decorated with tiny pits and bulges, unlike the larger tubercles present in Lagopus. As in many extant members of Phasianidae (e.g., Alectoris), a long and slim ossified tendon is present adjacent to the major metacarpal. The alular digit is displaced adjacent to the carpometacarpus (Fig. 3, ad-1). The extensor process of the carpometacarpus is stout (Fig. 3).
Pelvis and hindlimb
The dorsoventral height of the ischium is proportionally similar to that of other phasianids, but much higher than that of Tetraoninae (e.g., Dendragapus, Lagopus, and Tetrao). The fenestra between the ischium and pubis is a narrow slit with the pubis closely aligned with the ventral edge of the ischium (Figs. 2 and 3). The terminal process of the ischium is missing and appears to have been pointed and straight based on the impression of the missing bone preserved in the matrix. A notch is present in the distal end of the ischium in grouse (Tetraoninae)40, but does not appear to have been present in the new specimen. The dorsolateral crests of the ilia are projected slightly. The antitrochanter rises into a small tubercle, but it is largely obscured (Fig. 2). The preacetabular tubercle (or pectineal process) appears to be small (Fig. 2) and shorter than that seen in Phasianus25.
The femur of the new specimen is slightly curved. The short and stout appearance of the femur is quite distinct from the much more slender and elongate femur of Cracidae and Megapodiidae. A femur longer than the humerus is in contrast to the state present in Cracidae, Numididae, Odontophoridae, and Tetraoninae25. The projection of the trochanteric crest of the left femur in the fossil is greater than that of Megapodiidae, but less than in Cracidae27. The four scars on the proximolateral surface of the femoral shaft are mostly similar to other members of Phasianinae (e.g., Lophophorus) (Fig. 4). In lateral view, the central scar is round and associated with the attachment of puboischiofemoralis muscle (PIFE in Fig. 4). A longer groove is cranially positioned and deeply marked, and it is associated with the attachment of the iliotrochantericus caudalis muscle (Fig. 4: ITC). A few smaller trough-like ridges associated with the ischiofemoralis and iliotrochantericus cranialis muscles (Fig. 4: ISF and ITCR) are positioned distal to the ITC (Fig. 4). The medial and lateral condyles are well projected as seen in the right femur, with the medial one wider than the lateral one; a deep groove (intercondylar incisure39) is present in between the condyles.
The right tibiotarsus is visible in cranial and medial views (Fig. 5). The femoral articular facet of the tibiotarsus is visible (Fig. 6), and the facet is slightly narrower mediolaterally than its craniocaudal length. A ball-shaped interarticular area bulges in the center of the facet39. Cranial to this bulge, the retropatellaris fossa is developed. Medial to the bulge, the medial articular facet is convex. The tibial incisure is significantly concave, and the patellar crest protrudes distinctly (Fig. 5). The cnemial crests do not extend far proximally relative to the proximal end of the tibiotarsus. The fibular crest projects laterally with a long intermuscular ridge extending distally. Distally, the medial and lateral condyles of the tibiotarsus are subcircular in outline. The ossified superatendinal bridge is present but broken. A narrow extensor sulcus is present on the distal end of the tibiotarsus. The distal medial condyle projects cranial to the lateral condyle. A crescent-shaped sesamoid is preserved positioned against the distal ends of the left and right tibiotarsi (Fig. 5).
The tarsometatarsi are long and are similar to the length of the femur (Fig. 5; see Table 1 for measurements). They are relatively stouter than those of the long-legged Megapodiidae and Numididae. Comparatively, the tarsometatarsus in the fossil is longer than that of most other phasianids (e.g., Tetraoninae). The left tarsometatarsus is visible in plantolateral view and the right, in medial view (Figs. 2 and 5). The plantar opening of the distal vascular foramen is rounded. The hypotarsal crests are short overall, but the medial hypotarsal crest extends distal to the lateral crest. A blunt tarsometatarsal callosity that potentially associated with further attachment of the spur is preserved on the left tarsometatarsus just distal to the proximodistal midpoint of the bone. A spur is present in males of several galliform taxa including Meleagridinae and Phasianinae, and it is absent in Odontophoridae, Numididae, and Tetraoninae41. The lateral cotyle of the tarsometatarsus is slightly deeper than the medial one. The medial parahypotarsal fossa is deep (Fig. 5).
The metatarsal trochleae are not widely splayed (Fig. 2). Metatarsal trochlea II is plantarly deflected (relative to metatarsal trochlea III), and extends approximately as far distally as metatarsal trochlea IV. The trochlea of metatarsal II bears a plantarly projected wing (Fig. 5). Metatarsal trochlea III is almost symmetric in distal view. Metatarsal trochlea IV bears rounded lateral depression (fovea. lig. collat.39). The hallux is elevated proximally on the tarsometatarsal shaft, near the distal end of the medial plantar ridge along the tarsometatarsus shaft. The phalanges are well preserved with digit III being the longest, and despite its length, that digit is slightly shorter than the length of tarsometatarsus (Fig. 5). Generally, the pedal (non-ungual) phalanges decrease in length distally, and the ungual phalanges are shorter than the penultimate phalanx in length. The pedal ungual phalanges are somewhat curved with a lateral sulcus (Figs. 2 and 5). Phalangeal proportions indicate that this bird likely was terrestrial in its habits42.
Approximately 40 ossified tracheal rings are preserved around the pectoral region of the skeleton with some additional isolated rings in the abdominal region of the body. The exterior surface of the tracheal rings appears tuberculated in subparallel patches (Fig. 2, pat). The color of these mineralized tracheal rings is reddish-grey and is distinguished from the white color of other skeletal features (Fig. 2). This difference would be consistent with the comparatively weakly ossified or mineralized cartilage of tracheal rings in extant species and subsequent diagenetic changes. There does not appear to be a large variation in tracheal diameter over the length of preserved trachea, or any isolated rings (n = 12, mean = 6.0 mm, Stdv = 0.9). Given the missing tracheal rings that would have been anatomically closer to the head or within the body cavity (adjacent to the lungs), it is not clear if the trachea had a greater diameter in the elongated region, as in some extant species. If the more cranial portion of the trachea is present in the fossil, it likely would be located along the right side of the cervical vertebrae as in most crown birds and all known galliforms, when in life position43,44.
Cranial to the sternal margin and keel, approximately 25 tracheal rings are aligned to form coiled shapes. We believe that this coiling of the trachea is not an artifact of preservation because the uncoiled length of the trachea as preserved (minimum of ~36.2 cm) would extend beyond the position of the head, and clearly points to the presence of a hyper-elongated trachea. The particular coiling pattern preserved in the fossil (Fig. 2) is most similar to that seen in grouse11. The proximodistal width of individual tracheal rings ranges between 3–4 mm, significantly larger than that of most other Galliformes, but rather close to some large-sized Anseriformes with tracheal elongation, such as the ring diameters of Anseranas semipalmata ranged from 5–7 mm (American Museum of Natural History 1772, 1873, and 2771).
The coiled section of the trachea appears to form at least two complete loops with approximately half of another loop preserved. The great diameter of the individual tracheal rings in the coiled, elongated portion of the trachea is consistent with the increased diameter and greater tracheal ring width present in living birds with elongate tracheas12,13. The more caudal section of the trachea preserved on the sternal keel and the isolated tracheal rings in the abdominal region also display similar wide tracheal rings and a diameter consistent with the coiled section cranial to the sternal keel, rather than the smaller and incomplete tracheal rings that likely would be present adjacent to the lungs in extant species13,43. In addition, it would also be extremely difficult to have broken the trachea close the lungs and have it moved outside the thoracic cavity, then laterally and cranially to position them on the sternum (without disturbing other parts of the body/skeleton). Thus, the sternal keel section of the trachea could be a caudally displaced section that broke away from the main coiled part. However, that section might also be a representative of an elongate subcutaneous coil along the side of the body, and its position and orientation (in line with the long axis of the body) are consistent with that conclusion (Fig. 2). The additional isolated rings in the abdominal region suggest that such a loop could have extended further caudally as in some cracids and birds of paradise, where the trachea reaches the abdominal and rear areas of the body13,14. If that caudal section of trachea derived from a long subcutaneous loop, it would indicate that the trachea was at least ~13 cm longer than our conservative estimate based on the coiled region (maximum length over 49 cm). For comparison, the length of the trachea in chicken ranges from 15.5 to 18 cm45. It appears that the trachea was at a minimum longer than the length of the bird’s body (36 cm vs. 30 cm, an estimate of length from head to tail). Lastly, the coiling of the trachea and its close application to the cranial edge of the sternal keel (and cranial to the furcula) suggests that the crop and interclavicular air sacs would have been displaced or restricted in some way.
Discussion
The phylogenetic analysis (see methods) resulted in 24 most parsimonious trees (with a length of 11792 steps) in PAUP, and the strict consensus of those trees places the new species within a polytomy comprised of species of Phasianinae, Tetraoninae, and Meleagridinae (node ‘a’ in Fig. 7). Other major clades of crown Galliformes (Megapodiidae, Cracidae, Numididae, Tetraoninae, and Odontophoridae) were recovered as monophyletic, and their relationships largely are consistent with results of prior analyses using either morphological or molecular datasets, or both28,46. Within Phasianidae, the clades Tetraoninae, Pavoninae, Coturnicinae, and Gallininae are all well supported (see bootstrap support in Fig. 7).
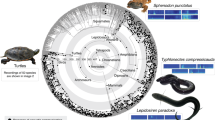
Strict consensus of the most parsimonious trees showing the phasianin affinity of Panraogallus hezhengensis. Major clades of galliforms are colored. Tracheal elongation is labeled on terminal branches with dots, and its distribution suggests multiple origins within Galloanserae (closed squares indicate that both sexes have the trait, and open squares designate that only males have the trait).
The new specimen is referred to crown Galliformes by the following characters: two caudal incisures present on the sternum27,28; cranial apex of the sternal keel shifted caudally; the rami of the furcula narrow; and a tarsometatarsus with two hypotarsal crests, between which a long groove is present27,28. The specimen is supported as part of Phasianidae by the presence of following synapomorphies: a relatively wide intermetacarpal space; metatarsal trochleae II and IV closely spaced and subequal in distal extent; hallux proximally situated on the tarsometatarsus; and four distinct muscular scars are present on the lateral surface of the proximal femur26. This specimen displays a tarsometatarsal process for spur attachment that is only known to be present in Numididae, Phasianinae, and Meleagridinae30. The new specimen can additionally be differentiated from Tetraoninae by the absence of a dorsoventrally narrow ischium40 and from Odontophoridae by the arrangement of proximolateral muscular scars on the femur26. Both of these taxa also lack the tarsometatarsal spur37. Finally, the length of the humerus is less than that of the femur (Figs 2 and 3; see Table 1), a synapomorphy of crown Phasianinae38,47. The holotype specimen of Panraogallus is distinguished from other galliform species not only by discrete characters, but also by size and limb proportions.
Mineralized or ossified cartilaginous tracheal rings are preserved and reveal a highly elongated trachea that is rarely present in phasianids. This trait has evolved independently at least four other times among galliform species, including in grouse (e.g., Tetrao urogallus and Lagopus mutus), cracids (e.g., Ortalis vetula), and guineafowl (e.g., Guttera plumifera)11,32. The reconstructed coiling pattern of Panraogallus is more similar to Tetrao urogallus in forming a ‘zigzag’ shape, rather than to other simpler looped coiling patterns present in cracids and guineafowl32,48 (Fig. 2). The tracheal coiling preserved here represents the earliest fossil evidence of this acoustically important trait. The point-like projections on the exterior surface of the tracheal rings have not been described in extant species (and may be an autapomorphy of this extinct species). These pointed structures in the fossil trachea might serve to increase the attachment between cartilaginous rings and the surrounding epithelium. The close contact between cartilage and surrounding tissues provides better heat exchange function than a simple, smooth surface.
The independent evolution of tracheal modification in pheasants is consistent with the broader convergent pattern among galliforms, suggesting possible similar underlying factors in selection. We further examined five traits in comparative analyses (using ‘R,’ see Electronic supplementary material), including TE, esophageal inflation, mating system, and habitat openness. However, our results demonstrate that none of those characteristics exhibit any significant correlation to one another on the most parsimonious trees in our analyses (Methods and Supplementary Figs 1 and 2). Therefore, the evolution of TE in galliforms does not appear to have been directly influenced by those factors. The body mass of the new species is estimated to be approximately 2.5 kg based on regression of femoral length49. That mass estimate is much greater than the average mass of galliform species without a TE (~0.83 kg) and similar to the mean value of galliform taxa with a TE (~2.26 kg11). Previous work did not treat body mass and TE in a comparative framework11. Here we show that evolutionary origins of TE are linked to increases in body size (see Electronic supplementary material). More anatomical work may identify further cases of TE in birds, but our results are robust to additional gains of TE (Supplementary Fig. 3).
Panraogallus likely produced a lower frequency vocalization and/or louder call than its close phasianine relatives (of similar size) implied by its elongate, coiled trachea and larger body size. Such lower frequencies likely occurred later in ontogeny during what has been termed ‘voice breaking,’ and that change is not necessarily directly linked to reaching a certain size or ontogenetic stage because TE (growth) is complete before the change in vocalization pattern50. Panraogallus represents an independent evolution of an elongate trachea and the third record of such elongation in the diverse clade Phasianidae (Fig. 7). Its independent evolution in a savannah/open habitat setting may be consistent with the greater frequency bandwidth produced by some birds in open habitats relative to their body size and phylogenetic constraints17,51. The elongate trachea52 most often co-occurs with larger body sizes (Supplementary Figs 4 and 5) that are associated with lower frequencies and potentially louder calls11. Given the preference of the ‘size exaggeration’ hypothesis for the evolution of the TE in birds11, and that Panraogallus has a body mass consistent with other galliforms with elongate tracheae, it is possible (if not likely) that this extinct Chinese species produced calls of a lower fundamental frequency and possibly louder calls than its similar sized close relatives. Potential selective advantages of low frequency calls include territory defense and mate attraction52,53. Such vocalizations should have carried a long distance in the reconstructed temperate savannah habitat where Panraogallus lived in the Linxia Basin23,24. While the data are consistent with the ‘size exaggeration’ hypothesis, we cannot reject other hypotheses and functions for tracheal elongation in this extinct species such as it being a compensative mechanism for heat conservation and water retention in the late Miocene54.
The co-occurrence of a bony process for attachment of the spur on the tarsometatarsus and an elongate trachea, features frequently present in males of sexually dimorphic species of galliforms and other birds13,15,41,55,56 may indicate that the holotype specimen is of a male individual. The evolutionary gain of vocal modification (and potential vocal dimorphism) in an extinct lineage of pheasants is more consistent with the tracheal modification acting as a competitive trait (more frequently gained by males) rather than a signaling trait (e.g., plumage color) that are lost more commonly in phasianid evolution55. The presence and degree of sexual dimorphism among phasianids is evolutionarily labile41 and the discovery of Panraogallus demonstrates that the evolutionary history of pheasants is more complex than previously hypothesized.
Conclusions
The nearly complete skeleton of Panraogallus hezhengensis includes an elongate, coiled trachea that also appears to have possibly extended subcutaneously over the pectoral muscles to the posterior abdominal region. The preserved tracheal rings are wider and thicker than typical galliform rings but consistent with the morphology present in other bird species with tracheal elongation13. The unexpected preservation of the coiled elongate trachea in the new species likely indicates an acoustic adaptation independently evolved within an extinct galliform lineage, and that this type of vocal modification likely was more widespread across birds in the past. The co-occurrence of an elongate trachea and a tarsometatarsal projection for the attachment of the spur in this individual might represent sexually dimorphic features only present in males. Given the diversity of birds that have evolved TE and the absence of correlation of its evolution with many other biological and environmental factors, future work should examine the genetic and developmental modifications that lead to the formation of TE in order to determine the underlying mechanism for its production. Such data likely will shed more light on the reason(s) why this vocal modification has repeatedly evolved among crown group birds.
Methods
Phylogenetic analysis
In order to test the phylogenetic position of the holotype specimen of Panraogallus hezhengenesis, we analyzed a combined molecular and morphological character matrix28 in PAUP57 containing 120 morphological characters (with six characters ordered, 35–37, 55, 67, and 109) and 5022 BP of molecular sequences (Supplementary data). Paleognaths and anseriforms were used as the outgroups to the galliform taxa. A heuristic search was done using 3000 random taxon addition sequences and tree bisection and reconnection branch swapping. Zero length branches were collapsed. 1000 replicates of bootstrap were conducted in PAUP, and support numbers larger than 50% are reported.
Comparative analyses
We reconstructed joint ancestral states for four life history and morphological characters using squared-change parsimony since branch lengths are not in units of time. Branch lengths were transformed to length 1 using compute.brlen() in the R package ape58. The four traits used in analyses are: tracheal elongation, esophageal inflation, mating system, and habitat openness (see Supplementary Methods for details). We used a maximum likelihood approach in the corDISC() function of the R package corHMM59 because it allowed us to test for trait correlations with missing data (coded as ‘0&1’) and to incorporate polytomies. We tested for correlated evolution using a modified Pagel’s test60. Briefly, we fit two models: one in which all rates between different trait combinations were allowed to vary independently and one in which trait evolution was not affected by the state of the other traits60. We then selected the best model used AICc, an adjusted version of Akaike information criterion (AIC) suitable for small sample sizes61. We ran this analysis over all 24 parsimony trees and averaged the results.
To test for a correlation between body size (ln mass in grams) and tracheal elongation (presence or absence), we used phylogenetic logistic regression in the phylolm package62. We used the phylogeny of ref.5 and data from ref.11. This analysis indicates that tracheal elongation is significantly correlated with the evolutionary increase in body size (beta = 0.49, p = 0.04, 100 bootstrap replicates). Given the limited amount of morphological data confirming presence or absence of TE in birds11, we further assessed the influence of missing data on our body mass analyses (see Supplementary Information).
Data availability
The list of species and dataset is available in the supplemental dataset.
References
Clarke, J. A. et al. Fossil evidence of the avian vocal organ from the Mesozoic. Nature 538, 502–505 (2016).
Degrange, F. J., Tambussi, C. P., Taglioretti, M. L., Dondas, A. & Scaglia, F. A new Mesembriornithinae (Aves, Phorusrhacidae) provides new insights into the phylogeny and sensory capabilities of terror birds. J. Vert. Paleontol. 35, e912656 (2015).
Mayr, G. & Mourer-Chauviré, C. Rollers (Aves: Coraciiformes) from the middle Eocene of Messel (Germany) and the upper Eocene of the Quercy (France). J. Vert. Paleontol. 20, 533–546 (2000).
Fisher, S. E. & Scharff, C. FOXP2 as a molecular window into speech and language. Trends in Genetics 25, 166–177 (2009).
Riede, T., Eliason, C. M., Miller, E. H., Goller, F. & Clarke, J. A. Coos, booms, and hoots: The evolution of closed‐mouth vocal behavior in birds. Evolution 70, 1734–1746 (2016).
Suthers, R. A., Fitch, W. T., Fay, R. R. & Popper, A. N. Vertebrate Sound Production and Acoustic Communication Vol. 53 (ed. Suthers, R. A., Fitch, W. T., Fay, R. R. & Popper, A. N.) (Springer, Heidelberg, 2016).
Ohms, V. R., Beckers, G. J., Ten Cate, C. & Suthers, R. A. Vocal tract articulation revisited: the case of the monk parakeet. J. Exp. Biol. 215, 85–92 (2012).
Warner, R. W. The structural basis of the organ of voice in the genera Anas and Aythya (Aves). J. Zool. Lond. 164, 197–207 (1971).
Pierko, M. Structural analysis of upper respiratory tract in Anas platyrhynchos (l., 1758) and Clangula hyemalis (l., 1758). Elect. J. Pol. Agri. Univ. 13, 22 (2010).
Daley, M. & Goller, F. Tracheal length changes during zebra finch song and their possible role in upper vocal tract filtering. J Neurobiol. 59, 319–330 (2004).
Fitch, W. T. Acoustic exaggeration of size in birds via tracheal elongation: comparative and theoretical analyses. J. Zool. 248, 31–48 (1999).
Worthy, T. H. & Holdaway R. N. The Lost World of the Moa (Indiana, 2002).
Amadon, D. Variation in the trachea of the Cracidae (Galliformes) in relation to their classification. Nat. Hist. Bull. Siam. Soc. 23, 239–248 (1969).
Clench, M. H. Tracheal elongation in birds-of-paradise. Condor 80, 423–430 (1978).
Whitehead, P. J. Boofheads with deep voices: sexual dimorphism in the magpie goose Anseranas semipalmata. Wildfowl 49, 72–91 (1998).
Owerkowicz, T., Musinsky, C., Middleton, K. M. & Crompton, A. W. Respiratory turbinates and the evolution of endothermy in mammals and birds. Great transformations in vertebrate evolution.143–165 (Chicago, 2015).
Jones, M. R. & Witt, C. C. Migrate small, sound big: functional constraints on body size promote tracheal elongation in cranes. J. Evol. Biol. 27, 1256–1264 (2014).
Johnsgard, P. A. Vocalization: Cranes of the world Ch. 3 (Bloomington, 1983).
Ohms, V. R., Snelderwaard, P. C., ten Cate, C. & Beckers, G. J. L. Vocal tract articulation in Zebra Finches. PLoS ONE 5, e11923 (2010).
Hou, L., Zhou, Z., Zhang, F. & Wang, Z. A Miocene ostrich fossil from Gansu Province, northwest China. Chi. Sci. Bull. 50, 1808–1810 (2005).
Zhang, Z., Zheng, X., Zheng, G. & Hou, L. A new Old World vulture (Falconiformes: Accipitridae) from the Miocene of Gansu Province, northwest China. J. Ornith. 151, 401–408 (2010).
Li, Z., Zhou, Z., Deng, T., Li, Q. & Clarke, J. A. A falconid from the Late Miocene of northwestern China yields further evidence of transition in Late Neogene steppe communities. The Auk 131, 335–350 (2014).
Li, Z., Clarke, J. A., Zhou, Z. & Deng, T. A new Old World vulture from the late Miocene of China sheds light on Neogene shifts in the past diversity and distribution of the Gypaetinae. The Auk 133, 615–625 (2016).
Deng, T., Qiu, Z. X., Wang, B. Y., Wang, X. M. & Hou, S. K. Fossil Mammals of Asia: Neogene Biostratigraphy and Chronology. 243–273 (New York, 2013).
Holman, J. A. Osteology of gallinaceous birds. Quart. J. Flor. Acad. Sci. 27, 230–252 (1964).
Olson, S. L. Telecrex restudied: a small Eocene guineafowl. The Wilson Bulletin, 246–250 (1974).
Dyke, G. J., Gulas, B. E. & Crowe, T. M. Suprageneric relationships of galliform birds (Aves, Galliformes): a cladistic analysis of morphological characters. Zool. J. Linn. Soc. 137, 227–244 (2003).
Ksepka, D. T. Broken gears in the avian molecular clock: new phylogenetic analyses support stem galliform status for Gallinuloides wyomingensis and rallid affinities for Amitabha urbsinterdictensis. Cladistics 25, 173–197 (2009).
Mayr, G. Variations in the hypotarsus morphology of birds and their evolutionary significance. Acta Zoologica 97, 196–210 (2016a).
Mayr, G. Avian evolution: the fossil record of birds and its paleobiological significance (John Wiley & Sons, 2016b).
Yeh, H. K. Fossil birds from Linqu, Shandong. Vertebrata PalAsiatica. 18, 116–125 (1980).
Frith, C. B. Adaptive significance of tracheal elongation in manucodes (Paradisaeidae). The Condor 96, 552–555 (1994).
Hou, L. H. Upper Miocene birds from Lufeng, Yunnan. Acta Anthropol. Sin. 4, 118–126 (1985).
Zelenkov, N. V. & Kurochkin, E. N. Neogene phasianids (Aves: Phasianidae) of Central Asia: 1. Genus Tologuica gen. nov. Paleontol. J. 43, 208–215 (2009).
Zelenkov, N. V., & Kurochkin, E. N. Neogene Phasianids (Aves: Phasianidae) of Central Asia: 2. Genera Perdix, Plioperdix, and Bantamyx. Paleontol. J. 43, 318–325 (2009).
Zelenkov, N. V., & Kurochkin, E. N. Neogene phasianids (Aves: Phasianidae) of Central Asia: 3. Genera Lophogallus gen. nov. and Syrmaticus. Paleontol. J. 44, 328–336 (2010).
Pavia, M., Göhlich, U. B. & Mourer-Chauviré, C. Description of the type-series of Palaeocryptonyx donnezani Depéret, 1892 (Aves: Phasianidae) with the selection of a lectotype. Comptes. Rendus. Palevol. 11, 257–263 (2012).
Mayr, G., Poschmann, M. & Wuttke, M. A nearly complete skeleton of the fossil galliform bird Palaeortyx from the late Oligocene of Germany. Acta Ornithologica. 41, 129–135 (2006).
Baumel, J. J. & Witmer, L. M. Osteologia. Handbook of avian anatomy: Nomina Anatomica Avium. (ed. Baumel, J. J., King, A. S., Breazile, J. E., Evans, H. E. & Vanden Berge, J. C.) (Publications of the Nuttall Ornithological Club 23, 45–132, 1993).
Kuzmina, M. A. & Siegel-Causey, D. Tetraonidae and phasianidae of the USSR: Ecology and morphology (No. 598.2 K8)(ed. Kuzmina, M. A. & Siegel-Causey, D.) (1992).
Kimball, R. T., Colette, M. S. M. & Braun, E. L. A macroevolutionary perspective on multiple sexual traits in the Phasianidae (Galliformes). Inter. J. Evol. Biol. 211, 1–16 (2011).
Hopson, J. A. In New perspectives on the origin and early evolution of Birds (ed. Gauthier, J. & Gall, L. F.) 211–235 (New Haven, 2001).
Klingler, J. J. On the morphological description of tracheal and esophageal displacement and Its phylogenetic distribution in Avialae. PLoS ONE 11, e0163348 (2016).
Rajathi, S., Lucy, K. M., Maya, S. & Chungath, J. J. Anatomical studies on the trachea in Japanese quail (Coturnix coturnix japonica). J. Vet. Anim. Sci. 40, 17–19 (2009).
McLelland, J. In Sisson and Grossman’s The Anatomy of the Domestic Animals, Vol. II. (ed. Getty, R.) 2063–2095 (W. B. Saunders Co., Philadelphia, 1975).
Wang, N., Kimball, R. T., Braun, E. L., Liang, B. & Zhang, Z. Assessing Phylogenetic Relationships among Galliformes: a multigene phylogeny with expanded taxon sampling in Phasianidae. PLoS ONE 8, e64312 (2013).
Mayr, G. A new basal galliform bird from the Middle Eocene of Messel Hessen,Germany. Senckenbergiana lethaea 80, 45–57 (2000).
McLelland, J. In Form and Function in Birds, Vol. 4, 69–103 (ed. King, A. S. & McLelland, J.) (New York, 1989).
Zeffer, A., Johansson, L. C. & Marmebro, A. Functional correlation between habitat use and leg morphology in birds (Aves). Biol. J. Linn. Soc. 79, 461–484 (2003).
Klenova, A. V., Volodin, I. A. & Volodina, E. V. The vocal development of the Red-crowned Crane. Grus japonensis. Ornith. Sci. 6, 107–119 (2007).
Bertelli, S. & Tubaro, P. L. Body mass and habitat correlates of song structure in a primitive group of birds. Biol. J. Linn. Soc. 77, 423–430 (2002).
Gaunt, A. S., Gaunt, S. L., Prange, H. D. & Wasser, J. S. The effects of tracheal coiling on the vocalizations of cranes (Aves: Gruidae). J. Com. Physio. A 161, 43–58 (1987).
Searcy, W. A., & Nowicki, S. The evolution of animal communication: reliability and deception in signaling systems (Princeton University Press, 2005).
Liu, Y. S. C., Utescher, T., Zhou, Z. & Sun, B. The evolution of Miocene climates in North China: preliminary results of quantitative reconstructions from plant fossil records. Palaeogeogr. Palaeoclimatol. Palaeoecol. 304, 308–317 (2011).
Crowe, T. M. et al. Phylogenetics, and biogeography of, and character evolution in, gamebirds (Aves: Galliformes): effects of character exclusion, data partitioning and missing data. Cladistics 22, 495–532 (2006).
Davison, G. W. H. Avian spurs. J. Zool. 206, 353–366 (1985).
Swofford, D. L. PAUP*: phylogenetic analysis using parsimony (* and other methods). Sunderland, MA (2002).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinform. 20, 289–290 (2004).
Beaulieu, J. M., O’Meara, B. C. & Donoghue, M. J. Identifying hidden rate changes in the evolution of a binary morphological character: The evolution of plant habit in campanulid angiosperms. Syst. Biol. 62, 725–737 (2013).
Pagel, M. Detecting correlated evolution on phylogenies: a general method for the comparative analysis of discrete characters. Proc. R. Soc. London B 255, 37–45 (1994).
Posada, D. & Crandall, K. A. Selecting the best-fit model of nucleotide substitution. Syst. Biol. 50, 580–601 (2001).
Ho, L. S. T. & Ané, C. Intrinsic inference difficulties for trait evolution with Ornstein‐Uhlenbeck models. Methods Ecol. Evol. 5, 1133–1146 (2014).
Hutchinson, J. R. The evolution of pelvic osteology and soft tissues on the line to extant birds (Neornithes). Zool. J. Linn. Soc. 131, 123–168 (2001).
Acknowledgements
Hezheng Paleozoological Museum is graciously thanked for providing support during study of the holotype specimen. He W. and Chen S. are thanked for facilitating access to the specimen. National Museum of Natural History and American Museum of Natural History are thanked for access to extant avian skeletons. The work is supported by the Strategic Priority Research Program of the Chinese Academy of Sciences Grant No. XDA19050102 and Grant No. XDA 20070203. ZL is supported by the one hundred talents project from the Chinese Academy Sciences (KC 217113) and the National Natural Science Foundation of China (NSFC 41772013; with T.S.). Z.L. and T.S. also are funded by a grant from the Chinese Academy of Sciences (XDPB05). Funding from the Gordon and Betty Moore Foundation (Grant 4498; J.C. and Z.L.) and Craton destruction and terrestrial life evolution (NSFC 41688103) is also gratefully acknowledged. T.S. is funded further by grant NSFC 41472025.
Author information
Authors and Affiliations
Contributions
Z.L., J.C., T.S., D.T., Z.Z: Contributed to the design of the study, interpretation of results, and manuscript preparation. Z.L., J.C., E.C., and T.S.: Performed the data analyses. All authors contribute to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Z., Clarke, J.A., Eliason, C.M. et al. Vocal specialization through tracheal elongation in an extinct Miocene pheasant from China. Sci Rep 8, 8099 (2018). https://doi.org/10.1038/s41598-018-26178-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-26178-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.