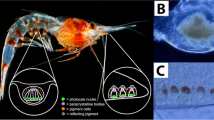
Abstract
Ocular reflectors enhance eye sensitivity in dim light, but can produce reflected eyeshine when illuminated. Some fish can occlude their reflectors during the day. The opposite is observed in cryptic sit-and-wait predators such as scorpionfish and toadfish, where reflectors are occluded at night and exposed during the day. This results in daytime eyeshine, proposed to enhance pupil camouflage by reducing the contrast between the otherwise dark pupil and the surrounding tissue. In this study, we test this hypothesis in the scorpionfish Scorpaena porcus and show that eyeshine is the result of two mechanisms: the previously described Stratum Argenteum Reflected (SAR) eyeshine, and Pigment Epithelium Transmitted (PET) eyeshine, a newly described mechanism for this species. We confirm that the ocular reflector is exposed only when the eye is light-adapted, and present field measurements to show that eyeshine reduces pupil contrast against the iris. We then estimate the relative contribution of SAR and PET eyeshine to pupil brightness. Visual models for different light scenarios in the field show that daytime eyeshine enhances pupil camouflage from the perspective of a prey fish. We propose that the reversed occlusion mechanism of some cryptobenthic predators has evolved as a compromise between camouflage and vision.
Similar content being viewed by others
Introduction
An ocular reflector behind the retina is a common feature of vertebrate eyes. Their presence and diversity across taxa is linked with increased visual sensitivity under dim light1. This is achieved by reflecting light not captured by the retina during its first pass back through the photoreceptors, allowing for an increased photon catch2,3,4. However, ocular reflectors come with two disadvantages. First, visual acuity in bright environments might be reduced by backscatter5,6,7. Second, an animal may become more conspicuous because of eyeshine caused by reflection of light out of the pupil7. In some elasmobranchs, teleost fish and a few reptiles, these side effects are minimized by occlusion mechanisms that cover the reflector in the light-adapted eye with black melanin pigmentation1,8. Hence, strong eyeshine can only be induced when the eye is dark-adapted; it is weak or absent when the eye is light-adapted.
In contrast to this general pattern, a few families of highly cryptic fishes such as toadfishes (Batrachoididae) and scorpionfishes (Scorpaenidae) feature strong eyeshine when the eye is light-adapted, but not when dark-adapted6,9,10. In these species, the reflector is a stratum argenteum located in the outer part of the choroid1,6,11, rather than the common tapetum lucidum. This stratum argenteum is a bi-laminate reflective structure with an inner layer of pentameric uric acid crystals and an outer layer of yellow birefringent granules of unknown material10,12. In the light-adapted state of the eye, the melanosomes of the retinal pigment epithelium enter the cell processes between the cones, clearing the way for light that passed through the receptor layer to also penetrate the translucent choroid1,6 and reach the stratum argenteum. This light is reflected out of the pupil, generating a type of eyeshine termed Stratum Argenteum Reflected (SAR) eyeshine13. Another consequence of choroid translucency is that down-welling light that penetrates the dorsal part of the eye can be transmitted through the sclera, choroid and the retinal pigment epithelium and then out through the pupil. This can result in a second type of eyeshine described as Pigment Epithelium Transmitted (PET) eyeshine13. While the translucency of the sclera and choroid has been already documented for these fishes10,14, pupil eyeshine has always been assumed to be the exclusive result of SAR eyeshine. The possibility that PET eyeshine also contributes to total eyeshine has not been considered. Both eyeshine types might explain why scorpionfishes, toadfishes and some stonefishes have bright pupils when exposed to ambient light14. The possible function of this counter-intuitive occlusion mechanism, however, is yet unclear. Here, we investigate the hypothesis that it conceals the pupil by allowing daytime eyeshine, thus reducing contrast with the surrounding tissue10 (video S1).
The well-defined, often circular, dark pupil that stands out against the body in most animals makes the vertebrate eye difficult to hide15. Eyes are indeed considered key features for face recognition of predators, prey or conspecifics16,17,18,19,20,21,22. Consequently, mechanisms for pupil camouflage are widespread. Pupillary closure (e.g. elasmobranchs) reduces pupil size and shape in response to fluctuations in ambient light, but also minimizes pupil conspicuousness15. Lidless species, such as fishes and snakes, often feature an eye mask (e.g. vertical stripes in lionfishes) that embeds the pupil in a dark skin pattern15. For example, vertical dark stripes have been shown to effectively decrease the visibility of an eyelike pattern23. In some fish species, iridescent corneal reflectors have been proposed to reduce an eye’s detectability24. Skin flaps (e.g. flatheads), where the pupil is partly covered by an irregular extension of the cryptic iris, are another adaptation to reduce pupil conspicuousness15. Some fishes simply have very small pupils for their body size (e.g. frogfishes), which may be a strategy to minimize eye detection at the cost of visual acuity. Thus far, the use of eyeshine for enhancing daytime eye camouflage has only been proposed for invertebrates: pelagic stomatopod larvae reduce the conspicuousness of their dark retinas by eyeshine that matches the light field of the background25. Scorpionfishes, toadfishes and stonefishes are sit-and-wait predators that strongly rely on crypsis. Since featuring a dark pupil could disrupt their camouflage, diurnal pupil eyeshine might help to hide their eyes.
Methods
After introducing our model species, the methods follow a specific logic: first, in the laboratory we confirm a reversed occlusion mechanism in the black scorpionfish. Second, we assess in the field if a bright pupil enhances eye concealment by reducing contrast with the surrounding iris. Third, we characterise the components of daytime eyeshine quantitatively in the laboratory (Table 1 for definitions) and test their validity by reconstructing known pupil radiance measurements from the field. Finally, we developed a visual model to compare the natural pupil and an artificial model pupil contrast against the iris and skin from the perspective of a prey fish under three light scenarios. This allowed us to assess to what extent daytime eyeshine could enhance pupil camouflage.
Model species
The black scorpionfish Scorpaena porcus is common in coastal marine hard-substrate and seagrass habitats, occurring from fully exposed to heavily shaded light environments, in the eastern Atlantic Ocean and Mediterranean Sea26. It is a generalist sit-and-wait predator that relies on crypsis to catch naϊve prey, ambushing them only when close to its mouth over distances of few centimetres. Like most scorpionfish, it features prominent eyes with a partially translucent retinal pigment epithelium resulting in daytime eyeshine (Fig. 1)6,13. We caught 15 individuals in Calvi (Corsica, France) between 5 and 20 m depth under the general permit of STARESO (Station de Recherches Sous Marines et Océanographiques). At STARESO, fish were kept in two 300 L tanks with a continuous fresh seawater flow. For field measurements, three individuals were used and subsequently set free. The remaining twelve were transported to the University of Tübingen (Germany) in individual plastic canisters filled with 1.5 L seawater and oxygen enriched air. In Tübingen, fish were held individually in 160 L tanks (20 °C, salinity 35 ppt, pH 8.2, 12 h light/dark cycle, fed once every two days). Animal husbandry was carried out in accordance with German animal welfare legislation. Because the individuals were not experimentally manipulated, a formal permit was not required for this study (confirmed by the Animal Care Officer at the Biology Department of the University of Tübingen).
Spectroradiometry
Occlusion of the stratum argenteum during dark-adaptation (laboratory)
To confirm that the stratum argenteum is occluding while the eye is dark-adapting, an individual was placed in a 12 L tank (L × W × H: 30 × 20 × 20 cm3) positioned on a cooling plate and equipped with an aeration stone. Before measuring, we light-adapted the fish for 2 h using a sun-simulating Plasma-i AS1300 Light Engine (Plasma International, Mühlheim am Main, Germany) pointed upwards to a diffuse reflector (#273 soft silver reflector, LEE Filters, Andover, England) attached to the ceiling to illuminate the whole room. Three polytetrafluoroethylene (PTFE) white reflectance standards (Lake Photonics, Uhldingen-Mühlhofe, Germany) were positioned in the tank for ambient light measurements. We then measured the change in reflectance of the pupil in relation to time spent in darkness, on a total of three individuals.
The radiance of the left eye pupil was measured at normal incidence (90° angle from surface) through the glass of the tank using a calibrated SpectraScan PR 740 spectroradiometer (Photo Research, NY, USA). This device uses Pritchard optics and measures the absolute spectral radiance of an area with known solid angle. The glass tank was tilted at an angle of 5° to reduce external reflection. We used a cold light source KL2500 LCD (Schott, Mainz, Germany) equipped with a blue filter (insert filter 258302, Schott, Mainz, Germany) to coaxially illuminate the pupil of the fish. The light was led through liquid light guides (LLG 380, Lumatec, Deisenhofen, Germany) to a mechanical shutter and then on to a 90° elbow-shaped glass-fibre light guide (Heine Optotechnik, Herrsching, Germany) with an exit diameter of 3 mm. The light exit was aligned coaxially with the spectroradiometer’s optical axis, 15 cm in front of the lens. The distance between the lens and the fish was fixed at ∼50 cm. Slight distance adjustments were necessary to assure complete coverage of the pupil with the cross-section of the measuring area. Since scorpionfish tend to sit passively on the substrate, there was no need for the use of anaesthesia, which is known to affect fish pigmentation27.
Measurements started immediately after turning off the plasma light source. The room was kept completely dark except for brief moments (< 10 s) during which the shutter of the coaxial source was opened for measuring, approximately once every 10 min for 2 h. The radiance of the PTFE white standard best aligned with the fish’s pupil was measured orthogonal to its surface at the beginning and the end of the experiment. SAR eyeshine reflectance was calculated as the photon radiance of the pupil normalized (i.e. divided) by the average radiance of the PTFE white standard. Because it was exclusively generated by a coaxial light source in an otherwise dark environment, a condition that would be uncommon in the field and that was explicitly staged to strictly describe the reflective properties of the stratum argenteum, we subsequently refer to this measurement as “narrow-sense” SAR eyeshine.
We analysed the data using a Generalized Linear Model (gamma distribution, link = log) with total reflectance (reflectance integrated over the wavelength range from 380 to 700 nm) as response variable, and dark adaptation time, individual ID and their interaction as predictors using R v3.2.028. Model assumptions were verified by plotting residuals against fitted values and against each covariate in the model. Significance of predictors was tested by using the 95% Credible Interval (CrI), a Bayesian analogue of the confidence interval29. For the response variable time, the factor individual ID, and their interaction, we computed the model estimates from the back-transformed effect sizes. The associated 95% CrI were then obtained from 10000 simulations of the mean and variance of each estimate, using the sim function of the R package arm, with non-informative priors30. If the CrI of one individual did not overlap with the mean of another, we concluded that their intercepts were significantly different. We considered time and its interaction with ID to be significant if the CrI values of the regression coefficient did not include zero.
Contrast of natural and model pupil against iris (field)
To confirm that S. porcus produces enough daytime eyeshine to reduce pupil contrast against the iris in the field if compared to a dark model pupil, we measured pupil and iris radiance of three fish in situ in STARESO under shaded and exposed conditions. Each fish was measured in a transparent plastic terrarium (L × W × H: 20 × 15 × 15 cm3) placed underwater on a bright sandy substrate. The side of the container through which measurements were taken consisted of Evotron optically neutral Plexiglas® (Evonik Performance Materials, Essen, Germany). A black PVC sheet was added behind the fish to encourage the scorpionfish to face the spectroradiometer. The other three lateral sides of the container were transparent, exposing the scorpionfish to natural light. Such configuration should minimise possible skin colour adaptations caused by the black PVC sheet placed in the back of the container. Holes in the sides ensured water circulation. To cast an optional shade over the entire fish, we used a black plastic slate. All measurements were taken using a SpectraScan PR 740 in a custom-built underwater housing (BS Kinetics, Achern, Germany) with the spectroradiometer facing south and the fish north, at noon on clear, sunny days. The focal distance of the instrument was fixed at 80 cm. The external dimensions of the underwater housing are 35 × 25 × 25 cm3 (L × W × H), excluding the length of the port (10 cm). The port is located at the upper edge of the housing. It is hard to evaluate the influence of the housing on the light field, but even if a partial obstruction would occur this would hardly influence our estimates since every radiance measurement was obtained under comparable geometries.
We measured pupil and iris radiance (top, bottom, left and right) at normal incidence (90° from surface) in the same three fish at 7 m and 15 m depth. Under shaded conditions, pupil radiance involves SAR reflected eyeshine only, as PET transmitted eyeshine is prevented. Under exposed conditions, both mechanisms contribute to eyeshine. We also estimated the radiance of the skin patches surrounding the iris (top, bottom, right and left of the eye) and the ambient light field for visual modelling (see below). To estimate the ambient light fields, we measured three PTFE white standards, one parallel to the water surface measured from a 45° angle as a proxy for down-welling light, and two perpendicular to the water surface, one of them exposed and the other shaded, measured at normal incidence (90° from standard surface) as a proxy for side-welling light. Finally, we also measured a model pupil (Fig. S1). The latter consisted of a black, hollowed-out PVC block (L × W × H: 6 × 3 × 3 cm3) with a pupil-like opening at the front, and internally filled with black cloth to absorb as much light as possible. The resultant pupil is practically black, mimicking a pupil with no eyeshine. We cycled through all measurements two times, both under shaded and exposed conditions. White standards were also measured twice to control for light fluctuations between measurements.
To compare the contrast of the natural and dark model pupil against the iris, we calculated the average photon radiance of all three structures for each wavelength and exposure. We then calculated the Michelson contrasts Cm as follows:
where Lx1 is the radiance of the pupil, Lx2 the radiance of the iris. There is no contrast when C m = 0. When C m > 0, the pupil is brighter than the iris, and vice versa for C m < 0.
Measurement of SAR and PET eyeshine in light-adapted fish (laboratory)
To describe the relative contributions of reflected (SAR) and transmitted (PET) eyeshine to pupil radiance in light-adapted scorpionfish (Table 1 for a summary), we measured individuals in a similar setup to the one used to confirm occlusion during dark-adaptation. We used a down-welling tungsten source (650+ ARRI, Munich, Germany) attached to the ceiling pointing downward at the glass tank where the scorpionfish was placed. The light reflected by the diffuse grey metal walls of the room generated the side-welling light. To estimate the exclusive contribution of narrow-sense SAR eyeshine, induced only by the coaxial light entering through the pupil, we measured the pupil radiance of 10 individuals using the coaxial illumination system described earlier while only briefly turning off the main source (ARRI) for the duration of the measurement to keep fish light-adapted. Narrow-sense SAR eyeshine reflectance was calculated as the coaxially induced pupil radiance divided by the radiance of a coaxially illuminated PTFE white standard normal to the coaxial source. Since the stratum argenteum behind the eye lens acts as a retroreflector, and reflectance is expressed relative to a diffuse reflecting surface, reflectance values readily exceed 1 under this coaxial geometry, but are much lower than 1 when looking at the eye from a non-coaxial direction. In the field, purely coaxial light conditions as used to characterise narrow-sense SAR eyeshine are very rare. Hence, to properly reconstruct the contribution of SAR eyeshine assessed in the field, we also estimated “broad-sense” SAR eyeshine by measuring the pupil of nine fish and the radiance of a PTFE white standard in the shade (to suppress PET eyeshine), under a general side-welling light field in the absence of a coaxial light source. Broad-sense SAR eyeshine reflectance was calculated as pupil radiance of the shaded fish normalized by the radiance of a shaded PTFE white standard. We assessed the exclusive contribution of PET eyeshine by measuring the pupil radiance of 10 individuals under exposed and shaded conditions. By subtracting the second measurement, which represents the contribution of SAR eyeshine only, from the first, which consists of the contributions of PET plus SAR eyeshine, we obtained the contribution of PET eyeshine only. This value was subsequently expressed as PET eyeshine transmittance dividing by the radiance of an exposed PTFE white standard positioned parallel to the water surface and measured from an angle of 45°. For visual modelling, we also measured the reflectance of the iris and the dark model pupil under exposed conditions.
Reconstructing field values using laboratory estimates
To determine if our laboratory estimates of broad-sense SAR, PET eyeshine, and iris reflectance could be used to reliably predict field observations, we used these measurements to ‘reverse engineer’ the pupil and iris radiance for three fish for which complete field data were available: one at 7 m and two at 15 m. We used down- and side-welling light measurements from the field as the illuminants. By combining them with the broad-sense SAR, PET eyeshine and iris relative radiances estimated in the laboratory, we then predicted pupil and iris radiance under exposed or shaded conditions for each of the three fish. We then assessed the match between the two curves by calculating the mean of the ratio between the logarithm on base 10 of the predicted and real radiances at each wavelength for iris and pupil of each fish under both exposures. Finally, we computed the averaged ratio for all three fish.
Visual models to assess pupil camouflage as perceived by prey fish
To test if PET and SAR eyeshine enhance pupil camouflage in the black scorpionfish, we determined how well the pupil matches the appearance of the iris as perceived by one of its prey species, the triplefin Tripterygion delaisi31 under three light scenarios where this prey-predator interaction is likely to occur in the field. These scenarios are based on a specific geometry commonly found in the field and that was also used for all our measurements (in the lab and in the field). It assumes orthogonal view of the eye, with the scorpionfish sitting on a horizontal substrate. We calculated the chromatic contrasts between the pupil and the iris using the receptor-noise model32. We informed the model using species-specific visual system parameters including the visual sensitivity of the photoreceptors (SWS: 468 nm, MWS: 517 nm, LWS: 530 nm), the relative photoreceptor densities in the fovea of 1:4:4 (SWS: MWS: LWS), the ocular media transmittance, and setting the Weber fraction at 0.0531,32,33. The achromatic contrasts were calculated as the absolute Michelson contrast between the luminance photon catches (sum input of the two members of the double-cone) perceived by the prey species. Using the light field measurements at 7 and 15 m depth, we reconstructed three scenarios in which scorpionfish and triplefin were likely to interact: (1) the scorpionfish and triplefin in the open against bright backgrounds where both PET and SAR eyeshine are present, (2) the scorpionfish in the open and the triplefin in the shade or against a dark background where only PET eyeshine is present (SAR eyeshine is weak or absent because of reduced side-welling illumination), and (3) the scorpionfish in the shade and the triplefin against a bright background in the open where only SAR eyeshine is induced (see Table S1 for details). The calculations generate values of chromatic contrast in just-noticeable-differences (JNDs), where values greater than one indicate discernible differences32, and values of achromatic contrasts as absolute Michelson contrasts (ranging from 0 to 1). To enhance camouflage, the contrast between the pupil and the iris should not only be reduced, but should also be comparable to the contrast of the overall patterning of the fish’s body. To assess the contrast between different skin patches on the body, we used the reflectance of four skin patches around the iris (calculated as the photon radiance of the skin normalized by the respective PTFE white standard facing the spectroradiometer) measured in the field on the three scorpionfish. We calculated the absolute Michelson achromatic contrasts as perceived by the prey species (chromatic contrasts between the two pupils and the iris were not showing substantial differences) for all pairwise comparisons of the skin patches under the three scenarios at both depths. We then compared the achromatic contrasts between the irides and the natural and model pupils with the distribution of the skin patches achromatic contrasts. Visual models were implemented using the R package ‘pavo’ 1.034.
Units, statistics and data availability
Reflectance and transmittance are expressed in relation to the radiance of PTFE white standards, as proportions, not percentages. Means are shown ± standard deviation unless specified otherwise. All data are available in the Dryad repository (https://doi.org/10.5061/dryad.dp4kt5t), and R scripts are available upon request.
Results
Occlusion of the stratum argenteum during dark-adaptation (laboratory)
Model validation did not show any violation of the model assumptions. The reflectance of the light-adapted pupil of Scorpaena porcus was on average up to 30 times as strong as a white standard (27.3 ± 7.4). This value fell significantly during dark-adaptation to 4.6 ± 0.7 (Fig. 2) with a regression coefficient of 0.0099 (95% Credible Interval (CrI): from 0.0078 to 0.0120). This implies that pupil reflectance decreased by 1% per minute. At the beginning of the experiment, one fish showed significantly higher reflectance, up to 35 times as strong as a white standard (95% CrI: from 0.1800 to 0.8089). The overall model fit was very high, R2 = 0.86.
Reduction in narrow-sense SAR eyeshine with increased dark-adaptation in Scorpaena porcus. Narrow-sense SAR eyeshine total reflectance (pupil radiance normalized by white standard radiance) under coaxial illumination in the dark as function of time. At t = 0 the fish was light-adapted and the light was switched off resulting in total darkness. The small, coaxial source used to induce eyeshine was only switched on for a brief moment for each measurement. Radiance is integrated over 380–700 nm. Line styles and symbols indicate three different individuals.
Contrast of natural and model pupil against iris (field)
The natural pupil showed reduced contrast against the iris relative to the model pupil, which was always considerably darker than the iris. At 7 m depth, this was true for both shaded and exposed conditions (Fig. 3). At 15 m depth, the difference between the contrasts of the two pupils against the iris was strong only when exposed. Under shaded conditions both the natural and model pupil showed low contrast.
Michelson contrast at each wavelength between natural pupil or dark model pupil against the iris in the field. Measurements are from exposed and shaded Scorpaena porcus (n = 3 ind.) at 7 and 15 m depth. Positive values indicate that pupils are brighter than the irides. Shaded areas represent the standard error of the mean. (a) Exposed and (b) shaded fish at 7 m. (c) Exposed and (d) shaded fish at 15 m.
Measurement of SAR and PET eyeshine in light-adapted fish (laboratory)
Narrow-sense reflected (SAR) eyeshine (n = 10 ind.) was stronger than a PTFE white standard, showing an average reflectance of 12.01 ± 3.82 (Fig. 4a). Broad-sense SAR eyeshine (n = 9 ind.) yielded an average reflectance of 0.06 ± 0.01 (Fig. 4b). Transmitted (PET) eyeshine (n = 10 ind.) had an average transmittance of 0.033 ± 0.028 (Fig. 4b) with a shift towards the long wavelength part of the spectrum. The iris (n = 9 ind.) showed an average reflectance of 0.15 ± 0.05 (Fig. 5) and the model pupil an average reflectance of the negligible value of 0.0008 ± 0.0001 (not shown in the figure).
Wavelength dependent contribution of eyeshine types in Scorpaena porcus. (a) Narrow-sense SAR eyeshine reflectance. (b) Broad-sense SAR eyeshine reflectance and PET eyeshine transmittance. Y-values are expressed as proportions of the pupil photon radiance normalized by the radiance of the associated PTFE white standard. Shaded areas represent the standard error of the mean.
Reconstructing field values using laboratory estimates
Using the laboratory estimates, we predicted the pupil and iris radiance under natural light conditions and compared them to the actual field measurements (Fig. 6). Overall, in fish exposed to ambient light, the averaged ratio between the measured and reconstructed values was 1.004 ± 0.007 for the pupil and 1.012 ± 0.012 for the iris, where a value of 1 represents the perfect match. When the fish was shaded, pupil and iris radiances were predicted with a ratio of 1.024 ± 0.009 and 1.008 ± 0.008.
Reconstruction of known field pupil and iris radiance. Photon radiance of pupil (a, e, i) and iris (b, f, j) in exposed Scorpaena porcus individuals. Panels (c, g, k) and (d, h, l) show the same for shaded individuals. Solid lines show radiance measured in the field. Dotted lines show radiance reconstructed using the ambient light measurements for each fish combined with the eyeshine and iris relative radiances estimated in the laboratory. Photon radiance values are expressed on a log-scale. Each colour indicates a different individual, placed at 7 or 15 m depth.
Visual models to assess pupil camouflage as perceived by prey fish
A natural pupil with PET and/or SAR eyeshine showed a similar chromatic contrast against the iris if compared to a model pupil in all scenarios, except for a slight increase when only PET eyeshine was present (triplefin shaded, scorpionfish exposed) (Table 2). However, achromatic contrast was substantially reduced (Table 2). Whereas the model pupil without eyeshine against the iris showed absolute achromatic contrasts around 1, the contribution of one or both eyeshine mechanisms reduced this contrast to less than 0.4. This value is well within the range (from 0 to 0.6) of the achromatic contrasts between skin patches found near the irides (Fig. 7).
Predicted absolute achromatic contrast of the pupil with and without eyeshine against the iris of Scorpaena porcus compared to the contrast among body patches as perceived by a prey species. The achromatic contrast between a natural pupil with eyeshine and its iris (black lines) was small and fell within the distribution of the achromatic contrasts between skin patches (all combinations of four patches in each of the three fish measured; histogram bars). This was not the case when eyeshine was prevented, as in the dark model pupil (red lines). Black lines indicate contrast values between the natural pupil and the iris when Tripterygion delaisi is shaded (dashed) or exposed (solid) for four S. porcus scenarios: (a) exposed and (b) shaded at 7 m depth, (c) exposed and (d) shaded at 15 m depth. Red lines indicate contrast values between the dark model pupil and the iris when T. delaisi is shaded (dashed) or exposed (solid) for the same four S. porcus scenarios (note that in scenario (a) and (c) two lines overlap).
Discussion
Only few families of cryptobenthic fish occlude their ocular reflector in the dark and expose it during the day, which is opposite to what is known from fish featuring an occlusible ocular reflector. Reversed occlusion can make a pupil bright during the day, but keeps it dark at night, even when illuminated. We confirmed the presence of reversed occlusion in the scorpionfish Scorpaena porcus and show that two different eyeshine types contribute to daytime pupil radiance: Stratum Argenteum Reflected (SAR) and Pigment Epithelium Transmitted (PET) eyeshine. Spectroradiometry in the field shows that daytime eyeshine reduces the contrast between pupil and iris, which may help to conceal an otherwise dark pupil during the day. Visual modelling confirmed this observation: daytime eyeshine reduces pupil contrast against the surrounding tissue, decreasing the detectability of S. porcus pupil from the perspective of a prey species under three light scenarios where SAR and PET eyeshine differ in their contribution to pupil radiance.
The presence of an occluded stratum argenteum in the dark-adapted eye was first observed in S. porcus during night dives (pers. obs.). The occlusion mechanism responsible for this phenomenon may be similar to the one described for other species of toadfishes and scorpionfishes6,10. Why these fishes suppress eyeshine when dark-adapted remains unclear. Using the stratum argenteum as a reflector in dim light conditions would allow for an increased quantal catch and thus increased visual acuity10. Covering it with pigmentation, however, forgoes this option. Enhanced pupil camouflage by reversed occlusion may come at the expense of reduced visual acuity due to a possible increase in internal scatter in the light-adapted eye and a reduced photon catch in the dark-adapted eye. However, pupil concealment by means of daytime eyeshine may explain why scorpionfish possess unobstructed, large pupils. Most other cryptobenthic predators feature fringes, skin flaps, or have small pupils to reduce eye conspicuousness.
Daytime eyeshine due to the absence of melanocytes in the choroid has been already described for toadfishes and scorpionfishes6,10,14. Until now, however, the focus was exclusively on reflected (SAR) eyeshine6,10,14. Our measurements in the laboratory show that eyeshine in S. porcus has a second component in the form of transmitted (PET) eyeshine. These two eyeshines could be mechanistically linked because they both rely on the translucent choroid and the exposed stratum argenteum. Since the combination of PET and SAR eyeshine could enhance pupil camouflage, the bright pupils of this fish should not be seen just as a side-effect of eye anatomy, but rather as a possible adaptive trait.
For a sit-and-wait predator, successful prey capture strongly depends on its crypsis35. In this study, we show that daytime eyeshine enhances the camouflage of the pupil in a cryptobenthic predator. Eyeshine does not perfectly camouflage the eye, but it likely reduces the probability of detection compared to a situation in which the pupil is dark. The contribution of SAR eyeshine to pupil brightness had already been summarily described6, but we also showed that transmitted PET eyeshine is an additional component that makes a significant contribution, regardless of its low transmittance, particularly because it is induced by the relatively strong down-welling light rather than the relatively weak side-welling light. A similar concealing process has been described for pelagic stomatopod crustacean larvae that use a photonic structure external to the optical pathway of the eye to hide their dark retinas25. Since in S. porcus the stratum argenteum is located in the optical pathway, this ocular reflector may also improve visual sensitivity in dim light environments10. However, reversed occlusion of the stratum argenteum strongly suggests that this function is secondary to camouflage. Future behavioural studies could be implemented to strengthen these findings.
As alternative explanation, bright pupils could be rather used as a lure for attracting potential prey, a mechanism that is similar to the one proposed for the bright lures of some deep-sea organisms36. However, since this type of eyeshine is diurnal, it is unlikely that the pupil could become bright enough to stand out against the surroundings, especially considering the amount of bright structures and surfaces already present in the field.
We did not inform our visual models with the true physical properties of (narrow-sense) SAR eyeshine. This parameter showed extremely high reflectance values (Fig. 4), as expected in the presence of a retroreflector, and it would only have to be considered if the observer possesses a light source close to its eyes, or if sunlight is shining parallel to the observer’s visual axis in an otherwise dark environment, e.g. a narrow crevice. Under these conditions, the target pupil would appear so bright that it may stand out against the iris, potentially decreasing eye camouflage. Our visual models instead focused on the broad-sense SAR eyeshine reflectance, which represents the more general situation in which the complete side-welling light field is considered.
This is the first study showing that a vertebrate features daytime eyeshine by means of two complementary mechanisms to reduce the conspicuousness of its large pupils to the perspective of one of its prey species. We propose that the unusual reversed occlusion of the ocular reflector has evolved in response to selection to optimize the trade-off between camouflage and vision.
References
Ollivier, F. et al. Comparative morphology of the tapetum lucidum (among selected species). Vet. Ophthalmology 7, 11–22 (2004).
Greene, N. R. & Filko, B. J. Animal-eyeball vs. road-sign retroreflectors. Ophthalmic Physiol. Opt. 30, 76–84 (2010).
Land, M. F. & Nilsson, D.-E. Animal eyes (Oxford University Press, 2012).
Schwab, I. R., Yuen, C. K., Buyukmihci, N. C., Blankenship, T. N. & Fitzgerald, P. G. Evolution of the tapetum. Transactions Am. Ophthalmol. Soc. 100, 187 (2002).
Barton, M. & Bond, C. E. Bond’s biology of fishes (Thomson, 2007).
Best, A. & Nicol, J. Eyeshine in fishes. a review of ocular reflectors. Can. J. Zool. 58, 945–956 (1980).
Denton, E. On the organization of reflecting surfaces in some marine animals. Phil. Trans. R. Soc. Lond. B 258, 285–313 (1970).
Denton, E. & Nicol, J. The chorioidal tapeta of some cartilaginous fishes (chondrichthyes). J. Mar. Biol. Assoc. United Kingd. 44, 219–258 (1964).
Grover, D. A. & Zigman, S. Funduscopic morphology of the light-and dark-adapted eye of the toadfish (Opsanus tau) in vivo. Can. J. Zool. 69, 2498–2500 (1991).
Nicol, J. Studies on the eyes of toadfishes opsanus. structure and reflectivity of the stratum argenteum. Can. J. Zool. 58, 114–121 (1980).
Herring, P. J. Reflective systems in aquatic animals. Comparative Biochemistry and Physiol. Part A: Physiol. 109, 513–546 (1994).
Nicol, J. Uric acid in the stratum argenteum of toadfishes opsanus. Can. J. Zool. 58, 492–496 (1980).
Fritsch, R., Ullmann, J. F., Bitton, P.-P., Collin, S. P. & Michiels, N. K. Optic-nerve-transmitted eyeshine, a new type of light emission from fish eyes. Front. Zool. 14, 14 (2017).
Nicol, J. A. C. & Somiya, H. The eyes of fishes (Oxford University Press, USA, 1989).
Cott, H. B. Adaptive coloration in animals (Methuen & Co., Ltd., 1940).
Gothard, K. M., Brooks, K. N. & Peterson, M. A. Multiple perceptual strategies used by macaque monkeys for face recognition. Animal Cogn. 12, 155–167 (2009).
Jones, R. Reactions of male domestic chicks to two-dimensional eye-like shapes. Animal Behav. 28, 212–218 (1980).
Kano, F. & Tomonaga, M. Face scanning in chimpanzees and humans: Continuity and discontinuity. Animal Behav. 79, 227–235 (2010).
Kjernsmo, K., Grönholm, M. & Merilaita, S. Adaptive constellations of protective marks: eyespots, eye stripes and diversion of attacks by fish. Animal Behav. 111, 189–195 (2016).
Satoh, S., Tanaka, H. & Kohda, M. Facial recognition in a discus fish (cichlidae): Experimental approach using digital models. PloS ONE 11, e0154543 (2016).
Scaife, M. The response to eye-like shapes by birds. i. the effect of context: a predator and a strange bird. Animal Behav. 24, 195–199 (1976).
Trnka, A., Prokop, P. & Grim, T. Uncovering dangerous cheats: how do avian hosts recognize adult brood parasites? PLoS ONE 7, e37445 (2012).
Karplus, I. & Algom, D. Visual cues for predator face recognition by reef fishes. Ethol. 55, 343–364 (1981).
Lythgoe, J. The structure and function of iridescent corneas in teleost fishes. Proc. Royal Soc. Lond. B: Biol. Sci. 188, 437–457 (1975).
Feller, K. D. & Cronin, T. W. Hiding opaque eyes in transparent organisms: a potential role for larval eyeshine in stomatopod crustaceans. J. Exp. Biol. 217, 3263–3273 (2014).
Louisy, P. & Ade, C. Meeresfische: Westeuropa und Mittelmeer (Ulmer, 2002).
Gray, S. M., Hart, F. L., Tremblay, M. E., Lisney, T. J. & Hawryshyn, C. W. The effects of handling time, ambient light, and anaesthetic method, on the standardized measurement of fish colouration. Can. J. Fish. Aquatic Sci. 68, 330–342 (2011).
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria). https://www.R-project.org/ (2017).
Bolker, B. M. et al. Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol. & Evol. 24, 127–135 (2009).
Korner-Nievergelt, F. et al. Bayesian data analysis in ecology using linear models with R, BUGS, and Stan (Academic Press, 2015).
Bitton, P.-P. et al. Red fluorescence of the triplefin Tripterygion delaisi is increasingly visible against background light with increasing depth. Royal Soc. Open Sci. 4, 161009 (2017).
Vorobyev, M. & Osorio, D. Receptor noise as a determinant of colour thresholds. Proc. Royal Soc. Lond. B: Biol. Sci. 265, 351–358 (1998).
Douglas, R. & Djamgoz, M. The visual system of fish (Springer Science & Business Media, 2012).
Maia, R., Eliason, C. M., Bitton, P.-P., Doucet, S. M. & Shawkey, M. D. pavo: an r package for the analysis, visualization and organization of spectral data. Methods Ecol. Evol. 4, 906–913 (2013).
Stevens, M. & Merilaita, S. Animal camouflage: current issues and new perspectives. Philos. Transactions Royal Soc. Lond. B: Biol. Sci. 364, 423–427 (2009).
Haddock, S. H., Dunn, C. W., Pugh, P. R. & Schnitzler, C. E. Bioluminescent and red-fluorescent lures in a deep-sea siphonophore. Sci. 309, 263–263 (2005).
Acknowledgements
We warmly thank the STARESO staff for hosting and supporting our research group in their field station, Oeli Oelkrug for fish maintenance at the University of Tübingen. We also thank Dr. Martin J. How and Dr. Yakir Gagnon for fruitful comments and suggestions and Dr. Nils Anthes and four anonymous reviewers for useful comments on previous drafts of the manuscript. We acknowledge support by Deutsche Forschungsgemeinschaft and Open Access Publishing Fund of University of Tübingen. The project was funded by the by the German Science Foundation Koselleck grant (Mi482/13-1) and the Volkswagen Foundation (Az. 89148 and Az. 91816) awarded to NKM. P-PB was funded by a Postdoctoral Fellowship from the Natural Science and Engineer Research Council of Canada.
Author information
Authors and Affiliations
Contributions
M.S. conceptualised the study. M.S. and N.K.M. designed the experimental setups. M.S. performed spectroradiometry in the laboratory. M.S., U.K.H. and N.K.M. performed spectroradiometry in the field. M.S. analysed the data. M.S. and P.-P.B. implemented the visual models. M.S. drafted the manuscript. M.S., N.K.M. and P.-P.B. edited the manuscript and all authors commented on the completed manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Santon, M., Bitton, PP., Harant, U.K. et al. Daytime eyeshine contributes to pupil camouflage in a cryptobenthic marine fish. Sci Rep 8, 7368 (2018). https://doi.org/10.1038/s41598-018-25599-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-25599-y
This article is cited by
-
Scorpionfish rapidly change colour in response to their background
Frontiers in Zoology (2023)
-
Visual modelling supports the potential for prey detection by means of diurnal active photolocation in a small cryptobenthic fish
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.