Abstract
The mental representation of pitch structure (tonal knowledge) is a core component of musical experience and is learned implicitly through exposure to music. One theory of congenital amusia (tone deafness) posits that conscious access to tonal knowledge is disrupted, leading to a severe deficit of music cognition. We tested this idea by providing random performance feedback to neurotypical listeners while they listened to melodies for tonal incongruities and had their electrical brain activity monitored. The introduction of random feedback was associated with a reduction of accuracy and confidence, and a suppression of the late positive brain response usually elicited by conscious detection of a tonal violation. These effects mirror the behavioural and neurophysiological profile of amusia. In contrast, random feedback was associated with an increase in the amplitude of the early right anterior negativity, possibly due to heightened attention to the experimental task. This successful simulation of amusia in a normal brain highlights the key role of feedback in learning, and thereby provides a new avenue for the rehabilitation of learning disorders.
Similar content being viewed by others
Introduction
Tonality is the complex hierarchical structure that governs the organization of pitch in Western music. This structure organizes the 12 chromatic tones into keys, forming a schema that places all the possible tones at varying distances from a central stable pitch known as the tonic. This schema is the basis of the cognition of musical pitch, and informs the perception of tones in musical contexts. The processing of tonality (key) is a core component of music experience and is learned implicitly1,2. Despite the implicit nature of its mental representation, listeners can perform tasks that require conscious access to tonal representations. For instance, implicit knowledge about tonal pitch regularities helps listeners to detect out-of-key notes within musical excerpts. This implicit knowledge allows non-musicians to rate the belongingness of probe tones within a tonal context in a manner consistent with music theoretical descriptions of tonality, despite a lack of explicit knowledge of these rules3. It is precisely this lack of explicit knowledge that makes access to tonal knowledge prone to experimental manipulation. The main goal of this study was to manipulate conscious access to tonal knowledge in order to model congenital amusia (tone deafness) in neurotypical individuals.
Congenital amusia is a neurodevelopmental disorder causing a lifelong deficit in melodic perception and production that cannot be explained by hearing loss, brain damage, intellectual deficiencies, or lack of music exposure4. Recent research on congenital amusia refers to this condition as a disorder of conscious access or awareness5,6,7. Amusics fail to consciously detect tonal violations in melodies. Yet, they seem to have access to tonal knowledge when probed non-consciously8. For example, amusics exhibit faster and more accurate responses to a target phoneme when that phoneme is sung on a tonally expected chord compared to an unexpected chord, just like neurotypical controls do9. The most compelling evidence comes from studies measuring brain responses to tonal violations. When a pitch falls outside the key of the presented melody, an early negativity is observed in both amusic and control participants6,10. This brain response, often called the early right anterior negativity (ERAN), is related to the automatic, non-conscious perception of a tonal hierarchical violation11,12 (although it can also be modulated in specific circumstances by attention13,14,15,16). In contrast, when amusics are asked to detect these pitch violations, the late positivity (i.e., P3, P300, or P600) typically observed in controls is not observed in amusics6. This late positivity is related to the conscious, voluntary detection of a tonal violation17, and the lack thereof is associated with chance performance6. This overall pattern has been repeatedly observed in a number of neurophysiological studies18,19,20,21,22. Thus, the amusic deficit seems to be rooted in a lack of conscious access to tonal knowledge that is nevertheless represented by the brain.
Neuroimaging studies of amusia have reported impoverished connectivity with the frontal lobe8,18,23,24,25. Interestingly, both experimental26,27,28,29 and patient30,31 studies of consciousness have reported that the frontal lobe plays a vital role in conscious access to information. In the case of amusia, converging evidence from magnetoencephalography18, functional magnetic resonance imaging24, voxel-based morphometry32, cortical thickness23, and diffusion tensor imaging8 suggests that disordered connectivity between the right superior temporal gyrus and the right inferior frontal gyrus is the source of the amusic disorder. These studies provide a neurophysiological basis for the lack of conscious access to pitch information. Accordingly, the current view of amusia is that impoverished fronto-temporal connectivity reduces the ability of the frontal lobes to modulate pitch processing in an otherwise normally functioning auditory cortex18,22.
Here, we attempted to simulate the disconnection between the auditory cortex, which is taken to represent pitch information (and is associated with the generation of early auditory responses), and the inferior frontal gyrus, which is critical for conscious access to hierarchical representations of pitch (and is associated with the generation of late auditory responses)11,33,34,35, by creating a mismatch between external and internal cues. External cues were manipulated by altering performance feedback during the detection of an out-of-key note in a task originally used by Peretz et al.6 to study electrical brain responses in amusia. After acquiring baseline measurements of brain activity and task performance without feedback, we interfered with participants’ conscious access to tonal knowledge. This interference was achieved by presenting random feedback on each trial, such that a participant’s responses were reported as 50% “correct” and 50% “incorrect”, regardless of the actual response. To ensure the effects of random feedback were due specifically to its random nature and not simply the occurrence of feedback, a control group of participants was tested. This group went through exactly the same procedure, but received accurate feedback in the blocks following baseline.
For the control group in all blocks and the experimental group during baseline, we predicted that accuracy and confidence would be significantly above chance. Additionally, we expected an ERAN and a late positivity in response to pitch violations (out-of-key notes). These neural responses would indicate a match between the early computation of a tonal violation in the temporal lobe and higher-level conscious integration with prior knowledge in the inferior frontal gyrus. For the experimental group, we predicted that random feedback would induce a top-down perturbation in these cortical interactions. Thus, the administration of random feedback over two blocks of trials should lead to a decrease in both confidence and accuracy as compared to baseline, paralleled by a reduction of the late positive brain responses, while leaving the ERAN unaffected. This pattern of results would mirror the behavioural and neurophysiological expression of amusia. More generally, if we can model the amusic phenotype using false feedback, we could establish whether there is a causal link between unreliable feedback, conscious access to tonal knowledge, and the amusic disorder. If this effect is due specifically to random feedback, we should not observe any change in the behavioural performance or brain activity of control participants across the blocks.
Methods
Ethical Approval and Informed Consent
All experimental protocols were approved by the Arts and Sciences Research Ethics Committee (CERAS) at l′Université de Montréal, and the methods were carried out in accordance with this committee’s guidelines. All participants provided informed consent prior to participating in the experiment.
Participants
Participants were recruited via advertisement on the internet and Université de Montréal campus, and compensated $10/hour for their participation. Participants were recruited first for the experimental group (participants who received false feedback after the baseline), followed by the control group (participants who did not receive false feedback). The experimental and control groups comprised 18 and 22 university students who were matched for age, education, and musical training (Table 1). All participants were non-musicians, with less than 5 years of formal training. Twelve experimental participants and 12 control participants reported no formal musical training, and no participant reported having ever taken music theory or harmony classes. All participants were right-handed, had no known neurological problems, and had normal hearing (according to self-report). Participants were excluded if they failed to score more than 57% correct (2 SD below the mean of a large control sample36) in the first block of the experiment. This criterion resulted in the exclusion of three participants who were initially recruited for the control group and five participants who were initially recruited for the experimental group.
Materials
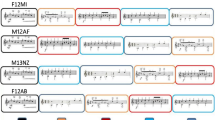
A set of 40 melodies, drawn from a corpus used previously with amusic participants6,22,37, served as stimuli. All of these were melodies in a major key and varied in rhythm. Melodies contained between 7 and 15 successive tones (\(\bar{{\rm{x}}}\) = 10.3, s = 1.90) and were played at 120 beats per minute (500 ms per beat). The whole set of melodies contained notes ranging in pitch from B4 to C5 (two octaves). Stimulus files are available in MIDI format at https://osf.io/f2tu9/. These melodies were synthesized in four versions, varying with regard to instrumental timbre (guitar or piano) and condition (in-key or out-of-key), resulting in 160 melodies in total. Importantly, the changed pitch always affected the same critical tone, which was 500 ms in duration and fell on the first downbeat in the third bar of the four-bar melody. For in-key melodies, all tones fell within the key of the melody, and for out-of-key melodies, the target tone was shifted by a semitone to fall out of the key of the melody, but remained close in pitch and respected the contour (see Fig. 1 for an example). In an effort to decrease the sensory novelty of out-of-key targets compared to in-key targets, the melodies were presented in eight different keys (A, Bb, B, C, D, Eb, F, or G). Furthermore, ten pitches were used as out-of-key targets (A, Bb, B, C, Db, Eb, E, F, G, Ab), and nine pitches were used as in-key targets (A, Bb, B, C, Db, D, E, F, Gb). Given the significant overlap between conditions, there was no significant difference in frequency of occurrence between in-key and out-of-key targets, t(15) = 0.09, p = 0.93.
The order of the melodies was set using a pseudorandomization technique to ensure that there was a relatively even distribution of in-key vs. out-of-key melodies and guitar vs. piano timbres across the trials, and that different versions of the same melody were not played back-to-back. The 160 melodies were divided into 20 sets of eight melodies each. Each of these sets contained four in-key melodies and four out-of-key melodies; four of these were produced with a piano sound and the other four with a guitar sound. Only one version (i.e., guitar/piano or in-key/out-of-key) of any of the original 40 melodies could occur in each set.
Participants completed four blocks of 40 trials each. These blocks were created for each participant by randomizing the 8 melodies in each set and then randomly combining the 20 sets into four blocks of five sets. For each melody, participants were asked to judge if the melody contained an anomalous note, and how confident they were of their judgment. Judgments were measured on a four-point scale (1 – anomalous note/sure; 2 – anomalous note/not sure; 3 – no anomalous note/not sure; 4 – no anomalous note/sure). These response choices appeared on the screen during each trial. Participants were required to respond using their right hand.
Apparatus
Stimuli were presented to participants using a personal computer running Windows XP, with code written and run in MATLAB 2011b, using the Psychophysics toolbox38,39. Visual components of the experiment were presented on a Dell Trinitron monitor. The auditory components of the experiment were presented through a pair of Etymotic ER-2 insert earphones connected to a Fireface 800 soundcard, with the volume of the experimental stimuli calibrated to 70 dB SPL. Responses were collected using the computer keyboard.
Procedure
Prior to the experiment, participants were given written instructions (in addition to the verbal ones just described) and were presented with two examples of each melody type (i.e., melodies containing and not containing an out-of-key note). None of the example melodies were used during the experimental task. Participants were allowed to ask questions and listen to the examples as many times as they wished, until they were comfortable with the task.
Experimental Group
Once participants were ready to begin, they were informed that they would hear three blocks of melodies (a deception). The first block (Baseline) would constitute practice trials, and they would receive no feedback. Any individual who did not perform significantly above chance (hits − false alarms = 0) was excluded. The second and third blocks (Random 1, Random 2) would constitute test trials, during which they should expect to receive feedback. This feedback would occur in the form of game-show inspired sounds, with “correct” responses being signaled with a ringing bell, and “incorrect” responses being signaled with a low-pitched buzzer. For the experimental group, “correct” vs. “incorrect” feedback was determined randomly (50% trials signaled correct, 50% incorrect), regardless of the actual response of the participant.
After the third block, experimental participants were given a short survey asking whether they had noticed anything strange about the feedback they had received, and if so, what the nature of the alteration might have been. Following this survey, the experimenter debriefed the participant regarding the random feedback they had been receiving. The participant was then informed that we would record a fourth block of trials (Recovery), in which correct feedback would be provided.
Control Group
The procedure for control participants was identical to the procedure for the experimental group, with a few important changes. First, participants were told that they would hear four blocks of trials (one block of practice – Baseline; three blocks of test trials with correct feedback – Correct 1, Correct 2, Correct 3) from the outset of the experiment. For the second through fourth blocks, “correct” vs. “incorrect” feedback was congruent with participant performance on each trial. Finally, for control participants, there was no break between the third and fourth blocks. Rather, participants were debriefed at the end of the experiment following all four blocks.
The entire experimental session was approximately 1.5 hours in duration.
Recording and Averaging of Electrical Brain Activity
Encephalography (EEG) was digitized continuously over all four experimental blocks, from 70 active electrodes at a sampling rate of 1024 Hz, using a Biosemi ActiveTwo system. Six electrodes were placed bilaterally at mastoid, inferior ocular, and lateral ocular sites (M1, M2, IO1, IO2, LO1, LO2).
EEG processing was accomplished using Brain Electrical Source Analysis software (BESA, version 5.2). Before the start of the experiment, prototypical eye blinks and horizontal and vertical and eye movements were recorded. A principal component analysis of these prototypical recordings provided a set of components that best explained the eye movements. These components were then decomposed into a linear combination along with topographical components that reflected brain activity. This linear combination allowed the scalp projections of the artifact components to be subtracted from the continuous EEG to minimize ocular contamination such as blinks, vertical and lateral eye movements for each individual average with minimal effects on brain activity40. Trials containing excessive noise (>120 µV) at electrodes not adjacent to the eyes (i.e., IO1, IO2, LO1, LO2, FP1, FP2, FPz, FP9, and FP10) were rejected before averaging. On average 87.5% of trials were accepted across all blocks and conditions. To determine whether the number of accepted trials differed across conditions, an ANOVA that included Block (1, 2, 3, 4), and Condition (In-Key, Out-of-Key) was calculated. Neither the main effects of Block and Condition nor their interaction was significant, all p > 0.18.
After this correction, continuous EEG was averaged into ERPs based on the onset of the target tone (i.e., in- or out-of-key note). The averaged ERP epoch included 1500 ms of post-target activity. ERP trials were separated for each condition and each block, into eight event-related potentials (ERPs) corresponding to the two conditions (in-key, out-of-key) and four blocks (baseline, random feedback 1, random feedback 2, recovery). Each ERP was corrected using a 50 ms pre-stimulus baseline, band-pass filtered to attenuate frequencies below 0.1 Hz (forward, 6 dB/octave) and above 30 Hz (zero-phase, 12 dB/octave), and referenced to the averaged mastoid. The short 50 ms baseline was used to reduce the amount of noise contained in the baseline period, due to the highly variable but ecologically valid melody stimuli.
Results
Data Availability
The datasets generated during and analyzed for the current study are available online at https://osf.io/f2tu9/.
Statistics
All reported statistical tests were two-tailed. All dependent variable distributions were indistinguishable from a normal distribution, according to the Shapiro-Wilk test.
Behaviour
Accuracy
Accuracy was calculated by subtracting the percentage of false alarms (i.e., incorrect judgment that a melody contains an anomalous note when it does not) from the percentage of hits (i.e., correct judgment that a melody contains an anomalous note) for each block (i.e., hits minus false alarms). As a first step, baseline accuracy was compared between the experimental and control groups. There were no differences in performance between the two groups during the baseline block, t(38) = 0.81, p = 0.43.
Accuracy was submitted to mixed effects ANOVA, with Block (1, 2, 3, 4) as a within-subjects factor and Group (Experimental, Control) as a between-subjects factor. Accuracy changed significantly across the four blocks, F(3,114) = 7.50, p < 0.001, η p 2 = 0.17, and the control group outperformed the experimental group overall, F(1,38) = 5.97, p = 0.02, η p 2 = 0.14. There was also a significant interaction between Block and Group, F(3,114) = 3.51, p = 0.02, η p 2 = 0.09, indicating that accuracy changed across blocks for the experimental, but not the control group. Indeed, a quadratic polynomial trend significantly predicted the pattern of accuracy across the four blocks for the experimental group, F(1,17) = 14.76, p = 0.001, η p 2 = 0.47, indicating that accuracy fell from baseline during the two random feedback blocks and increased towards baseline levels in the recovery block (Fig. 2). There was no such quadratic effect present in the control group, F(1,17) = 1.62, p = 0.22, η p 2 = 0.07.
Confidence
Confidence was quantified by calculating the percentage of trials reported as “sure” (regardless of the perceptual judgment) for each block and condition. Confidence was submitted to mixed effects ANOVA, with Block (1, 2, 3, 4) and Condition (In-Key, Out-Of-Key) as within-subjects factors and Group (Experimental, Control) as a between-subjects factor. Participants were more confident in their judgments of melodies in which all notes were in-key compared to melodies containing out-of-key notes, F(1,38) = 6.86, p = 0.01, η p 2 = 0.15. Confidence changed across blocks, F(3,114) = 4.49, p = 0.005, η p 2 = 0.11, but there was no overall confidence difference between groups, F(1,38) < 0.001, p = 0.99, η p 2 < 0.001. However, there was a significant interaction between Block and Group, F(3,114) = 3.99, p = 0.01, η p 2 = 0.10, indicating that confidence changed across blocks for the experimental, but not the control group. Indeed, a quadratic polynomial trend significantly predicted the pattern of confidence across the four blocks for the experimental group, F(1,17) = 7.45, p = 0.01, η p 2 = 0.31, indicating that confidence fell from baseline during the two random feedback blocks and increased towards baseline levels in the recovery block (Fig. 2). There was no such quadratic effect present in the control group, F(1,17) = 0.14, p = 0.71, η p 2 = 0.01. Finally, there was a significant three-way interaction between Group, Block, and Condition, F(3,114) = 3.66, p = 0.02, η p 2 = 0.09 (Fig. 2). No other interaction effect was significant, all F values < 2.34, all p values > 0.07.
Overall, this pattern of results suggests that the random feedback manipulation was effective. Both accuracy and confidence decreased significantly as a result of the introduction of random feedback, and rebounded back towards baseline with the introduction of accurate feedback during the recovery block.
Next, we assessed the effect of false feedback on behaviour in the experimental group. Participants received false feedback when they were told their correct response was incorrect or when they were told their incorrect response was correct. During the random feedback blocks, participants had a 50% chance of being told their response was “correct” or “incorrect” on each trial, regardless of their response. Because of individual differences in task performance, this fixed probability of “correct” vs. “incorrect” feedback led to different participants receiving varying proportions of false feedback. On average, participants in the experimental group received false feedback on 35.17 ± 11.25% of random feedback trials (range = [16%, 56.3%]). In order to assess whether this inter-participant variability in false feedback rate was related to accuracy and confidence, the false feedback rate for each participant was correlated with accuracy and confidence during both random feedback and recovery blocks. There were no significant correlations between the amount of false feedback a participant received and their behavioural performance.
Event-related Potentials
ERAN
The ERAN was measured as the mean amplitude difference between out-of-key and in-key notes during the 100–250 ms epoch post-stimulus onset, over twelve right-frontal electrodes (Fz, F2, F4, F6, FCz, FC2, FC4, FC6, Cz, C2, C4 & C6). ERAN amplitude was submitted to mixed effects ANOVA, with Block (1, 2, 3, 4) and Condition (In-Key, Out-Of-Key) as within-subject factors and Group (Experimental, Control) as a between-subjects factor. As can be seen in the difference wave plotted in Fig. 3A, there was greater negativity for the out-of-key note compared to the in-key note, F(1,38) = 37.54, p < 0.001, η p 2 = 0.50. The only other significant effect was a three-way interaction between Condition, Block and Group, F(3,114) = 2.94, p = 0.036, η p 2 = 0.07.
Experimental and control group ERP difference waveforms (out-of-key minus in-key) for the ERAN at electrode F4 (top) and the P3 at electrode POz (bottom). Grey lines highlight the ERAN, P300, and P600. For the experimental group, Block 1 = Baseline, Block 2 = Random Feedback 1, Block 3 = Random Feedback 2, Block 4 = Recovery. For the control group, Block 1 = Baseline, Block 2 = Correct Feedback 1, Block 3 = Correct Feedback 2, Block 4 = Correct Feedback 3.
Follow-up simple two-way interactions revealed a significant Condition by Block interaction for the Random Feedback group, F(3,33) = 2.91, p = 0.043, η p 2 = 0.15, but not for the Control group (p = 0.61; Fig. 3B). Polynomial decompositions calculated on the difference waves (i.e., out-of-key minus in-key) as a function of Block in the Random Feedback group revealed a quadratic trend, F(1,17) = 3.79, p = 0.07, η p 2 = 0.18, with the ERAN being largest during the random feedback blocks (−2.6 µV and −1.4 µV, respectively) compared to blocks 1 and 4 (baseline and recovery blocks; −0.8 µV and −0.5 µV, respectively). The topographical distribution of the difference wave exhibits a right-frontal topography (Fig. 4), suggesting that this component has been correctly identified as an ERAN11.
In order to determine if the false feedback rate impacted ERAN amplitude (as was done with accuracy and confidence), the false feedback rate for each participant was correlated with the ERAN amplitude in the random feedback and recovery blocks. ERAN amplitude was not related to the false feedback rate, all p > 0.34, although the ERAN increased in amplitude in response to the application of random feedback. Therefore, despite the global effect of random feedback on ERAN amplitude, the ERAN response was insensitive to inter-subject variation in false feedback.
To investigate whether ERAN amplitude was related to task performance a series of bivariate correlations were calculated between accuracy, confidence, and ERAN amplitude, separately for each group. In the experimental group, ERAN amplitude was related to accuracy during the second random feedback block, r(18) = −0.49, p = 0.04, with increased accuracy being associated with a larger ERAN. No other correlations between ERAN amplitude and accuracy were significant, all p > 0.13. Confidence in detecting an out-of-key note was also related to ERAN amplitude in the second random feedback block, r(18) = 0.47, p = 0.048, with increased confidence being associated with a larger ERAN amplitude. No other correlations between ERAN amplitude and confidence were significant in the experimental group, all p > 0.15. In the control group ERAN amplitude was not related to accuracy nor to confidence in any block, all p > 0.19.
P3
The late positivity was measured as the mean amplitude difference between out-of-key and in-key notes during the 350–700 ms epoch post-stimulus onset, over six parieto-occipital electrodes (CP1, CPz, CP2, P1, Pz, P2). The electrode montages for the ERAN and positivity were chosen based on previous work11,17, and a visual inspection of the overall scalp-topography of the responses. Additionally, we divided the late positivity epoch in half, and separately calculated the mean amplitude for the 350–525 ms epoch and the 525–700 ms epoch. Past research suggests that these two epochs may represent two distinct processing stages. The first epoch might correspond to a traditional P3b, or P300, component representing the orientation of attention to a target sound17, whereas the second may correspond to a P600 component, related to the the attentive process of integrating the incongruous note into the musical context41,42,43,44. Thus we refer to the overall late positivity as the P3, the early epoch of the positivity as the P300, and the late epoch as the P600. Electrode was included as a factor in both analyses, but main effects and interactions with Electrode are not reported because multiple electrode sites were used to obtain a stable estimate of the ERAN and P3 components.
The impact of random feedback on the P300 and P600 was quantified using two separate mixed effects ANOVAs, each with Block and Condition as within-subject factors and Group as a between-subjects factor The topographical distribution of the overall P3 was maximal over midline parietal sites (Fig. 4), suggesting that this component has been correctly identified as a P317.
P300
Overall there was a greater early positivity (P300) for the out-of-key notes compared to the in-key note, F(1,38) = 36.29, p < 0.001, η p 2 = 0.49. No other effects were significant for this comparison. Next, the effect of false feedback was assessed; the amount of false feedback received by a participant was not related to the amplitude of their P300 component in any block, all p > 0.33.
To investigate if P300 amplitude was related to task performance a series of bivariate correlations were calculated between accuracy and P300 amplitude and confidence and P300 amplitude, separately for each group. For the experimental group, P300 amplitude was correlated with accuracy in the baseline block, r(18) = 0.62, p = 0.006, and the first random feedback block, r(18) = 0.48, p = 0.045, with increased accuracy associated with a larger P300. This relationship was not present in the second random feedback block nor the recovery block (p > 0.36). P300 amplitude was correlated with confidence in detecting an out-of-key note during the baseline block, r(18) = 0.47, p = 0.049, with a larger P300 being associated with greater confidence. No other correlations between confidence and P300 amplitude were significant for the experimental group (all p > 0.28). For the control group, increased accuracy was correlated with increased P300 amplitude in the baseline block, r(22) = 0.64, p = 0.001, the first feedback block, r(22) = 0.45, p = 0.034, and the last feedback block, r(22) = 0.53, p = 0.011. P300 amplitude was associated with increased confidence when identifying an in-key melody during the first feedback block, r(22) = 0.46, p = 0.03 and the second feedback block, r(22) = 0.42, p = 0.049. No other relationships between P300 amplitude and confidence were observed for the control group (p > 0.07).
P600
Overall there was a greater late positivity (P600) for the out-of-key notes compared to the in-key note, F(1,38) = 50.82, p < 0.001, η p 2 = 0.57. There was also a marginally significant interaction between Condition, Block, and Group, F(3,114) = 2.47, p < 0.066, η p 2 = 0.06. Follow-up simple two-way interactions revealed a marginally significant Block by Condition interaction in the experimental group F(3,51) = 2.63, p < 0.06, η p 2 = 0.13, but not in the Control group (p = 0.42). Polynomial decompositions calculated on the difference waves (i.e., in-key minus out-of-key) as a function of Block in the experimental group revealed a quadratic trend, F(1,17) = 6.20, p = 0.02, η p 2 = 0.27, with the P600 being smallest during the two random feedback blocks (1.91 µV & 0.95 µV, respectively) compared to the baseline and recovery blocks (3.51 µV & 2.30 µV, respectively; see Fig. 3B). Next, the effect of false feedback was assessed; the amount of false feedback received by a participant was not related to the amplitude of their P600 component in any block, all p > 0.33.
To investigate if P600 amplitude was related to task performance a series of bivariate correlations were calculated between accuracy and P600 amplitude, and confidence and P600 amplitude, separately for each group. For the experimental group, P600 amplitude was correlated with accuracy in the baseline block, r(18) = 0.64, p = 0.004, with higher accuracy being associated with a larger P600. In the first random feedback block this relationship weakened, r(18) = 0.45, p = 0.06, and then disappeared for the second random feedback and recovery blocks, both p > 0.52. P600 amplitude was not correlated with confidence, all p > 0.30. For the control group, P600 amplitude was not related to task performance all p > 0.12.
Although the P600 occurred during the same timeframe as the N1-P2 for the post-target tone, it was unlikely to be related to that response. For more details see Supplementary Note S1.
Awareness of Random Feedback (Experimental Group Only)
When asked in the debriefing survey whether they had noticed strange feedback, only seven of the 18 experimental participants answered in the affirmative. To quantify participants’ awareness of the random feedback, a variable was coded such that 0 corresponded to “unaware” (i.e., those who responded that they were unaware or unsure of whether they had noticed strange feedback), and 1 corresponded to “aware”.
Behaviour
Awareness of random feedback was not significantly related to any other measured demographic variable (participant age, gender, education, musical training), all p > 0.05. To test if awareness of the random feedback impacted accuracy, accuracy data were reanalyzed in the experimental group, using Awareness (aware of random feedback, not aware of random feedback) as a between-subjects factor, and Block as a within-subjects factor. Confidence data were similarly treated, with the addition of Condition (In-Key, Out-Of-Key) as a within-subject variable. There was no effect of Awareness on accuracy, all F < 0.59, all p > 0.45, nor confidence, all F < 1.83, all p > 0.19.
ERPs
To test if awareness of the random feedback impacted the ERPs, the amplitude of the ERAN, P300 and P600 were reanalyzed in the experimental group, using Awareness (aware of random feedback, not aware of random feedback) as a between-subjects factor, and block as a within-subjects factor (Fig. 5).
The ERAN was larger in participants who were aware of the random feedback, F(1,16) = 8.22, p = 0.011, η p 2 = 0.34. This effect was consistent across all blocks as the Awareness by Block interaction was not significant (p = 0.35). In order to determine if the impact of random feedback on the ERAN (see above) was driven by awareness of the feedback, we examined the impact of Block, Condition, and Group. For this analysis, the Group factor compared the control group with experimental group participants who were unaware of random feedback. The interaction between Block and Group was not significant, F(3,93) = 2.01, p = 0.12, η p 2 = 0.06. Note that the size of the Group effect for this analysis was similar to the size of the Group effect observed in the omnibus analysis that included all participants (η p 2 = 0.06 vs. 0.07). The stable effect size, but non-significant interaction suggests that this follow-up analysis was underpowered, due to the nearly half the experimental participants being excluded from this analysis. Given that there was a differential impact of Block on the ERAN on the experimental group as a whole (and the trend was identical when only aware participants were included), it is likely that the impact of random feedback on the ERAN was due to the feedback and not awareness of the feedback.
For the P300, neither the main effect of Awareness nor its interaction with Block was significant (p = 0.43 & 0.11, respectively). For the P600, the main effect of Awareness was not significant (p = 0.79), nor was its interaction with Block, F(1,16) = 2.43, p = 0.08, η p 2 = 0.13.
Discussion
Random feedback disturbs the musical judgments of ordinary listeners. The presentation of random feedback lowered participants’ accuracy and confidence on a tonal judgment task and decreased the amplitude of the P600 brain response to tonality violations. In contrast, the amplitude of the ERAN increased during random feedback. When correct feedback was finally provided in the final block, behaviour and brain measures recovered toward baseline levels. These changes in behaviour and brain activity were not observed in the control group, who received correct feedback. Thus, the detrimental experimental effects could not be due to the simple occurrence of feedback, and rather can be attributed to the disrupting effects of random feedback specifically.
Previous studies investigating the relation between feedback and performance have predominantly focused on the behavioural effects of (accurate) feedback being present or absent, mostly in the visual modality45. The current study extends the limited literature on the effects of random or false feedback on performance from the domain of visual perceptual learning46,47 to auditory perception, and confirms previous findings in vision with respect to its disruptive behavioural effects. Furthermore, this study constitutes the first experimental induction of the amusic phenotype in a normal brain. The observed decrease in accuracy and suppression of the P600 mimic the signature of amusia in the current task22.
Exposure to random feedback likely increased participants’ attention to the task, which may have activated explicit attempts to access tonal knowledge. Some authors have argued that ERAN amplitude increases with task-directed attention48, whereas others have shown that the ERAN is relatively stable, and only changes under very specific manipulations of attention13,14,15,16. The increase in ERAN amplitude observed in the experimental group might be linked to enhanced attention due to overlap with the N1. N1 amplitude is normally enhanced when attention is directed to the stimulus evoking the N149,50. The random feedback can be seen as introducing noise into participants’ conscious access to tonal knowledge, as demonstrated by declining task performance and suppression of the P600 brain response. The finding that ERAN amplitude continued to increase across both random feedback blocks in participants who were aware of the feedback manipulation supports this idea. These participants likely experienced the most conflict between their tonal knowledge and the feedback, and the ERAN amplitude increase may indicate the strategic drawing of attention to the task in order to amplify conscious access to tonal knowledge.
In contrast, participants who were unaware of the feedback manipulation showed a suppression of this component. This converges with a recent study in which amusics showed an inhibited ERAN in participants with amusia22. The decrease in ERAN amplitude for unaware participants may reflect a drawing of attention away from the target tone, due to the random feedback compromising their understanding of the task and therefore their ability to focus on the critical event (the target tone). Importantly, the lack of differences in behaviour between aware and non-aware participants in the experimental group indicates that the noise introduced by random feedback effectively decreased conscious access to tonal knowledge, regardless of the differing strategies used by each group to cope with that noise (as indexed by the differing impact on the ERAN).
Future work will continue to explore this hypothesis by experimentally manipulating awareness of the false feedback manipulation, by titrating the exact amount of false feedback received by participants. These experiments will increase the power to observe graded effects of false feedback over time, which may have gone undetected in the current study due to low inter-subject variability in the percentage of false feedback trials received.
In sum, we successfully simulated the amusic profile using random feedback in typical brains as an experimental model for the conscious access deficit in amusia. The use of this transient experimental model can be compared, for instance, to the use of transcranial magnetic stimulation (TMS) to temporarily “induce” brain lesions51. Our successful modeling of the amusic phenotype using false feedback suggests a causal link between unreliable feedback, conscious access to tonal knowledge, and the amusic disorder.
Reliable feedback is a powerful tool for learning. Many studies have shown that learning is more effective when participants attend to (accurate) external feedback52. The current results raise the possibility that amusia results from uncertainty due to inappropriate feedback. Since external feedback rarely occurs in ordinary musical activities, the faulty mechanism is more likely to be internal and to arise from the malfunctioning communication between implicitly learned (bottom-up) tonal knowledge and conscious (top-down) access to that intact knowledge7. The predictive coding framework provides a useful model for this process.
The predictive coding model sees the brain as a hierarchically organized system in which each level strives to attain an optimal balance between bottom-up sensory information and top-down predictions53. Electrophysiological data suggests that this balance is disrupted in amusia, as amusic brains display a normal bottom-up response to tonal deviants (i.e., ERAN), but lack the typical top-down response required to consciously detect those deviants (i.e., P600)6,22. In the current non-amusic participants, the enhanced ERAN observed after the administration of random feedback likely represents an amplification of top-down predictive coding. This response could be a neural attempt to amplify the perception of the tonal deviant in order to resolve the anticipated conflict with the potentially false upcoming feedback. In other words, because the external feedback has been inconsistent, the brain attempts to amplify the bottom-up perceptual input to aid in making accurate tonal predictions. Interestingly, the P600, which represents conscious access to the tonal deviant, decreased in amplitude most in the second block of random feedback. This suggests that after abandoning the amplification of the sensory input, the brain recalibrates in order to use the feedback to learn a new (tonal) system. Importantly, this response parallels the behavioural and brain responses observed in amusia.
One clear implication of the current study is that (correct) feedback may be a useful rehabilitative strategy for amusia, as well as other learning disorders, such as dyslexia and prosopagnosia. Indeed, all three learning disorders seem to result from deficient conscious access to intact implicit knowledge. A recent neuroimaging study reveals that adult dyslexics have intact but less accessible phonological representations of speech54. Similarly, adults who suffer from prosopagnosia show covert recognition of faces in the absence of overt recognition55. Like in amusia, deficient neural communication between sensory and frontal cortices seems to underlie lack of conscious access in dyslexia and prosopagnosia.
However, the case of dyslexia suggests that external feedback, as provided by explicit instruction, is not enough to correct the deficient feedback loop underlying the disorder. Neurofeedback56 might be a better strategy. Neurofeedback training is a form of conditioning in which brain activity is rewarded or repressed without requiring conscious access to knowledge. Recent research has successfully applied neurofeedback to children with dyslexia57. Future studies of the neural communication between temporal and frontal cortices will help to understand the neurophysiological (and neuroplastic) mechanism responsible for conscious access to knowledge in learning disorders. The manipulation of feedback in a normal brain provides a complementary experimental model to test theories of learning disorders.
References
Collins, T., Tillmann, B., Barrett, F. S., Delbé, C. & Janata, P. A combined model of sensory and cognitive representations underlying tonal expectations in music: From audio signals to behavior. Psychol. Rev. 121, 33 (2014).
Tillmann, B., Bharucha, J. J. & Bigand, E. Implicit learning of tonality: A self-organizing approach. Psychol. Rev. 107, 885–913 (2000).
Cuddy, L. L. & Badertscher, B. Recovery of the tonal hierarchy: Some comparisons across age and levels of musical experience. Percept. Psychophys. 41, 609–620 (1987).
Peretz, I. Brain specialization for music. Ann. N. Y. Acad. Sci. 930, 153–165 (2001).
Loui, P., Guenther, F. H., Mathys, C. & Schlaug, G. Action–perception mismatch in tone-deafness. Curr. Biol. 18, R331–R332 (2008).
Peretz, I., Brattico, E., Järvenpää, M. & Tervaniemi, M. The amusic brain: In tune, out of key, and unaware. Brain 132, 1277–1286 (2009).
Peretz, I. Neurobiology of Congenital Amusia. Trends Cogn. Sci. 20, 857–867 (2016).
Loui, P., Alsop, D. & Schlaug, G. Tone deafness: A new disconnection syndrome? J. Neurosci. 29, 10215–10220 (2009).
Tillmann, B., Gosselin, N., Bigand, E. & Peretz, I. Priming paradigm reveals harmonic structure processing in congenital amusia. Cortex 48, 1073–1078 (2012).
Omigie, D., Pearce, M. T., Williamson, V. J. & Stewart, L. Electrophysiological correlates of melodic processing in congenital amusia. Neuropsychologia 51, 1749–1762 (2013).
Koelsch, S. Toward a neural basis of music perception- A review and updated model. Front. Psychol. 2, 110 (2011).
Koelsch, S., Rohrmeier, M., Torrecuso, R. & Jentschke, S. Processing of hierarchical syntactic structure in music. Proc. Natl. Acad. Sci. 110, 15443–15448 (2013).
Guo, S. & Koelsch, S. The effects of supervised learning on event-related potential correlates of music-syntactic processing. Brain Res. 1626, 232–246 (2015).
Maidhof, C. & Koelsch, S. Effects of selective attention on syntax processing in music and language. J. Cogn. Neurosci. 23, 2252–2267 (2011).
Sammler, D. et al. Co-localizing linguistic and musical syntax with intracranial EEG. Neuroimage 64, 134–146 (2013).
Koelsch, S. Neural Correlates of Music Perception. In Language, Music, and the Brain: A Mysterious Relationship 141–172 (MIT Press, 2013).
Polich, J. Updating P300: an integrative theory of P3a and P3b. Clin. Neurophysiol. 118, 2128–2148 (2007).
Albouy, P. et al. Impaired pitch perception and memory in congenital amusia: The deficit starts in the auditory cortex. Brain 136, 1639–1661 (2013).
Moreau, P., Jolicœur, P. & Peretz, I. Pitch discrimination without awareness in congenital amusia: evidence from event-related potentials. Brain Cogn. 81, 337–344 (2013).
Moreau, P., Jolicoeur, P. & Peretz, I. Automatic brain responses to pitch changes in congenital amusia. Ann. N. Y. Acad. Sci. 1169, 191–194 (2009).
Peretz, I., Brattico, E. & Tervaniemi, M. Abnormal electrical brain responses to pitch in congenital amusia. Ann. Neurol. 58, 478–482 (2005).
Zendel, B. R., Lagrois, M.-É., Robitaille, N. & Peretz, I. Attending to pitch information inhibits processing of pitch information: The curious case of amusia. J. Neurosci. 35, 3815–3824 (2015).
Hyde, K. L. et al. Cortical thickness in congenital amusia: When less is better than more. J. Neurosci. 27, 13028–13032 (2007).
Hyde, K. L., Zatorre, R. J. & Peretz, I. Functional MRI evidence of an abnormal neural network for pitch processing in congenital amusia. Cereb. Cortex 21, 292–299 (2011).
Peretz, I., Champod, A. S. & Hyde, K. Varieties of musical disorders. Ann. N. Y. Acad. Sci. 999, 58–75 (2003).
Kouider, S., Dehaene, S., Jobert, A. & Le Bihan, D. Cerebral bases of subliminal and supraliminal priming during reading. Cereb. Cortex 17, 2019–2029 (2007).
Marois, R., Yi, D.-J. & Chun, M. M. The neural fate of consciously perceived and missed events in the attentional blink. Neuron 41, 465–472 (2004).
McIntosh, A. R., Rajah, M. N. & Lobaugh, N. J. Interactions of prefrontal cortex in relation to awareness in sensory learning. Science 284, 1531–1533 (1999).
Stephan, K. M. et al. Conscious and subconscious sensorimotor synchronization—Prefrontal cortex and the influence of awareness. Neuroimage 15, 345–352 (2002).
Boly, M. et al. Preserved feedforward but impaired top-down processes in the vegetative state. Science 332, 858–862 (2011).
Stuss, D. T., Picton, T. W. & Alexander, M. P. Consciousness, self-awareness and the frontal lobes. In The frontal lobes and neuropsychiatric illness (eds Salloway, S. P., Malloy, P. F. & Duffy, J. D.) 101–109 (American Psychiatric Publishing, 2001).
Hyde, K. L., Zatorre, R. J., Griffiths, T. D., Lerch, J. P. & Peretz, I. Morphometry of the amusic brain: A two-site study. Brain 129, 2562–2570 (2006).
Maess, B., Koelsch, S., Gunter, T. C. & Friederici, A. D. Musical syntax is processed in Broca’s area: An MEG study. Nat. Neurosci. 4, 540–545 (2001).
Janata, P. et al. The cortical topography of tonal structures underlying western music. Science (80−.). 298, 2167–2170 (2002).
Norman-Haignere, S. V. et al. Pitch-Responsive Cortical Regions in Congenital Amusia. J. Neurosci. 36, 2986–94 (2016).
Peretz, I. & Vuvan, D. T. Prevalence of congenital amusia. Eur. J. Hum. Genet.
Peretz, I. et al. On-line identification of congenital amusia. Music Percept. An Interdiscip. J. 25, 331–343 (2008).
Brainard, D. H. The psychophysics toolbox. Spat. Vis. 10, 433–436 (1997).
Kleiner, M., Brainard, D. H. & Pelli, D. What’s new in Psychtoolbox-3? Perception 36, ECVP Abstract Supplement (2007).
Berg, P. & Scherg, M. A multiple source approach to the correction of eye artifacts. Electroencephalogr. Clin. Neurophysiol. 90, 229–241 (1994).
Besson, M. & Faïta, F. An event-related potential (ERP) study of musical expectancy: Comparison of musicians with nonmusicians. J. Exp. Psychol. Hum. Percept. Perform. 21, 1278 (1995).
Brattico, E., Tervaniemi, M., Näätänen, R. & Peretz, I. Musical scale properties are automatically processed in the human auditory cortex. Brain Res. 1117, 162–174 (2006).
Koelsch, S. Neural substrates of processing syntax and semantics in music. Curr. Opin. Neurobiol. 15, 207–212 (2005).
Patel, A., Gibson, E., Ratner, J., Besson, M. & Holcomb, P. Processing syntactic relations in language and music: An event-related potential study. J. Cogn. Neurosci. 10, 717–733 (1998).
Seitz, A. R., Nanez, J. E., Holloway, S., Tsushima, Y. & Watanabe, T. Two cases requiring external reinforcement in perceptual learning. J. Vis. 6, 966–973 (2006).
Herzog, M. H. & Fahle, M. The role of feedback in learning a vernier discrimination task. Vision Res. 37, 2133–2141 (1997).
Shibata, K., Yamagishi, N., Ishii, S. & Kawato, M. Boosting perceptual learning by fake feedback. Vision Res. 49, 2574–2585 (2009).
Loui, P., Grent-’t-Jong, D., Torpey, D. & Woldorff, M. Effects of attention on the neural processing of harmonic syntax in Western music. Cogn. Brain Res. 25, 678–687 (2005).
Hansen, J. C. & Hillyard, S. A. Endogeneous brain potentials associated with selective auditory attention. Electroencephalogr. Clin. Neurophysiol. 49, 277–290 (1980).
Näätänen, R., Gaillard, A. W. K. & Mäntysalo, S. Early selective-attention effect on evoked potential reinterpreted. Acta Psychol. 42, 313–329 (1978).
Royal, I., Zendel, B. R., Desjardins, M.-È., Robitaille, N. & Peretz, I. Modulation of electric brain responses evoked by pitch deviants through transcranial direct current stimulation. Neuropsychologia 109, 63–74 (2018).
Bangert-Drowns, R. L., Kulik, C.-L. C., Kulik, J. A. & Morgan, M. The instructional effect of feedback in test-like events. Rev. Educ. Res. 61, 213–238 (1991).
Garrido, M. I., Kilner, J. M., Stephan, K. E. & Friston, K. J. The mismatch negativity: A review of underlying mechanisms. Clin. Neurophysiol. 120, 453–463 (2009).
Boets, B. et al. Intact but less accessible phonetic representations in adults with dyslexia. Science 342, 1251–1254 (2013).
Eimer, M., Gosling, A. & Duchaine, B. Electrophysiological markers of covert face recognition in developmental prosopagnosia. Brain 135, 542–554 (2012).
Florin, E., Bock, E. & Baillet, S. Targeted reinforcement of neural oscillatory activity with real-time neuroimaging feedback. Neuroimage 88, 54–60 (2014).
Nazari, M. A., Mosanezhad, E., Hashemi, T. & Jahan, A. The effectiveness of neurofeedback training on EEG coherence and neuropsychological functions in children with reading disability. Clin. EEG Neurosci. 43, 315–322 (2012).
Acknowledgements
This research was supported by grants from the Fonds de Recherche du Québec - Nature et Technologies, the Fonds de Recherche du Québec - Santé, and the Canadian Institutes for Health Research. The authors would like to thank Natalie Harvey and Mihaela Felezeu for their help with data collection, and Marion Cousineau for her insightful comments on the manuscript.
Author information
Authors and Affiliations
Contributions
D.T.V. and I.P. designed the study. D.T.V. and B.R.Z. analyzed the data, created figures, and wrote the main manuscript text. All three authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vuvan, D.T., Zendel, B.R. & Peretz, I. Random Feedback Makes Listeners Tone-Deaf. Sci Rep 8, 7283 (2018). https://doi.org/10.1038/s41598-018-25518-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-25518-1
This article is cited by
-
Electrophysiological and behavioral indicators of musical knowledge about unfamiliar music
Scientific Reports (2022)
-
Improvisation is a novel tool to study musicality
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.