Abstract
Sarcoidosis commonly affects the lung. Lung transplantation (LT) is required when there is a severe and refractory involvement. We compared post-transplant survival rates of sarcoidosis patients with chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF). We also explored whether the race and age of the donor, and double lung transplant have any effect on the survival in the post transplant setting. We analyzed 9,727 adult patients with sarcoidosis, COPD, and IPF who underwent LT worldwide between 2005–2015 based on United Network for Organ Sharing (UNOS) database. Survival rates were compared with Kaplan-Meier, and risk factors were investigated by Cox-regression analysis. 469 (5%) were transplanted because of sarcoidosis, 3,688 (38%) for COPD and 5,570 (57%) for IPF. Unadjusted survival analysis showed a better post-transplant survival rate for patients with sarcoidosis (p < 0.001, Log-rank test). In Cox-regression analysis, double lung transplant and white race of the lung donor showed to have a significant survival advantage. Since double lung transplant, those who are younger and have lower Lung Allocation Score (LAS) at the time of transplant have a survival advantage, we suggest double lung transplant as the procedure of choice, especially in younger sarcoidosis subjects and with lower LAS scores.
Similar content being viewed by others
Introduction
Sarcoidosis is a multiorgan granulomatous disorder of unknown etiology associated with significant morbidity and mortality. The lung is the organ most commonly involved and the disease is characterized by frequent pulmonary exacerbations and remissions, particularly within the first 2 to 5 years of the diagnosis1,2.
Sarcoidosis can affect all age groups but is more frequent in middle age populations, and women are two times more likely to have the disease3. There is a known difference in prevalence according to race, with Caucasians having a 0.85% lifetime risk of developing sarcoidosis, while this risk for African Americans is 2.5%4. Racial differences in disease severity are also observed. African Americans have a more acute and severe presentation in contrast to a typically slow insidious process in European Americans5.
Medical treatment of symptomatic sarcoidosis is based on immunomodulation. Corticosteroids are usually the first line of therapy, but other medications including methotrexate, leflunomide, infliximab and other anti-TNF agents are commonly used6. The selection of the best treatment option is based on the degree of lung involvement, ranging from mild interstitial pulmonary disease to severe disabling pulmonary fibrosis and pulmonary hypertension3. Eligible patients with severe lung involvement, refractory to medical therapy, are referred for lung transplantation (LT).
Since the first successful lung transplant was performed in 1983, thousands procedure have been performed worldwide7. Lung transplantation has been recommended in patients with advanced chronic obstructive pulmonary disease (COPD), bronchiectasis, cystic fibrosis (CF), primary pulmonary hypertension, idiopathic pulmonary fibrosis (IPF), sarcoidosis, and other end stage interstitial lung diseases8. Advanced COPD and IPF are the most common indications for lung transplants worldwide7. Advances in lung transplant techniques, post-transplant patient care, and immunosuppressive therapies during the last 30 years have significantly improved survival9. The median survival of adults who undergo lung transplantation is reported to be 5.6 years, with an unadjusted survival rate of 53% at 5 years, and 31% at 10 years10. The highest post-lung transplant survival rate per specific disease occurs in CF patients, with a 67% survival rate at 5 years11. Meanwhile 5 year survival is reported to be 45% for COPD and 44% for IPF patients12. There are limited information regarding survival rates following LT in patients with sarcoidosis13. Taimeh et al. recently found no difference in lung transplant outcomes between sarcoidosis and non-sarcoidosis lung transplant recipients in the last 30 years14. However, given the significant improvement in LT outcome in the last 10 years, the sarcoidosis survival after LT may have also been improved. Moreover, information on the factors that influence on sarcoidosis survival in lung transplantation are still unclear. The aim of this study was to compare the post-transplant survival rates of sarcoidosis patients with the two most common indications for LT: COPD and IPF. We also explore whether some independent factors, such as race of the donor, age of the donor and double lung transplant have any effect on the survival of sarcoidosis patients in the post transplant setting.
Materials and Methods
We analyzed adult sarcoidosis subjects who underwent LT in the United States from December 30th, 2005, to December 30th, 2015 based on the United Network for Organ Sharing (UNOS)/OPTN standard transplant analysis and research files database. All methods were conducted in accordance with pertinent guidelines and regulations. We used open resource de-identified national dataset, therefore no Institutional Review Board (IRB) or informed consent was required. From the same dataset, all subjects with diagnosis of COPD and IPF were selected for survival rate comparisons. The variables of interest were age, sex, race, body mass index (BMI), and outcomes including death and re-transplantation.
A log-rank test was utilized to determine if there were any statistically significant differences in survival between the three groups of interest (p < 0.05) and Kaplan-Meier curves were plotted to depict ordinality of any found differences. A cox regression model was performed to find if variables including age of transplant recipient, age of donor, race of donor, double lung transplant, Lung Allocation Score (LAS), cold ischemic time (time of removal of the organ from the donor to the time the organ is transplanted into the recipient), mechanical ventilation, and male gender are independently associated with mortality in sarcoidosis subjects. We developed multivariate analysis model based on variables which have previously been used by Taimeh’s et al.14. Factors were selected based on clinically important variables which was found in previous study. Missing data in the 10-year dataset was also studied, and collinearity between covariates was also explored. A second cox regression model was performed only in the overall black population to determine if the race of the donor had a persistent effect on the outcome. Moreover, a separate cox regression model was performed in non-white patients. Adjusted hazard ratio (HR) and 95% confidence interval (CI) were estimated. All analyses were performed in R (version 3.2.2) and the Survival package (version 2.38-3) and results were considered significant at P < 0.05.
Results
A total of 9727 patients with sarcoidosis, COPD and IPF underwent LT from 2005 to 2015. Of these, 469 (4.8%) were transplanted because of advanced sarcoidosis, 3688 (37.9%) for COPD and 5570 (57.3%) for IPF.
Table 1 summarizes the demographic characteristics of transplanted subjects with sarcoidosis, COPD, and IPF. Age at time of transplantation for sarcoidosis was 52.91 ± 8.29 years, showing statistical difference with that of COPD (60.92 ± 6.20 years; p = < 0.0001) or IPF (60.91 ± 8.60 years; p = < 0.0001). About half of the sarcoidosis subjects were female, similar to what was observed in COPD but not in IPF, where an overall significant difference was observed (p = < 0.0001), as most IPF subjects were male. In the sarcoidosis group, 64% (302) of the subjects were African American while 90% (3346) and 82% (4582) were European American in the COPD and IPF group, respectively. Finally, lung re-transplant rates were relatively low but statistically significantly higher in patients with IPF.
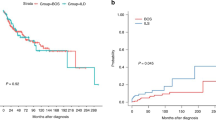
The survival analysis in the three groups showed a better survival rate for patients with sarcoidosis compared with patients with COPD and IPF (p < 0.001, log-rank test). Figure 1 displays the significant post-transplant survival difference between sarcoidosis and the COPD and IPF groups. In Cox-regression analysis, double lung transplant and white race of the lung donor showed to have a significant survival advantage, with an HR of 0.83 (95%CI: 0.77–0.90) and 0.86 (95%CI: 0.81–0.92), respectively. Figure 2 illustrates a plot of the estimates and the 95% confidence intervals obtained from Cox regression analysis representing independent factors associated with mortality in patients with sarcoidosis who underwent lung transplantation. Moreover, higher age of both lung- recipient and -donor plus higher LAS were independently associated with increased risk of mortality among the sarcoidosis group. Figure 3 represents Kaplan-Meier curves where a significant post-transplant survival difference is appreciated in the double lung transplant group (p < 0.001, log-rank test).
A second Cox-regression model was made only in African American subjects to explore if the Caucasian donor effect persisted. In this model we couldn’t find any significant differences concerning the impact of the race of the donor on the survival rate. Moreover, as illustrated in Table 2, male donor and higher age of the donor were associated with a negative outcome in this population.
A third Cox-regression model was made in the non-white population. We couldn’t find any impact concerning the race of the donor regarding survival. Significant statistical differences were found in recipients and donors with higher age, being associated with poor outcome, as showed in Table 3. It is important to mention that LAS score was not longer significantly associated anymore. This may suggest that the scoring system needs further investigation for its effectiveness to rank non-white patients, including African Americans.
Collinearity
Despite age being a component of LAS score, no strong collinearities were detected between these variables that would impede the estimation of this model, as shown in Fig. 4.
Handling Missing data
Table 4 displays the missing data in the 10-year dataset. Of note, the highest rate of missing for any single variable was about 3% out of approximately 10,000 observations in the 10-year dataset. This suggests that a very complete data was utilized in the study.
Discussion
This 10-year lung-transplant database review of patients with a primary diagnosis of pulmonary sarcoidosis in the US shows there is a higher number of females and African American patients who underwent lung transplant due to sarcoidosis compared to IPF. There are also a higher number of African Americans in the sarcoidosis group in contrast to the COPD group. This study demonstrates that sarcoidosis subjects had a significantly higher survival rate compare to patients with COPD and IPF in an adjusted survival analysis.
Sarcoidosis comprised only a 4.8% of subjects when compared to other transplant indications, such as COPD and IPF in the last 10 years. It has not been changed in the last 30 years14. The high prevalence of COPD and higher severity of pulmonary fibrosis in IPF may explain such a high proportion of LT in the US. The prevalence of sarcoidosis in the United States is estimated at 1 to 40 per 100,000 people15, while COPD is reported to occur in as high as 15.2% of the US population16. The IPF prevalence is less than sarcoidosis (8.2 cases per 100, 000 people in 2010) in the US17, However, IPF causes severe pulmonary fibrosis and advanced lung disease in almost all subjects, while fibrotic lung disease develops in up to 20% of patients with sarcoidosis18.
The significant higher prevalence of females and African Americans in the sarcoidosis group highlights the gender and race disparities. Females and African Americans are more susceptible to sarcoidosis for still unknown reasons. African Americans also suffer from higher severity of disease and have a higher mortality rate19. Recently, we reported that the overall mortality rate of sarcoidosis was eight times higher in African Americans than in European Americans5.
In contrast with our study, Taimeh et al. could not demonstrate any association between donor age and race, and survival in the sarcoidosis cohort14. We can hardly compare these studies together for several reasons. We analyzed the outcomes of lung transplant in the last 10 years to achieve the current status. We also aimed to compare sarcoidosis patients who underwent lung transplantation with the two most common causes for lung transplant: COPD and IPF. Taimeh et al. analyzed a 30-year cohort for their analysis, comparing sarcoidosis to all non-sarcoidosis subjects. In the last 30 years, significant improvement in outcome of lung transplant has been achieved including surgical techniques, post-operative care and immunosuppressive therapy. Those factors may affect the outcome results between two studies. We considered this may be a major driven factor to see an association between donor age and race with a better outcome in sarcoidosis subjects in the current study.
Our observation of 53% survival of sarcoidosis subjects at 10 years post-lung transplant is better than previously report by Walker et al.20 that was 56% at 5 years. Interestingly, sarcoidosis subjects had a better unadjusted survival compared to COPD and IPF subjects in Kaplan–Meier analysis. The Cox regression analysis revealed that better survival in sarcoidosis subjects was secondary to their younger age, higher number of double lung transplant and receiving lungs from European-American donors. Sarcoidosis patients were on average 8 years younger than their COPD and IPF counterparts, and this was an independent risk factor in the adjusted survival analysis. The effect of age on lung transplant outcome has been studied before. Gutierrez and colleagues showed that subjects with advanced age who underwent lung transplant have an increased mortality rate even after adjusting for expected advanced age-related mortality21. Our study also showed that age and race of donors are independently associated with post transplant survival in sarcoidosis subjects. Sommer and colleagues could not find significant difference in 1-year survival rate when using of donor lungs of aged population (≥70 years old). But, noted significantly lower forced expiratory in 1 second values in subjects with lung transplantation of aged donor lungs in IPF recipients22. Obviously, our 10 years follow up study has higher power to show survival effect of aged donor lungs on post transplantation survival.
Recipients of lungs from Caucasian donors had better survival. We speculate that socioeconomic factors influence on this observation. To our knowledge, no other studies have reported a survival advantage related to race and ethnicity of donor lungs. However, studies in kidney transplant patients have found that recipients of African American kidney donors had lower graft survival23. Further investigation is needed to determine the nature of the relationship between donor and recipient race and survival in LT.
Our analysis showed that higher lung allocation scores are associated with worse post-LT outcomes in sarcoidosis. LAS has been universally used to prioritize LT candidates by balancing expected post-transplant survival and predicted waitlist mortality24. According to our analyses, sarcoidosis patients with a lower LAS at the time of transplant should have a higher post-LT survival. Therefore, a new scoring system should be developed to determine the most appropriate time for lung transplantation for patients with advanced refractory sarcoidosis.
Double lung transplant was associated with higher survival in our study. It was performed in higher percentage in sarcoidosis subjects compared to IPF and COPD, perhaps due to severe bilateral lung involvement, presence of bronchiectasis, and pulmonary hypertension. Our study suggests that double lung transplantation should be the procedure of choice in sarcoidosis patients.
Sarcoidosis recurrence after lung transplantation is an important topic. We reported no significant increasing risk for re-transplantation among sarcoidosis subjects when compared to others. The rate of re-onset of sarcoidosis after lung transplant was reported by Banga A et al. previously25. They found granulomatous reaction recurrence in 7 (23%) out of 30 sarcoidosis subjects who undergone lung transplant in Cleveland Clinic, Ohio, USA. However, they found no impact of granulomas recurrence on overall outcomes. Schultz and colleagues also reported that 7 (28%) of 25 subjects who undergone lung transplant for sarcoidosis in Copenhagen, Denmark showed recurrence of granulomas in new lungs with no impact of outcomes26. UNOS does not provide information concerning recurrence of sarcoidosis in post-transplanted patients. Therefore, we would not able to analyze rate of sarcoidosis recurrence after lung transplantation. This topic requires further investigation.
This study consists of a big cohort comprised of 9,727 patients that underwent lung transplant because of sarcoidosis, COPD and IPF, in a period of 10 years. To our knowledge, this is the first time that the role of the ethnicity of the donor was analyzed as an independent factor for the outcome in lung transplantation.
Limitations
Despite the use of the largest available database for lung transplantation, our study has several limitations. We are unable to analyze the impact of many other variables that play a role in post lung transplant survival including nutritional status, psychological factors, adherence to medications, pre existent pulmonary arterial hypertension (PAH), spirometric values including total lung capacity (TLC), and other co-morbidities. Even though PAH may be a potential cofounder in post lung transplant outcome, we had no access to PAH data of subjects from UNOS database. Moreover, only a total of 912 subjects were African-American among 9,727 patients (9.4%). This represents an important limitation because we were not able to analyze the correlation between African-American recipients undergoing lung transplant from African-American donors. A more powered sample size is required to further investigate the impact of other ethnicities in the outcome of lung transplant.
Sarcoidosis patients have a reasonable survival rate and re-transplant rate that are comparable with other common LT indications such as COPD and IPF. Given that double lung transplant is associated with better survival in sarcoidosis subjects, and those who are younger and have a lower LAS score at the time of transplant have a survival advantage, we would suggest double lung transplant as the procedure of choice, especially in younger sarcoidosis subjects and preferably those with lower LAS scores.
References
Mirsaeidi, M. et al. Plasma metabolomic profile in fibrosing pulmonary sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis 33, 29–38 (2016).
Mortaz, E. et al. Association of serum TNF-alpha, IL-8 and free light chain with HLA-DR B alleles expression in pulmonary and extra-pulmonary sarcoidosis. J Inflamm (Lond) 12, 21, https://doi.org/10.1186/s12950-015-0066-3 (2015).
Newman, L. S., Rose, C. S. & Maier, L. A. Sarcoidosis. N Engl J Med 336, 1224–1234, https://doi.org/10.1056/NEJM199704243361706 (1997).
Baughman, R. P. et al. Sarcoidosis in America. Analysis Based on Health Care Use. Ann Am Thorac Soc 13, 1244–1252, https://doi.org/10.1513/AnnalsATS.201511-760OC (2016).
Mirsaeidi, M., Machado, R. F., Schraufnagel, D., Sweiss, N. J. & Baughman, R. P. Racial difference in sarcoidosis mortality in the United States. Chest 147, 438–449, https://doi.org/10.1378/chest.14-1120 (2015).
Korsten, P., Mirsaeidi, M. & Sweiss, N. J. Nonsteroidal therapy of sarcoidosis. Current opinion in pulmonary medicine 19, 516–523, https://doi.org/10.1097/MCP.0b013e3283642ad0 (2013).
Yusen, R. D. et al. The Registry of the International Society for Heart and Lung Transplantation: Thirty-second Official Adult Lung and Heart-Lung Transplantation Report–2015; Focus Theme: Early Graft Failure. J Heart Lung Transplant 34, 1264–1277, https://doi.org/10.1016/j.healun.2015.08.014 (2015).
Orens, J. B. et al. International guidelines for the selection of lung transplant candidates: 2006 update–a consensus report from the Pulmonary Scientific Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 25, 745–755, https://doi.org/10.1016/j.healun.2006.03.011 (2006).
Watson, C. J. & Dark, J. H. Organ transplantation: historical perspective and current practice. Br J Anaesth 108(Suppl 1), i29–42, https://doi.org/10.1093/bja/aer384 (2012).
Puri, V., Patterson, G. A. & Meyers, B. F. Single versus bilateral lung transplantation: do guidelines exist? Thorac Surg Clin 25, 47–54, https://doi.org/10.1016/j.thorsurg.2014.09.007 (2015).
Stephenson, A. L. et al. Clinical and demographic factors associated with post-lung transplantation survival in individuals with cystic fibrosis. J Heart Lung Transplant 34, 1139–1145, https://doi.org/10.1016/j.healun.2015.05.003 (2015).
Titman, A., Rogers, C. A., Bonser, R. S., Banner, N. R. & Sharples, L. D. Disease-specific survival benefit of lung transplantation in adults: a national cohort study. Am J Transplant 9, 1640–1649, https://doi.org/10.1111/j.1600-6143.2009.02613.x (2009).
Nathan, S. D. Lung transplantation: disease-specific considerations for referral. Chest 127, 1006–1016, https://doi.org/10.1378/chest.127.3.1006 (2005).
Taimeh, Z., Hertz, M. I., Shumway, S. & Pritzker, M. Lung transplantation for pulmonary sarcoidosis. Twenty-five years of experience in the USA. Thorax 71, 378–379, https://doi.org/10.1136/thoraxjnl-2015-207497 (2016).
Erdal, B. S., Clymer, B. D., Yildiz, V. O., Julian, M. W. & Crouser, E. D. Unexpectedly high prevalence of sarcoidosis in a representative U.S. Metropolitan population. Respir Med 106, 893–899, https://doi.org/10.1016/j.rmed.2012.02.007 (2012).
Adeloye, D. et al. Global and regional estimates of COPD prevalence: Systematic review and meta-analysis. J Glob Health 5, 020415, https://doi.org/10.7189/jogh.05-020415 (2015).
Raghu, G., Chen, S. Y., Hou, Q. & Yeh, W. S. & Collard, H. R. Incidence and prevalence of idiopathic pulmonary fibrosis in US adults 18-64 years old. Eur Respir J 48, 179–186, https://doi.org/10.1183/13993003.01653-2015 (2016).
Patterson, K. C. & Strek, M. E. Pulmonary fibrosis in sarcoidosis. Clinical features and outcomes. Ann Am Thorac Soc 10, 362–370, https://doi.org/10.1513/AnnalsATS.201303-069FR (2013).
Sartwell, P. E. & Edwards, L. B. Epidemiology of sarcoidosis in the U.S. Navy. Am J Epidemiol 99, 250–257 (1974).
Walker, S. et al. Medium term results of lung transplantation for end stage pulmonary sarcoidosis. Thorax 53, 281–284 (1998).
Gutierrez, C. et al. The effect of recipient’s age on lung transplant outcome. Am J Transplant 7, 1271–1277, https://doi.org/10.1111/j.1600-6143.2006.01681.x (2007).
Sommer, W. et al. Survival and spirometry outcomes after lung transplantation from donors aged 70 years and older. J Heart Lung Transplant 34, 1325–1333, https://doi.org/10.1016/j.healun.2015.06.002 (2015).
Callender, C. O. et al. Effect of donor ethnicity on kidney survival in different recipient pairs: an analysis of the OPTN/UNOS database. Transplant Proc 41, 4125–4130, https://doi.org/10.1016/j.transproceed.2009.06.182 (2009).
Egan, T. M. et al. Development of the new lung allocation system in the United States. Am J Transplant 6, 1212–1227, https://doi.org/10.1111/j.1600-6143.2006.01276.x (2006).
Banga, A., Sahoo, D., Lane, C. R., Farver, C. F. & Budev, M. M. Disease Recurrence and Acute Cellular Rejection Episodes During the First Year After Lung Transplantation Among Patients With Sarcoidosis. Transplantation 99, 1940–1945, https://doi.org/10.1097/TP.0000000000000673 (2015).
Schultz, H. H. et al. Recurrence of sarcoid granulomas in lung transplant recipients is common and does not affect overall survival. Sarcoidosis Vasc Diffuse Lung Dis 31, 149–153 (2014).
Acknowledgements
The authors acknowledge the support of Dr. Golnaz Ebrahimi. This work was supported in part by Health Resources and Services Administration contract 234-2005-37011C, and Mallinckrodt Pharmaceuticals for research grant support. The content is the responsibility of the authors alone and does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government.
Author information
Authors and Affiliations
Contributions
Conception, review literature, design and modeling for review writing manuscript: M.M., S.R., The review literature, design and modeling for review writing manuscript: M.M., S.R., O.S., A.M., Writing the article or substantial involvement in its revision before submission: M.M., O.S., S.R., M.D.S., A.M., S.S., M.C.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salamo, O., Roghaee, S., Schweitzer, M.D. et al. White donor, younger donor and double lung transplant are associated with better survival in sarcoidosis patients. Sci Rep 8, 6968 (2018). https://doi.org/10.1038/s41598-018-25144-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-25144-x
This article is cited by
-
Double lung transplantation is better than single lung transplantation for end-stage chronic obstructive pulmonary disease: a meta-analysis
Journal of Cardiothoracic Surgery (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.