Abstract
The 1-hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate synthase (HDS) gene and the 1-hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate reductase (HDR) gene are two important genes in the 2-C-methyl-D-erythritol 4-phosphate (MEP) pathway. In this study, we reported the isolation and characterization of full-length HDS (MF101802) and HDR (MF159558) from Pyropia haitanensis. Characteristics of 3-D structures of the PhHDS and PhHDR proteins were analysed respectively. The results showed that the full-length cDNA of PhHDS, which is 1801 bp long, contained a 1455 bp open reading frame (ORF) encoding a putative 484 amino acid residue protein with a predicted molecular mass of 51.60 kDa. Meanwhile, the full-length cDNA of PhHDR was 1668 bp and contained a 1434 bp ORF encoding a putative 477 amino acid 2 residue protein with a predicted molecular mass of 51.49 kDa. The expression levels of the two genes were higher in conchocelis than that in leafy thallus. Additionally, the expression levels could be influenced by light, temperature and salinity and induced by methyl jasmonate (MJ) and salicylic acid (SA). This study contributed to our in-depth understanding of the roles of PhHDS and PhHDR in terpenoid biosynthesis in Pyropia haitanensis and the regulation of the two genes by external environments.
Similar content being viewed by others
Introduction
Pyropia haitanensis (Bangiales, Rhodophyta) is one of the most important economic algae in China and has great value in research1. This alga is distributed in the intertidal zones from the temperate rocky shores and has a biphasic life cycle including gametophytic blade and filamentous conchocelis phases2,3. As a typical warm temperate zone species, it is cultivated along the coast of the South China Sea4, and its output accounts for 75%-80% of the total production of Pyropia in China every year5. Because of tidal effects, light, temperature, salinity and other environmental factors that may result in cyclical changes in intertidal zone, Pyropia haitanensis has a unique physiological adaptation mechanism. When the tide comes, the algae is exposed to air for up to 4 h or more, and its water loss rate reaches up to 90% or more, which reveals its strong resistance to the environment. Therefore, Pyropia haitanensis is regarded as an ideal material for studying adaptability to complex intertidal zone environments6.
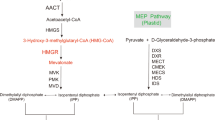
Terpenoids are a kind of biological secondary metabolite that are widespread in nature. The metabolic product is composed of isoprene as a unit. There are more than 23000 different isoprenoids in nature, and half of them are found in plants7. Terpenoids are synthesized by two biological pathways: the MVA pathway and the MEP pathway. The MEP pathway is not dependent on mevalonic acid (MVA) for the IPP synthesis. It is known that the deoxyxylulose-5-phosphate pathway (DXP) or methylerythritol 4-phosphate pathway (MEP) is an important way to synthesize terpene compounds (Fig. 1). The MEP pathway exists in pathogens, algae and higher plants, including important human pathogens, such as Mycobacterium tuberculosis and malaria parasites8,9. The two pathways both use isopentenyl pyrophosphate (IPP) as a precursor for downstream terpene biosynthesis, and the differences between the two pathways are the formation mechanisms for IPP and its isomer diallylene pyrophosphate (DMAPP). Moreover, the MEP pathway exists in plastids, and 1-xyloxyl-5-phosphate (DXP) is its key precursor material10.
The MEP pathway for isoprenoid biosynthesis in plants. Seven enzymes participate in the MEP pathway. HDS and HDR are the enzymes that regulate the last two steps in terpenoid metabolism in the MEP pathway. Isopentenyl pyrophosphate (IPP) as a precursor for the downstream terpene biosynthesis and the difference between the two is formation mechanisms of IPP and its isomer diallylene pyrophosphate (DMAPP). Geranyhlgeranyl diphosphate (GGPP) is produced by plastidic GGPP synthase (GGPS) and serves as a precursor for metabolic branches, including chlorophylls, carotenoids.
The 1-hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate synthase (HDS) and 1-hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate reductase (HDR) genes participate in the last two steps of terpenoid metabolism in the MEP pathway. HDR is a key enzyme in the MEP pathway11. HDS has been fully characterized in Solanum lycopersicum, Hevea brasiliensis, Arabidopsis thaliana and Ginkgo biloba 12,13,14,15. HDS is considered a potential regulator of carbon flux in the MEP pathway; HDS also participates in plant defence mechanisms16. HDR simultaneously synthesizes IPP and DMAPP in the last step of the MEP pathway17 and acts as a restricting factor for isoprenoid biosynthesis in Escherichia coli 18. HDR has also been cloned and analysed from many species, such as Hevea brasiliensis, Arabidopsis thaliana, Ipomoea batatas and Camptotheca acuminata 19,20,21,22. However, neither the HDS nor HDR genes have been cloned and analysed from Pyropia.
Our research group aimed to explore the metabolic status of the MEP pathway in Pyropia haitanensis by studying the expression of PhHDS and PhHDR under different environmental stresses and elicitor treatments. In this study, we obtained the full-length cDNA sequence of the two genes, which are the last two steps of the terpenoid metabolism in the MEP pathway, and we analysed the putative proteins encoded by the two genes using bioinformatics analysis. The results helped further our understanding of these two important genes in the MEP pathway and provided a practical way to study the molecular mechanism associated with terpenoid biosynthesis in Pyropia haitanensis. Moreover, analysis of the expression levels of these terpenoid biosynthesis genes in different environments will prove that Pyropia haitanensis has a unique physiological adaptation mechanism to different environments.
Results
Cloning the full-length cDNA of PhHDS and PhHDR
The primers for cloning PhHDS (HDSF/HDSR) were designed based on the conserved region in HDS genes from pyropia yezoensis. A 446 bp DNA fragment amplified with this primer pair was proven to be a partial HDS sequence from pyropia haitanensis by Blast analysis. Similar methods and results were observed when cloning PhHDR. A 435 bp PhHDR DNA fragment amplified with the HDRF/HDRR primer pair was proven to be a partial sequence from HDR from pyropia haitanensis. Then, the HDS-5/HDS-3 and HDR-5/HDR-3 primer pairs were designed and used from 5′- and 3′-RACE for PhHDS and PhHDR, respectively. Sequence analysis indicated that the full-length cDNA of PhHDS was 1801 bp and contained a 192 bp 5′-untranslated region (UTR) and a 153 bp 3′-UTR. The cDNA contained a 1455 bp open reading frame (ORF) and encoded a putative 484 amino acid protein with a molecular weight of 51.60 kDa. The full-length cDNA of PhHDR was 1668 bp and contained a 115 bp 5′− untranslated region (UTR) and a 119 bp 3′-UTR. The cDNA contained a 1434 bp open reading frame (ORF) and encoded a putative 477 amino acid protein with a molecular weight of 51.49 kDa. The two cDNA sequences were submitted to GenBank with accession numbers of PHHDS (MF101802) and PHHDR (MF159558).
Comparative analysis of PhHDS and PhHDR
We compared the amino acid sequences of PhHDS and PhHDR with that of other proteins in GenBank using Protein BLAST (Figs 2 and 3). The results revealed that PhHDS shared 95% sequence identity with HDS from Pyropia yezoensis (ACI45961.1), 83% similarity with Porphyra umbilicalis (OSX72194.1), 64% similarity with Chondrus crispus (XP005715044), 72% similarity with Cyanidioschyzon merolae strain 10D (XP005536899), 72% similarity with Cyanobacterium aponinum (WP015219624), 73% similarity with Galdieria sulphuraria (XP005706298), 73% similarity with Hapalosiphonaceae (WP026723850) and 72% similarity with Pseudanabaena sp. PCC 6802 (WP019498559). Meanwhile, PhHDR had 82% homology with HDR from Porphyra umbilicalis (OSX71166.1), 96% identity with HDR from Pyropia yezoensis (ACI45962.1), 68% identity with Chondrus crispus (XP 005715383), 63% identity with Cyanidioschyzon merolae strain 10D (XP 005534886), 60% identity with Fischerella sp. NIES-3754 (BAU08587), 60% identity with Fischerella (WP062250827), 64% identity with Galdieria sulphuraria (XP005707572) and 61% identity with Scytonema hofmannii (WP017745471). Phylogenetic trees for PhHDS and PhHDR were constructed to explore the evolutionary relationship between PhHDS and PhHDR among the HDSs and HDRs in other algae (Figs 4 and 5), respectively. The 3-D structures of the PhHDS and PhHDR proteins were predicted by SWISS-MODEL and different 3-D structure models are shown in Fig. 6A and B, respectively. For PhHDS, the modelled residue range was 86–470; this structure was based on template 4g9p. The sequence identity for the template was 37.67% and the quality information QMEAN4 was −4.49. For PhHDR, modelled residue range was 123–465; this range was based was based on template 4h4c. The sequence identity with the temple was 28.72% and the quality information QMEAN4 was −3.00.
Multiple alignment of the HDS amino acid sequences from different species. Black indicates the amino acids from these species that were the same. Red indicates amino acids where one or two species were different from the other species. Cyan indicates amino acids where three or more species were different from the other species. The aligned HDSs were from Chondrus crispus (CHHDS, GenBank accession no.: XP005715044), Cyanidioschyzon merolae strain 10D (CYHDS, GenBank accession no.: XP005536899), Cyanobacterium aponinum (CYAHDS, GenBank accession no.: WP015219624), Galdieria sulphuraria (GAHDS, GenBank accession no.:XP005706298), Hapalosiphonaceae (HAHDS, GenBank accession no.: WP026723850), Porphyra umbilicalis (PUHDS, GenBank accession no.: OSX72194), Pseudanabaena sp. PCC 6802 (PSHDS, GenBank accession no.: WP019498559), Pyropia yezoensis (PYHDS, GenBank accession no.: ACI45961), and Pyropia haitanensis (PHHDS, GenBank accession no.: ATE45997).
Multiple alignment of HDR amino acid sequences from different species. Black indicates the amino acids from these species were all the same. Red indicates amino acids where one or two species were different from the other species. Cyan indicates amino acids where three or more species were different from the other species. The aligned HDRs were from Chondrus crispus (CHHDR, GenBank accession no.: XP 005715383), Cyanidioschyzon merolae strain 10D (CYHDR, GenBank accession no.: XP 005534886), Fischerella sp. NIES-3754 (FISHDR, GenBank accession no.: BAU08587), Fischerella (FIHDR, GenBank accession no.: WP062250827), Galdieria sulphuraria (GAHDR, GenBank accession no.: XP005707572), Scytonema hofmannii (SCHDR, GenBank accession no.: WP017745471), Porphyra umbilicalis (PUHDR, GenBank accession no.: OSX71166), Pyropia yezoensis (PYHDR, GenBank accession no.: ACI45962), and Pyropia haitanensis (PHHDR, GenBank accession no.: ATE45998).
Phylogenetic tree for PHHDS and its related sequences. The tree was constructed with the MEGA programme using the neighbour-joining method. Numbers at nodes represent bootstrap support values after 1000 replicates. The result showed that the PHHDS from Pyropia haitanensis which we studied was grouped with Porphyra umbilicalis and Pyropia yezoensis as Pyropia.
Phylogenetic tree for PHHDR and its related sequences. The tree was constructed with the MEGA programme using the neighbour-joining method. Numbers at nodes represent the bootstrap support values after 1000 replicates. The result showed that the PHHDR from Pyropia haitanensis which we studied was grouped with Porphyra umbilicalis and Pyropia yezoensis as Pyropia.
PhHDS and PhHDR gene expression profiles in different life histories
To detect PhHDS and PhHDR in different life stages, a qPCR approach was used to examine the gene expression patterns between leafy thallus and filamentous conchocelis, which are the two stages in heteromorphic life cycles (see Supplementary Fig. S1). PhHDS expression in filamentous conchocelis was 1.81 ± 0.22-fold higher than in leafy thallus, while the expression of PhHDR in filamentous conchocelis was 1.98 ± 0.25-fold higher than in leafy thallus. The results suggested that both PhHDS and PhHDR expression were approximately 2-fold higher in the conchocelis phase than in the thallus phase.
PhHDS and PhHDR gene expression profiles under different growth environments
Leafy thallus was subjected to different environmental factors, and the relative mRNA expression levels of PhHDS and PhHDR were investigated using qPCR. The results showed that both PhHDS and PhHDR were significantly influenced by temperature (see Supplementary Fig. S2). For PhHDS, higher temperatures lower its expression levels. PhHDS expression was the highest at 12 °C; this value was 1.96 ± 0.08-fold and 3.04 ± 0.39-fold higher than at 17 °C and 26 °C, respectively. However, the expression levels for PhHDR were the opposite, with higher temperature causing higher PhHDR expression levels. PhHDR expression was the lowest at 12 °C, which was 0.80 ± 0.05-fold and 0.56 ± 0.04-fold than lower than at 17 °C and 26 °C, respectively.
Both PhHDS and PhHDR were also influenced by light (see Supplementary Fig. S3). PhHDS expression was up-regulated by light; the 120 μmol photons m−2·s−1 level displayed the highest PhHDS expression. This was followed by 240 μmol photons m−2·s−1. PhHDS expression was lowest at 20 μmol photons m−2·s−1 levels, which was 0.57 ± 0.06-fold and 0.97 ± 0.07-fold less than at 120 μmol photons m−2·s−1 and 240 μmol photons m−2·s−1 respectively. Meanwhile, PhHDR expression levels were the highest under the light intensity of 120 μmol photons m−2·s−1, followed by 240 μmol photons m−2·s−1 and 20 μmol photons m−2·s−1, which were 0.43 ± 0.03-fold and 0.82 ± 0.05-fold less than 120 μmol photons m−2·s−1 and 240 μmol photons m−2·s−1, respectively.
After the treatment with the three different salinities, PhHDS expression was highest in medium seawater with 80 salinity, followed by 50 salinity and 30 salinity. PhHDS expression at 30 salinity was 0.55 ± 0.01-fold and 0.48 ± 0.05-fold less than at 50 salinity and 80 salinity, respectively. However, the expression levels of PhHDR among the three salinities showed few differences. PhHDR levels were the highest at 30 salinity, which was 1.07 ± 0.10-fold and 1.27 ± 0.18-fold more than 50 salinity and 80 salinity, respectively. (see Supplementary Fig. S4).
PhHDS and PhHDR gene expression due to elicitor treatments
Leafy thallus was placed in sterile distilled water and subjected to methyl jasmonate (MJ) and salicylic acid (SA) treatments to study the expression in response to different elicitors. For MJ treatment, thallus was dipped in either 100 μM or 200 mΜ MJ solutions and placed over a soaked filter paper. Similarly, thallus was dipped in either 1 mM or 2 mM SA solutions for the SA treatment. The control and elicitor-treated samples were harvested after 24 h. Then, the samples were subject to RNA isolation.
The effects of the different MeJA and SA concentrations on PhHDS and PhHDR expression were examined by qPCR. The data showed that PhHDS and PhHDR expression levels were up-regulated by MeJA treatment compared to the control group (see Supplementary Fig. S5); the expression levels were higher at the higher concentration. PhHDS expression was 1.70 ± 0.22-fold and 5.40 ± 0.46-fold, while PhHDR expression was 2.55 ± 0.16-fold and 4.13 ± 0.56-fold compared to the control groups without MeJA treatment. SA treatment also up-regulated the expression levels of the two genes, and their expression was higher at the higher SA concentration. PhHDS expression was increased 1.01 ± 0.12-and 4.66 ± 0.29-fold, while PhHDR expression was increased 1.21 ± 0.41-fold and 2.45 ± 0.49-fold compared to the control groups without SA treatment (see Supplementary Fig. S6).
Discussion
Terpenoids are a biological secondary metabolite that directly affect Pyropia yield23. In terms of their functions, plant terpenoids are divided into the following two categories: primary metabolites and secondary metabolites. The MEP pathway exists in plant plastids, and is one of the major metabolic pathways for the synthesis of secondary metabolites. In this study, we cloned PhHDS and PhHDR, the last two enzymes in the MEP pathway, from Pyropia haitanensis and we analysed the proteins encoded by the two genes using bioinformatics methods. The phylogenetic analysis results showed that both of the genes from Pyropia haitanensis were clustered with Pyropia yezoensis and Porphyra umbilicalis, which meant that the two genes were conserved in Pyropia.
Previous research has suggested that metabolic activity was distinctive in different Pyropia haitanensis life stages by analysing alternative oxidase (AOX) gene expression24. In this study, the expression patterns of PhHDS and PhHDR during different growth periods showed that PhHDS and PhHDR expression were approximately 2-fold higher in the conchocelis phase than in the leafy thallus phase. We speculated that there were 2 copies of the corresponding genes because conchocelis is diploid and thallus is haploid; thus, the gene expression differences monitored by qPCR were not considered significant.
Secondary metabolites are the result of biological and non-biological interactions between organisms and the environment during long-term evolution, and secondary metabolites play an important role in organisms in improving survival and coordinating with the environment. Therefore, the production of and changes to secondary metabolites are susceptible to the environment25,26. To simulate the complex environments associated with intertidal zones, light, temperature, and salinity were used as environmental factors to study PhHDS and PhHDR expression in leafy thallus. The results showed that the expression of the two genes was influenced by the various environments. Transcription levels of the two genes in the MEP pathway obviously increased when Arabidopsis thaliana leaves were irradiated by light27. The expression profile of PuGGPS in Porphyra umbilicalis, which is related to carotenoid biosynthesis, was influenced by light. The gene was more highly expressed under high light conditions28. Light is also an influencing factor for the expression of PhHDS and PhHDR, and their expression levels appeared to increase under higher light intensity. However, PhHDS and PhHDR was more highly expressed under 120 μmol photons m−2·s−1 than under 240 μmol photons m−2·s−1. This finding suggested that excessive light intensity would affect PhHDS and PhHDR expression. The relationship between the light and temperature dependency of isoprenoid production downstream of the MEP pathway was addressed29. The results showed that the metabolic flux of the MEP pathway in spinach increased with light intensity and temperature. At hot temperatures, 2- C-methyl- d -erythritol 2,4-cyclodiphosphate (MEcDP), an intermediate in the MEP pathway in different herbs and trees, accumulated30. The results also revealed that MEP pathway metabolic activity was influenced by temperature. In this study, PhHDS and PhHDR expression levels were diverse at the different temperatures. PhHDS was highly expressed at 12 °C. In contrast, PhHDR expression was highly expressed at 26 °C. Salinity stress can induce the terpenoid synthesis pathway and synthesize compounds such as monoterpenes and carotenoids31. Acetoacetyl-CoA thiolase is a regulatory enzyme in isoprenoid biosynthesis, and real-time RT-PCR analysis indicated that acetoacetyl-CoA thiolase expression levels were highly increased in the roots and leaves under salinity stress32. In this study, the expression of PhHDS was also influenced by salinity stress. Its expression was increased in both experimental groups compared with the control group; this conclusion was in agreement with the results for terpene synthesis in Helianthus annuus, Saltbushes and Disphyma austral 33,34,35. However, the PhHDR expression levels were almost the same threat the different salinities; this phenomenon showed that terpenoid biosynthesis remains the same Pyropia haitanensis even under high salinity. The expression levels of the two genes were affected by all three environmental factors, which strongly suggested that the effects of environmental cues on the transcription of the corresponding biosynthetic genes are significant23. Environmental stress could induce the emission of algal isoprenoids36 and Pyropia haitanensis could maintain normal terpenoid metabolic activity even under harsh environments.
MJ and SA have been used intensively either individually or in combination to affect phytoalexin and secondary metabolite synthesis and explore biomolecule biosynthesis37,38. MJ and SA can induce the overexpression of genes related to the production of plant secondary metabolites, such as terpenes39,40,41. In this study, PhHDS and PhHDR expression levels were increased by MJ and SA treatment, and higher concentration of MJ and SA elicited higher gene expression levels, indicating that MJ and SA may increase metabolite production. This conclusion was in agreement with the production of various metabolites in other species, such as sesquiterpene lactone in Cichorium intybus 42 and artemisinin in A. annua 43. The major roles of secondary metabolites are to protect organisms from natural enemies and promote survival against biotic and abiotic stresses. Thus, the expression levels of genes associated with terpenoid biosynthesis should increase in Pyropia haitanensis upon application of exogenous elicitors.
In conclusion, we first cloned and analysed the PhHDS and PhHDR genes, which are important for terpene biosynthesis in the MEP pathway in Pyropia haitanensis. We also studied their expression profiles under the influence of various environmental factors and through elicitor treatments. Such studies will not only be advantageous for understanding Pyropia haitanensis biosynthesis, but will also provide molecular value for studying the MEP pathway in other red algae.
Methods
Plant materials
The leafy thallus from pyropia haitanensis was collected from Putian, Fujian Province in China (25° 11′ N 119°28′ E) and the conchocelis were purchased from Jiangsu Research Institute of Laver. Before used, the materials were cultivated in the seawater medium, and cool-white fluorescent light was provided in a 12:12 L:D cycle. The cultivation environment for the leafy thallus from pyropia haitanensis was as follows: 12 °C, 20 μmol photons m−2·s−1 and 30 salinity, these conditions were used for the control group in real-time quantitative PCR.
RNA isolation and cDNA synthesis
Total RNA was isolated from leafy thallus and conchocelis using a MiniBEST Plant RNA Extraction Kit (Takara, Japan) according to the user’s manual. For reverse transcription PCR (Takara Reverse Transcription kit), single-strand cDNA was synthesized from 2 μg of total RNA and stored at −20 °C before used.
Cloning and sequencing of PhHDS and PhHDR
All primers used in this study are shown in Table 1. Two pairs of specific primers, HDSF and HDSR and HDRF and HDRR, were designed based on conserved regions from previously known pyropia yezoensis sequences (GenBank accession numbers FJ175682.1 and FJ175683.1, respectively) and used to amplify the core cDNA fragments. The reactions were both performed in a total volume of 50 μl containing 39.5 μl ddH2O, 2 μl 10 × PCR Buffer II (Mg2+ plus), 2 μl dNTP Mixture (10 mM), 2 μl cDNA, 1 μl forward primer (20 μM), 1 μl reverse primer (20 μM) and 0.5 μl TaKaRa LA Taq HS (5 U/μl, TaKaRa). The PCR products were cloned into the pEASY-T3 vector (TransGen, China) and sequenced. Rapid amplification of cDNA ends (RACE) PCR was used to obtain the full-length cDNA of PhHDS and PhHDR according to the manufacturer’s instructions (SMARTer RACE5′/3′ Kit User Manual Extraction Kit (TaKaRa, Japan)). The UPM (Univer Primer Mix) and HDS-5 primer pair was used for the 5′-RACE of PhHDS, whereas the HDS-3 and UPM primer pair was used for the 3′-RACE reaction. Meanwhile, the UPM and HDR-5 primer pair was used for the 5′-RACE of PhHDR, while the HDR-3 and UPM primer pair was used for the 3′-RACE reaction.
Multiple alignment and bioinformatics analysis of PhHDS and PhHDR
The deduced amino acid sequences of PhHDS and PhHDR were aligned with the sequences of other organisms from GenBank using the BLAST programmes (http:// www.ncbi.nlm.nih.gov/BLAST/) and DNAMAN software. The phylogenetic analysis was performed with the MEGA 5.1 software (USA) with the neighbour-joining (NJ) analyses option, and the bootstrap confidence intervals for the phylogenetic tree were based on 1000 replications.
Different growth environment treatments
To detect the impact of various growth environments on PhHDS and PhHDR expression patterns, the following three environmental factors were studied in our research: (1) temperature, (2) light and (3) salinity. For temperature, leafy thallus was subject to three different temperatures (12 °C, 17 °C, and 26 °C) in the same seawater medium for 48 h. For light, leafy thallus was subject to three different light intensities (20 μmol photons m−2·s−1, 120 μmol photons m−2·s−1, and 240 μmol photons m−2·s−1) for 48 h. Leafy thallus was also cultivated in the seawater with three different salinities (30, 50, and 80 salinity) for 2 h. The samples that were subject to different environmental factors were used for RNA isolation in the following experiments.
Elicitor treatments
Leafy thallus was placed in sterile distilled water and subjected to methyl jasmonate (MJ) and salicylic acid (SA) treatments to study gene expression in response to different elicitors. For MJ treatment, thallus was dipped in either 100 μM or 200 μM MJ solutions and placed over a soaked filter paper. Similarly, thallus was dipped in either 1 mM or 2 mM SA solutions for the SA treatment. The control and elicitor-treated samples were harvested after 24 h, and then the samples were used for RNA isolation.
Relative quantification by real-time quantitative PCR
The expression patterns of PhHDS and PhHDR were explored in different life stages. Additionally, the expression levels of PhHDS and PhHDR were also detected under different growth environmental and elicitor treatments. For RT-PCR analysis, a total of 1 μg RNA was used to synthesize cDNA with oligo (dT) primers using the RT-for-PCR kit (Takara, Japan). The expression levels were measured with a Baiyuan ASA-4800 Real Time PCR System using SYBR green fluorescence (TaKaRa) according to the instructions. The following cycling profile was used for amplification: (1) 95 °C for 30 s and (2) 40 cycles of 95 °C for 5 s and 60 °C for 34 s. The relative gene expression was calculated with the 2−ΔΔCt relative quantitative method. The primer sequences used for the amplification of specific genes are listed in Table 1. 18S rRNA primers were designed as an internal control.
RT-PCR data analysis
All the experiments were repeated at least three times and the data were analysed using the SPSS software (version 22.0 USA). All the data were assessed by one-way analysis of variance (ANOVA) and the significance level was set at P < 0.05. The highest significance level was set at P < 0.01.
References
Li, B., Chen, C., Yan, X. U., Dehua, J. I. & Xie, C. Validation of housekeeping genes as internal controls for studying the gene expression in Pyropia haitanensis (Bangiales, Rhodophyta) by quantitative real-time PCR. Acta Oceanol Sin. 33, 152–159 (2014).
Sutherland, J. E. et al. A new look at an ancient order: generic revision of the Bangiales (Rhodophyta). J Phycol. 47, 1131–1151 (2011).
Zhang, Y., Yan, X. H. & Aruga, Y. The sex and sex determination in Pyropia haitanensis (Bangiales, Rhodophyta). Plos One. 8, e73414 (2013).
Zhang, X. C., Qin, S., Ma, J. H. & Xu, P. The genetic of marine algae. China Agriculture Press, 184 (Beijing, 2005).
Xie, C. T. et al. Construction of DNA fingerprinting database of germplasm materials of Porphyra haitanensis. J Fish Chin. 34, 733–740 (2010).
Xie, J., Xu, Y., Ji, D. H., Chen, C. S. & Xie, C. T. Physiological response of the antioxidant system in Pyropia haitanensis to desiccation stress. JFSC. 21, 405–412 (2014).
Zhang, W. & Liang, C. W. Research progress of plant isoprenoids biosynthetic pathway. Shandong Chemical Industry. 5, 57–58 (2014).
Tang, G. et al. β-carotene in golden rice is as good as β-carotene in oil at providing vitamin a to children. Am J Clin Nutr. 96, 658–664 (2012).
Masini, T. & Hirsch, A. K. H. Development of inhibitors of the 2c-methyl-d-erythritol 4-phosphate (MEP) pathway enzymes as potential anti-infective agents. J Med Chem. 57, 9740–9763 (2014).
Rasulov, B., Talts, E., Kännaste, A. & Niinemets, Ü. Bisphosphonate inhibitors reveal a large elasticity of plastidic isoprenoid synthesis pathway in isoprene-emitting hybrid aspen. Plant Physiol. 168, 532–548 (2015).
Laule, O. et al. Crosstalk between cytosolic and plastidial pathways of isoprenoid biosynthesis in arabidopsis thaliana. Proc Natl Acad Sci USA 100, 6866–6871 (2003).
Rodríguez-Concepción, M., Querol, J., Lois, L. M., Imperial, S. & Boronat, A. Bioinformatic and molecular analysis of hydroxymethylbutenyl diphosphate synthase (GCPE) gene expression during carotenoid accumulation in ripening tomato fruit. Planta. 217, 476–482 (2003).
Sando, T. et al. Cloning and characterization of the 2–methyl–erythritol 4-phosphate (MEP) pathway genes of a natural-rubber producing plant. Biosci Biotech Bioch. 72, 2903–2917 (2014).
Gutiérreznava, M. L., Gillmor, C. S., Jiménez, L. F., Guevaragarcía, A. & León, P. Chloroplast biogenesis genes act cell and noncell autonomously in early chloroplast development. Plant Physiol. 135, 471–482 (2004).
Kim, S. M. & Kim, S. U. Characterization of 1-hydroxy-2-methyl-2-(e)-butenyl-4-diphosphate synthase (HDS) gene from ginkgo biloba. Mol Biol Rep. 37, 973–979 (2010).
Gil, M. J., Coego, A., Mauch-Mani, B., Jordá, L. & Vera, P. The arabidopsis csb3 mutant reveals a regulatory link between salicylic acid-mediated disease resistance and the methyl-erythritol 4-phosphate pathway. Plant J. 44, 155–166 (2010).
Hao, G. et al. Cloning, molecular characterization and functional analysis of 1-hydroxy-2-methyl-2-(e)-butenyl-4-diphosphate reductase (HDR) gene for diterpenoid tanshinone biosynthesis in salvia miltiorrhiza, bge. f. alba. Plant Physiol Bioch. 70, 21–32 (2013).
Cunningham, F. X., Lafond, T. P. & Gantt, E. Evidence of a role for lytb in the nonmevalonate pathway of isoprenoid biosynthesis. J Bacteriol. 182, 5841–5848 (2000).
Li, H. L., Lei, M. Y. & Peng, S. Q. Cloning and expression of HbHDR from Hevea brasiliensis. Genomics and Applied Biology. 28, 15–21 (2009).
Hsieh, M. H., Chang, C. Y., Hsu, S. J. & Chen, J. J. Chloroplast localization of methylerythritol 4-phosphate pathway enzymes and regulation of mitochondrial genes in ispd and ispe albino mutants in arabidopsis. Plant Mol Biol. 66, 663–673 (2008).
Wang, G. et al. Molecular cloning and characterization of the HDR gene involved in the methyl-erythritol phosphate (MEP) pathway from sweet potato [Ipomoea batatas (L.) Lam]. Afr j Agr Res. 7, 205–211 (2012).
Wang, Q. et al. Molecular cloning and characterization of 1-hydroxy-2-methyl-2-(e)-butenyl 4-diphosphate reductase (CaHDR) from Camptotheca acuminata and its functional identification in Escherichia coli. Bmb Reports. 41, 112–118 (2008).
Yang, Y. et al. Environmental cues induce changes of steviol glycosides contents and transcription of corresponding biosynthetic genes in Stevia rebaudiana. Plant Physiol Bioch. 86, 174–180 (2015).
Zhang, B. Y., Zhu, D. L., Wang, G. C. & Peng, G. Characterization of the AOX gene and cyanide-resistant respiration in Pyropia haitanensis, (rhodophyta). J Appl Phycol. 26, 2425–2433 (2014).
Duan, C. R. & Wang, B. C. & Chongqing. The effects of the environment stress on the plant secondary metabolites. Journal of Chongqing University. 26, 67–71 (2003).
Kliebenstein, D. J. Secondary metabolites and plant/environment interactions: a view through Arabidopsis thaliana tinged glasses. Plant Cell Environ. 27, 675–684 (2010).
Hsieh, M. H. & Goodman, H. M. The Arabidopsis IspH homolog is involved in the plastid nonmevalonate pathway of isoprenoid biosynthesis. Plant Physiol. 138, 641–653 (2005).
Yang, L. E., Huang, X. Q., Lu, Q. Q., Zhu, J. Y. & Lu, S. Cloning and characterization of the geranylgeranyl diphosphate synthase (GGPS) responsible for carotenoid biosynthesis in Porphyra umbilicalis. J Appl Phycol. 28, 1–8 (2016).
Mongélard, G. et al. Measurement of carbon flux through the MEP pathway for isoprenoid synthesis by (31) P-NMR spectroscopy after specific inhibition of 2- C-methyl- d -erythritol 2,4-cyclodiphosphate reductase. Effect of light and temperature. Plant Cell Environ. 34, 1241–1247 (2011).
Rivasseau, C. et al. Accumulation of 2-c-methyl-d-erythritol 2,4-cyclodiphosphate in illuminated plant leaves at supraoptimal temperatures reveals a bottleneck of the prokaryotic methylerythritol 4-phosphate pathway of isoprenoid biosynthesis. Plant Cell Environ. 32, 82–92 (2009).
Shi, L. P. et al. The expression and analysis of terpene synthesis related genes in Maize under the condition of salt stress. China Biotechnology. 36, 31–37 (2016).
Soto, G. et al. Acetoacetyl-coa thiolase regulates the mevalonate pathway during abiotic stress adaptation. J Exp Bot. 62, 5699–5711 (2011).
Santos, C. V. Regulation of chlorophyll biosynthesis and degradation by salt stress in sunflower leaves. Sci Horticulturae. 103, 93–99 (2004).
Joseph, S., Murphy, D. & Bhave, M. Glycine betaine biosynthesis in saltbushes (Atriplex spp.) under salinity stress. Biologia. 68, 879–895 (2013).
Jain, G. & Gould, K. S. Functional significance of betalain biosynthesis in leaves of Disphyma australe, under salinity stress. Environ Exp Bot. 109, 131–140 (2015).
Meskhidze, N., Sabolis, A., Reed, R. & Kamykowski, D. Quantifying environmental stress induced emissions of algal isoprene and monoterpenes using laboratory measurements. Biogeosciences Discussions. 11, 13533–13570 (2015).
Namdeo, A. G. Plant cell elicitation for production of secondary metabolites: a review. Pharmacogn Rev. 1, 69–79 (2007).
Agarwal, A. V. et al. Comprehensive assessment of the genes involved in withanolide biosynthesis from Withania somnifera: chemotype-specific and elicitor-responsive expression. Funct Integr Genomics. 17, 477–490 (2017).
Newberger, N. C., Ranzer, L. K., Boehnlein, J. M. & Kerr, R. G. Induction of terpene biosynthesis in dinoflagellate symbionts of Caribbean gorgonians. Phytochemistry. 67, 2133–2139 (2006).
Cheng, A., Wang, L., Sun, Y. & Lou, H. Identification and expression analysis of key enzymes of the terpenoids biosynthesis pathway of a liverwort Plagiochasma appendiculatum by EST analysis. Acta Physiol Plant. 35, 107–118 (2013).
Xiang, L. et al. Enhancement of artemisinin content and relative expression of genes of artemisinin biosynthesis in Artemisia annua by exogenous MeJA treatment. Plant Growth Regul. 75, 435–441 (2015).
Malarz, J., Stojakowska, A. & Kisiel, W. Effect of methyl jasmonate and salicylic acid on sesquiterpene lactone accumulation in hairy roots of Cichorium intybus. Acta Physiol Plant. 29, 127–132 (2007).
Wang, H. et al. Effects of exogenous methyl jasmonate on artemisinin biosynthesis and secondary metabolites in Artemisia annua L. Ind Crops Prod. 31, 214–218 (2010).
Acknowledgements
This work was supported by National Key R&D Program of China (2016YFC1402102, 2016YFC1402104) and National Natural Science Foundation of China (project nos. 41276134).
Author information
Authors and Affiliations
Contributions
Yuan He wrote the main manuscript text, Zhihong Yan provided the leafy thallus of pyropia haitanensis, Yu Du prepared figures S2–S4, Yafeng Ma prepared figures S5–S6. Songdong Shen modified the text. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
He, Y., Yan, Z., Du, Y. et al. Molecular cloning and expression analysis of two key genes, HDS and HDR, in the MEP pathway in Pyropia haitanensis . Sci Rep 7, 17499 (2017). https://doi.org/10.1038/s41598-017-17521-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-17521-9
This article is cited by
-
RNA-seq analysis reveals narrow differential gene expression in MEP and MVA pathways responsible for phytochemical divergence in extreme genotypes of Thymus daenensis Celak
BMC Genomics (2024)
-
Proliferating cell nuclear antigen of Ulva prolifera is involved in the response to temperature stress
Journal of Oceanology and Limnology (2024)
-
3-Hydroxy-3-methylglutaryl coenzyme A reductase genes from Glycine max regulate plant growth and isoprenoid biosynthesis
Scientific Reports (2023)
-
Identification and functional study of oleoresin terpenoid biosynthesis-related genes in masson pine (Pinus massoniana L.) based on transcriptome analysis
Tree Genetics & Genomes (2020)
-
Effects of light and salinity on carotenoid biosynthesis in Ulva prolifera
Acta Oceanologica Sinica (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.