Abstract
Here, we propose an environmentally benign removal technique for heavy metal ions based on selective and spontaneous transfer to oleic acid. The ions can be removed via (1) the selective and rapid complexation with the carboxylic end of oleic acid at an oleic acid/water interface, and (2) the diffusion of such complex into the oleic acid layer. A wide variety of heavy metal ions such as Cu2+, Pb2+, Zn2+, and Ni2+ can be selectively removed over K+ and Na+. For example, the concentration of Cu2+ is reduced to below 1.3 ppm within 24 h, which corresponds to the level of Cu2+ permitted by the Environmental Protection Agency. The addition of ethylenediamine ligand to the metal ion solutions is also shown to enhance the phase transfer. The removal efficiency is increased by up to 6 times when compared with that in the absence of the ligand and follows the order, Cu2+ (99%) > Pb2+ (96%) > Zn2+ (95%) > Ni2+ (65%). Moreover, the removal time can be shortened from 24 h to 1 h. The effect of an emulsion induced by a mechanical agitation on the removal of heavy metal ion is also studied.
Similar content being viewed by others
Introduction
A few years ago, accidental lead (Pb2+) poisoning in drinking water in the city of Flint, Michigan, caused many people, especially children younger than 5 years, to suffer from skin rashes, as well as cognitive and behavioral disorders1. This incident serves to demonstrate that the removal of heavy metal ions is still one of critical issues in modern public health2,3.
To date, the removal of heavy metal ions in water has been performed by a variety of techniques4,5,6,7,8,9. One of the most popular techniques for this purpose is chemical precipitation due to its operational simplicity and low cost10,11. Precipitation method utilizes basic precipitants such as NaOH and Ca(OH)2 in order to produce insoluble metal salts12. However, after precipitation, an acid neutralization process is required to lower the pH of the solution. Adsorption is an alternative technique to chemical precipitation13,14. Porous materials such as activated carbon, mesoporous oxide and zeolite are widely used as adsorbents after surface modification15,16,17,18,19,20,21. Carbon nanotubes have been also tested as adsorbents, but they have several issues such as potential toxicity and mass production to be addressed for fieldwork applications22,23,24.
Here, we report a facile method for the removal of heavy metal ions by taking advantage of spontaneous and selective phase transfer into an oil phase. The phase transfer is driven by interfacial complexation and diffusion. Biocompatible oleic acid is chosen as the oil phase since its carboxylic end is known to form stable complexes with many heavy metal ions25,26. Our proposed method has several key advantages over chemical precipitation and adsorption. First, it requires neither specially-designed adsorbent nor toxic precipitant. Second, oleic acid is one of lipids and is known to be biocompatible27,28. Lastly, oleic acid which captures aqueous heavy metal ions is quite easy to be separated from water due to the density difference between water and oleic acid. Water and oleic acid are immiscible. Therefore, our method does not require an additional filtration to remove insoluble precipitates or fine solid adsorbents.
Results and Discussion
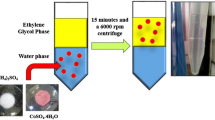
Our idea for the removal of heavy metal ions is schematically illustrated in Fig. 1. Upon the addition of oleic acid to an aqueous solution of heavy metal ions, the carboxylic end of oleic acid at an oleic acid/water interface would rapidly capture the metal ion via a complex formation at the interface. Then, the metal-carboxylate complex would diffuse from the interface into the oleic acid layer, resulting in the removal of the aqueous heavy metal ions (Fig. 1a). In addition, the phase transfer of the metal ions could be chemically enhanced by the addition of ethylenediamine (EN) ligand to an aqueous phase (Fig. 1b) since more stable complex is known to be produced with the aid of the EN ligand (Fig. 1c)29,30. The formation of an emulsion by mechanical agitation could also improve removal rate and efficiency due to increased interfacial area and concomitant shorter diffusion time (Fig. 1b).
Schematic illustration of the spontaneous phase transfer-mediated selective removal of heavy metal ions using oleic acid. (a) Complex formation between heavy metal ions and the carboxylic end of oleic acid (top) and experimental procedure for the removal of heavy metal ions by using oleic acid (bottom). (b) Chemical and mechanical enhancements for heavy metal ions removal. (c) Complex formation of heavy metal ions with ethylenediamine ligand.
First, removal performance by oleic acid for aqueous Cu2+ ion is investigated since Cu2+ ion is environmentally and biologically important31. After 1.5 ml of oleic acid is added to 3.0 ml of 48 ppm Cu2+ solution, transparent oleic acid layer gradually turns blue (Fig. 2a). The result implies that the aqueous Cu2+ ion is spontaneously transferred into the oleic acid layer. To quantitatively analyze Cu2+ removal by oleic acid, the concentration of the aqueous Cu2+ over time is recorded. As shown in Fig. 2b (black dot), the concentration is decreased from 48 to 37 ppm in 24 h. Most of the change in the concentration occurs within the first 1 h, followed by a gradual decrease to 37 ppm.
Removal performance for heavy metal ions by oleic acid. (a) Photographs showing the phase transfer of Cu2+ from water to oleic acid layer over time. (b) The concentration of Cu2+ in water over time (black dot) and simulated fit (red line). (c) Removal capacity for Cu2+ by changing the volume of oleic acid. (d) Simulated three-dimensional images of Cu2+ concentrations at different time. (e) Removal efficiencies for various metal ions (Pb2+, Cu2+, Zn2+, and Ni2+) after 24 h.
Red line in Fig. 2b shows the simulation result for the removal kinetics of aqueous Cu2+. We assume both diffusion and electrostatic interaction to be mainly responsible for the mass flux of the aqueous Cu2+ 32,33. Consequently, the total mass flux (J tot ) can be expressed as follows:
where D, z, and u are the diffusion constant, the valence, and the ion mobility of Cu2+ ion. F and ϕ represent Faraday’s constant and the electrostatic potential, respectively34. The law of conservation of mass along with the equation (1) produces the following governing equation:
The interfacial concentrations of Cu2+ (c Cu ), oleic acid (c OA ), and their complex (c Cu·OA ) can be determined from the following equilibrium constant (K eq ) (Table S1):
The diameter of a container is 19 mm and the volumes of water and oleic acid are fixed at 3.0 and 1.5 ml, respectively. The simulation in Fig. 2b shows that the concentration of the aqueous Cu2+ is drastically decreased at an early stage due to rapid complexation. This tendency is in good agreement with the experimental observation.
Next, the effect of the volume of oleic acid on the removal of Cu2+ is examined while maintaining the concentration at 48 ppm. Interestingly, the amounts of Cu2+ removed are found to be similar irrespective of the volume of oleic acid (Fig. 2c). The photographs in Fig. 2c (inset) also show that the blue color appears near the interface. The simulated three-dimensional images of Cu2+ concentrations over time in Fig. 2d also show significant accumulation of the Cu2+ complex around the interface. Both the experiment and simulation suggest that oleic acid near the interface would participate in the complex formation. It is understandable that the poor solubility of the complex in the oleic acid layer could retard the diffusion into the oleic acid layer.
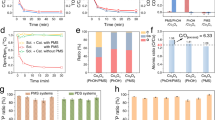
To further test the utility of our method, the removal of other heavy metal ions (Pb2+, Zn+, and Ni2+) is examined at a similar initial concentration as Cu2+. For a comparison, removal efficiency, defined as percentage removal ratio, is calculated. Fig. 2e shows that the removal efficiency at a given concentration follows the order, Pb2+ (49%) > Cu2+ (24%) > Zn2+ (16%) > Ni2+ (11%). This result is in good agreement with the order of stability constants for the metal-carboxylate complexes25. At lower initial concentrations, the concentration of aqueous Cu2+ is reduced to below 1.3 ppm which is the level of the Environmental Protection Agency’s regulation for drinking water (Table S2)35.
Since bidentate EN ligand is known to form a more stable complex with Cu2+ than oxalate which contains two carboxylates, the removal of Cu2+ by oleic acid with the aid of EN is examined. After the addition of EN into an aqueous Cu2+ solution, we observe that the color of the solution immediately turns violet (Fig. 3a top). The violet color would result from the d-orbital splitting of the complex between Cu2+ and EN36. Upon exposure to oleic acid layer, the intense violet color of the aqueous solution disappears while the color of the oleic acid layer changes to greenish blue (Fig. 3a bottom). This color change is further confirmed by using a UV–vis spectrophotometer. As shown in Fig. 3b, the maximum absorbance wavelength of the solution is around 548 nm. The intensity at 548 nm is rapidly attenuated by approximately 75% within 30 min and then gradually decreased until 24 h.
Removal performance for heavy metal ions by oleic acid with the addition of EN into water. (a) Photographs showing the phase transfer of Cu2+ from water to oleic acid layer over time in the presence of EN. (b) Changes of UV-vis spectrum over time after adding oleic acid onto the aqueous Cu2+ solution with EN. (c) The concentrations of Cu2+ in the presence of EN. (d) Removal efficiencies for various heavy metal ions (Cu2+, Pb2+, Zn2+, and Ni2+) with EN.
The change in an aqueous Cu2+ concentration over time is also measured, as shown in Fig. 3c. Similar to the previous UV–vis result, the concentration in water is decreased from 54 to 0.5 ppm after 24 h. The removal capacity is increased by ca. 4 times in comparison with that without EN. The removal rate by oleic acid with the aid of EN becomes faster. For example, the amount of Cu2+ removed in the first 30 min is increased by ca. 10 times.
The removal efficiencies for other heavy metal ions (Cu2+, Pb2+, Zn2+, and Ni2+) by oleic acid with the aid of EN are also evaluated (Fig. 3d). The removal efficiencies for the metal ions are increased by up to ca. 6 times relative to those without EN. For example, 95% of Zn2+ in the presence of EN is removed within 24 h, whereas only 16% of Zn2+ is removed without EN. The removal efficiencies follow the order, Cu2+ (99%) > Pb2+ (96%) > Zn2+ (95%) > Ni2+ (65%). Note that the removal efficiency of Cu2+ is higher than that of Pb2+. This can be contributed to a higher stability constant of Cu2+ with EN37.
Since complex formation at an oleic acid/water interface and diffusion across the interface depends on an interfacial area, the effect of an emulsion (i.e., oleic acid) on the removal rate and efficiency is addressed. Mechanical stirring for the formation of the emulsion would also reduce the diffusion time of an aqueous metal ion into the interface (Fig. 4a). The photographs in Fig. 4b show the representative emulsion of oleic acid by mechanical agitation. The average emulsion diameter is decreased as the stirring speed is increased (Fig. S1). The removal efficiency at different stirring speeds is shown in Fig. 4c. The removal efficiency is linearly increased from 12 to 35% with increasing the stirring speed from 0 to 1000 rpm.
In this study, we have demonstrated the environment-friendly removal of aqueous heavy metal ions by utilizing spontaneous and selective phase transfer of such ions into oleic acid. This phase transfer is based on metal–carboxylate complexation and diffusion. Upon the addition of oleic acid onto the aqueous metal ion solution, the ions begin to diffuse toward an oleic acid/water interface by either random diffusion or electrostatic interaction. Stable complex between the metal ion and carboxylic end of oleic acid is thus spontaneously formed at the interface, followed by slow diffusion into the oleic acid layer. By using oleic acid alone, it is possible to reduce the concentration of Cu2+ below the regulation level by the Environmental Protection Agency for drinking water (i.e., 1.3 ppm). Other heavy metal ions such as Pb2+, Ni2+, and Zn2+ can also be removed. Removal performance by oleic acid can also be improved by the EN ligand due to the formation of more stable complex. In the presence of EN, removal efficiency and rate by oleic acid are increased up to ca. 6 and 10 times, respectively. In addition, the removal efficiency is increased by ca. 3 times by the formation of an emulsion of oleic acid. We believe that our method will have a large impact on a wide variety of environmental applications ranging from environmental monitoring to water purification.
Methods
Chemicals and materials
Copper sulfate pentahydrate (99.5%) and nickel nitrate hexahydrate (97%) were obtained from Junsei chemical Co., Ltd. Lead nitrate (99%), ethylenediamine (EN) (99.5%), and oleic acid (technical grade, 90%) were purchased from Aldrich. Zinc chloride (98%) was purchased from Kanto chemical Co., Inc. Deionized (DI) water was used throughout the experiments.
Preparation of simulated heavy metal ion solutions
Simulated aqueous heavy metal ion solutions were prepared with the 50 ppm CuSO4∙5H2O, Pb(NO3)2, Ni(NO3)2∙6H2O, ZnCl2 solution with DI water, respectively. In case of adding ligand, these powders were dissolved in 100 ppm of EN solution because it is known that a heavy metal ion generally forms complex with EN of 1:2 stoichiometry38.
Heavy metal ions removal by oleic acid
Heavy metal ions removal experiments were performed in 10 ml vial (1.9 cm diameter). 1.5 ml of oleic acid was added to 3.0 ml of the above simulated solution to examine the removal kinetics and efficiency. To investigate examine the maximum removal capability of oleic acid, we added oleic acid at different volumes (0.6, 1.2, 1.8, 2.4, and 3.0 ml) while fixing of oleic acid to fixed the volume (5.4 ml) of water layer.
Formation of an emulsion
Mechanical agitation is performed by magnetic stirrer (HS15-26p, Misung Scientific Co., Ltd.) and stirring bar. The maximum rpm of the magnetic stirrer is 1500 rpm. Pictures of emulsion with different stirring speed (0, 500, 1000, 1250, and 1500 rpm) are taken to determine average emulsion sizes statistically. Then we calculated average interfacial area from average emulsion size.
Determination of heavy metal ion concentrations
The concentrations of aqueous heavy metal ions were analyzed by inductively coupled plasma-optical emission spectrophotometer (ICP-OES, Perkin Elmer Optima 8300). Absorbance spectra were also obtained from UV-vis spectrophotometer (Agilent 8453 G1103A).
Simulation
In order to quantify the removal kinetics of aqueous Cu2+, the local concentration of Cu2+ was numerically investigated by solving Fick’s second law of diffusion. The reaction of Cu2+ with oleic acid molecules at the oil/water interface was considered with an assumption that the equilibrium is always satisfied at the interface. Diffusivity of Cu2+ in water and self-diffusivity of oleic acid itself were set to be 0.7 × 10−9 m2/s and 0.47 × 10−10 m2/s, respectively39,40,41,42. The equilibrium constant without additional ligand was measured from the experimental results and set to be 5.4 × 10−4 (Table S1). Numerical simulations are performed by using commercial finite element analysis software (COMSOL Multiphysics 4.4, COMSOL Inc.).
References
Hanna-Attisha, M., LaChance, J., Sadler, R. C. & Champney Schnepp, A. Elevated blood lead levels in children associated with the Flint drinking water crisis: a spatial analysis of risk and public health response. Am. J. Public Health. 106, 283–290 (2016).
Huber, M., Welker, A. & Helmreich, B. Critical review of heavy metal pollution of traffic area runoff: Occurrence, influencing factors, and partitioning. Sci. Tot. Environ. 541, 895–919 (2016).
Baby, J. et al. Toxic effect of heavy metals on aquatic environment. Int. J. Biol. Chem. Sci. 4 (2010).
Heidmann, I. & Calmano, W. Removal of Zn (II), Cu (II), Ni (II), Ag (I) and Cr (VI) present in aqueous solutions by aluminium electrocoagulation. J. Hazard. Mat. 152, 934–941 (2008).
Hua, M. et al. Heavy metal removal from water/wastewater by nanosized metal oxides: a review. J. Hazard. Mat. 211, 317–331 (2012).
Zhao, G., Wu, X., Tan, X. & Wang, X. Sorption of heavy metal ions from aqueous solutions: a review. Open Colloid Sci. J. 4 (2010).
Dermentzis, K., Christoforidis, A. & Valsamidou, E. Removal of nickel, copper, zinc and chromium from synthetic and industrial wastewater by electrocoagulation. Int. J. Environ. Sci. 1, 697 (2011).
Bilal, M. et al. Waste biomass adsorbents for copper removal from industrial wastewater—a review. J. Hazard. Mat. 263, 322–333 (2013).
Gupta, V. K. et al. Chemical treatment technologies for waste-water recycling—an overview. Rsc Adv. 2, 6380–6388 (2012).
Huisman, J. L., Schouten, G. & Schultz, C. Biologically produced sulphide for purification of process streams, effluent treatment and recovery of metals in the metal and mining industry. Hydrometallurgy. 83, 106–113 (2006).
Fu, F. et al. Application of a novel strategy—Advanced Fenton-chemical precipitation to the treatment of strong stability chelated heavy metal containing wastewater. Chem. Eng. J. 189, 283–287 (2012).
Mirbagheri, S. A. & Hosseini, S. N. Pilot plant investigation on petrochemical wastewater treatmentfor the removal of copper and chromium with the objective of reuse. Desalination. 171, 85–93 (2005).
Lakherwal, D. Adsorption of heavy metals: a review. Int. J. Environ. Res. Dev. 4, 41–48 (2014).
Salam, O. E. A., Reiad, N. A. & ElShafei, M. M. A study of the removal characteristics of heavy metals from wastewater by low-cost adsorbents. J. Adv. Res. 2, 297–303 (2011).
Ali, I. The quest for active carbon adsorbent substitutes: inexpensive adsorbents for toxic metal ions removal from wastewater. Sep. Purif. Rev. 39, 95–171 (2010).
Saleh, T. A. & Gupta, V. K. Column with CNT/magnesium oxide composite for lead (II) removal from water. Environ. Sci. Pollut. Res. 19, 1224–1228 (2012).
Hui, K. S., Chao, C. Y. H. & Kot, S. C. Removal of mixed heavy metal ions in wastewater by zeolite 4A and residual products from recycled coal fly ash. J. Hazard. Mat. 127, 89–101 (2005).
Gupta, V. K. & Tawfik, A. S. Sorption of pollutants by porous carbon, carbon nanotubes and fullerene-An overview. Environ. Sci. Pollut. Res. 20, 2828–2843 (2013).
Gupta, V. K. et al. “Design parameters for fixed bed reactors of activated carbon developed from fertilizer waste for the removal of some heavy metal ions”. Waste Manag. 17, 517–522 (1998).
Karnib, M., Kabbani, A., Holail, H. & Olama, Z. Heavy metals removal using activated carbon, silica and silica activated carbon composite. Energy Procedia. 50, 113–120 (2014).
Erdem, E., Karapinar, N. & Donat, R. The removal of heavy metal cations by natural zeolites. J. Colloid. Interface. Sci. 280, 309–314 (2004).
Abbas, A. et al. Heavy metal removal from aqueous solution by advanced carbon nanotubes: critical review of adsorption applications. Sep. Purif. Technol. 157, 141–161 (2016).
Davoren, M. et al. In vitro toxicity evaluation of single walled carbon nanotubes on human A549 lung cells. Toxicol. In Vitro. 21, 438–448 (2007).
Gupta, V. K., Agarwal, S. & Tawfik, A. S. Synthesis and characterization of alumina-coated carbon nanotubes and their application for lead removal. J. Hazard. Mater. 185, 17–23 (2011).
Bunting, J. W. & Thong, K. M. Stability constants for some 1: 1 metal–carboxylate complexes. Can. J. Chem. 48, 1654–1656 (1970).
Mahdavi, M. et al. Synthesis, surface modification and characterisation of biocompatible magnetic iron oxide nanoparticles for biomedical applications. Molecules. 18, 7533–7548 (2013).
Kalhapure, R. S. & Akamanchi, K. G. A novel biocompatible bicephalous dianionic surfactant from oleic acid for solid lipid nanoparticles. Colloids Surf. B. Biointerfaces 105, 215–222 (2013).
Zhang, J. et al. Self-assembled polymeric nanoparticles based on oleic acid-grafted chitosan oligosaccharide: biocompatibility, protein adsorption and cellular uptake. J. Mater. Sci. Mater. Med. 23, 1775–1783 (2012).
Cai, J. et al. Variation in the coordination mode of arenedisulfonates to copper (II): synthesis and structural characterization of six copper (II) arenedisulfonate complexes. J. Chem. Soc. Dalton Trans. 7, 1137–1142 (2001).
Schaap, W. B. & McMasters, D. L. A Polarographic Study of Mixed-ligand Complex Formation; Complexes of Copper and Cadmium with Oxalate Ion and Ethylenediamine. J. Am. Chem. Soc. 83, 4699–4706 (1961).
Wu, M. et al. The effect of copper from water and food: changes of serum nonceruloplasmin copper and brain’s amyloid-beta in mice. Food. Funct. 7, 3740–3747 (2016).
Fundamentals of electrochemical deposition (Vol. 45); Paunovic, M. & Schlesinger, M. John Wiley & Sons (2006).
Dynamics of adsorption at liquid interfaces: theory, experiment, application (Vol. 1); Dukhin, S. S., Kretzschmar, G. & Miller, R. Elsevier, (1995).
Griffiths, S. K. et al. Modeling electrodeposition for LIGA microdevice fabrication. Microsyst. Technol. 4, 98–101 (1998).
Table of Regulated Drinking Water. https://www.epa.gov/ground-water-and-drinking-water/table-regulated-drinking-water-contaminants#one (accessed Aug 31, 2017).
Gray, H. B. Molecular orbital theory for transition metal complexes. J. Chem. Educ. 41, 2 (1964).
Powell, B. D. & Sheppard, N. Infrared spectra and the stabilities of chelate metal–ethylenediamine complexes. J. Chem. Soc. (Resumed) 224, 1112–1114 (1961).
Metal ammine formation in aqueous solution; Bjerrum, J. & Andersen, P. Munksgaard (1945).
Kariuki, S. & Dewald, H. D. Evaluation of diffusion coefficients of metallic ions in aqueous solutions. Electroanalysis. 8, 307–313 (1996).
Quickenden, T. I. & Jiang, X. The diffusion coefficient of copper sulphate in aqueous solution. Electrochim. Acta. 29, 693–700 (1984).
Iwahashi, M. et al. Self-diffusion, dynamical molecular conformation, and liquid structures of n-saturated and unsaturated fatty acids. J. Phys. Chem. B. 104, 6186–6194 (2000).
Yamamoto, S. et al. Dynamic molecular behavior of semi-fluorinated oleic, elaidic and stearic acids in the liquid state. J. Oleo. Sci. 61, 649–657 (2012).
Acknowledgements
This research was supported by the Mid-Career Researcher Support Program (No. 2016R1A2B3014157) and by Basic Science Research Program (No. 2016R1A6A1A03012845) through the National Research Foundation of Korea funded by the Ministry of Science, ICT, and Future Planning.
Author information
Authors and Affiliations
Contributions
T.K. conceived the concept. J.C., S.Y., and T.K. designed and organized the experiments. J.C., S.Y. performed the experiments. W.L. and D.K. contributed to the simulations. J.C., S.Y., and T.K. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chang, J., Yoo, S., Lee, W. et al. Spontaneous Phase Transfer-Mediated Selective Removal of Heavy Metal Ions Using Biocompatible Oleic Acid. Sci Rep 7, 16727 (2017). https://doi.org/10.1038/s41598-017-17092-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-17092-9
This article is cited by
-
Decontamination of water co-polluted by copper, toluene and tetrahydrofuran using lauric acid
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.