Abstract
Orthrozanclus is a shell-bearing, sclerite covered Cambrian organism of uncertain taxonomic affinity, seemingly representing an intermediate between its fellow problematica Wiwaxia and Halkieria. Attempts to group these slug-like taxa into a single ‘halwaxiid’ clade nevertheless present structural and evolutionary difficulties. Here we report a new species of Orthrozanclus from the early Cambrian Chengjiang Lagerstätte. The scleritome arrangement and constitution in this material corroborates the link between Orthrozanclus and Halkieria, but not with Wiwaxia — and calls into question its purported relationship with molluscs. Instead, the tripartite construction of the halkieriid scleritome finds a more compelling parallel in the camenellan tommotiids, relatives of the brachiopods and phoronids. Such a phylogenetic position would indicate the presence of a scleritome in the common ancestor of the three major trochozoan lineages, Mollusca, Annelida and Brachiozoa. On this view, the absence of fossil Ediacaran sclerites is evidence against any ‘Precambrian prelude’ to the explosive diversification of these phyla in the Cambrian, c. 540–530 million years ago.
Similar content being viewed by others
Introduction
The Cambrian fossil record is renowned for the morphologically puzzling organisms that it preserves. Such taxa often represent long-extinct combinations of characters, offering a unique perspective on the early origin of modern body plans – presuming, of course, that relationships with modern groups can be established1. The reconstructed origins of the molluscan lineage, for example, have been overhauled in order to accommodate two emblematic Cambrian taxa, Halkieria and Wiwaxia 2,3,4,5,6,7,8,9. These two genera bear superficially similar sclerites, which occur the world over as carbonaceous and mineralized microfossils10,11,12,13; the grouping Sachitida was erected to reflect this perceived commonality14. The case for phylogenetic proximity was strengthened by the discovery of articulated specimens in the Burgess Shale and Sirius Passet Lagerstätten, which showed that the sclerites of both taxa were dorsal and imbricating2,15,16. This arguably overlooks some notable differences between the two genera – Halkieria has dorsal valves, Wiwaxia bears elongate spines, and the sclerites of the two groups are far from identical – but suggestions that these differences might denote a degree of phylogenetic separation17,18 were soon countered by the description of the Burgess Shale animal Orthrozanclus reburrus, which incorporates a single Halkieria-like valve within a spiny non-mineralized scleritome19. The ‘halwaxiid’ clade, incorporating Wiwaxia, Orthrozanclus, Halkieria and other sachitids, was erected on the basis that the scleritomes of these taxa were consequently homologous. A new species of Orthrozanclus from the Chengjiang lagerstätten, however, prompts a re-evaluation of the basis for a halwaxiid grouping, and calls into question the position of Halkieria and Orthrozanclus in molluscan evolution.
Results
Systematic Palaeontology
Superphylum Lophotrochozoa
Family Halkieriidae Poulsen 196720.
Remarks
Orthrozanclus falls within the emended diagnosis of Halkieriidae provided by Conway Morris and Peel 199516, negating the need for a separate family Orthrozanclidae19.
Orthrozanclus Conway Morris and Caron 200719
Orthrozanclus elongata Zhao et Smith n. sp. Figs 1 and 2.
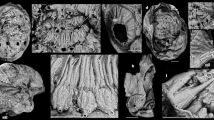
Orthrozanclus elongata n. sp. (a–e) NIGPAS164893, paratype. (a,b) part and counterpart of entire specimen. (c) part, anterior region, dorsal sclerites exhibit relief. (d) counterpart, showing ‘fanning’ of spines at posterior. (e) counterpart, showing arrangement of spines and ventrolateral sclerites. (f–l) NIGPAS164892, holotype. (f,g) part and counterpart of entire specimen. (h) part, anterior region, dark field illumination emphasizes relief of ventrolateral sclerites. (i) counterpart, anterior region, bright field illumination emphasizes sclerite margins. (j) counterpart, left lateral region showing inclination of spines relative to the bedding plane – the anterior edge (+) is raised above the posterior edge (−) – and ribs on ventrolateral sclerites (k) and dorsolateral spines (l). Abbreviations: ds, dorsal sclerites; sp, spines; valv, valve; vls, ventrolateral sclerites. Bars = 1 mm except k, 100 µm.
Type material
NIGPAS 164892 (Fig. 1f–l), holotype; 164893 (Fig. 1a–e), paratype, each comprising part and counterpart and preserved in the characteristic Chengjiang fashion21 as weathered aluminosilicate films associated with superficial iron oxides.
Provenance
Maotianshan Shale, Yu’anshan Formation, Eoredlichia-Wutingaspis Zone, Cambrian Series 2, Stage 3. The holotype was collected from Jiucun, near Chengjiang (24°41’33” N, 102°59’26” E); the paratype from Yuanbaocun, Chenggong, Kunming (24°49’24” N, 102°49’14” E), Yunnan, southwest China.
Diagnosis
Species of Orthrozanclus with elongate (c. 1:7) aspect ratio. Dorsal sclerites mineralized, oblong in aspect, occurring in regular rows. Dorsolateral spinose sclerites flat, ribbed and blade-like, without central cavity.
Description
The two specimens of Orthrozanclus elongata n. sp. (Fig. 1) are 20 mm long and a uniform 3 mm in width. Their dorsal scleritome bears an anterior valve and three zones of sclerites: a medial zone covers the flattened dorsal surface of the organism, and inner and outer peripheral zones surround its flanks. Its rectangular outline, rounded anterior and posterior ends and overall architecture resemble that of O. reburrus.
The medial sclerite zone comprises transverse chevron-like rows, each containing fourteen sclerites, seven on each side (Figs 1c and 2). These sclerites measure 220 × 90 µm, are oblong to teardrop shaped, and lie flat to the body. Their pronounced three-dimensionality distinguishes these sclerites from those in other zones, and – in view of the well-defined margins of the individual sclerites – indicates an originally mineralized composition. Neither phosphatization of labile tissue22 nor secondary infilling of original cavities (as observed in Wiwaxia and O. reburrus 8,19) are consistent with the observed preservation. The enhanced relief of the dorsal elements relative to the dorsolateral and ventral sclerites presumably reflects original three-dimensional structure.
The spinose dorsolateral sclerites reach 6 mm in length, and form a c. 45° angle to the body, with their tips directed posteriad (Figs 1 and 2). They are regularly spaced (Fig. 1h–j) in a single series that encircles the body, surrounding the anterior margin of the valve and the posterior of the dorsal area (Fig. 1d). The spines bear ribs, but are otherwise flat in cross-section; in contrast to O. reburrus, there is no evidence of a central cavity (Fig. 1j–l). Their flat surfaces lie at an angle of 20–45° to the bedding surfaces – indicating a high original angle (Fig. 1j). Apparent differences in width between spines can be attributed to differential angles of burial relative to the bedding surface. The proximal configuration of the spines (Fig. 1l) has a putative similarity to the auricle of certain Halkieria sclerites10.
Dagger-shaped (cultrate) sclerites occupy the lateral surfaces of the organism, extending to partly enclose the ventral surface (Fig. 1h). The best-preserved sclerites bear a bilaterally symmetrical series of ribs (Fig. 1k). These sclerites (but not the spines or dorsal sclerites) encircle the valve to enclose the anterior margin of the organism (Fig. 1h); the tips of the sclerites were originally directed dorsally, rather than radially as depicted for O. reburrus.
The valve is denoted by a region of pronounced relief, presumably reflecting a robustly mineralized original constitution (Fig. 1c–d,h). The shape of the valve suggests a posterior umbo: though the opposite has been interpreted in O. reburrus, the umbo is difficult to locate with certainty in either taxon. The posterior and anterior margins of the valve are overlapped by sclerites of the medial and outer peripheral zones respectively (Fig. 1h).
A three-dimensionally preserved structure, presumably representing the digestive tract, follows the main body axis (Figs. 1d,h and 3a). As with the presumed gut of O. reburrus, this begins slightly posterior to the shell; the gap between the gut and the shell marks a 90° bend in the axis of NIGPAS 164892, reminiscent of an equivalent bend in many Halkieria fossils (see ref.16 and Fig. 3b).
Scleritome arrangement in Orthrozanclus elongata n. sp. (a, NIGPAS164892), Halkieria evangelista (b, Sedgwick Museum of Earth Sciences X24914.2) and Wiwaxia corrugata (c, Royal Ontario Museum 61510). The Orthrozanclus (d) and Halkieria (e) scleritomes are arranged in three concentric zones: a medial zone of oblique transverse rows (vermillion); a dorsolateral ‘inner peripheral’ zone (purple), containing long spines in Orthrozanclus and cultrate sclerites in Halkieria; and a ventrolateral ‘outer peripheral’ zone, containing cultrate (Orthrozanclus) or siculate (Halkieria) sclerites. Dailyatia bacata (f) is reconstructed as having a medial region containing A and B sclerites and a single peripheral zone of C sclerites. The approximately 4:1 ratio of C1:A sclerites and 4:2 ratio of C2:B sclerites35 is taken to indicate that C sclerites occur at twice the frequency of elements in the medial zone. The Wiwaxia scleritome (g) comprises eight transverse rows (shaded) intersected by two rows of intermittently spaced spines.
Discussion
The new material strengthens the case for a close relationship between Orthrozanclus and Halkieria (Fig. 3). Mineralized dorsal sclerites, occurring in oblique transverse rows behind an anterior shell, are now evident in both taxa (Fig. 3a–b,d–e) – even if Orthrozanclus has no counterpart to the posterior shell of Halkieria. And each taxon exhibits two peripheral sclerite zones: the inner, dorsolateral zone contains long spines in Orthrozanclus and short cultrate sclerites in Halkieria; the outer, ventrolateral zone bears regularly spaced cultrate sclerites in Orthrozanclus and siculate sclerites in Halkieria (Fig. 3a–b,d–e). Homology of the zones is thus recognized based on their position, rather than the shape or constitution of the sclerites that they contain.
An equivalent sclerite arrangement was once envisaged in Wiwaxia 15,23, but recent studies8,9 have shown that the Wiwaxia scleritome conforms to a metameric architecture, comprising 8–9 transverse rows (Fig. 3c,g). Even though the most lateral sclerites are morphologically distinct in certain Wiwaxia species, they belong to the same transverse rows as the medial sclerites, rather than forming a distinct peripheral zone that surrounds the entire circumference of the organism9,24 (Fig. 3c). The two dorsal rows of spines in Wiwaxia are highly variable in their number, size, spacing, and orientation, both within and between species8,15,25, so do not form a distinct region of the scleritome architecture. As such, the peripheral sclerite zones in Orthrozanclus (Fig. 3a) and Halkieria (Fig. 3b) have no counterpart in Wiwaxia, and it is not clear that the two scleritome layouts are equivalent in any meaningful way – undermining the case for a ‘halwaxiid’ clade.
Are halkieriids molluscs?
At a broader taxonomic level, perceived similarities in scleritome construction are said to indicate a close relationship between halkieriids and aculiferan molluscs4,6,19,26,27,28. This position has most recently been propounded based on the Ordovician aculiferan Calvapilosa, which has been interpreted as a close relative of halkieriids28. The evidence that Calvapilosa is an aculiferan is strong; the evidence that it is a halkieriid warrants more careful consideration.
Sclerites – a likely inheritance from the ancestral lophotrochozoan17,18,29,30 – have been assembled into scleritomes on multiple occasions: the scleritomes of the scaly-footed gastropods31 and chrysopetalid annelids32,33, for example, represent independent innovations that are demonstrably unique to the respective clades31. Indeed, multiple groups incorporate both shell-like valves and mineralized plates into dorsal imbricating skeletons – witness machaeridians, Pelagiella and certain tommotiids, who have affinities with annelids, gastropods and brachiopods, respectively34,35,36,37.
It is therefore significant that the Calvapilosa scleritome prominently lacks the differentiated sclerite morphologies and peripheral morphological zones that characterize halkieriids. Halkieriid sclerites exhibit a broad range of morphologies, but none resemble the slender, spinose sclerites of Calvapilosa 28. The central cavity present in both halkieriid and Calvapilosa sclerites has little taxonomic value (discussed in ref.8). The shell of Calvapilosa is a markedly different shape to that of Halkieria, and bears depressions (interpreted as aesthete canals) that have no counterpart in halkieriid shells.
In the absence of any demonstrably equivalent constructional features or an unambiguously close genetic relationship, it is difficult to defend the homology of the halkieriid scleritome with that of Calvapilosa.
One thing that Calvapilosa (and Wiwaxia 7) does establish is that where a radula is present, it preserves readily in Burgess Shale-type conditions. But importantly, this robust and distinctive multi-row mouthpart is prominently absent in both Orthrozanclus and Halkieria. (A potentially radula-like structure evident in a single specimen of Halkieria 16 corresponds in angle and dimensions with diagonal displacements of sclerites elsewhere in the scleritome, and is not associated with any diagnostically radular characteristics, such as teeth38; its identification as a radula must be considered unproven.) As a radula was present in the ancestral mollusc39, and perhaps deeper in the trochozoan lineage9, its absence in halkieriids is difficult to reconcile with a molluscan affinity.
Could halkieriids be tommotiids?
One set of organisms whose scleritomes exhibit an intriguing similarity with those of halkieriids are the camenellan tommotiids, a group that is implicated in the earliest ancestry of brachiopods40,41,42,43. The scleritome of the kennardiid camenellan Dailyatia 35 has been reconstructed as comprising median and peripheral fields (Fig. 3f). The medial region bears a series of transverse ‘rows’ of one or two sclerites (A and B sclerites); the peripheral field bears dorsally-directed sclerites with a distinct morphology (C sclerites). As no fully articulated camenellan scleritomes have yet been found, this comparison does of course warrant a degree of caution, particularly in view of the tube-like configuration of other tommotiid scleritomes37,44,45,46 – but the general arrangement reconstructed from sclerite asymmetry, fused arrays of sclerites, morphological proportions and relative sclerite frequency is fundamentally compatible with a halkieriid-like construction. Taking this further, sclerites in the peripheral zones of camenellan scleritomes occur in dextral and sinistral forms35,47, as do the sclerites of Halkieria 10 and – in view of the symmetrical scleritome arrangement revealed by O. elongata n. sp. – those of Orthrozanclus. Camenellan sclerites show continuous variation within a particular morphological category47 – as do spines in the dorsolateral zone of the Orthrozanclus scleritome. Certain camenellan sclerites48 exhibit a tuberculate ornament and apical tip that correspond closely to the sclerites of, for example, Halkieria mira (see Figs 4, 6 in ref.49). More speculatively, the camerate construction of certain halkieriid sclerites6,10 might find a parallel in the internal chambers of Kelanella sclerites or Micrina valves47,50.
Looking more widely, the paired muscle scars and shelly internal projections evident in Morph A valves of Oikozetetes 51,52, some of the best documented halkieriid shells, have possible parallels in the equivalent paired muscle scars and internal processes present in the mitral sclerite of the tommotiids Micrina 44 and Dailyatia 35 and the operculum of hyolithids53 (potential relatives of tommotiids54).
In view of these similarities, we therefore propose that halkieriids and camenellans may be closely related (Fig. 4). If camenellans are derived from an ancestrally tube-dwelling tommotiid55, then a vagrant, slug-like habit would represent an apomorphy of a halkieriid + camenellan clade; alternatively, the halkieriid condition may be ancestral for the tommotiid + brachiopod lineage16,40, with the bivalved condition perhaps arising through paedomorphic retention of an ancestral state41.
One obvious objection to this taxonomic hypothesis is that camenellan elements are composed of calcium phosphate, whereas halkieriids secreted calcium carbonate, probably in the form of aragonite56. This said, tommotiids and early brachiopods deploy a wide variety of biominerals (Fig. 4): examples exist of non-mineralized, agglutinated, aragonitic, calcitic, phosphatic, and mixed calcite-phosphate shells54,57,58.
Switching from one biomineral to another is generally the exception rather than the rule59,60, but members of the brachiopod lineage have nevertheless changed their primary biomineral from phosphate to calcite61,62, from calcite to aragonite63, and from phosphate to a non-mineralized configuration64; indeed, some living brachiopods switch from using silica to calcite as they grow65.
On a broader view, biomineralization has evolved multiple times within Metazoa66, seemingly coming and going in Ediacaran lineages according to prevailing environmental conditions67. If this situation persisted into the early Cambrian, it is possible to envision a predominantly non-mineralised brachiopod stem lineage that obtained biomineralization on multiple occasions, each time reflecting the prevailing seawater chemistry. The aragonite mineralogy of halkieriids and hyoliths arose in the aragonite seas of the Fortunian; the calcitic and phosphatic mineralogies of tommotiids and crown-group brachiopods arose in the calcite seas of the Tommotian59. Linguliforms and tommotiid-like specimens from Burgess Shale-type deposits64,68,69 attest to the persistence of non-mineralized skeletons across the brachiopod total group into the mid-Cambrian. In any case, whether modification or multiple innovations account for the diversity of biomineral use in brachiopods and tommotiids, the carbonate elements of halkieriids clearly fit within this gamut.
Conclusion
Because halkieriid-like sclerites occur so early in the Cambrian period70,71, their affinity has profound implications for the timing of early trochozoan evolution. Removing halkieriids from Mollusca would shift the origin of this phylum significantly later: notwithstanding hyoliths (now interpreted as brachiozoans, i.e. brachiopods or phoronids54) and helcionellids (which lack any compelling molluscan apomorphies), there are no strong candidates for crown group molluscs until the Tommotian, and no unequivocal cases until the Late Cambrian1.
If, on the other hand, brachiozoans evolved from a halkieriid-like ancestor, then multi-element scleritomes characterise the earliest brachiozoans as well as molluscs and annelids9 (Fig. 4). The absence of such sclerites among Ediacaran and earliest Cambrian fossil assemblages55 either requires special taphonomic pleading or genuinely denotes that Trochozoans had not yet originated. The subsequent appearance of a rich diversity of exoskeletal elements in the early Cambrian fossil record12,72 points to a very rapid origin and divergence of the key lophotrochozoan phyla in the first few million years of the Cambrian period – representing a truly ‘explosive’ evolutionary radiation.
Methods
The paratype was prepared with a fine blade. Photographs were taken using a Zeiss Stereo Discovery V16 microscope system and processed using TuFuse and the GNU image manipulation program.
Data availability
Specimens are accessioned at the Nanjing Institute of Geology and Palaeontology, Chinese Academy of Sciences (NIGPAS); high resolution images are available at the FigShare repository73.
References
Budd, G. E. & Jensen, S. A critical reappraisal of the fossil record of the bilaterian phyla. Biol. Rev. 75, 253–295 (2000).
Conway Morris, S. & Peel, J. S. Articulated halkieriids from the Lower Cambrian of north Greenland. Nature 345, 802–805 (1990).
Bengtson, S. The cap-shaped Cambrian fossil Maikhanella and the relationship between coeloscleritophorans and molluscs. Lethaia 25, 401–420 (1992).
Vinther, J. & Nielsen, C. The Early Cambrian Halkieria is a mollusc. Zool. Scr. 34, 81–89 (2005).
Caron, J.-B., Scheltema, A. H., Schander, C. & Rudkin, D. A soft-bodied mollusc with radula from the Middle Cambrian Burgess Shale. Nature 442, 159–163 (2006).
Vinther, J. The canal system in sclerites of Lower Cambrian Sinosachites (Halkieriidae: Sachitida): significance for the molluscan affinities of the sachitids. Palaeontology 52, 689–712 (2009).
Smith, M. R. Mouthparts of the Burgess Shale fossils Odontogriphus and Wiwaxia: implications for the ancestral molluscan radula. Proc. R. Soc. B 279, 4287–4295 (2012).
Smith, M. R. Ontogeny, morphology and taxonomy of the soft-bodied Cambrian ‘mollusc’. Wiwaxia. Palaeontology 57, 215–229 (2014).
Zhang, Z.-F., Smith, M. R. & Shu, D.-G. New reconstruction of the Wiwaxia scleritome, with data from Chengjiang juveniles. Sci. Rep. 5, 14810 (2015).
Bengtson, S. & Conway Morris, S. A comparative study of Lower Cambrian Halkieria and Middle Cambrian Wiwaxia. Lethaia 17, 307–329 (1984).
Porter, S. M. Skeletal microstructure indicates chancelloriids and halkieriids are closely related. Palaeontology 51, 865–879 (2008).
Kouchinsky, A. V. et al. Chronology of early Cambrian biomineralization. Geol. Mag. 149, 221–251 (2012).
Slater, B. J., Harvey, T. H. P., Guilbaud, R. & Butterfield, N. J. A cryptic record of Burgess Shale-type diversity from the early Cambrian of Baltica. Palaeontology 60, 117–140 (2017).
Hao, T.-G. Lower Cambrian (Meishucunian) sachitids and their stratigraphic significance [in Chinese]. J. Chengdu Coll. Geol. 2, 84–90 (1981).
Conway Morris, S. The Middle Cambrian metazoan Wiwaxia corrugata (Matthew) from the Burgess Shale and Ogygopsis Shale, British Columbia, Canada. Phil. Trans. R. Soc. Lond. B 307, 507–582 (1985).
Conway Morris, S. & Peel, J. S. Articulated halkieriids from the Lower Cambrian of North Greenland and their role in early protostome evolution. Phil. Trans. R. Soc. B 347, 305–358 (1995).
Butterfield, N. J. A reassessment of the enigmatic Burgess Shale fossil Wiwaxia corrugata (Matthew) and its relationship to the polychaete Canadia spinosa Walcott. Paleobiology 16, 287–303 (1990).
Butterfield, N. J. Hooking some stem-group ‘worms’: fossil lophotrochozoans in the Burgess Shale. BioEssays 28, 1161–1166 (2006).
Conway Morris, S. & Caron, J.-B. Halwaxiids and the early evolution of the lophotrochozoans. Science 315, 1255–1258 (2007).
Poulsen, C. Fossils from the Lower Cambrian of Bornholm. Danske Vidensk. Selsk. Mat. Meddelelser 36, 1–48 (1967).
Zhu, M.-Y., Babcock, L. E. & Steiner, M. Fossilization modes in the Chengjiang Lagerstätte (Cambrian of China): testing the roles of organic preservation and diagenetic alteration in exceptional preservation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 220, 31–46 (2005).
Butterfield, N. J. Leanchoilia guts and the interpretation of three-dimensional structures in Burgess Shale-type fossils. Paleobiology 28, 155–171 (2002).
Eibye-Jacobsen, D. A reevaluation of Wiwaxia and the polychaetes of the Burgess Shale. Lethaia 37, 317–335 (2004).
Yang, J., Smith, M. R., Lan, T., Hou, J.-B. & Zhang, X.-G. Articulated Wiwaxia from the Cambrian Stage 3 Xiaoshiba Lagerstätte. Sci. Rep. 4, 4643 (2014).
Conway Morris, S., Selden, P. A., Gunther, G., Jamison, P. M. & Robison, R. A. New records of Burgess Shale-type taxa from the middle Cambrian of Utah. J. Paleontol. 89, 411–423 (2015).
Sigwart, J. D. & Sutton, M. D. Deep molluscan phylogeny: synthesis of palaeontological and neontological data. Proc. R. Soc. B 274, 2413–2419 (2007).
Sutton, M. D., Briggs, D. E. G., Siveter, D. J., Siveter, D. J. & Sigwart, J. D. A Silurian armoured aplacophoran and implications for molluscan phylogeny. Nature 490, 94–97 (2012).
Vinther, J., Parry, L., Briggs, D. E. G. & Van Roy, P. Ancestral morphology of crown-group molluscs revealed by a new Ordovician stem aculiferan. Nature 542, 471–474 (2017).
Schiemann, S. M. et al. Clustered brachiopod Hox genes are not expressed collinearly and are associated with lophotrochozoan novelties. Proc. Natl Acad. Sci. USA 114, E1913–E1922 (2017).
Peterson, K. J. & Eernisse, D. J. Animal phylogeny and the ancestry of bilaterians: inferences from morphology and 18S rDNA gene sequences. Evol. Dev. 3, 170–205 (2001).
Chen, C., Copley, J. T., Linse, K., Rogers, A. D. & Sigwart, J. How the mollusc got its scales: convergent evolution of the molluscan scleritome. Biol. J. Linn. Soc. 114, 949–954 (2015).
Watson Russell, C. Description of a new species of Arichlidon (Chrysopetalidae: Polychaeta) from the West Atlantic and comparison with the East Atlantic species Arichlidon reyssi. Bull. Mar. Sci. 67, 465–477 (2000).
Watson Russell, C. Patterns of growth and setal development in the deep-sea worm, Strepternos didymopyton (Polychaeta: Chrysopetalidae). Bull. Mar. Sci. 60, 405–426 (1997).
Vinther, J., Van Roy, P. & Briggs, D. E. G. Machaeridians are Palaeozoic armoured annelids. Nature 451, 185–188 (2008).
Skovsted, C. B., Betts, M. J., Topper, T. P. & Brock, G. A. The early Cambrian tommotiid genus Dailyatia from South Australia. Mem. Assoc. Australas. Palaeontol. 48, 1–117 (2015).
Thomas, R. D. K., Vinther, J. & Matt, K. Structure and evolutionary implications of finely preserved chaetae associated with Pelagiella, a stem-group gastropod from the Kinzers Formation (Early Cambrian) at Lancaster, Pennsylvania. In International Palaeontological Congress 3, London, U.K. Programme & Abstracts 375 (2010).
Larsson, C. M. et al. Paterimitra pyramidalis from South Australia: scleritome, shell structure and evolution of a lower Cambrian stem group brachiopod. Palaeontology 57, 417–446 (2014).
Smith, M. R. Putative ‘radula’ in Halkieria evangelista. FigShare. https://doi.org/10.6084/m9.figshare.5501596 (2017).
Scheltema, A. H. The original molluscan radula and progenesis in Aplacophora revisited. J. Nat. Hist. 48, 2855–2869 (2014).
Holmer, L. E., Skovsted, C. B. & Williams, A. A stem group brachiopod from the lower Cambrian: support for a Micrina (halkieriid) ancestry. Palaeontology 45, 875–882 (2002).
Holmer, L. E., Skovsted, C. B., Larsson, C., Brock, G. A. & Zhang, Z. First record of a bivalved larval shell in Early Cambrian tommotiids and its phylogenetic significance. Palaeontology 54, 235–239 (2011).
Balthasar, U., Skovsted, C. B., Holmer, L. E. & Brock, G. A. Homologous skeletal secretion in tommotiids and brachiopods. Geology 37, 1143–1146 (2009).
Harper, D. A. T., Popov, L. E. & Holmer, L. E. Brachiopods: origin and early history. Palaeontology 60, 609–631 (2017).
Holmer, L. E., Skovsted, C. B., Brock, G. A., Valentine, J. L. & Paterson, J. R. The Early Cambrian tommotiid Micrina, a sessile bivalved stem group brachiopod. Biol. Lett. 4, 724–728 (2008).
Skovsted, C. B., Clausen, S., Álvaro, J. J. & Ponlevé, D. Tommotiids from the early Cambrian (Series 2, Stage 3) of Morocco and the evolution of the tannuolinid scleritome and setigerous shell structures in stem group brachiopods. Palaeontology 57, 171–192 (2014).
Skovsted, C. B., Brock, G. A., Topper, T. P., Paterson, J. R. & Holmer, L. E. Scleritome construction, biofacies, biostratigraphy and systematics of the tommotiid Eccentrotheca helenia sp. nov. from the Early Cambrian of South Australia. Palaeontology 54, 253–286 (2011).
Devaere, L. et al. The tommotiid Kelanella and associated fauna from the early Cambrian of southern Montagne Noire (France): implications for camenellan phylogeny. Palaeontology 57, 979–1002 (2014).
Devaere, L. & Skovsted, C. B. New early Cambrian sclerites of Lapworthella schodakensis from NE Greenland: advancements in knowledge of lapworthellid taxonomy, sclerite growth and scleritome organization. Geol. Mag. 154, 1061–1072 (2017).
Conway Morris, S. & Chapman, A. J. Lower Cambrian halkieriids and other coeloscleritophorans from Aksu-Wushi, Xinjiang, China. J. Paleontol. 71, 6–22 (1997).
Li, G.-X. & Xiao, S.-H. Tannuolina and Micrina (Tannuolinidae) from the Lower Cambrian of eastern Yunnan, South China, and their scleritome reconstruction. J. Paleontol. 78, 900–913 (2004).
Paterson, J. R., Brock, G. A. & Skovsted, C. B. Oikozetetes from the early Cambrian of South Australia: implications for halkieriid affinities and functional morphology. Lethaia 42, 199–203 (2009).
Jacquet, S. M., Brock, G. A. & Paterson, J. R. New data on Oikozetetes (Mollusca, Halkieriidae) from the lower Cambrian of South Australia. J. Paleontol. 88, 1072–1084 (2014).
Martí Mus, M. & Bergström, J. The morphology of hyolithids and its functional implications. Palaeontology 48, 1139–1167 (2005).
Moysiuk, J., Smith, M. R. & Caron, J.-B. Hyoliths are Palaeozoic lophophorates. Nature 541, 394–397 (2017).
Budd, G. E. & Jackson, I. S. C. Ecological innovations in the Cambrian and the origins of the crown group phyla. Phil. Trans. R. Soc. B 371, 20150287 (2016).
Porter, S. M. Halkieriids in Middle Cambrian phosphatic limestones from Australia. J. Paleontol. 78, 574–590 (2004).
Balthasar, U. An Early Cambrian organophosphatic brachiopod with calcitic granules. Palaeontology 50, 1319–1325 (2007).
Zhang, Z.-F. et al. An early Cambrian agglutinated tubular lophophorate with brachiopod characters. Sci. Rep. 4, 4682 (2014).
Porter, S. M. Calcite and aragonite seas and the de novo acquisition of carbonate skeletons. Geobiology 8, 256–77 (2010).
Zhuravlev, A. Y. & Wood, R. A. Eve of biomineralization: Controls on skeletal mineralogy. Geology 36, 923–926 (2008).
Balthasar, U. Mummpikia gen. nov. and the origin of calcitic-shelled brachiopods. Palaeontology 51, 263–279 (2008).
Skovsted, C. B. A silicified tommotiid from the lower Cambrian of Greenland. Bull. Geosci. 91, 553–559 (2016).
Balthasar, U. et al. Relic aragonite from Ordovician-Silurian brachiopods: Implications for the evolution of calcification. Geology 39, 967–970 (2011).
Holmer, L. E. & Caron, J.-B. A spinose stem group brachiopod with pedicle from the Middle Cambrian Burgess Shale. Acta Zool. 87, 273–290 (2006).
Williams, A., Lüter, C. & Cusack, M. The nature of siliceous mosaics forming the first shell of the brachiopod Discinisca. J. Struct. Biol. 134, 25–34 (2001).
Knoll, A. H. In Reviews in Mineralogy and Geochemistry (eds Dove, P. M., DeYoreo, J. J. & Weiner, S.) 54, 329–356 (Mineralogical Society of America, 2003).
Wood, R., Ivantsov, A. Y. & Zhuravlev, A. Y. First macrobiota biomineralization was environmentally triggered. Proc. R. Soc. B 284, 20170059 (2017).
Balthasar, U. & Butterfield, N. J. Early Cambrian ‘soft-shelled’ brachiopods as possible stem-group phoronids. Acta Pal. Pol. 54, 307–314 (2009).
Zhang, Z.-F. et al. A sclerite-bearing stem group entoproct from the early Cambrian and its implications. Sci. Rep. 3, 1066 (2013).
Zhu, M.-Y., Zhuravlev, A. Y., Wood, R. A., Zhao, F. & Sukhov, S. S. A deep root for the Cambrian explosion: Implications of new bio- and chemostratigraphy from the Siberian Platform. Geology 45, 459–462 (2017).
Kouchinsky, A. et al. Terreneuvian stratigraphy and faunas from the Anabar Uplift, Siberia. Acta Pal. Pol. 62, 311–440 (2017).
Telford, M. J., Budd, G. E. & Philippe, H. Phylogenomic insights into animal evolution. Curr. Biol. 25, R876–R887 (2015).
Zhao, F.-C., Smith, M. R., Yin, Z.-J., Zeng, H. & Zhu, M.-Y. High resolution images of Orthrozanclus elongata. FigShare. https://doi.org/10.6084/m9.figshare.c.3841387 (2017).
Acknowledgements
Research was supported by the Strategic Priority Research Program (B) of the Chinese Academy of Sciences (XDB18000000, XDB10010101); and the National Natural Science Foundation of China (41472012, 41661134048). Lars Holmer and Tim Topper provided constructive thoughts on an early draft of the manuscript. We thank Mr. Huihong Zhang for field assistance and finding the paratype specimen. We are grateful to reviewers for invaluable suggestions and comments.
Author information
Authors and Affiliations
Contributions
Conceptualization, F.C.Z., M.R.S.; Investigation, F.C.Z., M.R.S., Z.J.Y., H.Z., G.X.L., M.Y.Z.; Resources: F.C.Z.; Writing – Original Draft, M.R.S.; Writing – Review & Editing, F.C.Z., M.R.S., M.Y.Z.; Visualization, F.C.Z., M.R.S.; Funding Acquisition, F.C.Z.; Supervision, F.C.Z., M.R.S., M.Y.Z.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, F., Smith, M.R., Yin, Z. et al. Orthrozanclus elongata n. sp. and the significance of sclerite-covered taxa for early trochozoan evolution. Sci Rep 7, 16232 (2017). https://doi.org/10.1038/s41598-017-16304-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-16304-6
This article is cited by
-
Protomelission is an early dasyclad alga and not a Cambrian bryozoan
Nature (2023)
-
Exceptionally preserved early Cambrian bilaterian developmental stages from Mongolia
Nature Communications (2021)
-
Death march of a segmented and trilobate bilaterian elucidates early animal evolution
Nature (2019)
-
Sclerite-bearing annelids from the lower Cambrian of South China
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.