Abstract
Phaeocystis globosa, one of the most typical red tide-forming species, is usually mixed in the food composition of rotifers. To explore how rotifers respond by adjusting life history strategy when feeding on different quality foods, we exposed the rotifer Brachionus plicatilis to cultures with 100% Chlorella, a mixture of 50% P. globosa and 50% Chlorella, or 100% P. globosa. Results showed that rotifers exposed to 100% Chlorella or to mixed diets produced more total offspring and had higher age-specific fecundity than those exposed to 100% P. globosa. Food combination significantly affected the net reproduction rates of rotifers. By contrast, rotifers that fed on 100% P. globosa or on mixed diets had a longer lifespan than those fed on 100% Chlorella. The overall performances (combining reproduction and lifespan together) of rotifers cultured in 100% Chlorella or mixed diets were significantly higher than those cultured in 100% P. globosa. In general, Chlorella favors rotifers reproduction at the cost of shorter lifespan, whereas P. globosa tends to extend the lifespan of rotifers with lower fecundity, indicating that trade-off exists between reproduction and lifespan under different food conditions. The present study also suggests that rotifers may have the potential to control harmful P. globosa.
Similar content being viewed by others
Introduction
Rotifer, as an important component of zooplankton community, plays an important role in linking the primary producers and higher trophic consumers1. Rotifers are considered as heterophagous microplanktonic filter feeder and can live in environments where resource types, quality, and abundance change dramatically. Under favorable food condition, rotifers generally reproduce rapidly2, that is, Brachionus calyciflorus had a maximal production rate when fed on Chlorella vulgaris at 750 µg mL−1 (wet weight). Facing unfavorable environments, rotifers can survive by adjusting their life history strategies. For example, B. calyciflorus would extend lifespan by reducing the growth and reproduction when feeding on bad food3. Kirk4 found the trade-off between reproduction and lifespan in the rotifers Keratella cochlearis and Platyius quadricornus under limited food. Based on the population abundance, reproduction and survival, rotifers are recently used for monitoring the toxicity of harmful algae, such as Prorocentrum micans and Heterosigma akashiwo 5. Some harmful algae at low concentrations could sustain the individual survival of rotifers, as well as the reproduction and population growth5.
Harmful algal blooms have become global environmental problems, which affect the balance of aquatic ecosystem and development in aquaculture and fishing industries6,7. The haptophyte Phaeocystis globosa, one of the most widespread marine algae, is a harmful bloom-forming phytoplankton8. It can grow well in the entire marine phytoplankton community from polar to temperate waters during the spring–summer transition9. P. globosa exists in two major morphotypes as follows: mucilaginous colonies and small single cells10. Like most Chrysophyta species, the small single cells have two flagella that help the cells move fast. The harmful effect of P. globosa derives from its ability to form red tide and produce hemolytic toxins11,12. Eight toxic compounds from P. globosa have been separated, some of which were lethal to aquatic animals, such as the brine shrimp Artemia salina, the juvenile Epinephelus akaara fish13, and the Gadus morhua larva14. P. globosa is generally not a good food for most zooplankton species15. However, during the early stage of red tide development, some of the zooplankton species, such as the copepods Temora longicornis and Acartia clause, could have high abundances16, indicating that copepods can survive well when fed with P. globosa. Some protozoa, such as ciliates could well graze the solitary cells of P. globosa 17,18.
To our knowledge, no report exists about the possible trade-off between reproduction and lifespan in the rotifers that fed on P. globosa to date. Considering that rotifer usually co-exists with P. globosa in nature and some zooplanktons survive on P. globosa, we hypothesized that (1) the rotifer Brachionus plicatilis can survive but reproduction may be affected when feeding on P. globosa, (2) trade-off exists between reproduction and lifespan of B. plicatilis under different food conditions, and (3) B. plicatilis may have the potential to control the harmful P. globosa. To test the hypotheses, we used both P. globosa (generally not a good food for zooplankton) and Chlorella sp. (generally a good food for rotifers) as experimental food to culture the widespread rotifer B. plicatilis under three different food combinations with the same dry weight, namely, 100% Chlorella sp. (100%C), a mixture of 50% P. globosa and 50% Chlorella sp. (50%C + 50%P), and 100% P. globosa (100%P), and recorded the life-history parameters. Results supported the hypotheses and indicated that B. plicatilis may have a potential to control the harmful P. globosa.
Results
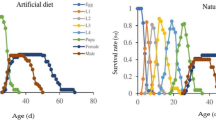
Survival rate and lifespan
No significant difference was found in the three treatments before 150 h. After 150 h, the survival rate of rotifers in 100% C decreased sharply and was significantly lower than those in the other treatments (Fig. 1). However, no significant effect was observed between the rotifers fed with P. globosa and mixed food (P = 0.152).
The mean lifespan of rotifers cultured with 100% C (196 h) was significantly shorter than those in both 50% C + 50% P (P = 0.020) and 100% P (P = 0.016), but no significant difference was observed between lifespans in rotifers fed on 50% C + 50% P (268 h) and 100% P (264 h) (P = 0.860) (Fig. 2a). The median lethal time of rotifers cultured with 100% P was significantly longer than that of rotifers cultured with 100% C (P = 0.024). No significant difference in the median lethal time was observed between 50% C + 50% P and 100% P (P = 0.547) or between 100% C and 50% C + 50% P (P = 0.09) (Fig. 2b).
Reproduction
The age-specific fecundity in all treatments increased with time, and then decreased after a plateau was reached (Fig. 3a). The accumulated total offspring of rotifers cultured with 100% P (maximum value: 7.33) significantly decreased by approximately 50% compared with those cultured in 100% C (maximum value: 14.44) and 50% C + 50% P (maximum value: 13.50) (P < 0.001), but no significant difference was observed between the rotifers cultured in 50% C + 50% P and 100% C (P = 0.091) (Fig. 3b).
Age-specific fecundity (a) of B. plicatilis at different culture times (12-h interval) with different food combinations (100% C, 50% C + 50% P, 100% P) and the accumulated offspring per rotifer (b) of B. plicatilis cultured with different food combinations. Vertical lines represent standard error (n = 3). The significant differences are indicated by different lowercase letters (one-way ANOVA, P < 0.05).
The net reproduction rate (R 0 ), generation time (T), and intrinsic rate of population increase (r m ) are shown in Table 1. The R 0 of rotifers in 100% P was lower than that of the rotifers in 50% C + 50% P (P = 0.016) or 100% C (P = 0.009), but no significant difference was observed in R 0 of rotifers cultured with 50% C + 50% P and 100% C (P = 0.870). Similarly, the r m of rotifers fed on 100% P was lower compared with that of the rotifers cultured with 50% C + 50% P (P = 0.019) or 100% C (P = 0.005), but no significant difference was observed between the rotifers in 50% C + 50% P and 100% C (P = 0.473). The generation time of the three food treatments had no significant difference (P = 0.622).
Overall performance
As for the overall performance, the rotifers cultured with 100% P was significantly lower than those in other two treatments (P < 0.001), but no significant difference was observed between the rotifers fed on 50% C + 50% P and 100% C (P = 0.082) (Fig. 4).
Discussion
The main results of the current study showed that Chlorella sp. favored rotifer reproduction with shorter lifespan, whereas P. globosa tended to extend lifespan with lower reproduction, which demonstrated that trade-off exists between the reproduction and lifespan of the rotifer B. plicatilis under different food conditions. However, the overall performance of the rotifers completely cultured with P. globosa was significantly lower than rotifers fed on Chlorella sp. or mixed foods.
Changes in reproduction and lifespan of rotifer under different foods in our experiments were comparable to the study by Yoshinaga et al.3, which suggested that the rotifer B. plicatilis would have a longer lifespan with the simultaneously decreased reproduction rate when they were badly fed or starved. Energy allocation models provide a very useful framework to investigate the changes in rotifer life history19,20. As all energy used to new growth and reproductive effort come from the ingested food, rotifers should achieve a two-way trade-off when nutrition is limited21, in which the lifespan and reproduction were clearly negatively related22. Our result indicated that when the reproduction rate was high, the mean lifespan was short, confirming the trade-off between reproduction and lifespan when rotifers were fed on different types of food, even under sufficient food conditions. As this trade-off generally occurs when rotifers confront stressful environments23, P. globosa was not as good food as the Chlorella to B. plicatilis.
As food for zooplankton, the quality of algae depends on the nutrition, toxicity, and morph24. In the present study, 100% Chlorella or mixed alga caused the rotifers to produce more offspring. Conversely, 100% P. globosa caused the rotifers to have a lower reproduction rate. Algal size influences the filtration and ingestion rate of zooplankton. Rotifers feeding with large algae generally had a better reproduction than that feeding with small algae25. However, in the present study, P. globosa is larger than the Chlorella sp. (7.2 µm vs. 4.8 µm in diameter), indicating other characters of P. globosa rather than the algal size affecting the rotifers. P. globosa lacks unsaturated fatty acid, which is necessary for the growth and reproduction of rotifers26,27,28. In addition, Phaeocystis sp. were found to be toxic toward Artemia salina, Epinephelus akaara fish13, and Gadus morhua larvae14. Thus, the poor quality and toxic property of P. globosa may have contributed to the inhibited reproduction of rotifers5,29.
When the rotifers were fed on 50% Chlorella + 50% P. globosa, they had longer lifespan compared with those fed on 100% Chlorella and higher accumulated offspring compared with those fed on 100% P. globosa. The overall performance of rotifers under 50% Chlorella + 50% P. globosa was nearly similar to those feeding on 100% Chlorella, indicating that the energy that these rotifers obtained is comparable under the two food conditions. The rotifers under this food combination may partly obtain both the advantages from the two species of algae. For example, some algal extracts and antioxidants in P. globosa could extend the lifespan of rotifers30,31. From the above results, it was concluded that rotifers may have excellent potential to control P. globosa, especially under mixed algal community.
In summary, food combination significantly affected the reproduction and lifespan of rotifers. Chlorella favors rotifers reproduction at the cost of shorter lifespan, whereas P. globosa tends to extend the lifespan of rotifers with lower fecundity, indicating that trade-off exists between reproduction and lifespan under different food conditions. This study also suggests that rotifers may have the potential to control the harmful P. globosa.
Materials and Methods
Plankton and cultivation
Both P. globosa and Chlorella sp. were obtained from Xiamen University and were cultivated in autoclaved seawater (NaCl 24.54 g, KBr 0.1 g, KCl 0.7 g, H3BO3 0.003 g, Na2SO4 4.09 g, NaHCO3 0.185 g, NaF 0.003 g, CaCl2∙2H2O 1.54 g, MgCl2∙6H2O 11.10 g, and SrCl2∙6H2O 0.017 g in every 1 L deionized water; salinity 33‰; pH = 8.3) with f/2 medium at 25 °C under a 14 h:10 h light:dark cycle at 50 µmol photons m−2s−1. The salinity of seawater The resting eggs of B. plicatilis were obtained from the Chinese Academy of Sciences and hatched in beakers with the autoclaved seawater (salinity 33‰, DO > 6.0 mg/L) at 25 °C in a 14 h:10 h light:dark cycle at 20 µmol photons m−2 s−1. Before the experiment, B. plicatilis were fed on Chlorella sp. (3.0 × 106 cells mL−1) under similar conditions as described above.
Experimental design
Three treatments were arranged, and each treatment contained six neonates (<8 h old). The three different food treatments comprised 100% C (3.0 × 106 cells mL−1), 50% C + 50% P (1.5 × 106 cells mL−1 + 2.75 × 106 cells mL−1), or 100% P (5.5 × 106 cells mL−1) with a same dry weight (0.22 mg mL−1). Each neonate in every treatment was raised in 1 mL of autoclaved seawater. The experiment was conducted in 24-welled culture plates. Every 12 h, the living rotifers and the new-born neonates were counted, and the dead maternal individuals and new-born neonates were removed. Every 24 h, the maternal rotifers were transferred into fresh seawater with the corresponding foods. The experiments were run in triplicates and under similar environmental conditions as described above. The experiments proceeded until all individuals of each cohort died. All 24-welled culture plates were shaken every 12 h to avoid the sedimentation of algae. We calculated the net reproduction rate (R 0 ), generation time (T), intrinsic rate of population increase (r m ), and mean lifespan (L) according to Krebs32. To assess the total energy output of rotifers that were cultured in different food combinations, we adopted an index, overall performance (PRL), combining reproduction and lifespan together, based on the fitness formula. The demography parameters were calculated using the formula:
where x is the culture time, l x is age-specific survival, m x is the age-specific fecundity, and N is the total offspring per rotifer.
Statistical analyses
Data were presented as the mean values ± standard error. The effect of food combination on mean lifespan, the accumulated offspring per rotifer, the median lethal time, R 0 , t, R m , and PRL were analyzed by one-way ANOVA. All statistical analyses were performed in Sigmaplot 11.0.
References
Lubzens, E. Raising rotifers for use in aquaculture. Hydrobiologia 147, 245–255 (1987).
Rajendiran, A. & Subramanian, P. Mass production of freshwater rotifer, Brachionus calyciflorus, under different diets and regimes. J. Appl. Aquac. 19, 101–111 (2007).
Yoshinaga, T., Hagiwara, A. & Tsukamoto, K. Life history response and age-specific tolerance to starvation in Brachionus plicatilis of Muller (Rotifera). J. Exp. Mar. Biol. Ecol. 287, 261–271 (2003).
Kirk, K. L. Life-history responses to variable environments: starvation and reproduction in planktonic rotifers. Ecology 78, 434–441 (1997).
Yan, T. et al. Application of rotifer Brachionus plicatilis in detecting the toxicity of harmful algae. Chin. J. Oceanol. Limnol. 27, 376–382 (2009).
Anderson, D. M., Cembella, A. D. & Hallegraeff, G. M. Progress in understanding harmful algal blooms: Paradigm shifts and new technologies forResearch, Monitoring, and Management. Ann. Rev. Mar. Sci. 4, 143–176 (2012).
Smith, W. O. et al. Giantism and its role in the harmful algal bloom species Phaeocystis globosa. Deep-sea Res. PT II 101, 95–106 (2014).
Baudoux, A. C., Noordeloos, A. A. M., Veldhuis, M. J. W. & Brussaard, C. P. D. Virally induced mortality of Phaeocystis globosa during two spring blooms in temperate coastal waters. Aquat. Microb. Ecol. 44, 207–217 (2006).
Dutz, J., Breteler, W. C. M. K. & Kramer, G. Inhibition of copepod feeding by exudates and transparent exopolymer particles (PEP) derived from a Phaeocystis globosa dominated phytoplankton community. Harmful Algae 4, 929–940 (2005).
Rousseau, V., Chretiennot-Dinet, M. J., Jacobsen, A., Verity, P. & Whipple, S. The life cycle of Phaeocystis: state of knowledge and presumptive role in ecology. Biogeochemistry 83, 29–47 (2007).
Tan, S., Hu, X. L., Yin, P. H. & Zhao, L. Photosynthetic inhibition and oxidative stress to the toxic Phaeocystis globosa caused by a diketopiperazine isolated from products of algicidal bacterium metabolism. J. Microbiol. 54, 364–375 (2016).
Cai, G. J. et al. Lysing bloom-causing alga Phaeocystis globosa with microbial algicide: An efficient process that decreases the toxicity of algal exudates. Sci. Rep. 6, 20081 (2016).
Long, C., Luo, W. C., Zhou, H. Y., Gao, C. H. & Huang, R. M. Isolation of toxic compounds from wild Phaeocystis globosa. Chin. Chem. Lett. 27, 247–250 (2016).
Stabell, O. B., Aanesen, R. T. & Eilertsen, H. C. Toxic peculiarities of the marine alga Phaeocystis pouchetii detected by in vivo and in vitro bioassay methods. Aquat. Toxicol. 44, 279–288 (1999).
Nejstgaard, J. C. et al. Zooplankton grazing on Phaeocystis: a quantitative review and future challenges. Biogeochemistry 83, 147–172 (2007).
Grattepanche, J. D., Breton, E., Brylinski, J. M., Lecuyer, E. & Christaki, U. Succession of primary producers and micrograzers in a coastal ecosystem dominated by Phaeocystis globosa blooms. J. Plankton Res. 33, 37–50 (2011).
Verity, P. G. Grazing experiments and model simulations of the role of zooplankton in Phaeocystis food webs. J. Sea Res. 43, 317–343 (2000).
Weisse, T., Tande, K., Verity, P., Hansen, F. & Gieskes, W. The trophic significance of Phaeocystis blooms. J. Marine Syst. 5, 67–79 (1994).
Heino, M. & Kaitala, V. Evolution of resource allocation between growth and reproduction in animals with indeterminate growth. J. Evol. Biol. 12, 423–429 (1999).
Kozlowski, J. Optimal allocation of resources to growth and reproduction: implications for age and size at maturity. Trends Ecol. Evol. 7, 15–19 (1992).
Shertzer, K. W. & Ellner, S. P. State-dependent energy allocation in variable environments: life history evolution of a rotifer. Ecology 83, 2181–2193 (2002).
Snell, T. W. & King, C. E. Lifespan and fecundity patterns in rotifers: the cost of reproduction. Evolution 31, 882–890 (1977).
Bouchnak, R. & Steinberg, C. E. W. Algal diets and natural xenobiotics impact energy allocation in cladocerans. I. Daphnia magna. Limnologica 43, 434–440 (2013).
Nandini, S. & Rao, T. R. Somatic and population growth in selected cladoceran and rotifer species offered the cyanobacterium it Microcystis aeruginosa as food. Aquat. Ecol. 31, 283–298 (1997).
Hotos, G. N. Growth, filtration and ingestion rate of the rotifer Brachionus plicatilis fed with large (Asteromonas gracilis) and small (Chlorella sp.) celled algal species. Aquac. Res. 34, 793–802 (2015).
Tang, K. W., Jakobsen, H. H. & Visser, A. W. Phaeocystis globosa (Prymnesiophyceae) and the planktonic food web: feeding, growth and trophic interactions among grazers. Limnol. Oceanogr. 46, 1860–1870 (2001).
Muller-Navarra, D. C., Brett, M. T., Liston, A. M. & Goldman, C. R. A highly unsaturated fatty acid predicts carbon transfer between primary producers and consumers. Nature 403, 74–77 (2000).
Koch, U., Martin-Creuzburg, D., Grossart, H. P. & Straile, D. Single dietary amino acids control resting egg production and affect population growth of a key freshwater herbivore. Oecologia 167, 981–989 (2011).
Soares, M. C. S., Lürling, M. & Huszar, V. L. M. Responses of the rotifer Brachionus calyciflorus to two tropical toxic cyanobacteria (Cylindrospermopsis raciborskii and Microcystis aeruginosa) in pure and mixed diets with green algae. J. Plankton Res. 32, 999–1008 (2010).
Snare, D. J., Fields, A. M., Snell, T. W. & Kubanek, J. Lifespan extension of rotifers by treatment with red algal extracts. Exp. Gerontol. 48, 1420–1427 (2013).
Snell, T. W., Fields, A. M. & Johnston, R. K. Antioxidants can extend lifespan of Brachionus manjavacas (Rotifera), but only in a few combinations. Biogerontology 13, 261–275 (2012).
Krebs, C. J. Ecology: the experimental analysis of distribution and abundance (2nd edition). 116–157 (Harper & Row, New York, 1978).
Acknowledgements
This study was supported by the National Natural Science Foundation of China (31730105) and the Priority Academic Program Development of Jiangsu Higher Education Institutions.
Author information
Authors and Affiliations
Contributions
Z.Y. and Y.F.S. conceived this study and designed the experiments; Y.F.S., X.Y.H., X.F.X., and L.Z. performed the experiments; Y.F.S., Z.Y., X.X.Z., and Y.H. conducted the data analysis and prepared figures and tables. Y.F.S., Z.Y., and Y.F.C. wrote the manuscript. All of the authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, Y., Hou, X., Xue, X. et al. Trade-off between reproduction and lifespan of the rotifer Brachionus plicatilis under different food conditions. Sci Rep 7, 15370 (2017). https://doi.org/10.1038/s41598-017-15863-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15863-y
This article is cited by
-
Linking genome size variation to population phenotypic variation within the rotifer, Brachionus asplanchnoidis
Communications Biology (2021)
-
Chronic Effects of Bromate on Sexual Reproduction of Freshwater Rotifer Brachionus calyciflorus
Bulletin of Environmental Contamination and Toxicology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.