Abstract
The aim of this study was to investigate whether a novel physiologically relevant marker, InsuTAG (fasting insulin × fasting triglycerides) can predict insulin resistance (IR) and metabolic syndrome (MetS). Data of 618 participants from the Retirement Health and Lifestyle Study (RHLS) were evaluated for the current study. IR was defined by homeostatic model assessment (HOMA-IR) scores. Pearson correlations were used to examine the associations of InsuTAG with HOMA-IR and other markers. Predictions of IR from InsuTAG were evaluated using multiple regression models. Receiver operating characteristic curves (ROC) were constructed to measure the sensitivity and specificity of InsuTAG values and to determine the optimum cut-off point for prediction of IR. InsuTAG was positively correlated with HOMA-IR (r = 0.86; p < 0.0001). InsuTAG is a strong predictor of IR accounting for 65.0% of the variation in HOMA-IR values after adjusting for potential confounders. Areas under the ROC curve showed that InsuTAG (0.93) has higher value than other known lipid markers for predicting IR, with a sensitivity and specificity of 84.15% and 86.88%. Prevalence of MetS was significantly (p < 0.0001) higher in subjects with InsuTAG values greater than optimal cut-off value of 11.2. Thus, InsuTAG appears to be a potential feasible marker of IR and metabolic syndrome.
Similar content being viewed by others
Introduction
The rise in sedentary lifestyle and ready access to calorie-dense processed foods in the modern-day world has created a convenient environment for the development of complex metabolic abnormalities in humans. Insulin, secreted by the pancreas, is one of the key anabolic hormones that tightly regulates glucose and lipid homeostasis1. To maintain this homeostasis, insulin supresses hepatic glucose production, enhances glucose uptake in muscle and fat tissues, increases lipogenesis and regulates hepatic transport of very low-density lipoprotein (VLDL) associated triglycerides (TG)1. Chronic over-nutrition results in disruption of insulin signalling pathways leading to increased hepatic secretion of VLDL and possible decrease in the clearance of TG rich lipoproteins. As a result, levels of circulating TG are increased2, and this is commonly observed in insulin resistance (IR) and metabolic syndrome3. Insulin-mediated suppression of hepatic glucose production is still preserved in IR, however, suppression of VLDL-TG secretion is less pronounced4, indicating a rise in TG which may represent an early manifestation of IR. Cross-sectional studies5,6 and mechanistic studies7 have shown a positive association between circulating TG levels and both insulin sensitivity and action. Kriketos et al. 2003 has shown that both skeletal muscle TG and circulating TG were inversely associated with whole body insulin sensitivity7. Mingrone et al. 1997 showed reversibility of IR by lowering plasma TG in obese individuals with diabetes8; together these observations suggest that circulating TG may serve as a marker of IR and its associated complications.
Evidence of a relationship between TG and IR has escalated following the identification of “metabolic syndrome” (MetS), a condition representing a cluster of metabolic abnormalities with central obesity and IR postulated as core components9,10,11. A considerable body of evidence suggests that obesity-associated IR is an independent risk factor and a central component in the pathogenesis of type 2 diabetes and cardiovascular disease (CVD), both of which have reached epidemic proportions worldwide12,13. Evidence that IR causes type 2 diabetes comes from cross-sectional studies demonstrating the presence of IR in a majority of type 2 diabetes patients, and prospective studies demonstrating the development of IR long before the onset of type 2 diabetes14,15. Dyslipidaemia associated with IR increases the risk of developing CVD in type 2 diabetes16,17. It is therefore important to quantify IR and identify MetS for primary and secondary prevention of these metabolic diseases.
Recognition of the importance of IR have prompted the derivation of a number of indices and surrogate markers to quantify IR18,19. Among these, the hyperinsulinemic euglycaemic clamp (HEC) technique has been described as the gold standard, providing a direct measure of IR20. However, cost, expertise, and the requirement for intravenous insulin infusions and frequent blood sampling limits the application of HEC in epidemiological studies and routine clinical investigations. Homeostatic model assessment (HOMA) is a simple minimally invasive model that predicts IR using fasting steady-state glucose and insulin levels and that has been shown to be highly correlated with clamp insulin sensitive index values21. However, HOMA does not consider the level of blood lipids, such as TG or HDL-Cholesterol (HDL-C), despite lipid availability in circulation having an important role in IR and its metabolic complications. In the current study we propose and evaluate a novel tool for estimating IR, InsuTAG. This model is based on the product of fasting blood insulin and fasting blood lipid (TG) levels. InsuTAG is unique in its incorporation of a measure of hyperinsulinemia and a measure of circulating TG in the general population, for identification of IR and its metabolic complications.
Results
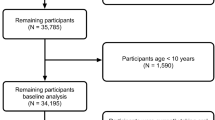
After screening the 618 participants recruited for the RHLS study with exclusion criteria, 486 participants were included in the analysis. One hundred and thirteen participants were excluded because of self-reported diabetes. An additional 19 participants were excluded because they had blood glucose levels ≥7 mmol/L (n = 9); self-reported use of oral anti-hyperglycaemic agents (n = 4); had no blood glucose values recorded (n = 3); and/or self-reported taking TG lowering medications (n = 3). Participant characteristics are outlined in Table 1. The study population was predominantly Caucasian (n = 469, 96.50%), included more females (n = 283, 58.23%) than males (n = 203, 41.77%), had a mean ± SD age of 77.78 ± 7.16 years, and a mean ± SD body mass index (BMI) of 28.05 ± 4.64 kg/m2. A total of 82 (16.87%) participants were categorised as IR according to HOMA-IR ≥2.5 and 167 (37.03%) were identified as having MetS according to the International Diabetes Federation (IDF) criteria. Fasting glucose, insulin, TG, weight, waist circumference (WC) and BMI were significantly (p < 0.0001) higher in individuals with IR compared with insulin-sensitive individuals (Table 1). InsuTAG scores ranged from 0.55 to 132.07 across the whole participant group with a median (25th–75th percentile) InsuTAG score of 7.22 (4.04–11.33)
There is a strong and highly significant positive correlation between InsuTAG and HOMA-IR (r = 0.86; p < 0.0001) (Fig. 1) when compared with the association between HOMA-IR and the other lipid indices, TyG index (triglycerides × glucose) (r = 0.43; p < 0.001) and TG/HDL-C ratio (r = 0.38; p < 0.001) (Table 2). InsuTAG is also positively associated with BMI (r = 0.51, p < 0.001) and WC (r = 0.48, p < 0.001).
Multiple regression analyses were used to evaluate the relationships between InsuTAG and other surrogate markers of IR with HOMA-IR (Table 3). Model 1 presents the unadjusted analyses. Model 2 present the analyses adjusted for age, gender, WC and C-reactive protein (CRP). All surrogate markers of IR were significant predictors of HOMA-IR, both independently and after adjusting for covariates (p < 0.0001), however, InsuTAG accounted for a greater proportion of variability in HOMA-IR (Model 1, R2 = 0.739; Model 2, R2 = 0.738) than did the TyG index (Model 1, R2 = 0.183; Model 2, R2 = 0.303) or the TG/HDL-C ratio (Model 1, R2 = 0.145; Model 2, R2 = 0.284). Evaluation of the partial correlations indicated that, after adjusting for covariates, InsuTAG accounted for 65.0% of the variability in HOMA-IR scores, whereas TyG and TG/HDL-C were independently associated with only 7.9% and 5.4% of the change in HOMA-IR scores, respectively. Examination of the regression coefficient revealed that, after adjusting for covariates, a 10% increase in InsuTAG resulted in a corresponding increase of 2.57% in HOMA-IR (p < 0.0001). In addition, logistic regression showed that after adjusting for age, gender, WC and CRP, each one-unit increase in InsuTAG increased the odds of having IR by 20% (OR (95%CI): 1.20 (1.15–1.26), p < 0.0001) and MetS by 16% (OR (95%CI): 1.16 (1.11–1.21), p < 0.0001).
ROC curves of InsuTAG and surrogate markers of IR were plotted to compare the predictive values for IR and MetS (Fig. 2). InsuTAG had significantly greater Area under the curve (AUC) values (AUC = 0.93, p < 0.001) than TyG index (AUC = 0.72) or TG/HDL-C (AUC = 0.70) for the identification of IR. For the identification of MetS, the AUC of InsuTAG (0.79) was significantly higher than HOMA-IR (0.73, p = 0.001) (Fig. 3), fasting insulin (0.69, p = 0.000) and WC (0.72, p = 0.009). The optimal cut off value for InsuTAG in identifying IR in the current study population was determined to be 11.2, with a sensitivity of 84.15% and specificity of 86.88%. At this cut-off, the sensitivity and specificity for MetS was 49.70% and 90.49% respectively (Table 4). Positive and negative predictive values along with likelihood ratios are also presented in Table 4. After determining the cut-off values for InsuTAG, the study population were categorised into two groups: InsuTAG < 11.2 (n = 364), and InsuTAG ≥11.2 (n = 122). 74.34% of participants with InsuTAG values ≥11.2 were identified with MetS compared to only 24.56% with InsuTAG values < 11.2 (Table 5, p < 0.0001). All the key components of the metabolic syndrome, WC (104.65 ± 12.45 vs 94.51 ± 11.79 cm); fasting glucose (5.46 ± 0.51 vs 5.14 ± 0.54 mmol/L); fasting TG [1.87 (1.43–2.31) vs 1.01 (0.79–1.30) mmol/L]; and Total-C/HDL-C [3.60 (3.00–4.70) vs 2.90 (2.40–3.50)] were significantly (p < 0.0001) higher in participants with InsuTAG values above the cut-off compared with participants InsuTAG values below the cut-off (Table 5). BMI, diastolic blood pressure, fasting insulin and HOMA-IR values were also significantly higher in the InsuTAG ≥11.2 group, whereas systolic blood pressure, glycosylated haemoglobin (HbA1c), total cholesterol and low density lipoprotein-cholesterol (LDL-C) did not differ between the two groups (Table 5). The lipid index values, TyG [10.29 (7.69–12.60) vs 5.25 (4.00–6.54)] and TG/HDL-C [1.41 (1.07–1.91) vs 0.66 (0.48–0.89)] were almost doubled in participants with InsuTAG values above the cut-off.
Discussion
Many studies have evaluated lipid ratios, homeostatic models and individual metabolic variables for predicting IR6,22,23, however, none account for fasting insulin and fasting TG in a single model or formula. The current study has evaluated the use of InsuTAG, a novel marker derived from the product of two key continuous variables, fasting insulin and fasting TG, as a predictor for IR and metabolic abnormalities. InsuTAG demonstrated a stronger positive association (0.86) with HOMA-IR than the other individual metabolic markers and lipid surrogate markers analysed in this study. InsuTAG, TyG and TG/HDL-C were all independent predictors of IR in regression models; InsuTAG was the highest (65.0%) contributor to prediction of IR in the study population. ROC analysis also indicated InsuTAG (AUC 0.93) was the favourable marker over TyG index and TG/HDL-C for predicting IR.
HOMA-IR is the most common or frequently used index for assessing IR and closely mirrors HEC values24. Given that it is not feasible to conduct HEC in large study populations, in this study HOMA-IR scores of ≥2.5 were used to identify IR. Previous studies proposed surrogate lipid-based markers such as TyG index and TG/HDL-C ratio as an alternate approach to predict IR6,25. Consistent with previous reports22,26,27, the current study found significant and positive associations between HOMA-IR and BMI, WC and lipid ratios. InsuTAG demonstrated a strong positive association with HOMA-IR, representing a close association with IR, higher than that of other lipid-based surrogate markers and anthropometric measurements analysed in this study population. Multivariate regression analysis indicated that both TyG index and TG/HDL-C were independent predictors for IR, findings that are consistent with previous published studies7,22. Amongst these independent predictors, InsuTAG accounted for the greatest variance in IR for this study population, accounting for 73.9% (without adjusting for covariates) and 65% (after adjusting for covariates) of variance in HOMA-IR. Along with the benefit of accounting for two key factors involved in the development of IR, these results indicate that InsuTAG may be a reliable and physiologically relevant marker for IR that can be easily calculated from routine clinical investigations. However, as HOMA-IR is not a gold standard method of determining IR, further validation studies of the predictive capacity of InsuTAG using clamp study insulin sensitivity index values are required.
AUC values from the ROC analysis showed that InsuTAG represented 93% probability of identifying individuals with IR in this study population, comparatively greater than the AUC’s of other lipid markers. The high sensitivity and specificity values of InsuTAG and positive likelihood ratio of 6.41 (Table 4) provides reasonable justification to explore InsuTAG as a diagnostic tool for IR. ROC analysis identified 11.2 as the optimal cut-off value for predicting IR in this study population. The corresponding value for identification of MetS was 8.0. The AUC for InsuTAG (0.79) suggests it is higher to HOMA-IR (0.73), fasting insulin (0.69) and WC (0.72) for predicting MetS in this study population. Metabolic characteristics of participants with InsuTAG scores above and below the suggested cut-off were compared. Significant differences in WC, BMI, fasting glucose, fasting insulin, TG, HDL-C and blood pressure, all of which are key components of MetS, were observed. There was no significant difference in participant characteristics between sub-groups stratified by the InsuTAG cut-off values for predicting IR (11.2) and MetS (8.0), other than minor changes in sensitivity and specificity values for MetS. No further conclusions on these associations can be made at this point warranting further exploration in prospective studies to determine whether InsuTAG alone can predict the development of MetS.
We do acknowledge some limitations to this study. The study population is older and predominantly Caucasian, limiting the generalisability of InsuTAG to younger people and other ethnicities. Since it is less feasible to conduct HEC in studies with larger sample sizes, InsuTAG values were compared with HOMA-IR values rather than insulin sensitivity index values from clamp studies. Additional exploration of InsuTAG using glucose clamp studies is required to further validate this marker. Furthermore, because of the cross-sectional design of this study, no causal relationships can be determined. Longitudinal studies are required to evaluate whether InsuTAG can predict development of IR and MetS.
Surrogate markers for IR are less invasive and closely mirror correlation with HEC for metabolic and CVD risk. A recently published paper (20) on correlation of surrogate indices with HEC, concluded that surrogate markers including fasting insulin provided the most information relating to IR, compared with other complex and invasive procedures. The combination of fasting insulin and an indicator of reduced lipid clearance could provide more reliable information on IR and metabolic abnormalities.
In conclusion, we have proposed and evaluated a novel marker for IR that accounts for both fasting insulin and TG. It is simple to calculate and feasible for large cohort studies. This study substantiates and shows InsuTAG as a predictor of IR and a predictor of metabolic syndrome with higher sensitivity and specify values over other anthropometry and existing lipid surrogate indices. Further research is required to validate InsuTAG against HEC and determine whether it can accurately predict the development of IR and MetS in prospective studies.
Methods
This study is a sub-study of the Retirement Health and Lifestyle (RHLS), a cross-sectional study of Australians aged 65 years and older, living in the Central Coast of New South Wales, Australia. Inclusion and exclusion criteria for participants in RHLS study has been described in detail elsewhere28,29. In brief, participants were invited to participate in the RHLS if they were: aged between 65 years or over; living in the Wyong Shire or Gosford City local government areas and living independently in retirement villages or within the community. Participants were included in the present study if: their diabetic status could be determined; their plasma fasting glucose, insulin and TG levels were available; and if they were not taking any TG-lowering medication. Participants provided written informed consent and ethics approval for this study was obtained from the University of Newcastle Human Research Ethics Committee (reference no. H-2008-0431) and the Northern Sydney Central Coast Health Human Research Ethics Committee (reference no. 1001–031 M). All testing was performed in accordance with the approved guidelines.
Demographic information, medical history, and information relating to medication and supplement intake were obtained from participants via interviewer-administered and self-administered questionnaire. Height and weight of study participants were measured using a portable stadiometer (design no. 1013522 Surgical and Medical products) and digital scales (Tanita HD 316 or Wedderburn UWPM150). BMI was calculated using the standard formula [weight (kg)/height (m)2]. WC was measured with a non-elastic measuring tape at the midpoint between the iliac crest and coastal margin in the mid-auxiliary line. All anthropometric measurements were conducted by trained research officers.
Information on dietary intake was obtained from study participants using a self-administered semi-quantitative food frequency questionnaire (FFQ) adapted from a validated Commonwealth Scientific and Industrial Research Organisation Human Nutrition FFQ30. Diet, energy and nutrient intake information was analysed using Food Works Professional (2009 edition, version 6.0.2562, Xyris software, Brisbane, Australia) in association with the following databases; Australia (fatty acids), Abbott products, AusFoods (brand) 2006, AusNut (all foods) 2007, and the New Zealand Vitamin and Mineral Supplement 1999.
Physical activity was assessed during the interviewer-administered questionnaire using questions designed to capture the frequency, duration and intensity of physical activity undertaken during the previous seven days. The questions were adapted from validated questionnaires measuring physical activity, and captured both incidental (e.g. household chores, gardening) and intentional (e.g. recreational sports, strength training) physical activity. Blood pressure measurements were taken with an OMRON 1A2 digital automatic blood pressure monitor in accordance with the “Measuring Blood Pressure” protocol published by the National Heart Foundation in 2008.
Blood samples were collected by trained phlebotomists following an overnight fast of at least 10 hours. Blood glucose control related parameters [fasting glucose (mmol/L), fasting insulin (mIU/L) and HbA1c], lipid profile [TG, total cholesterol, LDL-C and HDL-C; mmol/L] and CRP (mg/mol) were analysed by Hunter Area Pathology Service using standard laboratory procedures. Fasting insulin and glucose were used to measure IR using HOMA-IR scores (fasting insulin × fasting glucose/22.5). Study participants with HOMA-IR values ≥2.5 were categorised as insulin resistant. TyG index (fasting glucose × fasting TG) and TG/HDL-C ratio were also assessed. InsuTAG was calculated by multiplying fasting insulin (mU/L) and fasting TG (mmol/L). Participants were categorised as having MetS according to IDF criteria for abdominal obesity (waist circumference ≥94 cm for males, or ≥80 cm for females) plus any two of the following conditions (or self-report of receiving treatment for any of those conditions): high TG (≥1.7 mmol/L); high fasting glucose (≥5.6 mmol/L); high BP (≥130 mmHg systolic or ≥85 mmHg diastolic); and/or low HDL-C (<1.03 mmol/L for males, <1. 29 mmol/L for females).
The data of all variables included in the analysis were tested for normality using histograms and Shapiro-Wilk’s tests and are presented as mean ± SD or median (IQR) as appropriate. Heavily skewed parameters were log-transformed (log base e) prior to correlation and regression analyses. Distributions were reassessed after transformation and in all instances the log transformation successfully achieved a normal distribution. Bivariate relationships between continuous variables were assessed using Pearson’s Product-moment correlation. Standard multiple regression was used to assess the relationship of HOMA-IR with InsuTAG and other surrogate markers of IR, with and without adjustment for the potentially confounding variables: age, gender, WC and blood levels of CRP. Logistic regression was used to determine whether an increase in InsuTAG score was associated with increased odds of having IR or MetS. Receiver operating characteristic (ROC) curves were constructed for InsuTAG and other surrogate markers of IR to assess whether they were effective in identifying either IR and/or MetS. For each marker, the area under the curve (AUC) was compared against HOMA-IR using the Stata command roccomp. Youden’s index was used to determine the optimum cut-off point, the point which has the greatest sensitivity and specificity, Participants were categorised according to whether their InsuTAG scores fell below or above the suggested cut-off point. Group differences were assessed using independent sample t-tests, Mann-Whitney U tests, or chi-squared analysis as appropriate. Significance was set at p < 0.05. All statistical analyses were conducted using Stata version 14.1 (StataCorp, Texas, USA).
References
Saltiel, A. R. & Kahn, C. R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414, 799–806 (2001).
Sparks, J. D., Sparks, C. E. & Adeli, K. Selective hepatic insulin resistance, VLDL overproduction, and hypertriglyceridemia. Arterioscler Thromb Vasc Biol 32, 2104–2112, https://doi.org/10.1161/atvbaha.111.241463 (2012).
Meshkani, R. & Adeli, K. Hepatic insulin resistance, metabolic syndrome and cardiovascular disease. Clinical Biochemistry 42, 1331–1346, https://doi.org/10.1016/j.clinbiochem.2009.05.018 (2009).
Sorensen, L. P. et al. Increased VLDL-triglyceride secretion precedes impaired control of endogenous glucose production in obese, normoglycemic men. Diabetes 60, 2257–2264, https://doi.org/10.2337/db11-0040 (2011).
Kahn, H. S. & Valdez, R. Metabolic risks identified by the combination of enlarged waist and elevated triacylglycerol concentration. The American journal of clinical nutrition 78, 928–934 (2003).
McLaughlin, T. et al. Use of metabolic markers to identify overweight individuals who are insulin resistant. Ann. Intern. Med. 139, 802–809 (2003).
Kriketos, A. D. et al. Multiple indexes of lipid availability are independently related to whole body insulin action in healthy humans. The Journal of clinical endocrinology and metabolism 88, 793–798, https://doi.org/10.1210/jc.2002-020848 (2003).
Mingrone, G. et al. Reversibility of insulin resistance in obese diabetic patients: role of plasma lipids. Diabetologia 40, 599–605, https://doi.org/10.1007/s001250050721 (1997).
Reaven, G. M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 37, 1595–1607 (1988).
Li, N. et al. Are hypertriglyceridemia and low HDL causal factors in the development of insulin resistance? Atherosclerosis 233, 130–138, https://doi.org/10.1016/j.atherosclerosis.2013.12.013 (2014).
Boden, G. & Laakso, M. Lipids and Glucose in Type 2 Diabetes. Diabetes Care 27, 2253 (2004).
Laakso, M. & Kuusisto, J. Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat. Rev. Endocrinol. 10, 293–302, https://doi.org/10.1038/nrendo.2014.29 (2014).
Harris, W. S. n-3 Fatty acids and serum lipoproteins: human studies. Am. J. Clin. Nutr. 65, 164S–154S (1997).
Warram, J. H., Martin, B. C., Krolewski, A. S., Soeldner, J. S. & Kahn, C. R. Slow glucose removal rate and hyperinsulinemia precede the development of type II diabetes in the offspring of diabetic parents. Ann. Intern. Med. 113, 909–915 (1990).
Lillioja, S. et al. Impaired glucose tolerance as a disorder of insulin action. Longitudinal and cross-sectional studies in Pima Indians. N. Engl. J. Med. 318, 1217–1225, https://doi.org/10.1056/nejm198805123181901 (1988).
Eddy, D., Schlessinger, L., Kahn, R., Peskin, B. & Schiebinger, R. Relationship of Insulin Resistance and Related Metabolic Variables to Coronary Artery Disease: A Mathematical Analysis. Diabetes Care 32, 361 (2009).
Kuusisto, J., Lempiäinen, P., Mykkänen, L. & Laakso, M. Insulin Resistance Syndrome Predicts Coronary Heart Disease Events in Elderly Type 2 Diabetic Men. Diabetes Care 24, 1629 (2001).
Singh, B. & Saxena, A. Surrogate markers of insulin resistance: A review. World J. Diabetes 1, 36–47, https://doi.org/10.4239/wjd.v1.i2.36 (2010).
ter Horst, K. W. et al. Methods for quantifying adipose tissue insulin resistance in overweight/obese humans. Int J Obes 41, 1288–1294, https://doi.org/10.1038/ijo.2017.110 (2017).
DeFronzo, R. A., Tobin, J. D. & Andres, R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am. J. Physiol. 237, E214–223 (1979).
Matthews, D. R. et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28, 412–419 (1985).
Du, T. et al. Clinical usefulness of lipid ratios, visceral adiposity indicators, and the triglycerides and glucose index as risk markers of insulin resistance. Cardiovasc. Diabetol. 13, 146, https://doi.org/10.1186/s12933-014-0146-3 (2014).
Simonson, D. C. Surrogate measures of insulin resistance: does one size fit all? Diabetologia 58, 207–210, https://doi.org/10.1007/s00125-014-3454-y (2015).
Sarafidis, P. A. et al. Validity and reproducibility of HOMA-IR, 1/HOMA-IR, QUICKI and McAuley’s indices in patients with hypertension and type II diabetes. J. Hum. Hypertens. 21, 709–716, https://doi.org/10.1038/sj.jhh.1002201 (2007).
Guerrero-Romero, F. et al. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J. Clin. Endocrinol. Metab. 95, 3347–3351, https://doi.org/10.1210/jc.2010-0288 (2010).
Er, L.-K. et al. Triglyceride Glucose-Body Mass Index Is a Simple and Clinically Useful Surrogate Marker for Insulin Resistance in Nondiabetic Individuals. PLoS One 11, e0149731, https://doi.org/10.1371/journal.pone.0149731 (2016).
Giannini, C. et al. The Triglyceride-to-HDL Cholesterol Ratio. Diabetes Care 34, 1869 (2011).
Abbott, K. A. et al. Sex-dependent association between erythrocyte n-3 PUFA and type 2 diabetes in older overweight people. Br. J. Nutr. 115, 1379–1386, https://doi.org/10.1017/S0007114516000258 (2016).
Ferguson, J. J. A. et al. Association between omega-3 index and blood lipids in older Australians. Journal of Nutritional Biochemistry 27, 233–240, doi:https://doi.org/10.1016/j.jnutbio.2015.09.010.
Lassale, C. et al. Estimating food intakes in Australia: validation of the Commonwealth Scientific and Industrial Research Organisation (CSIRO) food frequency questionnaire against weighed dietary intakes. J. Hum. Nutr. Diet. 22, 559–566, https://doi.org/10.1111/j.1365-277X.2009.00990.x (2009).
Acknowledgements
The research on which this paper is based was conducted as part of the Retirement Health and Lifestyle Study, The University of Newcastle. We are grateful to the Australian Research Council, Central Coast Local Health District Public Health Unit, UnitingCare Ageing NSW/ACT, Urbis Pty Ltd, Valhalla Village Pty Ltd, and Hunter Valley Research Foundation for funding the initial study and to the men and women of the Central Coast region who provided the information recorded.
Author information
Authors and Affiliations
Contributions
R.N.T. and M.L.G. designed research; R.N.T., M.L.G., and K.A.A., conducted research; M.L.G., M.L., and M.V. provided essential materials; K.A.A., and R.N.T., analysed data; R.N.T., K.A.A., M.L.G., J.J.A.F., M.L., S.N., and K.K., wrote the paper; R.N.T., and M.L.G., had primary responsibility for final content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thota, R.N., Abbott, K.A., Ferguson, J.J.A. et al. InsuTAG: A novel physiologically relevant predictor for insulin resistance and metabolic syndrome. Sci Rep 7, 15204 (2017). https://doi.org/10.1038/s41598-017-15460-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15460-z
This article is cited by
-
Understanding the predictive accuracy of the InsuTAG index over other surrogate indices in normoglycaemic, non-obese males from Southern India
Scientific Reports (2023)
-
Insulin resistance and cardiometabolic indexes: comparison of concordance in working-age subjects with overweight and obesity
Endocrine (2022)
-
Curcumin and/or omega-3 polyunsaturated fatty acids supplementation reduces insulin resistance and blood lipids in individuals with high risk of type 2 diabetes: a randomised controlled trial
Lipids in Health and Disease (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.