Abstract
Meaningful, reliable and valid mRNA expression analyses by real-time quantitative PCR (RT-qPCR) can only be achieved, if suitable reference genes are chosen for normalization and if appropriate RT-qPCR quality standards are met. Human periodontal ligament (hPDL) fibroblasts play a major mediating role in orthodontic tooth movement and periodontitis. Despite corresponding in-vitro gene expression studies being a focus of interest for many years, no information is available for hPDL fibroblasts on suitable reference genes, which are generally used in RT-qPCR experiments to normalize variability between samples. The aim of this study was to identify and validate suitable reference genes for normalization in untreated hPDL fibroblasts as well as experiments on orthodontic tooth movement or periodontitis (Aggregatibacter actinomycetemcomitans). We investigated the suitability of 13 candidate reference genes using four different algorithms (geNorm, NormFinder, comparative ΔCq and BestKeeper) and ranked them according to their expression stability. Overall PPIB (peptidylprolyl isomerase A), TBP (TATA-box-binding protein) and RPL22 (ribosomal protein 22) were found to be most stably expressed with two genes in conjunction sufficient for reliable normalization. This study provides an accurate tool for quantitative gene expression analysis in hPDL fibroblasts according to the MIQE guidelines and shows that reference gene reliability is treatment-specific.
Similar content being viewed by others
Introduction
Orthodontics and periodontology are specialties of dentistry tending to the treatment of misaligned teeth/jaws and bacterially induced inflammation of the periodontal tissues (periodontitis), respectively, with several interactive associations existing1. In orthodontics mechanical forces applied to the teeth result in tensile and pressure zones within the periodontal ligament (PDL)2. PDL fibroblasts react to this mechanical strain with an increased synthesis of proinflammatory enzymes, cytokines and chemokines2,3,4, triggering osteoclastogenesis. Bacterial toxins from periodontal pathogens in periodontitis, such as the gram-negative Aggregatibacter actinomycetemcomitans (Agac), the key pathogen in aggressive periodontitis5, can in a similar way stimulate PDL fibroblasts, which are thus essential both for mediating orthodontic tooth movement and bacterial periodontitis.
Real-time quantitative PCR (RT-qPCR) and DNA microarray analysis are the methods of choice to analyse transcription of cellular genes6,7. In contrast to microarray analysis, which allows expression profiling of a high number of genes, RT-qPCR enables a precise quantification of gene expression differences in physiological, pathological and various experimental states8,9,10. However, a reliable RT-qPCR setup is necessary to achieve valid results. To improve quality and reproducibility of RT-qPCR experiments, Bustin et al. published the MIQE guidelines11 in 2009, detailing the minimum information for publication of quantitative real-time PCR experiments. A view in current literature shows that many gene expression studies did not perform, consider or report important aspects such as RNA integrity, qPCR-efficiency, primer specifity or secondary structure analyses of primers and amplicons, thus limiting their scientific validity and reliability6,7,12. This is particularly the case in the field of dentistry, with RT-qPCR studies on cells of teeth and the surrounding periodontal tissue continuously increasing, particularly in orthodontics12,13,14,15,16,17,18 and periodontology19,20,21,22,23,24.
In RT-qPCR absolute quantification of gene expression is prone to errors due to intra- or interkinetic variations as well as variations in yield and efficiency during RNA isolation, reverse transcription and qPCR8,25. Therefore relative gene expression is usually calculated by normalization of a target gene expression to one or more reference genes7, which mostly regulate basic cellular functions and are deemed to be stably expressed in different experimental conditions as well as cell and tissue types7,26,27. But “perfect” reference genes do not exist6,7,28,29. Various studies have shown that the stability of reference genes can vary considerably between cell types, different tissues and even experimental conditions in the same specimen6,7,28. Thus an individual validation of suitable reference genes is required to allow a valid interpretation of relative gene expression data8,25. Otherwise relative gene expression of target genes may be over- or underestimated or even contrary to the expression actually occurring30. For various human cell types, tissues and experimental conditions valid reference genes have been identified27,31,32,33,34. However, no valid reference genes for gene expression studies on hPDL fibroblasts have been published so far despite many studies investigating periodontitis and orthodontic tooth movement using this cell type3,12,13,14,15,16,17,18,19,20,21,22,23,35. In most cases reference genes were not validated and normalization was performed using only one gene.
In the present study we wanted to introduce the MIQE guidelines11 to in-vitro experiments in the field of dentistry and to identify the ideal number and type of reference genes for qPCR gene expression studies on hPDL fibroblasts, particularly in experiments on orthodontic tooth movement and periodontitis, by determining the relative expression stability of 13 commonly used reference genes using four mathematical algorithms (geNorm29, NormFinder36, BestKeeper37, comparative ΔCq 31). In addition, we investigated the conformity and thus reliability of these algorithms for bioinformatical analyses of reference gene stability.
Results
In silico analysis of primer and amplicon quality and suitability
We selected 13 candidate reference genes based on their frequent usage for normalization in gene expression studies with differing functions in cell metabolism to minimize co-regulation (Table 1)7. All primers were newly designed by the authors with NCBI PrimerBLAST38 according to the MIQE guidelines11,39,40,41 (Supplementary Table 1) to minimize risk of bias. Intron-flanking primer pairs to prevent a co-amplification of genomic DNA could be designed for all candidate reference genes except RNA18S5 as well as sufficient absence of hairpin structures and self-/cross-dimer formation confirmed at annealing temperature (∆G ≥ −3,5 kcal/mol41, BeaconDesigner™ Free Edition, Premier BioSoft International, Palo Alto, CA, USA) (Supplementary Data 1). Target amplicon sequences were chosen to range from 60 to 150 bp with a GC content of 35–65% (Table 1) and no secondary structures were found at annealing temperature41 (60 °C, Supplementary Data 1), determined by UNAFold (Integrated DNA Technologies Inc., Coralville, IA, USA). In silico specifity of constructed primers was corroborated by PrimerBLAST38 and cross-checked using the UCSC Genome Browser (University of California, CA, USA) (Supplementary Data 1). Using NCBI PrimerBLAST38 and PrimerCheck (SpliceCenter of Genomics and Bioinformatics Group, LMP, CCR, NCI), we could confirm that primers also targeted possible splicing and transcript variants (except for RNA18S5), whereas no pseudogenes, retropseudogenes or other homologs were found to be amplified (Table 1, Supplementary Data 1), except for RNA18S5 (co-amplification of RNA45S5). For four of the 13 to be investigated candidate reference genes (UBC, GUSB, ACTB, TUBB), it was not possible to design a specific primer pair meeting all specified quality criteria11,39,40,41, which is why they were exempted from further qPCR analysis (Table 1). A commercially available, non-intron-flanking primer pair for TUBB (PPH17836A-200, Qiagen, Hilden, Germany), however, was tested alongside the custom primers to comparatively assess primer specifity and relative gene expression stability.
Primer specifity, RT-qPCR efficiencies and Cq expression levels
Primer specifity was confirmed by melting curve analysis (Fig. 1a, Supplementary Data 3) and agarose gel electrophoresis, which showed a single band at the expected molecular amplicon weight per primer pair (Fig. 1b). Primer (factor-specific) and amplification (sample-specific) efficiencies ranged from 91.7% to 100.3% (EP , Table 2, Supplementary Data 4) and 87.3% to 113.4% (EA, Table 2) with a minimum coefficient of determination in the linear dynamic range (LDR) of 0.9949. The highest SD of the arithmetic mean of Cq of the three technical replicates among all samples (n = 18) for each candidate gene was 0.53 Cq (EEF1A1) with a mean SD of 0.08–0.28 for the individual candidate reference genes (technical reliability, Table 2). In addition biological variation of Cq values was limited within experimental groups for each gene with SD ranging from 0.05 to 0.22 (Supplementary Table 3), except for YWHAZ and RNA18S5 (SD 0.31–0.62). Cq values of the investigated reference genes, which inversely correspond to the initial amount of cDNA template, ranged from 8.30 to 23.52 cycles (Fig. 2, Supplementary Table 3) with lowest values observed for RNA18S5 and highest for POLR2A, YWHAZ and TBP. Specifity, efficiencies and Cq expression levels for the commercially available primer pair for TUBB are given in Supplementary Data 5.
Specifity of RT-qPCR amplification as determined by (a) melting curve analysis and (b) agarose gel electrophoresis of RT-qPCR products. For each candidate reference gene/primer pair we found a single fluorescent band at the expected amplicon size. bp = base pairs. Gene names see Table 1. All RT-qPCR products were run concurrently and adjacently on the same gel, which was recorded with the gel documentation system Genoplex 2 (VWR International GmbH, Darmstadt, Germany) and its software GenoCapture (version 7.01, Synoptics Ltd., Cambridge, UK - automatic exposure, exposure time 80 ms, no binning, transillumination) as secure gel data (*.sgd) and exported as TIF image, which was inverted and cropped to encompass the relevant gel area. The uncropped original gel is provided as Supplementary Figure 2.
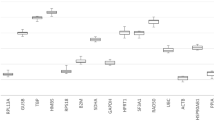
Expression levels of candidate reference genes across all experimental groups (n = 18). Values are presented as quantification cycle (Cq, mean of triplicate technical replicates) as second derivative maximum of the fluorescence curve and are inversely proportional to the initial amount of cDNA. Genes are ordered from left (highest expression) to right (lowest expression) according to their mean Cq values. Gene names see Table 1. Boxplots show median, interquartile range (box) and data range (whiskers).
Optimal number of reference genes for normalization
geNorm analysis revealed that the use of two reference genes for normalization in RT-qPCR was adequate for studies in hPDL fibroblasts in all experimental conditions (Fig. 3a). Average pairwise variation Vn/Vn+1 after inclusion of a third reference gene was below 0.15 for all tested conditions (Fig. 3a).
GeNorm expression stability analysis of the nine candidate reference genes, for which specific primers could be constructed. (a) Optimal number of reference genes for hPDL RT-qPCR data normalization in orthodontic studies (compressive orthodontic force vs. untreated control, n = 12), studies on periodontitis (Agac toxins vs. untreated control, n = 12) and pooled/overall (n = 18). (b) Average expression stability values of overall (pooled) specimens derived by stepwise exclusion of the least stable reference gene across all specimens and experimental conditions (n = 18). A smaller M value indicates a more stable gene expression. Gene names see Table 1.
Relative stability of candidate reference genes
With geNorm the most stably expressed reference genes for the pooled/overall conditions were found to be PPIB and TBP (Table 3, Fig. 3b). When we analysed the conditions for experimental orthodontic tooth movement and for periodontitis separately with geNorm, PPIB and RPL22 were most stable in orthodontic setups, whereas TBP and PPIB were most stably expressed in experiments on periodontitis (Agac toxins, Table 3). NormFinder confirmed geNorm findings and also identified RPL22 and PPIB as most stable genes in combined control and compressive force conditions and PPIB and TBP for combined control and Agac toxin treatment as well as overall combined experimental conditions (Table 3). The comparative ΔCq method31 was also in line with geNorm and NormFinder (Table 3). In contrast, the BestKeeper algorithm37 suggested RNA18S5 and YWHAZ for compressive force experiments, POLR2A and TBP for Agac toxins and RNA18S5 and TBP for combined experiments as the most stable reference genes (Table 3). Mean SD of mean Cq was ≤1 for each gene, as required for stable reference genes. For the three experimental groups (control, compressive orthodontic force, Agac toxins) separate stability rankings were also calculated and are given in Supplementary Table 4. When also considering RT-qPCR data obtained from the commercially available primer pair on TUBB, no influence on the top-ranking, most stable genes was detected (Supplementary Data 5).
Conformity of mathematical algorithms for reference gene stability analysis
Bivariate correlations of the pooled/overall (n = 18) gene ranking of the individual algorithms are presented in Fig. 4. geNorm, NormFinder and comparative ΔCq showed significant and pronounced gene ranking correlations. By contrast BestKeeper ranking did not correlate significantly with the other three tested algorithms (Fig. 4).
Correlation matrix of the stability values of the four different algorithms used for reference gene evaluation (geNorm, NormFinder, BestKeeper, comparative ΔCq). Scatterplots visualize bivariate correlations of the overall stability values of the nine assessed candidate reference genes as computed by two different algorithms including a linear regression line. r = Pearson’s correlation coefficient; **p ≤ 0.01.
Discussion
In general, PPIB, TBP and RPL22 performed best as reference genes with the highest stability values and good primer and amplification efficiency and reliability throughout for all experimental conditions and algorithms (mostly ranking top three). In addition, these genes have different cellular functions thus avoiding co-regulation7. PPIB is a protein binding cyclosporine in the endoplasmic reticulum, which plays a major role in the folding of collagen type I42 and was recently found to be associated pathological conditions, such as osteogenesis imperfecta43, which may also affect the periodontal apparatus. In contrast, TBP is a TATA-box-binding protein, which is required for the initiation of transcription by RNA polymerase II44, and RPL22 is a ribosomal protein45,46, which is involved in the control of morphogenesis by regulating Smad2 mRNA splicing47. Only subtle differences in their relative stability were detected among the three genes. A notable exception is RPL22 in experiments with Agac toxins (periodontitis), ranked as more unstable by all algorithms, indicating a regulation by Agac toxins. Also in orthodontic experiments BestKeeper found RNA18S5 and YWHAZ to be more stable than RPL22, TBP and PPIB. RNA18S5 and YWHAZ, however, are not suited as reference genes for hPDL fibroblasts, as discussed later. In a previous animal study on Fisher344 rats10, we also found PPIB to be one of the two most stably expressed reference genes for a conglomerate of dental-periodontal tissue. The second most stable reference gene identified (YWHAZ), however, did not perform well in the present study, which could be attributable to the difference in species.
Based on the results of this study, PPIB and RPL22 or TBP are thus recommended to be used in cell culture experiments with hPDL fibroblasts isolated from young and healthy donors treated with compressive force, as they are the most stably expressed reference genes under these conditions. PPIB and TBP are most stably expressed in hPDL fibroblasts stimulated with Agac toxins and should therefore be used for in vitro experiments on periodontitis. Other pre-existing pathological conditions such as osteogenesis imperfecta as well as the age of hPDL donors may also affect the performance of candidate reference genes7,48. The results of this study can thus only be safely generalized to hPDL cells from young and generally healthy donors, whereas different gene stability rankings may be expected for hPDL cells from older donors or during pathological conditions, as evidenced by the observed reduced stability of RPL22 in experimental periodontitis.
For evaluation of reference gene stability, four algorithms were used. geNorm29 calculates the average pairwise Cq variation of a one candidate reference gene with all other genes, which is given as expression stability M. The conceptual idea behind geNorm is the supposed constancy of the expression ratio of two ideal (stable) reference genes in all samples and experimental conditions29. Genes with higher M values are associated with a greater average pairwise variation in gene expression and should thus be excluded for normalization, since they indicate expression ratio inconstancy and thus expression instability29.
Despite generally increased normalization reliability7, the usage of a plethora of reference genes is time- and cost-demanding. Thus it is neither practical nor common to use more reference genes than necessary49. A reliable identification of the minimally necessary number of reference genes without risking distinct bias on target gene expression is therefore essential and could be achieved with the geNorm algorithm by calculating the average pairwise variation between normalization factors of n and n + 1 candidate genes (Vn/Vn+1)29. Since variation was not substantial after addition of a third reference gene (cut-off value V ≤ 0.15), no additional stabilizing effect was achieved for normalization, which is why two reference genes should be sufficient for normalization throughout29.
The NormFinder algorithm36 determines intra- and intergroup variation, creating a combined stability value for each candidate reference gene using a model-based approach36 with lower stability values associated with higher expression stability. The comparative ΔCq method compares the relative expression of gene pairs within each biological replicate and ranks reference genes according to the mean standard deviation of the mean ∆Cq differences of the respective gene from all other genes assessed with a lower SD indicating a more stable gene expression31. The underlying conceptual idea is that if ΔCq values between two assessed genes show variation between different samples, the expression level of one or both genes is bound to vary31. By performing repeated pairwise comparisons for all candidate gene combinations, the gene pairs with least variability and thus highest stability can be determined31.
BestKeeper37 determines stability based on the standard deviation (SD) of Cq means of each candidate reference gene as well as Pearson’s correlation coefficient r by pairwise bivariate correlations of Cq values of each gene with a “BestKeeper Index” as geometric mean of the individual Cq values of all reference genes with SD ≤ 1 (genes with SD > 1 are excluded as unsuitable/unstable). Higher r values, indicating a higher contribution of the respective gene to the “Index”, can thus be interpreted as more stably expressed genes.
The different stability values for individual candidate reference genes show that both orthodontic force application as well as bacterial Agac toxins have a distinct influence on the gene expression of basic cell metabolism, confirming that complex cellular-biological processes occur during both conditions directly affecting basic cell metabolism at a transcriptional level2,3,5,10. Interestingly, some of the more popular reference genes used previously, particularly in orthodontic and periodontitis experiments on hPDL fibroblasts12,13,14,15,16,17, performed with lower stability than anticipated. These include traditional and often used reference genes such as β-actin33 (ACTB), glucuronidase beta (GUSB), ubiquitin C (UBC) and tubulin beta class I (TUBB), for which no specific primer pair could be constructed according to the MIQE guidelines11. Several of them are commercially available, but these primers not in line with MIQE guidelines, as was the case with the comparatively tested primer pair for TUBB, which was gene-specific, but not intron-spanning/-flanking, allowing a co-amplification of genomic DNA, if no DNAse treatment is performed. Since sequences of these commercially available primers are often not published for corroboration, other problems such as secondary structure formation of primers and amplicons at annealing might be present as well, which is why we chose to exclude these genes and primers in our principal analysis. Furthermore the frequently used reference genes 18S-rRNA (RNA18S5)8, POLR2A15,16,17,50 and GAPDH13,14 also showed limited expression stability. Thus the usage of these common reference genes should be reconsidered in future gene expression studies on hPDL fibroblasts. In addition, both RNA18S5 and EEF1A1 show quite high absolute expression levels, which could pose a problem in relative quantification, since the expression levels of reference genes should approximate those of target genes for reliable results7. The usage of ribosomal RNA genes as reference has also been discouraged due to various other associated problems7. EEF1A1 did perform well in gene rankings for individual experimental groups, but showed both the highest technical as well as biological variation among all genes tested. YWHAZ also showed high biological variation and reduced stability, limiting its suitability.
The results indicate that RNA samples were of sufficient quality for RT-qPCR analysis. High intraassay and biological reliability as well as sufficient precision of the obtained data39 could be confirmed. Protein-free and intact RNA were indicated by purity and integrity assessment of total RNA. If protein contamination is present, it could result in an inhibition of the reverse transcription and qPCR reaction, thus leading to biased Cq values40. Primer efficiency (from standard curve11,37) ranged between 91.7% and 100.3% and amplification efficiency (from individual kinetic curves51, LinRegPCR52,53,54) between 83.2% and 119.3%. Thus formation of primer dimers, which can cause an efficiency beyond 100%39, was mostly at an acceptable level. Only for RNA18S5 we found primer and amplification efficiencies over 100%. This overestimation of efficiency can most likely be attributed to inhibitor traces in the RNA sample, which were further diluted for efficiency analyses, as well as the very low Cq values for RNA18S5, which make RNA18S5 an unsuitable reference gene, since absolute expression of reference genes should be similar to that of target genes7,10. Amplification efficiencies distinctly below 90% for POLR2A and RPLP0 also indicate limited suitability of these genes for normalization. Primer specifity as confirmed in silico and in vitro successfully prevented the co-amplification of pseudo-genes and homologues.
Although various studies on other tissues and species also used several algorithms to assess reference gene stability52,55,56, others only considered one or two algorithms27,34. We thus comparatively examined the various available statistical stability algorithms regarding their conformity to determine, whether the combined usage of several algorithms has advantages in reference gene stability determination. Our study showed a significant and high correlation between geNorm, NormFinder and comparative ΔCq algorithms as confirmed by the similar stability rankings of genes observed, which indicate that these could be used interchangeably. However, no significant correlations were found with the BestKeeper algorithm. Several reasons for this discrepancy of BestKeeper rankings to the rankings produced by the other algorithms can be assumed. BestKeeper was not particularly created to produce rankings of reference genes, but rather focuses on general suitability in a sequential two-step assessment (standard deviation of mean Cq and then correlation coefficient r). The three similarly performing algorithms are based on either performing pairwise comparisons of individual candidate reference genes with linear quantities (geNorm)/raw ΔCq values (comparative ΔCq method), ranking genes according to their expression profile similarity36, or a model-based approach using linear quantities (NormFinder), which is considered more robust, since it is less influenced by co-regulation of candidate reference genes36. By contrast, BestKeeper performs correlations of candidate genes with a single “BestKeeper Index”, identical for all correlations and not created from all candidate genes, but a selection hereof, pre-excluding those with higher Cq standard deviations than 1. Furthermore BestKeeper is based on raw Cq values instead of linear quantities, which are used by geNorm and NormFinder.
Conclusion
Using four different mathematical algorithms (geNorm, NormFinder, comparative ΔCq and BestKeeper) PPIB, TBP and RPL22 were identified as the most stable, reliable and suitable reference genes for normalization in relative RT-qPCR gene expression studies on human periodontal ligament fibroblasts, particularly in studies on orthodontic tooth movement (PPIB/RPL22) and periodontitis (Aggregatibacter actinomycetemcomitans, PPIB/TBP). Two reference genes were found to be sufficient for reliable normalization throughout. Many traditional and frequently used reference genes such as RNA18S5, POLR2A or GAPDH showed limited suitability and should be avoided in future experiments. The same is true for reference genes, for which no specific primers could be designed according to pre-specified quality criteria as described by the MIQE guidelines (ACTB, GUSB, UBC, TUBB). BestKeeper produced distinctly different stability rankings compared to the other algorithms, thus suggesting a rank-sum approach for stability evaluation.
Materials and Methods
In vitro cell culture experiments
Primary human periodontal ligament (hPDL) fibroblasts were cultivated from periodontal connective tissue isolated from the middle root section of human teeth free of decay, which had been freshly extracted for medical reasons at the authors’ dental facility. A pool of hPDL cell lines from four different patients was used (1 male, 3 female, age: 16–23 years). Collection and usage of hPDL fibroblasts from discarded patient biomaterial were approved by the ethics committee of the University of Regensburg, Germany (approval number 12-170-0150), and all experiments were carried out in accordance with the relevant guidelines and regulations. Informed consent was obtained from all participants and/or their legal guardian/s. Tissue samples were grown in 6-well cell-culture-plates until proliferation of adherently growing hPDL fibroblasts under normal cell culture conditions (37 °C, 5% CO2, water-saturated) in full media consisting of DMEM high glucose (D5796, Sigma–Aldrich®, Munich, Germany), 10% FCS (P30–3306, PAN-Biotech, Aidenbach, Germany), 1% L-glutamine (SH30034.01, GE-Healthcare-Europe, Munich, Germany), 100 µM ascorbic acid (A8960, Sigma-Aldrich®) and 1% antibiotics/antimycotics (A5955, Sigma-Aldrich®). hPDL fibroblasts were identified by their spindle-shaped morphology and hPDL-specific marker gene expression12,57,58 (Supplementary Table 5 and Supplementary Fig. 1). Until use they were then frozen in liquid nitrogen (90% FCS, 10% DMSO, freezing 1 °C/minute).
hPDL fibroblasts of the 6th passage24, quantified with Beckman Coulter Counter Z2™ (Beckman Coulter GmbH, Krefeld, Germany) according to the manufacturer’s recommendations, were randomly seeded onto 6-well cell-culture-plates at an initial density of 70,000 cells per well (Fig. 5a). We prepared three different experimental groups at 70% confluency (Fig. 5b) (37 °C, 5% CO2, 100% water-saturated, 2 ml DMEM/well) with 6 biological replicates (samples) each:
-
Untreated (physiological conditions, n = 6) - incubated for 48 h;
-
Compressive orthodontic force (n = 6) - mechanical stimulation for 24 h (2 g/cm2 pressure, sterilized glass plate of defined weight and size) according to Kanzaki et al.3 and Kirschneck et al.15,16 after a pre-incubation phase of 24 h;
-
Periodontitis (n = 6) - according to Proff et al.17, hPDL fibroblasts were incubated for 48 h with bacterial lysate of heat-inactivated Aggregatibacter actinomycetemcomitans (Agac, 107 cells/ml, DSM11123, German Collection of Microorganisms and Cell Cultures, Braunschweig, Germany), which was prepared as described before12,17.
Experimental setup for the hPDL fibroblast experiments. (a) 6-well cell culture plate with untreated controls and simulated periodontitis (left side) as well as simulated orthodontic compressive force (right side). (b) Experimental conditions tested (physiological cell culture conditions): untreated physiological controls (adherently growing hPDL cells at 70% confluence in full medium), simulated orthodontic compressive force of 2 g/cm2 applied by a 17.1 g glass disc, simulated periodontitis by adding bacterial lysate (toxins) of Aggregatibacter actinomycetemcomitans to the medium.
Isolation and purity assessment of total RNA
After washing the hPDL fibroblasts twice with phosphate-buffered saline, total RNA was extracted by applying peqGOLD TriFast™ (1 ml/well, PEQLAB-Biotechnology GmbH, Erlangen, Germany) and further processing according to the manufacturer’s instructions10,15,16. No DNAse treatment was performed, as all used primers were intron-flanking. We eluted the resulting RNA pellet in nuclease-free water (25 µl, T143, Carl-Roth GmbH, Karlsruhe, Germany) with immediate cooling on ice. To assess purity and quantity of the eluted total RNA, we determined optical density (OD) photometrically at 280 nm and 260 nm (NanoDrop ND-2000, Thermo-Fisher Scientific Inc., Waltham, MA, USA) with 1 OD260nm equalling 40 ng/µl total RNA10. An OD260nm/280nm ratio of >1.8 was considered protein-free RNA27,40. Mean concentration of extracted RNA (n = 18) was calculated from its optical density at 260 nm obtained with NanoDrop as 358.2 ng/µl (SD 104.7/Min. 218.6/Max. 495.4 - divergent results in capillary electrophoresis, Supplementary Data 2) with a mean NanoDrop OD260nm/280nm ratio of 1.90 (SD 0.03/Min. 1.82/Max. 1.96).We measured RNA integrity with an Agilent 2100 Bioanalyzer (Agilent Technologies Inc. Santa Clara, CA, USA) according to the manufacturer’s protocol. RIN values ranged from 9.50 to 10 (mean 9.85, SD 0.15) indicating absence of RNA degradation59 (Supplementary Data 2). Integrity of total RNA was confirmed by the non-proprietary 28 S/18 S ratio of ribosomal RNA in gel electrophoresis, which ranged from 1.6 to 1.8 (mean 1.71, SD 0.09; Supplementary Data 2). Reverse transcription negative control (-RT) and negative NTC reactions confirmed sufficient absence of genomic DNA, contamination and primer dimers with measured Cq values substantially higher than those of target samples (Supplementary Table 1).
Reverse transcription (cDNA synthesis)
To synthesize cDNA, we transcribed a standardized quantity of 1 µg RNA per sample using a random hexamer primer (0.1 nmol, 1 µl, SO142, Life Technologies GmbH, Darmstadt, Germany), an oligo-dT18 primer (0.1 nmol, 1 µl, SO131, Life Technologies), 5 × M-MLV-buffer (4 µl, M1705, Promega, Fitchburg, WI, USA) and dNTP mix (40 nmol, 1 µl–10 nmol/dNTP, Roti®-Mix PCR3, L785.2, Carl-Roth GmbH) ad 20 µl nuclease-free H2O (T143, Carl-Roth GmbH). After incubation (3 min, 70 °C) the mixture was quickly cooled on ice. We then added reverse transcriptase (200 U, 1 µl, M1705, Promega) and an RNase inhibitor (40 U, 1 µl, EO0381, Life Technologies), continued incubation (37 °C, 60 min) and heat-inactivated the reverse transcriptase (95 °C, 2 min). To minimize experimental variations, synthesis of cDNA, which was stored at −20 °C until use, was performed concurrently for all samples.
Quantitative real-time polymerase chain reaction (RT-qPCR)
Primer design was based on the official gene nucleotide sequences from the NCBI Nucleotide database (GeneBank, National Centre for Biotechnology Information, Bethesda MD, USA). They were constructed with NCBI PrimerBLAST38 considering the final concentration of qPCR components according to optimized criteria11,39,40,41. Primers received no terminal or other modifications and were synthesized and purified by Eurofins MWG Operon LLC (Huntsville, AL, USA; High Purity Salt Free Purification HPSF®). For qPCR amplification we used a Mastercycler® ep realplex-S thermocycler (Eppendorf AG, Hamburg, Germany) in conjunction with 96 well PCR plates (TW-MT, 712282, Biozym Scientific GmbH, Hessisch Oldendorf, Germany) and BZO Seal Filmcover sheeting (712350, Biozym Scientific GmbH). Into each well SYBR®Green JumpStart™ Taq ReadyMix™ (7.5 µl, Sigma–Aldrich®, S4438), consisting of Tris–HCl (20 mM, pH 8.3), KCl (100 mM), MgCl2 (7 mM), dNTPs (0.4 mM per dATP, dCTP, dGTP, dTTP), stabilizers, Taq-DNA-polymerase (0.05 U/µl), JumpStart™ Taq antibody and SYBR®Green I, as well as the respective cDNA solution (1.5 µl, dilution 1:10) and the respective primer pair (7.5 pmol, 0.75 µl–3.75 pmol/primer) were pipetted ad 15 µl nuclease-free H2O (T143, Carl-Roth GmbH). We amplified the cDNA in triplets (three technical replicates) per candidate reference gene and biological replicate (sample) and on the same qPCR plate in 45 cycles (initial heat activation 95 °C/5 min, per cycle 95 °C/10 s denaturation, 60 °C/8 s annealing, 72 °C/8 s extension, Supplementary Data 3), resulting in 6 (samples) × 3 (experimental conditions) × 3 (technical replicates) analysed PCR reactions (Cq values) per candidate reference gene. At the end of each extension step SYBR®Green I fluorescence was measured at 521 nm.
Amplification and primer efficiency and validation
RT-qPCR efficiency over all samples (n = 18)37 was calculated both sample-specific51 (amplification efficiency EA, LinRegPCR software53,54,60, http://LinRegPCR.HFRC.nl) and factor-specific (primer efficiency EP 61) according to the MIQE guidelines11,39,40. For primer efficiency determination a 6x log10 serial dilution series of a random cDNA sample (untreated group) was amplified in triplet (three technical replicates per dilution level) for each candidate reference gene and the limit of detection (LOD) as the highest dilution, at which 95% (all three) of the technical replicates are detectable, was determined. Standard curves were created by linear regression of the resulting Cq values with the relative cDNA dilution (Supplementary Data 4) within the linear dynamic range (LDR) and the corresponding coefficients of determination r2 as well as qPCR primer efficiencies (EP) derived from the slope of the standard curve: EP = (10−1/slope − 1) × 100%. Only primer pairs with a linear relation between Cq and the log-transformed cDNA copy number (r2 > 0.98) were considered as possible valid reference gene candidates39. In addition, only efficiencies E within the range of 90–110% were deemed acceptable. Specific amplification of target reference genes was assessed by agarose gel electrophoreses (single band, correct size)39 and a specific peak in melting curve analysis (95 °C/15 s, 60 °C/15 s, then continuous temperature increase to 95 °C with fluorescence measurement for 20 min, Supplementary Data 3). For agarose gel electrophoresis each qPCR product (7 µl) was mixed with sucrose loading buffer (3 µl) and loaded on a 1.5% agarose gel, which was prepared with Gel Red (6 µl, 41003, Biotrend Chemikalien GmbH, Köln, Germany). The amplification products were separated parallel to a 100 bp DNA ladder at 120 V for 40 min in TAE buffer. Fluorescent bands were visualized by the gel documentation system Genoplex 2 and its software GenoSoft (VWR International GmbH, Darmstadt, Germany). Intraassay reliability was assessed per gene via the standard deviation of the arithmetic mean of Cq of the technical replicates (triplets) across all biological replicates (samples). Repeatability was deemed sufficient, if maximum SD was <0.553 Cq 10. For each primer pair and qPCR run we also tested a no-template-control (NTC) without cDNA and a -RT control (cDNA synthesis without reverse transcriptase added) on the same plate to exclude possible bias by primer dimers, contaminating or genomic DNA.
Data analysis and statistics
Cq values, defined as the second derivative maximum of the fluorescence signal curve, were calculated with the realplex software (version 2.2, Eppendorf AG, CalqPlex algorithm, Automatic Baseline, Drift Correction On). An arithmetic mean of each Cq triplett per gene and sample was used for further analysis. The stability of each candidate gene was calculated with four different mathematical algorithms: geNorm29, NormFinder36, BestKeeper37 and the comparative ΔCq method31. Stability calculations were done with the official Microsoft-Excel-based software applets for geNorm, NormFinder and BestKeeper according to developers’ instructions. For the comparative ΔCq method manual calculations were performed31. The geNorm and NormFinder algorithms require the transformation of the raw Cq data to linear scale expression quantities Q10,26 corresponding to the qPCR (primer) efficiency (E) of each gene: Q = EP −(Cqmin-Cqsample). The genes were ranked according to their stability values (geNorm: M, NormFinder: ρig/σi, deltaCT: mean SD of ∆Cq; BestKeeper: Pearson’s r) for each algorithm and experimental condition as well as combined conditions and a rank sum of all algorithms calculated per gene for final stability assessment with the smallest rank sum indicating the most stable reference gene (Table 3). The geNorm algorithm allowed a calculation of the ideal number of reference genes for reliable RT-qPCR normalization29. If pairwise variation (Vn/Vn + 1) between two sets of reference genes with one set including an additional reference gene was ≤0.15, this additional gene was deemed unnecessary for normalization (Fig. 3a). To assess ranking variations between algorithms, we used IBM SPSS Statistics® 23 (IBM, Armonk, NY, USA) to create a correlation matrix of bivariate correlations (Pearson´s correlation coefficient r, two-sided, normality confirmed by Shapiro-Wilk tests and histogram evaluation) of the overall pooled stability values as calculated by two respective algorithms.
Data availability statement
All datasets are publically available either as supplementary information to this article or upon request from the corresponding author. RT-qPCR experiments are in agreement with the MIQE (Minimum Information for Publication of Quantitative Real-Time PCR Experiments) guidelines11. The MIQE checklist (http://www.rdml.org/miqe) is provided as supplementary information (Supplementary Table 1).
References
Kirschneck, C. et al. Interactive effects of periodontitis and orthodontic tooth movement on dental root resorption, tooth movement velocity and alveolar bone loss in a rat model. Ann Anat 210, 32–43 (2017).
Meikle, M. C. The tissue, cellular, and molecular regulation of orthodontic tooth movement: 100 years after Carl Sandstedt. Eur J Orthod 28, 221–240 (2006).
Kanzaki, H., Chiba, M., Shimizu, Y. & Mitani, H. Periodontal ligament cells under mechanical stress induce osteoclastogenesis by receptor activator of nuclear factor kappaB ligand up-regulation via prostaglandin E2 synthesis. J Bone Miner Res 17, 210–220 (2002).
d’Apuzzo, F. et al. Biomarkers of periodontal tissue remodeling during orthodontic tooth movement in mice and men: overview and clinical relevance. Scientific World J 2013, 105873 (2013).
Wolf, H. F. & Hassell, T. M. Periodontology (Georg Thieme, Stuttgart, 2006).
Jacob, F. et al. Careful selection of reference genes is required for reliable performance of RT-qPCR in human normal and cancer cell lines. PloS one 8, e59180 (2013).
Kozera, B. & Rapacz, M. Reference genes in real-time PCR. J Appl Genet 54, 391–406 (2013).
Huggett, J., Dheda, K., Bustin, S. & Zumla, A. Real-time RT-PCR normalisation; strategies and considerations. Genes Immun 6, 279–284 (2005).
Nygaard, V. & Hovig, E. Methods for quantitation of gene expression. Front Biosci 14, 552–569 (2009).
Kirschneck, C. et al. Reference genes for valid gene expression studies on rat dental, periodontal and alveolar bone tissue by means of RT-qPCR with a focus on orthodontic tooth movement and periodontitis. Ann Anat 204, 93–105 (2016).
Bustin, S. A. et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55, 611–622 (2009).
Römer, P., Köstler, J., Koretsi, V. & Proff, P. Endotoxins potentiate COX-2 and RANKL expression in compressed PDL cells. Clin Oral Investig 17, 2041–2048 (2013).
Wolf, M. et al. CD8+ T cells mediate the regenerative PTH effect in hPDL cells via Wnt10b signaling. Innate Immun 22, 674–681 (2016).
Lossdörfer, S., Kraus, D. & Jäger, A. Aging affects the phenotypic characteristics of human periodontal ligament cells and the cellular response to hormonal stimulation in vitro. J Periodontal Res 45, 764–771 (2010).
Kirschneck, C., Meier, M., Bauer, K., Proff, P. & Fanghänel, J. Meloxicam medication reduces orthodontically induced dental root resorption and tooth movement velocity: a combined in vivo and in vitro study of dental-periodontal cells and tissue. Cell Tissue Res 368, 61–78 (2017).
Kirschneck, C., Proff, P., Maurer, M., Reicheneder, C. & Römer, P. Orthodontic forces add to nicotine-induced loss of periodontal bone. An in vivo and in vitro study. J Orofac Orthop 76, 195–212 (2015).
Proff, P., Reicheneder, C., Faltermeier, A., Kubein-Meesenburg, D. & Römer, P. Effects of mechanical and bacterial stressors on cytokine and growth-factor expression in periodontal ligament cells. J Orofac Orthop 75, 191–202 (2014).
Nettelhoff, L. et al. Influence of mechanical compression on human periodontal ligament fibroblasts and osteoblasts. Clin Oral Investig 20, 621–629 (2016).
Im, J. et al. Enterococcus faecalis lipoteichoic acid suppresses Aggregatibacter actinomycetemcomitans lipopolysaccharide-induced IL-8 expression in human periodontal ligament cells. Int Immunol 27, 381–391 (2015).
Patil, C., Rossa, C. & Kirkwood, K. L. Actinobacillus actinomycetemcomitans lipopolysaccharide induces interleukin-6 expression through multiple mitogen-activated protein kinase pathways in periodontal ligament fibroblasts. Oral Microbiol Immunol 21, 392–398 (2006).
Kim, Y.-S. et al. Nicotine and lipopolysaccharide stimulate the production of MMPs and prostaglandin E2 by hypoxia-inducible factor-1α up-regulation in human periodontal ligament cells. J Periodontal Res 47, 719–728 (2012).
Liu, J. et al. Porphyromonas gingivalis promotes the cell cycle and inflammatory cytokine production in periodontal ligament fibroblasts. Arch Oral Biol 60, 1153–1161 (2015).
Mah, S.-J. et al. Induction of S100A4 in periodontal ligament cells enhances osteoclast formation. Arch Oral Biol 60, 1215–1221 (2015).
Jönsson, D., Nebel, D., Bratthall, G. & Nilsson, B.-O. The human periodontal ligament cell. A fibroblast-like cell acting as an immune cell. J Periodontal Res 46, 153–157 (2011).
Chervoneva, I. et al. Selection of optimal reference genes for normalization in quantitative RT-PCR. BMC Bioinformatics 11, 253 (2010).
Liu, D. et al. Validation of reference genes for gene expression studies in virus-infected Nicotiana benthamiana using quantitative real-time PCR. PloS one 7, e46451 (2012).
Tan, S. C. et al. Identification of valid housekeeping genes for quantitative RT-PCR analysis of cardiosphere-derived cells preconditioned under hypoxia or with prolyl-4-hydroxylase inhibitors. Mol Biol Rep 39, 4857–4867 (2012).
Bustin, S. A. Real-time, fluorescence-based quantitative PCR: a snapshot of current procedures and preferences. Expert Rev Mol Diagn 5, 493–498 (2005).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3, RESEARCH0034 (2002).
Dheda, K. et al. The implications of using an inappropriate reference gene for real-time reverse transcription PCR data normalization. Anal Biochem 344, 141–143 (2005).
Silver, N., Best, S., Jiang, J. & Thein, S. L. Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol Biol 7, 33 (2006).
Chey, S., Claus, C. & Liebert, U. G. Validation and application of normalization factors for gene expression studies in rubella virus-infected cell lines with quantitative real-time PCR. J Cell Biochem 110, 118–128 (2010).
Brugè, F., Venditti, E., Tiano, L., Littarru, G. P. & Damiani, E. Reference gene validation for qPCR on normoxia- and hypoxia-cultured human dermal fibroblasts exposed to UVA. Is β-actin a reliable normalizer for photoaging studies? J Biotechnol 156, 153–162 (2011).
Reuther, S., Reiter, M., Raabe, A. & Dikomey, E. Effect of irradiation on the expression of DNA repair genes studied in human fibroblasts by real-time qPCR using three methods of reference gene validation. Radiat Environ Biophys 52, 463–469 (2013).
Proff, P. & Römer, P. The molecular mechanism behind bone remodelling. A review. Clin Oral Investig 13, 355–362 (2009).
Andersen, C. L., Jensen, J. L. & Ørntoft, T. F. Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64, 5245–5250 (2004).
Pfaffl, M. W., Tichopad, A., Prgomet, C. & Neuvians, T. P. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol Lett 26, 509–515 (2004).
Ye, J. et al. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 13, 134 (2012).
Taylor, S., Wakem, M., Dijkman, G., Alsarraj, M. & Nguyen, M. A practical approach to RT-qPCR-Publishing data that conform to the MIQE guidelines. Methods 50, 5 (2010).
Taylor, S. C. & Mrkusich, E. M. The state of RT-quantitative PCR: firsthand observations of implementation of minimum information for the publication of quantitative real-time PCR experiments (MIQE). J Mol Microbiol Biotechnol 24, 46–52 (2014).
Thornton, B. & Basu, C. Real-time PCR (qPCR) primer design using free online software. Biochem Mol Biol Educ 39, 145–154 (2011).
Hoffmann, H. & Schiene-Fischer, C. Functional aspects of extracellular cyclophilins. Biol Chem 395, 721–735 (2014).
Gagliardi, A. et al. Cytoskeleton and nuclear lamina affection in recessive osteogenesis imperfecta: A functional proteomics perspective. J Proteomics 167, 46–59 (2017).
Savinkova, L. et al. An experimental verification of the predicted effects of promoter TATA-box polymorphisms associated with human diseases on interactions between the TATA boxes and TATA-binding protein. PloS one 8, e54626 (2013).
Yang, M. et al. Interaction of ribosomal protein L22 with casein kinase 2α: a novel mechanism for understanding the biology of non-small cell lung cancer. Oncol Rep 32, 139–144 (2014).
Yoshihama, M. et al. The human ribosomal protein genes: sequencing and comparative analysis of 73 genes. Genome Res 12, 379–390 (2002).
Zhang, Y. et al. Ribosomal Proteins Rpl22 and Rpl22l1 Control Morphogenesis by Regulating Pre-mRNA Splicing. Cell Rep 18, 545–556 (2017).
Uddin, M. J. et al. Age-related changes in relative expression stability of commonly used housekeeping genes in selected porcine tissues. BMC Res Notes 4, 441 (2011).
Li, B. et al. Identification of optimal reference genes for RT-qPCR in the rat hypothalamus and intestine for the study of obesity. Int J Obes (Lond) 38, 192–197 (2014).
Koretsi, V., Kirschneck, C., Proff, P. & Römer, P. Expression of glutathione peroxidase 1 in the spheno-occipital synchondrosis and its role in ROS-induced apoptosis. Eur J Orthod 37, 308–313 (2015).
Liu, W. & Saint, D. A. A New Quantitative Method of Real Time Reverse Transcription Polymerase Chain Reaction Assay Based on Simulation of Polymerase Chain Reaction Kinetics. Anal Biochem 302, 52–59 (2002).
Robledo, D. et al. RNA-seq analysis reveals significant transcriptome changes in turbot (Scophthalmus maximus) suffering severe enteromyxosis. BMC Genomics 15, 1149 (2014).
Ruijter, J. M. et al. Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res 37, e45 (2009).
Ruijter, J. M., Lorenz, P., Tuomi, J. M., Hecker, M. & van den Hoff, M. J. B. Fluorescent-increase kinetics of different fluorescent reporters used for qPCR depend on monitoring chemistry, targeted sequence, type of DNA input and PCR efficiency. Mikrochim Acta 181, 1689–1696 (2014).
Taki, F. A., Abdel-Rahman, A. A. & Zhang, B. A comprehensive approach to identify reliable reference gene candidates to investigate the link between alcoholism and endocrinology in Sprague-Dawley rats. PloS one 9, e94311 (2014).
Mahanty, A., Purohit, G. K., Mohanty, S., Nayak, N. R. & Mohanty, B. P. Suitable reference gene for quantitative real-time PCR analysis of gene expression in gonadal tissues of minnow Puntius sophore under high-temperature stress. BMC Genomics 18, 617 (2017).
Iwata, T. et al. Validation of human periodontal ligament-derived cells as a reliable source for cytotherapeutic use. J Clin Periodontol 37, 1088–1099 (2010).
Marchesan, J. T., Scanlon, C. S., Soehren, S., Matsuo, M. & Kapila, Y. L. Implications of cultured periodontal ligament cells for the clinical and experimental setting. A review. Arch Oral Biol 56, 933–943 (2011).
Schroeder, A. et al. The RIN: an RNA integrity number for assigning integrity values to RNA measurements. BMC Mol Biol 7, 3 (2006).
Tuomi, J. M., Voorbraak, F., Jones, D. L. & Ruijter, J. M. Bias in the Cq value observed with hydrolysis probe based quantitative PCR can be corrected with the estimated PCR efficiency value. Methods 50, 313–322 (2010).
Pfaffl, M. W. & Hageleit, M. Validities of mRNA quantification using recombinant RNA and recombinant DNA external calibration curves in real-time RT-PCR. Biotechnol Lett 23, 275–282 (2001).
Acknowledgements
The authors wish to thank Mrs Eva Zaglauer and Dipl.-Biol. Kathrin Bauer for their support in performing the RT-qPCR analyses as well as Prof. Dr. rer. nat. Susanne Grässel, Head of the Experimental Orthopedics Laboratory (ZMB/BioPark 1, Regensburg), for generously providing the Agilent 2100 Bioanalyzer and corresponding equipment, and Prof. Dr. rer. nat. Helmut Schweikl, Head of the Experimental Operative Dentistry Laboratory (University Medical Centre Regensburg) for providing the NanoDrop ND-2000. The authors also thank the German Orthodontic Society (DGKFO) for their financial support and funding (Kirschneck 12–01–2015). This work was supported by the German Research Foundation (DFG) within the funding programme Open Access Publishing.
Author information
Authors and Affiliations
Contributions
C.K. conceived the idea of the study and the study design as well as designed and validated the used primer pairs. S.B., P.P. and A.S. contributed to discussion and study design. A.S. and C.K. conducted the experiments. A.S., C.K. and S.B. analysed the results. J.K. produced and contributed the Agac bacterial lysate. G.S. provided the primary human periodontal ligament fibroblasts. C.K. and A.S. wrote the manuscript and created the figures, tables and the supplementary material. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kirschneck, C., Batschkus, S., Proff, P. et al. Valid gene expression normalization by RT-qPCR in studies on hPDL fibroblasts with focus on orthodontic tooth movement and periodontitis. Sci Rep 7, 14751 (2017). https://doi.org/10.1038/s41598-017-15281-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15281-0
This article is cited by
-
Effect of the antirheumatic medication methotrexate (MTX) on biomechanical compressed human periodontal ligament fibroblasts (hPDLFs)
BMC Oral Health (2024)
-
Validation of reliable reference genes for qPCR of CD4+ T cells exposed to compressive strain
Journal of Orofacial Orthopedics / Fortschritte der Kieferorthopädie (2024)
-
Impact of phosphorylation of heat shock protein 27 on the expression profile of periodontal ligament fibroblasts during mechanical strain
Journal of Orofacial Orthopedics / Fortschritte der Kieferorthopädie (2023)
-
Selection and validation of reference gene for RT-qPCR studies in co-culture system of mouse cementoblasts and periodontal ligament cells
BMC Research Notes (2022)
-
Xanthohumol exerts anti-inflammatory effects in an in vitro model of mechanically stimulated cementoblasts
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.