Abstract
Invasions are ecologically destructive and can threaten biodiversity. Trophic flexibility has been proposed as a mechanism facilitating invasion, with more flexible species better able to invade. The termite hunting needle ant Brachyponera chinensis was introduced from East Asia to the United States where it disrupts native ecosystems. We show that B. chinensis has expanded dietary breadth without shifting trophic position in its introduced range. Transect sampling of ants and termites revealed a negative correlation between the abundance of B. chinensis and the abundance of other ants in introduced populations, but this pattern was not as strong in the native range. Both termite and B. chinensis abundance were higher in the introduced range than in native range. Radiocarbon (14C) analysis revealed that B. chinensis has significantly younger ‘diet age’, the time lag between carbon fixation by photosynthesis and its use by the consumer, in the introduced range than in the native range, while stable isotope analyses showed no change. These results suggest that in the introduced range B. chinensis remains a termite predator but also feeds on other consumer invertebrates with younger diet ages such as herbivorous insects. Radiocarbon analysis allowed us to elucidate cryptic dietary change associated with invasion success.
Similar content being viewed by others
Introduction
Tens of thousands of species have been introduced, either intentionally or inadvertently, to locations outside their native ranges1. A subset of these species become established in the new locations as invasive species where they have negative impacts on native communities and ecosystems. These invasive species are now recognized as the second most important cause of species extinctions2. Species introductions have been the subject of an enormous body of research with thousands of published articles. However, why some species cause such impacts and others do not remains enigmatic.
Ants represent a particularly interesting taxonomic group for studying biological invasions mechanisms as they count among the most ecologically and economically important groups of biological invaders, with five species on the list of the 100 most problematic invaders3. Invasive ants often become highly abundant in their introduced range where they reduce native ant diversity and affect other organisms both directly and indirectly as has been well-documented for argentine ants4,5, fire ants6, crazy ants7,8,9, and Asian needle ants10 among others. The ecological effects of invasive ants in introduced ranges depend on a complex interaction of behavioural, ecological and genetic factors, each independently increasing the numerical abundance of the introduced species relative to native species11. Recently, a number of studies have highlighted the possibility of species that exhibit behavioral or ecological flexibility may be particularly likely to have strong effects on local communities12,13,14.
In particular, invasiveness may be facilitated by the ability of ants to shift their diets during invasion, whether to consume a wider breath of food items or even, potentially, to consume their competitors. Dietary shifts in introduced populations have been reported in the Argentine ant (Linepithema humile) and in the red imported fire ant (Solenopsis invicta), both considered among the world’s most destructive introduced species13,14. These omnivorous ants shifted trophic positions when they were introduced; in their invasive ranges both species shifted to diets with a greater dependence on honeydew-producing Hemiptera13,14. In the case of S. invicta, intense competition from other native ants, including L. humile, limits access to honeydew resources in its native range14, but it is able to dominate these resources in its introduced range in the U.S. It has been proposed that the access to carbohydrates provided by hemipterans contributes to competitive performance of these ants and enhances their ecological success in introduced ranges14,15, possibly through increased colony growth associated with greater honeydew consumption14. Although the importance of dietary flexibility in invasion success has been studied in hemipteran mutualist ants, nothing is known about potential dietary changes in ants less able to rely on honeydew.
The Asian needle ant, Brachyponera chinensis (formerly Pachycondyla chinensis), widely distributed from Far Eastern Asia to Southeast Asia, was introduced into the southeastern United States in the early 19th century16 (Fig. 1). It has since become an abundant invasive species, even within relatively undisturbed forests. The occurrence and increased abundance of B. chinensis diminishes the abundance and diversity of native ants10,17. Moreover, B. chinensis disrupts ant-seed dispersal mutualisms by displacing native ant species, especially the keystone mutualist Aphaenogaster species17,18. Unlike the Argentine ant and the red imported fire ant, B. chinensis does not rely on hemipteran-mutualist providing carbohydrates but is instead a predator of termites in the genus Reticulitermes in both its native19 and invaded20 ranges. It has been suggested that the availability of termite prey in the southeastern United States, combined with the absence of specialist predators of termites, might have facilitated the success of B. chinensis 10,20,21.
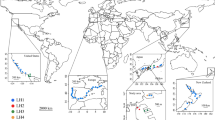
Map displaying the locations of study sites in the native (Japan) and introduced (United States) ranges of the Asian needle ant Brachyponera chinensis. The sampling design consisted of twelve 900-m line transects, seven in Japan (T1-T7) and five in the United States (T7-T12). Maps were created with the QGIS software (version 2.18; http://www.qgis.org/en/site/forusers/download.html).
An important limitation in this area of research concerns the absence of observational data of ant diets. To investigate dietary shifts in invasions of hemipteran-mutualist ants, earlier studies assessed how their trophic positions differ between native and introduced populations by comparing stable nitrogen isotope data (δ15N). However, the same approach may not be applicable to B. chinensis because this species does not tend honeydew excreting hemipterans and thus dietary changes of this predatory invasive ant may occur without shifts in trophic position. An alternative approach is to use other environmentally acquired elements to compare the composition of prey in native and introduced ranges. The use of the natural abundance of radiocarbon (14C) makes it possible to explore feeding habits of terrestrial consumers, which could not be revealed by more conventional analyses22,23,24. The testing of nuclear bombs after World War II doubled 14C concentration of atmospheric CO2. Concentrations have been, predictably, decreasing since the nuclear ban treaty in 196325. The steady decline of atmospheric 14CO2 with time makes it possible to estimate the ‘diet age’ of a consumer, which is defined as the average time lag between carbon fixation by photosynthesis and its use by the consumer, with an accuracy of 1–2 years26. Earlier studies showed a strong association between diet ages of consumers and their known feeding habits23,26,27,28,29. For example, wood-feeding termites that feed on wood have old diet ages (ca. 10 to > 50 years old). The diet ages of litter-feeding invertebrates (earthworms and termites) range from 3 to 6 years24,28. Ant species that consume honeydew and nectar (Polyrhachis sp.) have a younger diet age (just one year) than strictly predatory ants (e.g., Leptogenys diminuta) (6 years)24.
To examine dietary shifts of B. chinensis in its introduced range, we conducted stable carbon (13C) and nitrogen (15N) isotope analyses of termites and ants in both the native and introduced ranges of B. chinensis. We also measured diet ages of B. chinensis by using radiocarbon 14C. We found that B. chinensis has markedly changed its prey composition and now relies less on termites in introduced range and more on other non-decomposer invertebrates.
Results and Discussion
Diet ages of termites are associated with feeding habits: termites that inhabit and consume large-diameter wood have older diet ages than litter-feeding and soil-feeding species24. As expected, our carbon (14C) analysis demonstrated that wood-feeding Reticulitermes termites have old diet ages both in Japan (23–48 years, N = 18) and in the Unites States (17–36 years, N = 15), and did not differ between these regions (Fig. 2a; Dataset S1). The diet age of B. chinensis in both its native range (18–35 years, N = 14) and in its introduced range (3–18 years, N = 13) were much older than the oldest diet age of the ants reported in previous studies, i.e., Aenictus and Leptogenys genera (4–6 years)24. The extremely old diet ages of B. chinensis are in line with the hypothesis that B. chinensis relies on termites as prey10,19,20,21, particularly in its native range. However, our comparison of B. chinensis in its native and introduced range showed that the diet age of B. chinensis in the introduced range was significantly younger than that in the native range (Fig. 2a). We hypothesize, in light of these results, that the Asian needle ant has expanded its diet after invasion and now feeds on prey items that have younger diet ages than wood-feeding termites, most likely other invertebrates such as litter-feeding detritivores and herbivores.
Diet ages and trophic ecology of B. chinensis in the native range (Japan) and in the introduced range (United States). (a) Diet ages (least square means ± SE) of B. chinensis and termites in the native range (Japan) and in the introduced range (United States). Both the study sites and the sample types had significant effects on diet ages of B. chinensis and termites (F = 14.19, P = 0.0037 and F = 133.61, P < 0.0001, respectively). Interaction effect of native/introduced and sample type was also significant (F = 12.79, P = 0.0008), where the diet age of B. chinensis in the introduced range was significantly younger than that of B. chinensis in the native range, but there was no significant difference in the diet ages of termites. Different letters on the bars indicate significant differences among groups (P < 0.05, Tukey-Kramer test). (b) There was no significant difference between the native range and the introduced range in δ13C (least square means ± SE) (F = 0.0150, P = 0.904) and in δ15N (least square means ± SE) (F = 0.1809, P = 0.678), there was a while highly significant difference among sample types in δ13C (F = 218.47, P < 0.0001) and in δ15N (F = 287.68, P < 0.0001). The interaction term had no significant effect on δ13C (F = 1.81, P = 0.170) or δ15N (F = 1.94, P = 0.149).
Within a typical ecological community δ13C and δ15N values are higher in animals with more carnivorous diets, and lower in those that are herbivores (e.g.30,31,32). Earlier studies on trophic ecology of invasive ants showed that the Argentine ant L. humile and the red imported fire ant S. invicta increased reliance on plant-based resources and thus shifted to lower trophic positions after invasion13,14. In contrast to these cases, our stable isotope analysis demonstrated that there was no significant difference in δ15N and δ13C values of litter (native: N1 = 7, introduced: N2 = 5), termites (N1 = 23, N2 = 28) and B. chinensis (N1 = 21, N2 = 25) between the native and introduced range (Fig. 2b; Dataset S2), indicating no significant change in trophic position following its invasion; the ants are still highly predatory. Unlike L. humile and S. invicta, the Asian needle ant does not consume plant-based carbohydrates provided by honeydew-producing Hemiptera (Fig. 2b). However, even without shifting its trophic position in its introduced range B. chinensis appears to acquire carbon from younger sources in its introduced range than in its native range (Fig. 2a). This pattern suggests that native populations of B. chinensis are more dependent on prey of old diet age, i.e., termites, and introduced populations consume more prey of younger diet age such as herbivores and litter-feeding invertebrates. Although earlier studies illuminated the importance of exploitation of plant-based resources in ant invasions, our results suggest that increased dietary flexibility within the same trophic level might also facilitate large impacts.
There was no significant relationship between the number of B. chinensis nests and the abundance of termites in Japan (χ 2 = 1.01, P = 0.32; Table S1) or in the United States (χ 2 = 2.58, P = 0.11; likelihood ratio test; Table S2, Fig. 3a). Both termite nest density and B. chinensis nest density were higher in the introduced range than in native range (termite nest: χ 2 = 15.64, P < 0.001; B. chinensis nest: χ 2 = 7.74, P < 0.01, likelihood ratio test). The probability of termite presence in logs with B. chinensis was significantly higher than in those without B. chinensis both in the native (χ 2 = 4.126, P < 0.05) and introduced ranges (χ 2 = 5.243, P < 0.05; likelihood ratio test; Fig. 3b). Colony size of Reticulitermes termites is between tens of thousands to hundreds of thousands of individuals33, while nest size of B. chinensis ranges from 100 to several thousands of individuals34. As long as B. chinensis colonies live with termite colonies in the same piece of wood, termites are an abundant, high quality, renewable food supply, in many ways similar to the hemipteran honeydew exploited by most other invasive ant species.
The abundance of B. chinensis and termite nests collected at each sampling point. (a) Relationship between the number of B. chinensis nests and the abundance of termites in the native and introduced ranges of B. chinensis. We collected 73 R. speratus nests in Japan and 121 R. flavipes, 51 R. virginicus and one R. hageni nests in the United States. There was no significant relationship between the number of B. chinensis nests and the abundance of termites in Japan (χ 2 = 1.01, P = 0.32) and in the United States (χ 2 = 2.58, P = 0.11; likelihood ratio test). The size of each plot is proportional to the number of data points it represents. (b) Comparison of termite presence between the downed logs with and without B. chinensis colonies. The proportion of logs containing termites was higher than those without B. chinensis both in the native (χ 2 = 4.126, P < 0.05) and introduced ranges (χ 2 = 5.243, P < 0.05; likelihood ratio test).
On the other hand, the density of B. chinensis nests was highly negatively associated with nest density of log-nesting native ant species in the introduced range (χ 2 = 11.99, P < 0.001), while only marginally in the native range (χ 2 = 3.84, P = 0.050; Fig. 4a). Similarly to previous studies10,17,18, the presence of B. chinensis nests was strongly associated with lower native ant diversity in the introduced range (χ 2 = 29.68, P < 0.0001) but not within its native range (χ 2 = 1.51, P = 0.22; likelihood ratio test; Table 1, Fig. 4b). The low species diversity observed in areas invaded by B. chinensis, has previously been hypothesized as a consequence of B. chinensis increased predation on ants in its introduced range in comparison to its native range10. Our results, however, suggest that this scenario is unlikely in as much as we observed no differences between the δ15N and δ13C values of B. chinensis between native and introduced ranges (which would be expected if B. chinensis were feeding on ants since ants occupy a higher trophic level than do termites). We suggest instead that the strong effect of B. chinensis on native ants is more likely due to the ability of B. chinensis to compete for (and draw down) resources resulting from its expanded dietary breadth.
Effect of B. chinensis on other ant species. (a) There was a significant negative relationship between the number of B. chinensis nests and the number of nests of other ants found at the same point (χ 2 = 29.68, P < 0.0001), while marginally non-significant in Japan (χ 2 = 3.84, P = 0.05; likelihood ratio test). (b) There was a significant negative relationship between the number of B. chinensis nests and the number of the other ant species found at the same point (χ 2 = 11.99, P < 0.001), while there was no significant relationship in Japan (χ 2 = 1.51, P = 0.22; likelihood ratio test). The size of each plot is proportional to the number of data points it represents.
Overall, we demonstrate a marked expansion in feeding habits of an introduced ant. Even such a seemingly subtle dietary change in new environments can cause large impacts on the communities of introduced regions, impacts that cannot be easily predicted from the feeding habits in the native range.
Methods
Study sites
To compare colony density and feeding habits of B. chinensis between the native range (Japan) and the introduced range (United States), we conducted transect sampling in Okayama in Western Japan and in Raleigh in the United States. The climate of Raleigh is very similar to that of Okayama with mean annual precipitation around 1100 mm. The temperature in Raleigh ranges from mean annual minimum of −1 °C to mean annual maximum of 32 °C, and that in Okayama ranges from mean annual minimum of 1 °C to mean annual maximum of 33 °C.
In the native range, we conducted transect sampling at seven sites (T1-T7) in six secondary forests (Kasaiyama (T1: N34.718900, E133.934233), Washuzan (T2: N34.435047, E133.816467), Hattantoge (T3: N34.691147, E134.184311), Shimotakada (T4: N34.764981, E133.833881), Takahashi (T5: N34.795536, E133.576878), Ryuten (T6: N34.894858, E134.006953, T7: N34.893608, E134.001953)) in Okayama, Western Japan between 10am and 4pm (Fig. 1). These forests are dominated by Japanese red pine Pinus densiflora and Quercus serrata oak. One transect was run in each forest except for Ryuten where we ran two transects. The minimum distance between any two neighboring forests was 10 km.
In the introduced range, we conducted transect sampling at five sites (T8-T12) in four mature mesic deciduous forests (Schenck Memorial Forest (T8: N35.816322, W78.720964), Yates Mill Pond forest (T9: N35.721522, W78.689622), Lake Wheeler forest (T10: N35.693786, W78.698669) and Lake Johnson forest (T11: N35.762825, W78.716164, T12: N35.758286, W78.716264)) in Raleigh, North Carolina, USA between 10 am and 4 pm (Fig. 1). One transect was run in each forest except two transects were run in the Lake Johnson forest. The minimum distance between any two neighbouring forests was 3 km. This area is currently near the centre of the B. chinensis invaded range10,35.
Transect sampling
Ant and termite sampling was conducted along seven 900-m line transects in Japan (T1-T7) and five transects in the United States (T8-T12). Along each transect we sampled 10 points, one every 100 m. Sampling was conducted in a 10-m radius around each sampling point by six trained people. In order to standardize sampling effort, ten dead logs were randomly chosen in each circle. All termites and ants were carefully extracted from logs by using a hand axe, a knife and an aspirator (Dataset S3 and S4). We defined a B. chinensis nest as an aggregation of more than 10 individuals in the logs and excluded the individuals walking on the surface of the logs from nest counts. Litter was also sampled from the forest floor as the baseline of δ15N, δ13C, and ∆14C values at each sampling site. For stable isotope and radiocarbon analyses, samples were oven-dried at 60 °C for 24 h. The termites and the ants were also placed in vials filled with 95% ethanol for species identification.
Stable isotope analyses
For stable C and N isotope analyses, the samples (approximately 0.5 mg: a few individuals of termites and ants from each colony) were placed in folded tin capsules. Stable C and N isotope ratios were measured using a mass spectrometer (Deltaplus XP, Thermo Electron, Erlangen, Germany) coupled with an elemental analyser (Flash EA 1112, Thermo Electron, Erlangen, Germany). The precision of the on-line procedure was better than ± 0.2‰ for both isotope ratios. Natural abundances of 13C and 15N are expressed in per mil (‰) deviation from international standards: δ13C or δ15N = (R sample/R standard − 1) × 1000, where R in δ13C or δ15N is 13C/12C or 15N/14N, respectively. Pee Dee Belemnite and atmospheric nitrogen were used as the international standards for carbon and nitrogen, respectively.
Radio isotope analyses and diet age estimation
For radiocarbon analysis, the samples (estimated to contain approximately 1 mg C: several individuals of termites and ants from each colony) were combusted in evacuated and sealed Vycor tubes with CuO and Ag wire at 850 °C for 2h. After cooling, the Vycor tubes were cracked on a vacuum line, and the CO2 was cryogenically purified. The purified CO2 was graphitized under Fe catalysis at 650 °C for 4.5h36. The graphite samples were sent to Rafter Radiocarbon Laboratory, Institute of Geological and Nuclear Sciences, New Zealand, for accelerator mass spectrometry measurements of radiocarbon. Radiocarbon values were reported as ∆14C (‰), which is the part per thousand deviations from the activity of 19th century wood and were corrected for fractionation using stable C isotope ratios of the samples37,38. The average analytical error was ± 3.7‰.
We estimated the diet age by calculating the difference between the sample collection year and the year (t) when the ∆14C value of a sample matched that of atmospheric CO2, as described previously by Hyodo et al.27. The year (t) was calculated using the regression curve, year (t) = 2074 − 16.71 ln(∆14C) (R2 = 0.997, P < 0.0001), which is based on 14CO2 data for the northern hemisphere from 1977 to 199938. Because the deciduous leaf litter were collected both in Japan and United States, the ages of litter was expected to be 0 years. However, our results showed that the leaf litter samples collected both in Japan and United States showed negative ages (Dataset S1) likely due to the effect of fossil fuel burning on atmospheric 14CO2 39,40. Therefore, we corrected the diet ages of ants and termites for the fossil fuel effects by adding years so that leaf litter samples had ages of 0 years old. The analytical error of ∆14C indicated that the diet age could be estimated with a precision of 1−2 years. The time elapsed since the production of the litter was also estimated as diet age for comparison.
Statistical Analyses
To explore differences in diet ages of ants, termites, and litter between Japan and United States, we applied a generalized linear mixed model (GLMM) in which the diet ages of ants, termites, and litter were treated as response variables, the study sites (native and introduced ranges), the sample types (i.e., ants, termites, and litter), and the interaction term (i.e., the study sites × the sample types) as explanatory variables, and the sampling points in Japan and United States as a random effect. We also used GLMM to examine differences in feeding habits between the native and introduced ranges using stable C and N isotopes. In this model, the δ13C and δ15N of ants, termites, and litter were treated as response variables, the study sites (native and introduced ranges), the sample types (ants, termites, and litter), and the interaction term (the study sites × the sample types) as explanatory variables, and the sampling locations as a random effect. A post hoc test (Tukey-Kramer HSD test) was applied to examine the overall significant difference at P = 0.05.
We used a GLMM with binomial errors in which the proportion of logs (out of 10) with termites in a sampling site was treated as a response variable, and GLMM with Poisson errors in which the number of ant species (except B. chinensis) in a sampling site was treated as a response variable. In these models, the proportion of logs with B. chinensis colonies in a sampling point was treated as an explanatory variable, and the transect as a random effect. We used GLMM with binomial errors in which termite presence/absence in a log was treated as a response variable, the presence/absence of B. chinensis in the log as an explanatory variable, and the sampling site nested in the transect as a random effect.
References
Dawson, W. et al. Global hotspots and correlates of alien species richness across taxonomic groups. Nat. Ecol. Evol. 1, 0186 (2017).
Bellard, C., Cassey, P. & Blackburn, T. M. Alien species as a driver of recent extinctions. Biol. Lett. 12, 20150623 (2016).
Invasive Species Specialist Group. Global Invasive Species Database. http://www.iucngisd.org/gisd/100_worst.php (2017).
Ward, P. S. Distribution of the introduced Argentine ant (Iridomyrmex humilis) in natural habitats of the lower Sacramento Valley and its effects on the indigenous ant fauna. Hilgardia 55, 1–16 (1987).
Holway, D. A. Effect of Argentine ant invasions on ground-dwelling arthropods in northern California riparian woodlands. Oecologia 116, 252–258 (1998).
Porter, S. D. & Savignano, D. A. Invasion of polygyne fire ants decimates native ants and disrupts arthropod community. Ecology 71, 2095–2106 (1990).
Gillespie, R. G. & Reimer, N. The effect of alien predatory ants (Hymenoptera: Formicidae) on Hawaiian endemic spiders (Araneae: Tetragnathidae). Pac. Sci. 47, 21–33 (1993).
Feare, C. Ants take over from rats on Bird Island, Seychelles. Bird Conserv. Int. 9, 95–96 (1999).
Meek., P. D. The decline and current status of the Christmas Island shrew Crocidura attenuata trichura on Christmas Island, Indian Ocean. Aust. Mammal. 22, 49 (2000).
Guénard, B. & Dunn, R. A new (old), invasive ant in the hardwood forests of eastern North America and its potentially widespread impacts. PLoS ONE 7, e11614 (2000).
Holway, D. A., Lach, L., Suarez, A. V., Tsutsui, N. D. & Case, T. J. The causes and consequences of ant invasions. Annu. Rev. Ecol. Syst. 33, 181–233 (2002).
Sol, D., Timmermans, S. & Lefebvre, L. Behavioural flexibility and invasion success in birds. Anim. Behav. 63, 495–502 (2002).
Tillberg, C. V., Holway, D. A., LeBrun, E. G. & Suarez, A. V. Trophic ecology of invasive Argentine ants in their native and introduced ranges. Proc. Natl. Acad. Sci. USA 104, 20856–20861 (2007).
Wilder, S. M., Holway, D. A., Suarez, A. V., LeBrun, E. G. & Eubanks, M. D. Intercontinental differences in resource use reveal the importance of mutualisms in fire ant invasions. Proc. Natl. Acad. Sci. USA 108, 20639–20644 (2011).
Helms, K. R. & Vinson, S. B. Widespread association of the invasive ant with an invasive mealybug. Ecology 83, 2425–2438 (2002).
Smith, M. R. Ponerine ants of the genus Euponera in the United States. Ann. Entomol. Soc. Am. 27, 557–564 (1934).
Warren, R. J., McMillan, A., King, J. R., Chick, L. & Bradford, M. A. Forest invader replaces predation but not dispersal services by a keystone species. Biol. Invasions 17, 3153–3162 (2015).
Rodriguez-Cabal, M. A., Stuble, K. L., Guénard, B., Dunn, R. R. & Sanders, N. J. Disruption of ant-seed dispersal mutualisms by the invasive Asian needle ant (Pachycondyla chinensis). Biol. Invasions 14, 557–565 (2012).
Matsuura, K. Colony-level stabilization of soldier head width for head-plug defense in the termite Reticulitermes speratus (Isoptera: Rhinotermitidae). Behav. Ecol. Sociobiol. 51, 172–179 (2002).
Bednar, D. M. & Silverman, J. Use of termites, Reticulitermes virginicus, as a springboard in the invasive success of a predatory ant, Pachycondyla (=Brachyponera) chinensis. Insect. Soc. 58, 459–467 (2011).
Bednar, D. M., Shik, J. Z. & Silverman, J. Prey handling performance facilitates competitive dominance of an invasive over native keystone ant. Behav. Ecol. 24, 1312–1319 (2013).
Tayasu, I. et al. Termite ecology in a dry evergreen forest in Thailand in terms of stable (delta C-13 and delta N-15) and radio (C-14, Cs-137 and Pb-210) isotopes. Ecol Res 17, 195–206 (2002).
Briones, M. J. I., Garnett, M. H. & Piearce, T. G. Earthworm ecological groupings based on C-14 analysis. Soil Biol. Biochem. 37, 2145–2149 (2005).
Hyodo, F., Matsumoto, T., Takematsu, Y. & Itioka, T. Dependence of diverse consumers on detritus in a tropical rain forest food web as revealed by radiocarbon analysis. Funct. Ecol. 29, 423–429 (2015).
Levin, I. et al. Observations and modelling of the global distribution and long-term trend of atmospheric 14CO2. Tellus B 62, 26–46 (2010).
Hyodo, F., Tayasu, I. & Wada, E. Estimation of the longevity of C in terrestrial detrital food webs using radiocarbon (C-14): how old are diets in termites? Funct. Ecol. 20, 385–393 (2006).
Hyodo, F. et al. Gradual enrichment of N-15 with humification of diets in a below-ground food web: relationship between N-15 and diet age determined using C-14. Funct. Ecol. 22, 516–522 (2008).
Hyodo, F., Uchida, T., Kaneko, N. & Tayasu, I. Use of radiocarbon to estimate diet ages of earthworms across different climate regions. Appl. Soil Ecol. 62, 178–183 (2012).
Haraguchi, T. F., Uchida, M., Shibata, Y. & Tayasu, I. Contributions of detrital subsidies to aboveground spiders during secondary succession, revealed by radiocarbon and stable isotope signatures. Oecologia 171, 935–944 (2013).
Bocherens, H. & Drucker, D. Trophic level isotopic enrichment of carbon and nitrogen in bone collagen: Case studies from recent and ancient terrestrial ecosystems. Int. J. Osteoarchaeol. 13, 46–53 (2003).
Fox-Dobbs, K., Bump, J. K., Peterson, R. O., Fox, D. L. & Koch, P. L. Carnivore-specific stable isotope variables and variation in the foraging ecology of modern and ancient wolf populations: case studies from Isle Royale, Minnesota, and La Brea. Can. J. Zool. 85, 458–471 (2007).
Palmqvist, P., Perez-Claros, J. A., Janis, C. M. & Grocke, D. R. Tracing the ecophysiology of ungulates and predator-prey relationships in an early Pleistocene large mammal community. Palaeogeogr. Palaeocl. 266, 95–111 (2008).
Howard, R. W., Jones, S. C., Mauldin, J. K. & Beal, R. H. Abundance, distribution, and colony size estimates for Reticulitermes spp. (Isoptera: Rhinotermitidae) in Southern Mississippi. Environ. Entomol. 11, 1290–1293 (1982).
MacGown, J. A. The Asian Needle Ant, Pachycondyla chinensis (Emery) (Hymenoptera: Formicidae), Reported from Alabama. Midsouth Entomol. 2, 88–89 (2009).
Nelder, M. P., Paysen, E. S., Zungoli, P. A. & Benson, E. P. Emergence of the introduced ant Pachycondyla chinensis (Formicidae: Ponerinae) as a public health threat in the southeastern United States. J. Med. Entomol. 43, 1094–1098 (2006).
Kitagawa, H., Masuzawa, T., Makamura, T. & Matsumoto, E. A batch preparation method for graphite targets with low background for AMS (super 14) C measurements. Radiocarbon 35, 295–300 (1993).
Stuiver, M. & Polach, H. A. Discussion; reporting of C-14 data. Radiocarbon 19, 355–363 (1977).
Hua, Q. & Barbetti, M. Review of tropospheric bomb C-14 data for carbon cycle modeling and age calibration purposes. Radiocarbon 46, 1273–1298 (2004).
Pataki, D. E., Randerson, J. T., Wang, W. W., Herzenach, M. & Grulke, N. E. The carbon isotope composition of plants and soils as biomarkers of pollution. Isoscapes: Understanding Movement, Pattern, and Process on Earth through Isotope Mapping. (eds West, J. B., Bowen, G. J., Dawson, T. E. & Tu, K. P.) 407–423 (Springer, 2010).
Graven, H. D. Impact of fossil fuel emissions on atmospheric radiocarbon and various applications of radiocarbon over this century. Proc. Natl. Acad. Sci. USA 112, 9542–9545 (2015).
Acknowledgements
We thank Hiroyuki Konishi and Yuuka Yamamoto for help with sampling of ants and termites. This work was funded by the Japan Society for the Promotion of Science to K.T (Challenging Exploratory Research: 16K14865, Kiban Kenkyu B: 15H04425, 23405011, Kiban Kenkyu A: 15H02652) and to K.M. (Kiban Kenkyu S: 25221206), and partly by Special Coordination Funds for Promoting Sciences and Technology of the Ministry of Education, Sport, Culture, Science and Technology of Japan.
Author information
Authors and Affiliations
Contributions
K.M., E.L.V., W.S. and K.T designed research; W.S., C.H., T.Y., B.G., E.L.V., K.T. and K.M. performed transect sampling; F.H. and H.O.T. performed radio isotope and stable isotope analyses; W.S, S.D., F.H. and K.M performed statistical analysis; K.M., W.S., F.H., B.G., R.R.D., E.L.V. and K.T. wrote the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Suehiro, W., Hyodo, F., Tanaka, H.O. et al. Radiocarbon analysis reveals expanded diet breadth associates with the invasion of a predatory ant. Sci Rep 7, 15016 (2017). https://doi.org/10.1038/s41598-017-15105-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15105-1
This article is cited by
-
Variation in Argentine ant (Linepithema humile) trophic position as a function of time
Biological Invasions (2023)
-
Intraspecific differences in the invasion success of the Argentine ant Linepithema humile Mayr are associated with diet breadth
Scientific Reports (2021)
-
Host use diversification during range shifts shapes global variation in Lepidopteran dietary breadth
Nature Ecology & Evolution (2020)
-
Regional-scale environmental resistance to non-native ant invasion
Biological Invasions (2020)
-
Stable and radio-isotopic signatures reveal how the feeding habits of ants respond to natural secondary succession in a cool-temperate forest
Insectes Sociaux (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.