Abstract
In plants and parenchymatous brown algae the body arises through the activity of an apical meristem (a niche of cells or a single cell). The meristem produces lateral organs in specific patterns, referred to as phyllotaxis. In plants, two different control mechanisms have been proposed: one is position-dependent and relies on morphogen accumulation at future organ sites; the other is a lineage-based system which links phyllotaxis to the apical cell division pattern. Here we examine the apical patterning of the brown alga, Sargassum muticum, which exhibits spiral phyllotaxis (137.5° angle) and an unlinked apical cell division pattern. The Sargassum apex presents characteristics of a self-organising system, similar to plant meristems. In contrast to complex plant meristems, we were unable to correlate the plant morphogen auxin with bud positioning in Sargassum, nor could we predict cell wall softening at new bud sites. Our data suggests that in Sargassum muticum there is no connection between phyllotaxis and the apical cell division pattern indicating a position-dependent patterning mechanism may be in place. The underlying mechanisms behind the phyllotactic patterning appear to be distinct from those seen in plants.
Similar content being viewed by others
Introduction
In developmental biology, fate decisions (such as where to place a new organ) often exhibit characteristics of emergent phenomenon. Such decisions are often made based on a position-dependent patterning system where the position of a cell within a tissue or organ specifies its fate and a signal (or morphogen) acts as an instructive agent1. An alternative mechanism depends on cell lineage, although this seems less prevalent in walled organisms such as plants1. When one examines the processes behind areal organ positioning in plants, phyllotaxis, two major theories emerge: in some early diverging land plants, phyllotactic patterning is attributed to patterned divisions at the meristematic apical cell; in Spermatophytes (seed plants), they are attributed to a morphogen-based mechanism. The latter is position-dependent patterning and the former lineage-dependent.
Early diverging land plants, such as mosses and ferns, maintain a single apical cell which acts as a stem cell for the apex2,3,4. In mosses, the pattern of leaf production may be seen as lineage-dependent as it follows the apical cell patterning directly5,6. In horsetails and fern apices, the arrangement of the leaves is independent of the division pattern in the apical cell7,8. These latter two examples hint at a position-dependent patterning mechanism which takes place post apical-cell division. Further evidence for a self-organising and robust patterning mechanism comes from experiments where apical cell ablation does not lead to growth arrest, but instead to a new apical cell establishment and subsequent spiral phyllotaxis about the new centre9,10. Work from Wardlaw9 and Snow & Snow11 explored positional patterning mechanisms which were both physical (tissue tension) and morphogen (the phytohormone auxin) based; however, no further modern explorations have been conducted in these species to our knowledge.
In Spermatophytes the meristematic activity in the shoot apex is attributed to an organised group of cells. This niche serves as a reservoir for production of cells which then give rise to the lateral organs12,13. Phyllotactic patterning occurs independent from division patterns within the meristematic niche and evidence exists for a position/morphogen-based patterning mechanism: organs emerge due to local auxin accumulation14 followed by the softening of tissues at specific positions at the shoot apex15,16; stochastic fluctuations in auxin concentration can lead to coordinated polarisation of auxin transporters and result in a self-organising pattern of organs17. Ablation of the meristematic niche leads to re-establishment of a new niche and organised phyllotaxis lending weight to a robust self-organising mechanism rooted in the morphogen auxin12,18.
Plants are not the only organisms to display spiral organ arrangement: two genera of parenchymatous multicellular brown algae, in the order Fucales, arrange their organs in spirals: Sargassum and Cystoseira 19,20. Other members of the order tend to display dichotomous branching (e.g. Fucus). The body of parenchymatous brown algae is built through the meristematic activity of an apical cell21,22. In the Fucales, the apical cell presents as three or four sided in transverse view and divides from these faces23,24,25,26,27,28,29. In some cases, the apical cell is thought not to divide but rather stimulate the cells around it to do so26). In Cystophora, it has been proposed that the division pattern of the apical cell drives the observed branching pattern of the thallus, similar to the lineage-dependent theory for moss28. In Fucus, if the apical cell is removed growth of the branch ceases26, indicating a less robust patterning mechanism than seen in spermatophytes, ferns and lycophytes. As such, the literature indicates that phyllotactic patterns in parenchymatous brown algae may be lineage-dependant.
In Sargassum muticum, while a clear apical cell is present23 the shoot apex is similar in organisation to that seen in ferns and spermatophytes: a large central area is surrounded by emerging organs in a spiral pattern30. Since the brown algae have evolved completely independently from plants, it is fascinating to see similar spiral leaf patterns emerging in the shoots of Sargassum as are seen in that distant kingdom. Here we explore the apical organisation and spiral phyllotaxis observed in Sargassum muticum, and begin to investigate its robustness and underlying mechanisms by comparison to those proposed in plants.
Results
The arrangement of leaf buds in the Sargassum muticum meristem follows the golden angle
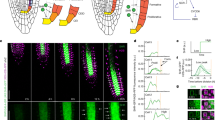
The S. muticum plant is attached to the substratum through a discoid holdfast from which the upper body arises. Its main body is formed of one main primary branch and a number of lateral branches which hold leaves, air bladders and reproductive structures (Fig. 1a). The apex of S. muticum has a striking ‘phyllotactic’ pattern, where subsequent branches are spirally organised with respect to each other19,20. At the apex, these branches begin as leaf buds31. In order to characterise the spiral pattern more fully, we performed detailed analysis of S. muticum apices collected in the field.
The Sargassum muticum apex displays distinct patterns which are independent of each other. (a) The morphology of an adult S. muticum alga. Abbreviations: lf = leaf, ab = air bladder, slb = secondary lateral branch. (b) Newly forming buds numbered by increasing age (P1 - > P10) with a representative divergence angle illustrated between the two consecutive buds. (c) Divergence angles distribution of measured apices (mean = 137.53 ± 2.08°; n = 260). (d) Division pattern in a longitudinal section of a Sargassum apex; AC divides to give rise to three tissues (meristoderm, cortex, medulla). (e) Apical cell division pattern in a transverse section of a Sargassum apex; first periclinal apical cell division (red; yellow star) followed by radial (orange, yellow; white arrowhead) and circumferential (blue; black arrowhead) anticlinal divisions. Schematic representation of the division in the longitudinal direction (f) and the transverse direction (g). (h,i) Clockwise phyllotaxis with a (l) clockwise or (m) counter-clockwise apical cell division orientation. (j,k) Counter-clockwise phyllotaxis with a (n) clockwise or (o) counter-clockwise apical cell division orientation (n = 27). *youngest daughter cell, **next-to-youngest daughter cell.
S. muticum apices were qualitatively divided into two zones: the apical pit-region, where pro-meristem cells were produced (Fig. 1b, pink), and the peripheral region where new leaf buds formed (Fig. 1b, yellow, Supplementary Fig. S1). In primary lateral apices the meristem size (proxied by presented area of the pit-region, Fig. 1b, pink) was not correlated with stipe length which is representative of apex age (n (individuals) = 7, n (branches) = 22, Supplementary Fig. S2). Within the peripheral region, the phyllotactic pattern was spiral and presented an average divergence angle (angle between two sequentially-aged buds) of 137.53 ± 2.08° (Fig. 1b,c; n (meristems) = 57). The organisation and phyllotactic pattern observed in the apices of S. muticum was highly regular and resembled that seen in complex multicellular plant apices.
The Sargassum muticum apical cell area suggests a highly organised division pattern
As the literature seemed to indicate that brown algae phyllotaxis might be lineage-dependent, we next examined the division patterns of the Sargassum apical cell to see if its pattern exhibited a golden angle, as in moss. In order to investigate the possible patterning of cell divisions in the promeristem, and any connection to the phyllotactic pattern, we examined transverse and longitudinal sections of S. muticum apices.
In sections, the apical cell of S. muticum presented as bi-convex and lenticular (longitudinally; Fig. 1d) and as three-sided (transversely; Fig. 1e) consistent with literature23,24,27,29. Unlike Fucales apical cells, which are reported to stimulate their neighbours to divide but refrain themselves26, evidence of apical cell division was observed. In the longitudinal view, divisions appeared to give rise to three tissues – outer layer (meristoderm) and two inner layers (cortex and medulla) (Fig. 1d,f, Supplementary Fig. S3).
In the transverse direction, the youngest (apical-cell-proximal) division always appeared to be asymmetric periclinal followed by sequential face divisions from the apical cell producing promeristem daughters at 119° angles to each other (Fig. 1e, red). In the first three rounds of division (a division round was defined as a pseudo-time progression that each daughter cell would undergo as it moved away from the apical cell; See Methods), the daughter cell underwent one or more anticlinal radial divisions (Fig. 1e, orange/yellow/blue; white arrowhead); the rounds of radial division never produced more than four cells (Fig. 1e, blue). The further divisions were anticlinal circumferential and created 8 cells in total (Fig. 1e, blue, purple; black arrowhead). After this point it became difficult to discern lineages in histological sections. The pattern described here was highly conserved although occasionally an anticlinal circumferential division was observed before the 4-cell stage (n = 1/30). From these data, it was concluded that the S. muticum apical cell divided asymmetrically from sequential faces, producing daughter cells at 120° angles, and that these promeristematic daughter cells further underwent a regimented division pattern.
The phyllotaxis pattern and the apical cell division pattern are not linked
In Cystophora, the apical cell division pattern (bifacial divisions) has been correlated with the apical branching pattern28). Our observations in S. muticum suggest that the apical cell divides from all three faces to produce promeristem daughter cells at an approximate 120° angle (α = 119.01 ± 6.11°, n = 74), while the phyllotactic pattern follows at ~137.5° spiral pattern. In order to examine whether these two patterns in S. muticum were linked, we examined the chirality in both the apical and phyllotactic patterns in the same meristems.
The spiral phyllotaxis in Sargassum muticum had either a clockwise or a counter-clockwise direction with a ratio of ~1:1 (58/118 clockwise, 60/118 counter-clockwise). In the clockwise and counter-clockwise orientation the older buds were located to the left side of the younger bud forming a right-handed or a left-handed spiral, respectively (Fig. 1h–k). This 1:1 ratio is observed in plants as well32,33. With respect to apical cell division patterning, two patterns were observed: moving out from the apical cell, daughter cells were produced to the left or the right yielding both counter- and clockwise patterns in a 1:1 ratio (Fig. 1l–o; 28/56 clockwise, 28/56 counter-clockwise).
In order to examine if a connection in chirality was observed, individuals were imaged under a light microscope and subsequently sectioned to check the orientation of the apical cell division. In either the counter or clockwise phyllotactic groups, the apical cells presented as ~1:1 counter- and clockwise (Fig. 1h–o, clockwise phyllotaxis – 8/16 clockwise, 8/16 counter-clockwise apical cell divisions; counter-clockwise phyllotaxis – 7/11 clockwise, 4/11 counter-clockwise apical cell divisions). This data strongly suggests that these two patterning mechanisms are unlinked and may be under separate control. This is highly similar to the patterning mechanisms seen in multicellular plant apices where the phyllotactic pattern and the stem cell niche are separately defined by two hormones; auxin and cytokinin14,34.
Ablation of the apical cell leads to formation of a new apical centre indicating pattern self-organisation
Given the observed similarities to multicellular plant meristems, we next examined whether the apical cell and phyllotaxis could re-establish after ablation of the apical cell. In plants, the stem cell region can re-establish in this way pointing to a robust self-organising patterning system18. In Fucus, such manipulations led to growth arrest and termination26.
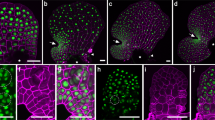
Apical cell ablations were performed on partially dissected apices using a thin needle aimed at the centre of the pit-area (Fig. 2a,b; white arrowhead). Apices were grown in culture, and re-dissected after a 3-week recovery period before a second imaging. Four scenarios were observed: in 30% (7/23) of the apices the growth of the central zone had stopped or they were dead (6/7 dead, 1/7 no new meristem formed, but the existing buds continued to grow); in 30% of the samples growth continued from what appeared to be a new pit-region (Fig. 2c,d; blue arrowhead, n = 7/23). In these apices, the phyllotactic pattern after recovery exhibited a spiral pattern. In another 20% of apices, the meristems seemed to split in two (Fig. 2e; n = 5/23) but again appeared to present spiral patterning. In the remaining 20% of the samples, the results we inconclusive as the imaging methods did not always produce sufficient quality data for pit-area positioning. Culturing itself did not alter the pattern of buds (Fig. 2f).
Removal of the apical cell can induce formation of a new central meristem. Sargassum meristem stabbed (white arrowhead) in the region of the apical cell in top view (a) and in subsequent longitudinal section (b). (c) Formation of a new central meristem; white arrowhead marks the spot of the stab, blue arrowhead shows the location of the new meristem. (d) Longitudinal section of a newly formed meristem (as in c) showing a new apical cell in the new meristem centre (blue arrowhead) and stabbed area (white arrowhead). Inset: magnified view of the new apical cell. (e) Apex presenting a split meristem; dashed line shows the separation of the two new meristems, centers indicated by blue arrowheads. (f) Control apex, not stabbed. Sample numbers: n = 23 stabbed (12 recovered, 7 ceased growth, 4 unclassified).
In the samples where a new pit-area appeared to establish, the wound had moved to the side of the meristem and the new pit-area was roughly centrally positioned (Fig. 2c,d). These data suggest that the meristematic region of the Sargassum meristem could re-establish itself indicating a self-organising system similar in nature to that in plant meristems. The data also imply that when a new apical cell is established, the spiral phyllotactic pattern can also re-establish.
A potential link between auxin and brown algal phyllotaxis is unlikely
In plants, auxin distribution within the peripheral zone dictates the phyllotactic pattern14. The brown algae Fucus vesiculosus and Ectocarpus siliculosus have both exhibited auxin-triggered morphogenetic changes35,36. Since the phyllotaxis in Sargassum muticum is spiral and highly resembles the one observed in higher plants and given the potential for auxin response in brown algae, we next examined whether auxin could alter, or be correlated with, the phyllotactic pattern.
Applying auxin externally in the artificial sea water cultivation medium in order to see if phyllotaxis could be altered had no effect on growth or the phyllotactic pattern (Supplementary Fig. S4).
In order to determine if auxin showed patterned distribution within the apex, we performed immunolocalisations on sectioned apices using the anti-IAA antibody. The anti-IAA signal was strongest in the meristoderm and mucilage external to the meristoderm with accumulation at apical pits and the bases of buds (Fig. 3a, white arrowhead). Internally, there were regions of high signal within the apex in the meristoderm although these did not correlate with bud size or position (Fig. 3a,b). Upon close examination, a large amount of signal originated from the mucilage external to the meristoderm (Fig. 3c). These data suggest that auxin may be accumulating in the meristoderm and mucilage, although its source is undetermined (see Discussion), and there was little correlation with bud position.
IAA and mannuronate immunolocalisation signals do not appear to correlate with new bud formation in the S. muticum apex. (a) Anti-IAA antibody localising to the buds, surface of the meristoderm and the apical pit (white arrowhead). Higher magnification view of the bud (b) and of the meristoderm cells (c). Dashed line in (c) delineates the meristoderm outer cell wall. (d) BAM10 antibody binds to the guluronic acid rich areas in the cell wall with a homogeneous distribution throughout the apex. (e) BAM6 antibody binds to mannuronic acid rich areas in the cell wall, localised at the surface and on cell-cell junctions in the inner tissues and with a slightly higher abundance in a young bud (green arrowhead). (f) BAM10 antibody signal is distributed throughout the apical cell and promeristem region in transverse sections. (g) BAM6 antibody signal is not detected in the apical cell and immediate neighbours. BAM6 localises mainly in the cell junctions around the apical cell. White arrowheads mark the pit-area (with an apical cell). ac = apical cell location.
Elongating organs are predicted to have softer walls and the apical cell to have stiffer cell walls
In plants, new organs form in the peripheral zone after auxin maxima lead to wall softening15. The central zone, containing the meristematic stem cells, exhibits stiffer cell walls than the peripheral zone or new primordia16. In most walled organisms, it is assumed that cell growth is limited by the cell wall and its mechanical properties, in turn linked to its biochemical composition37,38. Since Sargassum apices were so similar in pattern to the Arabidopsis apex, we checked whether they might follow similar mechanical rules. Alginate biochemistry was examined in muro using antibodies raised against different biochemical epitopes: the BAM10 antibody preferentially recognises guluronic acid within alginate39, which should be mechanistically rigid through calcium cross-linking40; conversely, the BAM6 antibody preferentially binds to mannuronic acid residues within alginate, which should be mechanically less rigid as they are unable to form calcium cross-links39. Control immunolocalisations may be found in the Supplementary Fig. S5.
In longitudinal sections, BAM10 showed a wide distribution of signal across the apex (Fig. 3d). BAM6 signal was found at the junctions of cells and on the outer periclinal walls of the meristoderm cells (Fig. 3e). There was no obvious pattern of mannuronic/soft alginate associated with young buds, however signal did appear higher in slightly older elongating buds (BAM6; Fig. 3e; green arrowhead). We next looked at the apical cell alginate biochemistry using immunolocalisations on transverse sections. The BAM10 signal was equally distributed across the apical cell, promeristem cells and the surrounding cells (Fig. 3f). The BAM6 signal was excluded from the apical cell and promeristem cells (Fig. 3g). BAM6 signal was detected in more mature tissues at the junctions between cells (Fig. 3g). These data suggest that the apical cell, young promeristem cells and the peripheral area in the apex have more guluronic acid residues than mannuronic, which may lead to stiffer cell walls.
Discussion
Here we report that the spiral phyllotactic pattern in S. muticum follows the Golden Angle (~137.5°) in a pattern almost identical to that found in many multicellular plants. Developmentally, this may not be so surprising since both organisms display indeterminate growth and produce new organs from an apical meristem-like region; both utilise their shoots and branches for light interception and reproduction. However, it is interesting to see a similar pattern when the building blocks of multicellularity are completely independent: while both organisms have cell walls and cell adhesion, the polysaccharides which make up the wall matrix (pectin and alginate) are distinct. These organisms have evolved multicellularity independently41 and therefore may have different communication strategies (morphogen identity). The data presented here indicate that complex parenchymatous brown algae have position-dependent patterning mechanism which results in spiral phyllotaxis, similar to that observed in plants.
The apical cell is the centre of the brown algal meristem; its sequential face divisions create a pool of cells which build the body of the adult alga. In the spiral meristem of S. muticum the division pattern of the algal apical cell does not relate to phyllotactic patterning. Firstly, the difference in the divergence angles between the two patterns does not support a causative relationship – in apical cell divisions, the angle of the newly produced daughter cell to the previous is 120°, whereas the observed phyllotactic angle centres on the golden angle of 137.5°. In the moss Atrichum undulatum, the triangular apical cell exhibits sequential face divisions but these occur at ~137.5°, and angle which is reflected directly in the phyllotactic angle42. In Physcomitrella patens, spiral apical cell divisions lead to spiral leaf arrangement6. As this correlation in pattern is not seen in S. muticum, apical cell division pattern and phyllotaxis appear to be unrelated. While growth distortion post-apical-cell cannot be discounted, we believe it is unlikely given the highly organised nature of divisions seen in the apex. A second piece of evidence comes from the observation that both patterns could follow either a left- or a right-handed rotation but the two could be disconnected. Our experiments suggest that apical cell division pattern and phyllotactic pattern are not correlated. We hypothesise that an apical-cell-independent patterning mechanism exists in Sargassum, that position is more instructive than lineage.
When a stem-cell niche or apical cell are destroyed in plants, there are two outcomes reported in the literature for ferns and plants: the apical growth can cease, or the apex will develop a new stem-cell centre and restart growth9,10,12,18. In examined brown algae, destruction of the apical cell in leads to the termination of apical growth26,43; no re-establishment of patterning has been reported to our knowledge.
In our experiments, S. muticum apices exhibited both outcomes upon apical-cell destruction: 30% of the apices ceased growing, while another 50% showed continued growth after re-organisation. Similar to plants, our data showed that the phyllotactic pattern in the Sargassum meristem was also re-established (or maintained) upon apical cell destruction. These data indicate that the S. muticum apex is more complex and capable of re-organisation after apical cell destruction, in a similar way to that seen in plants. This complexity, and its similarity to those of plants, may represent a more robust system when it comes to development. This further supports a morphogen-based position-dependent patterning mechanism.
Auxin’s potential as a morphogen has been established in plants14 and it has been found to have an effect in the development of Bryophytes44,45 and brown algae35,36,46. As such, auxin was a prime candidate for morphogen-like behaviour in the Sargassum apex. We were unable to detect an effect of exogenous auxin on phyllotactic patterning in S. muticum. It is possible that the auxins used (1-Naphthaleneacetic acid and 2, 4-Dichlorophenoxyacetic acid) were not effective or that their concentrations require further optimisation. It is also possible that local applications might have been more effective: local applications could alter phyllotaxis in the plants Lupinus and Epilobium 11 but when more broadly applied in Tropaeolum a lesser effect was seen47. In order to gain a more spatial view of possible endogenous auxin, we then switched to IAA-immunolocalisations. Our data suggest that there is no particular localisation of auxin to newly growing buds observed in sections of S. muticum apices, but a higher signal localized in the meristoderm cells and mucilage attached to the surface of the alga. We cannot completely rule out non-specific antibody reactions to the mucilage; however, the no primary antibody controls were negative (Fig. S5) and the cell wall antibodies did not show such signals (Fig. 3c,d). Detecting a small and highly dynamic molecule such as auxin using immunolocalisations is limiting; however, using a specific pre-fixation step auxin could be fixed and detected using this approach in plants and brown algae36,48. It is possible that IAA is not the functional auxin in Sargassum, although it has been detected by GCMS in the brown alga Ectocarpus 36.The data presented here suggest auxin is an unlikely morphogen for brown algal phyllotaxis, but without a tool such as the molecular reporter constructs in plants we cannot be certain. There is no evidence to date that brown algae have homologs of the auxin transporters found in plants36,49, although the algae may have completely different transport mechanisms, even diffusion. Classical phyllotactic patterning mechanisms have relied on reaction-diffusion equations in the past50,51, and as such it is possible that auxin diffusion may be instructive in brown algae. Another question that has been a topic of discussion is whether the auxin detected is being produced by the alga itself rather than being provided by auxin-producing associated bacteria on the surface of their thallus52. Bacteria have been described living in association with brown algae53,54, and having an effect on their development55. A similar process has been recently described in a sister class to brown algae – diatoms56. The recent interest in exploring the algal-bacterial interactions could lead to a better understanding if and how the bacteria might be affecting the development in a more “advanced” brown algal species such as Sargassum muticum.
The cell walls of brown algae are composed mostly out of matrix polysaccharides called alginates, sulphated fucans, and a small amount of cellulose57. In plants, the role of the cell wall and its relationship to auxin in organ formation has been previously explored15,37,58,59,60, but no information of such is available for the brown algae. Given the recent evidence for a role of pectin in organ emergence, and the predominant nature of alginate in the algal cell wall it seems plausible that alginate may be involved in bud formation. Alginate is formed of two residues, mannuronic and guluronic acid, the latter being able to cross-link with Ca2+ ions40, similarly to pectin in plants, and thus change its mechanical properties61. New techniques have been recently developed to look into the brown algal cell wall biochemistry by using immunolocalisations targeted towards specific epitopes in alginate chains39. Our data suggest that the guluronic acid rich areas are more abundant throughout the apex of S. muticum, but there was no clear distinction between growing and non-growing parts. The mannuronic acid (BAM6) signal tended to be higher in rapidly growing older buds without obviously marking young buds. These observations differ from the ones seen in A. thaliana meristems where pectin biochemical changes did mark new organ sites37. In slightly older, elongating buds, a stronger signal from BAM6 was detected, indicating a possible role for alginate biochemistry in elongation but not initiation of buds. In transverse sections, we observed that the guluronic acid signal (BAM10) was high, whereas that for mannuronic acid (BAM6) was barely detected. Based on these data, we could suggest that the area around the apical cell and the apical cell itself have stiffer walls. This is similar to the situation observed in plants, where the stem cells have been shown to be stiffer than the surrounding peripheral cells16. Taken together, it seems that the apical cell in Sargassum may behave mechanically similar to the stem-cell niche in spermatophyte plants and that softer alginate may be present in rapidly elongating new buds. However, it does not appear that new bud positions display biochemical markers of softer alginate and as such it is possible that alginate biochemistry is not related to bud positioning in Sargassum, only in outgrowth.
In the Sargassum apex, a new leaf will develop its own apical cell, and further cells along the meristoderm between this and the primary apical cell will follow suit, each giving rise to another organ on each branch31. All meristodermal cells have a meristematic ability which could indicate that any cell from this cell layer could “switch on” and become an apical cell and start producing its own bud25. This is not dissimilar to the specification seen in ferns for the production of new leaves from the epidermis8,62. Based on the robust, self-organising nature of the Sargassum apex and the lack of correlation between the apical cell division pattern and that of new buds, it seems likely that a positional mechanism is in place for phyllotactic patterning in Sargassum, possibly a morphogen. The secondary specification of further apical cells between the leaf and main apical cell also hints at a position-dependent specification of meristodermal conversion into apical cells. It has been observed that the apical cell can divide to potentially create a daughter apical cell which then continues to create a new branch29. In our experiments, we never observed an equal division of the apical cell which could explain the previously described situation. This is similar to the case in Cystophora where division to produce a second apical cell was rarely observed28. If we assume this hypothesis might be true and that we simply missed such special divisions, based on the observed patterns of longitudinal division it seems unlikely that this could produce a golden-angled spiral and would more likely produced a 120° spiral. The absence of cell wall biochemical marks associated with alginate softening correlated to new bud positions indicates that the physical events of initial bud outgrowth may be different than that in plants. This does not rule out a physically-based positioning system for the brown algal apex; physical buckling may give rise to phyllotactic patterns. If the underlying cortex tissue is growing at a differential rate to the meristoderm physical buckling may result through compression of the outer tissue63. This possibility is worthy of further investigation.
While the experiments presented here make a case for the Sargassum apex as being more plant-like in its patterning and organisation principals, we have many new questions. Is there a re-specification of a meristodermal cell into a new apical cell, and how is this regulated? If auxin is an unlikely morphogen, what might be the identity of the algal functional analogue (if it exists at all)? If auxin can in fact be instructive, is it produced by the algae or by associated bacteria (implying a more communal evolution of patterning in the brown algae)? The answers to many of these questions undoubtedly require advances in molecular techniques and genetics within the brown algae. These techniques are beginning to be developed in Ectocarpus and hopefully can be translated into other interesting algae35. Another hurdle is the inability to culture many brown algae for their full life cycle in vitro, thus limiting when questions might be asked (seasonally). Currently methods exist for Ectocarpus and Dictyota 34,64.
Materials and Methods
Sample collection and in vitro culture
The samples were collected in Rottingdean (East Sussex, United Kingdom) between November 2015 and February 2017. After collection, they were transported in seawater to the laboratory and kept at 4 °C. Processing was done shortly after returning back to the laboratory – the specimens were dissected using fine tweezers and used for further experiments. The medium used in the experiments was filter sterilised artificial seawater (ASW, Tropic Marin Sea Salt; Tropic Marin, Germany).
Imaging of the apices for divergence angle measurements
Sargassum apices were dissected using fine tweezers by removing all the leaves from their base, until the central region of the apex was clearly visible. The apices were then cut to a 1 cm length and anchored by insertion into Petri dishes containing 1% agarose melted in ASW and flooded with ASW to cover. Images were taken using a VHX 5000 microscope (Keyence (UK) Ltd, UK).
Measurements for the divergence angle were done using the VHX 5000 Keyence software; centres of each organ were used in reference to the centre of the apex.
Histology
The apices were fixed in a fixative containing 2.5% glutaraldehyde and 2% formaldehyde in artificial seawater. They were then dehydrated through 10% ethanol steps and embedded in resin. Samples were then cut in 1 µm slices using an ultramicrotome. Sections were placed onto slides and left to dry at room temperature. Sections were then stained with 0.05% Toluidine blue O solution for 5 minutes, washed, covered with a cover slip and imaged under a light microscope.
Cell division pattern quantification
As time-course analysis of division was technically impossible around the apical cell due to its internal position, we were limited to fixed and sectioned material and so needed to make assumptions on division time based on position, our so-called ‘pseudo-time progression’. We hypothesised that the three-sided apical cell divides by sequentially cutting off a daughter cell from each of its three faces in a progressive manner. The pattern described was based on this phenomena: the daughter cell with least cell divisions (usually just one or none) and closest to the mother apical cell was therefore determined to be the youngest newly formed daughter. From this daughter cell, the second youngest was determined based on the same parameters: least cell divisions (one, two or three) and still close to the mother cell, but at a rotated position. If the daughter cells on two sides of the mother apical cell had the same number of divisions, the age of them was assessed by looking at the next layer of cells, again based on how many cell divisions are present.
The apical cell resembles an equilateral triangle and because the divisions are predicted to occur at sequential faces of this triangle, the angle is roughly 120°. We calculated the central point of the triangle by drawing finding the crossing point of the three altitude lines. After calculating the centre, we drew lines from there to the interface of the two daughter cells and calculated the angle for each of these divisions. See Supplementary Fig. S1 for a pictorial description of this process.
Apex ablation
Sargassum apices were dissected and handled as described above. They were then precisely stabbed using a fine needle in the middle of the meristem, where the apical cell is located. The images of the stabbed meristems were taken using a VHX 5000 microscope (Keyence (UK) Ltd, UK). The samples were kept in culture under 16 °C, 12:12 hour day night cycle, 60 µmol m−2 s−1. After 20 days, they were dissected again to remove the newly grown leaves and imaged again. They were fixed, dehydrated, embedded in resin as previously described. The embedded apices were then cut into serial 1 µm sections (every 5 µm throughout the meristem), stained with TBO as above and imaged under a light microscope.
Alginate immunolocalisation
The apices were fixed, dehydrated and embedded in resin as described above. The sections were placed on Vectabond coated multitest 8-well slides, 2 sections per well (Vector Laboratories, USA). They were then incubated in a blocking solution of 5% milk for 2 hours. The sections were rinsed with phosphate buffered saline (PBS; 2.7 mM KCl, 6.1 mM Na2HPO4, and 3.5 mM KH2PO4) and incubated in the 60 µl of 1/5 (in 5% milk) monoclonal primary antibody for 1.5 hours. After the incubation, the slides were washed with PBS 3 times for 5 minutes each, followed by the incubation in the 60 µl of 1/100 (in 5% milk) IgG-FITC secondary antibody (F1763, Sigma-Aldrich). The sections were then washed 5 times for 5 minutes in PBS, mounted in Citifluor (Agar Scientific, UK), covered with a coverslip, sealed and imaged under a Leica SP8 confocal microscope (Leica Microsystems, Germany). The antibodies used were gifts from Prof. Paul Knox (University of Leeds).
Auxin immunolocalisation
The protocol was adapted from Le Bail et al. (2010). The Sargassum and Arabidopsis apices were dissected and prefixed in 3% of 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide (EDAC, Sigma-Aldrich, USA) followed by an overnight fixation in FAA ((47.5% ethanol, 5% acetic acid, and 10% formaldehyde in ASW). Samples were then dehydrated and embedded in resin as described above. 1 µm sections were cut using the Leica ultramicrotome and placed on Vectabond coated slides. The slides were placed into PBS for 5 minutes and then incubated in a blocking solution (0.1% [v/v] Tween 20, 1.5% [w/v] Glycine, and 5% [w/v] bovine serum albumin (BSA) in dH2O) for 45 minutes. The sections were rinsed in a salt rinse solution for 5 minutes, a quick wash with 0.8% (w/v) BSA in PBS and incubated in 60 µl of 1:100 monoclonal anti-IAA antibody (Sigma Aldrich, USA) overnight at 4 °C. The slides were vigorously washed three times for 10 minutes with a high salt rinse solution (2.9% [w/v] NaCl, 0.1% [v/v] Tween 20, and 0.1% [w/v] BSA in dH2O) and then washed for an additional 10 minutes in a salt rinse solution and a rinse with 0.8% (w/v) BSA and then in PBS. 60 µl of 1:100 (v/v) dilution of the 1 mgmL−1 goat anti-mouse IgG antibody Alexa Fluor® 488 (Invitrogen, USA) was added to each well and incubated for 4 h at room temperature. The slides were washed 5 times for 10 minutes in the salt rinse solution followed by a brief was in PBS, mounted in Citifluor (Agar Scientific, UK), covered, sealed and imaged under a Leica SP8 confocal microscope (Leica Microsystems, Germany).
Data availability
All raw data and images are available from the corresponding author upon request.
References
Scheres, B. Plant cell identity. The role of position and lineage. Plant Physiol. 125, 112–114 (2001).
Nägeli, C. Wachtumsgeschiehte von Delesseria hypoglossum. Zeitschrit. Wiss. Bot 2, 121–137 (1845).
Nägeli, C. Wachtumsgesehichte der Laub- und Lebermoose. Zeitschrit. Wiss. Bot 2, 138–210 (1845).
Schüepp, O. In Handbuch der Pflanzenanatomie (Gebrüder Borntraeger, 1926).
Renzaglia, K. S., Du¡, R. J., Nickrent, D. L. & Garbary, D. J. Vegetative and reproductive innovations of early land plants: implications for a unified phylogeny. Philos. Trans. R. Soc. B 769–793 (2000).
Harrison, C. J., Roeder, A. H. K., Meyerowitz, E. M. & Langdale, J. A. Local cues and asymmetric cell divisions underpin body plan transitions in the moss Physcomitrella patens. Curr. Biol. 19, 461–471 (2009).
Golub, S. J. & Wetmore, R. H. Studies of development in the vegetative shoot of Equisetum arvense L. I. The shoot apex. Am. J. Bot. 35, 755–767 (1948).
Bierhorst, D. W. On the Stem Apex, Leaf initiation and early leaf ontogeny in filicalean ferns. Am. J. Bot. 64, 125–152 (1977).
Wardlaw, C. W. Phyllotaxis and organogenesis in ferns. Nature 167–169 (1949).
Cutter, E. G. Recent experimental studies of the shoot apex and shoot morphogenesis. Bot. Rev. 31, 7–113 (1965).
Snow, M. & Snow, R. Experiments on phyllotaxis. Part III. Diagonal splits through decussate apices. Philos. Trans. R. Soc. B Biol. Sci. 225, 63–94 (1935).
Steeves, T. & Sussex, I. Patterns in Plant Development. (Cambridge University Press, 1989).
Meyerowitz, E. M. Genetic control review of cell dvision patterns in developing plants. Cell 88, 299–308 (1997).
Reinhardt, D. et al. Regulation of phyllotaxis by polar auxin transport. Nature 426, 255–260 (2003).
Braybrook, S. A. & Peaucelle, A. Mechano-chemical aspects of organ formation in Arabidopsis thaliana: the relationship between auxin and pectin. PLoS One 8, e57813 (2013).
Milani, P. et al. Matching patterns of gene expression to mechanical stiffness at cell resolution through quantitative tandem epifluorescence and nanoindentation. Plant Physiol. 165, 1399–1408 (2014).
Jönsson, H., Heisler, M. G., Shapiro, B. E., Meyerowitz, E. M. & Mjolsness, E. An auxin-driven polarized transport model for phyllotaxis. Proc. Natl. Acad. Sci. 103, 1633–1638 (2006).
Reinhardt, D., Frenz, M., Mandel, T. & Kuhlmeier, C. Microsurgical and laser ablation analysis of interactions between the zones and layers of the tomato shoot apical meristem. Development 130, 4073–4083 (2003).
Church, A. H. On the interpretation of phenomena of phyllotaxis. (Humphrey Milford and Oxford University Press, 1920).
Peaucelle, A. & Couder, Y. Fibonacci spirals in a brown alga [Sargassum muticum (Yendo) Fensholt] and in a land plant [Arabidopsis thaliana (L.) Heynh.]: a case of morphogenetic convergence. Acta Soc. Bot. Pol. 85, 3526 (2016).
Fritsch, F. E. Observations on the anatomical structure of the Fucales. New Phytol. 44, 1–16 (1945).
Katsaros, C. I. Apical cells of brown algae with particular reference to Sphacelariales, Dictyotales and Fucales. Phycological Research 43, 43–59 (1995).
Yoshida, T., Majima, T. & Marui, M. Apical organization of some genera of Fucales (Phaeophyta) from Japan. J. Fac. Sci. Hokkaido Univ. 13, 49–56 (1983).
Nizamuddin, M. Morphology and anatomy of Phyllospora, Scytothalia and Seirococcus (Fucales). Bot. Mar. 11, 81–105 (1967).
Clayton, M. N., Hallam, N. D., Luff, S. E. & Diggins, T. Cytology of the apex, thallus development and reproductive structures of Hormosira banksii (Fucales, Phaeophyta). Phycologia 24, 181–190 (1985).
Moss, B. The apical meristem of Fucus. New Phytol. 66, 67–74 (1967).
Moss, B. Apical meristems and growth control in Himanthalia elongata (S. F. Gray). New Phytol. 68, 387–397 (1969).
Klemm, M. F. & Hallam, N. D. Branching pattern and growth in Cystophora (Fucales, Phaeophyta). Phycologia 26, 252–261 (1987).
Kaur, I. Apical meristem of Sargassum vulgare C. Agardh (Phaeophyta, Fucales). Algae 14, 37–42 (1999).
Simons, E. B. A morphological study of Sargassum filipendula. Bot. Gazzette 41, 161–182 (1906).
Critchley, A. T. Experimental observations on variability of leaf and air vesicle shape of Sargassum muticum. J. Mar. Biol. Assoc. UK 63, 825–831 (1983).
Allard, H. A. Clockwise and counterclockwise spirality in the phyllotaxy of tobacco. J. Agric. Res. 73, 237–242 (1946).
Thompson, D. W. On Growth and Form. (Cambridge University Press, 1917).
Chickarmane, V. S., Gordon, S. P., Tarr, P. T., Heisler, M. G. & Meyerowitz, E. M. Cytokinin signaling as a positional cue for patterning the apical–basal axis of the growing Arabidopsis shoot meristem. Proc. Natl. Acad. Sci. 109, 4002–4007 (2012).
Basu, S., Sun, H., Brian, L., Quatrano, R. L. & Muday, G. K. Early embryo development in Fucus distichus is auxin sensitive. Plant Physiol. 130, 292–302 (2002).
Le Bail, A. et al. Auxin metabolism and function in the multicellular brown alga Ectocarpus siliculosus. Plant Physiol. 153, 128–44 (2010).
Peaucelle, A. et al. Pectin-induced changes in cell wall mechanics underlie organ initiation in Arabidopsis. Curr. Biol. 21, 1720–1726 (2011).
Braybrook, S. A. & Jönsson, H. Shifting foundations: The mechanical cell wall and development. Curr. Opin. Plant Biol. 29, 115–120 (2016).
Torode, T. A. et al. Dynamics of cell wall assembly during early embryogenesis in the brown alga Fucus. J. Exp. Bot. https://doi.org/10.1093/jxb/erw369 (2016).
Grant, G. T., Mon, E. R., Rees, D. A., Smith, P. J. C. & Thom, D. Biological interactions between polysaccharides and divalent cations: the egg-box model. FEBS Lett. 32, 195–198 (1973).
Baldauf, S. L. An overview of the phylogeny and diversity of eukaryotes. J. Syst. Evol. 46, 263–273 (2008).
Gola, E. M. & Banasiak, A. Diversity of phyllotaxis in land plants in reference to the shoot apical meristem structure. Acta Soc. Bot. Pol. 85, 3529 (2016).
Clayton, M. N. & Shankly, C. M. The apical meristem of Splachnidium rugosum (Phaeophyta). J. Phycol. 23, 296–307 (1987).
Bennett, T. A. et al. Plasma membrane-targeted PIN proteins drive shoot development in a moss. Curr. Biol. 24, 2776–2785 (2014).
Coudert, Y. et al. Three ancient hormonal cues co-ordinate shoot branching in a moss. Elife 4, e06808 (2015).
Sun, H., Basu, S., Brady, S. R., Luciano, R. L. & Muday, G. K. Interactions between auxin transport and the actin cytoskeleton in developmental polarity of Fucus distichus embryos in response to light and gravity. Plant Physiol. 135, 266–78 (2004).
Ball, E. The effects of synthetic growth substances on the shoot apex of Tropaeolum majus L. Am. J. Bot. 31, 316–327 (1944).
Avsian-Kretchmer, O., Cheng, J. C., Chen, L., Moctezuma, E. & R., S. Z. Indole acetic acid distribution coincides with vascular differentiation pattern during Arabidopsis leaf ontogeny. Plant Physiol. 130, 199–209 (2002).
Yue, J., Hu, X. & Huang, J. Origin of plant auxin biosynthesis. Trends Plant Sci. 19, 764–770 (2014).
Bernasconi, G. P. Reaction-diffusion model for phyllotaxis. Phys. D 70, 90–99 (1994).
Swinton, J. in Alan Turing: life and legacy of a great thinker (ed. Teuscher, C.) 477–498 (Springer Berlin Heidelberg, 2004).
Evans, L. V. & Trewavas, A. J. Is algal development controlled by plant growth Ssubstances? J. Phycol. 27, 322–326 (1991).
Hengst, M. B., Andrade, S., González, B. & Correa, J. A. Changes in epiphytic bacterial communities of intertidal seaweeds modulated by host, temporality, and copper enrichment. Microb. Ecol. 60, 282–290 (2010).
Lachnit, T., Meske, D., Wahl, M. & Harder, T. & Schmitz. Epibacterial community patterns on marine macroalgae are host-specific but temporally variable. Environ. Microbiol. 13, 655–665 (2011).
Tapia, J. E., González, B., Goulitquer, S., Potin, P. & Correa, J. A. Microbiota influences morphology and reproduction of the brown alga Ectocarpus sp. Front. Microbiol. 7 (2016).
Amin, S. A. et al. Interaction and signalling between a cosmopolitan phytoplankton and associated bacteria. Nature 522, 98–101 (2015).
Deniaud-Bouët, E. et al. Chemical and enzymatic fractionation of cell walls from Fucales: Insights into the structure of the extracellular matrix of brown algae. Ann. Bot. 114, 1203–1216 (2014).
Fleming, A. J. Induction of leaf primordia by the cell wall protein expansin. Science. 276, 1415–1418 (1997).
Reinhardt, D., Wittwer, F., Mandel, T. & Kuhlemeier, C. Localized upregulation of a new expansin gene predicts the site of leaf formation in the tomato meristem. Plant Cell 10, 1427–1437 (1998).
Peaucelle, A. et al. Arabidopsis phyllotaxis Is controlled by the methyl-esterification status of cell-wall pectins. Curr. Biol. 18, 1943–1948 (2008).
Mancini, M., Moresi, M. & Rancini, R. Mechanical properties of alginate gels: empirical characterisation. J. Food Eng. 39, 369–378 (1999).
Mueller, R. J. Shoot morphology of the climbing fern Lygodium (Schizaeaceae): general organography, leaf initiation, and branching. Bot. Gaz. 143, 319–330 (1982).
Dumais, J. Can mechanics control pattern formation in plants? Curr. Opin. Plant Biol. 10, 58–62 (2007).
Bogaert, K., Beeckman, T. & De Clerck, O. Abiotic regulation of growth and fertility in the sporophyte of Dictyota dichotoma (Hudson) J.V. Lamouroux (Dictyotales, Phaeophyceae). J. Appl. Phycol.. https://doi.org/10.1007/s10811-016-0801-z (2016).
Acknowledgements
We thank Dr. Thomas Torode for help with immunolocalisations, Dr. Paul Knox for the BAM10 and BAM6 antibodies, and the Braybrook Group for helpful feedback on the project. We thank our colleagues in the Phycomorph Network for their support and collaboration (COST Action FA1406; http://www.phycomorph.org/). Funding for the work presented here was provided by the Gatsby Charitable Foundation (GAT3396/PR4; M.L and S.A.B). M.L was supported by the R. Lewin and F.E. Fritsch Prize Studentship in Phycology.
Author information
Authors and Affiliations
Contributions
M.L. and S.B. designed the research. M.L. conducted the experiments and analysed the data. M.L. and S.B. wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Linardić, M., Braybrook, S.A. Towards an understanding of spiral patterning in the Sargassum muticum shoot apex. Sci Rep 7, 13887 (2017). https://doi.org/10.1038/s41598-017-13767-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-13767-5
This article is cited by
-
Phyllotaxis: from classical knowledge to molecular genetics
Journal of Plant Research (2021)
-
Three-dimensional growth: a developmental innovation that facilitated plant terrestrialization
Journal of Plant Research (2020)
-
Alginates along the filament of the brown alga Ectocarpus help cells cope with stress
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.