Abstract
Fibromyalgia is a refractory disease characterized by chronic intractable pain and psychological suffering, the cause of which has not yet been elucidated due to its complex pathology. Activation of immune cells in the brain called microglia has attracted attention as a potential underlying pathological mechanism in chronic pain. Until recently, however, technological and ethical considerations have limited the ability to conduct research using human microglia. To overcome this limitation, we have recently developed a technique to create human-induced microglia-like (iMG) cells from human peripheral blood monocytes. In this study, we created the iMG cells from 14 patients with fibromyalgia and 10 healthy individuals, and compared the activation of iMG cells between two groups at the cellular level. The expression of tumor necrosis factor (TNF)-α at mRNA and protein levels significantly increased in ATP-stimulated iMG cells from patients with fibromyalgia compared to cells from healthy individuals. Interestingly, there was a moderate correlation between ATP-induced upregulation of TNF-α expression and clinical parameters of subjective pain and other mental manifestations of fibromyalgia. These findings suggest that microglia in patients with fibromyalgia are hypersensitive to ATP. TNF-α from microglia may be a key factor underlying the complex pathology of fibromyalgia.
Similar content being viewed by others
Introduction
Fibromyalgia, a representative form of non-organic pain, is a chronic disease that causes severe systemic pain with psychological suffering, resulting in disability and a lowered quality of life. Its clinical picture has long been documented; yet, fibromyalgia remains a refractory disease of unknown etiology to this day1. In a clinical study using fMRI, patients with fibromyalgia showed hyper-responsiveness to stimuli compared with healthy participants2. In addition, recent reports suggest that neuroinflammation caused by immune cells and inflammatory cytokines is related to the pathophysiology of fibromyalgia3,4,5. Fibromyalgia is suggested to be caused by complex bio-psycho-social factors with the central nervous system (CNS) as the pathological base.
Microglia are immune cells in the CNS, and known to have inflammatory functions via releasing proinflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-1β6. In rodent studies, we have reported the abnormalities of microglia as the pathological basis of chronic pain7,8. These rodent studies have indicated the over-activation of microglial cells in patients with chronic pain, however clinical molecular data are lacking due to ethical and technical issues. Thus, techniques to develop human microglia-like cells from non-brain tissues have been warrented9. Just recently, a technique to induce microglia-like cells from human pluripotent stem (iPS) cells has been reported10,11,12. The iPS technology enables cell-based assays in many fields such as embryology, pharmacology, and regenerative medicine; however, the iPS technology has some limitations for the modeling of non-genetic disease and needs to have much amount of time and costs13. On the other hand, we have recently developed a novel technique to induce microglia-like (iMG) cells directly from human peripheral blood (monocytes) by applying only two cytokines IL-34 and granulocyte macrophage colony-stimulating factor (GM-CSF) within two weeks without any gene modificaitons (Fig. 1a)14,15. Compared to the iPS cells, our iMG technique is simpler and has advantages of time and cost management. Another merit is that we can produce iMG cells without any gene modification. Fresh blood is needed to produce iMG cells, and we can not stock iMG cells in the present stage, which is major limitations of our iMG technique. We have already confirmed abnormalities in cellular responses of iMG cells derived from patients with Nasu-Hakola disease, which is a known primary microglia disease14. Furthermore, we have analyzed the gene expression paterns of iMG cells in both manic and depressive state of patients with bipolar disorders, and revealed state-dependent microglial experession patterns16. More recently, gene expression analysis from other research group has revealed that iMG cells show the most similar characteristics with primary human microglia in comparison with other cell types such as immortalized human microglia (SV40) and human macrophage17. Our previous studies and this recent report strongly suggest that iMG technique is a powerful tool for analyzing dynamic molecular pathophysiologies of microglia in not only genetic diseases but also non-organic diseases (including fibromyalgia and majority of psychiatric disorders).
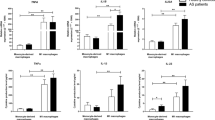
Human induced microglia-like (iMG) cells and gene expression of TNF-α during ATP stimulation. (a) Human induced microglia-like (iMG) cells. Scalebar, 50 μm. (b) Box-and-whisker plot showing gene expression of TNF-α during ATP stimulation in iMG cells from patients with fibromyalgia (n = 14; 25th percentile, 5.08; mean, 13.6; 75th percentile, 20.58) and healthy volunteers as a control group (n = 10; 25th percentile, 2.81; mean, 4.39; 75th percentile, 6.45). (c) TNF-α concentration of the supernatant released during ATP stimulation by iMG cells from patients with fibromyalgia (n = 14; 25th percentile, 6.20; mean, 7.13; 75th percentile, 7.74) and healthy volunteers (n = 10; 25th percentile, 4.83; mean, 5.73; 75th percentile, 6.86). (d) Basal gene expression of TNF-α in iMG cells from patients with fibromyalgia (n = 14; 25th percentile, 0.29; mean, 0.48; 75th percentile, 0.59) and healthy volunteers (n = 10; 25th percentile, 0.46; mean, 1.00; 75th percentile, 1.50).The y-axis represents the expression levels for each group normalized by the data of the non-ATP treatment group (NT: iMG cells without ATP stimulation) (a) or normalized by the data of healthy volunteers (b). As a result of Shapiro-Wilk normality test, statistical differences between groups were analyzed by Student’s t-test (two-tailed) (a and c) or Mann-Whitney U test (two-tailed) (b). *P < 0.05.
Here, we created iMG cells from both healthy volunteers as healthy control group (HC) and patients with fibromyalgia (Supplementary Tables 1 and 2) to test the hypothesis that microglia are hyperactive in patients with fibromyalgia.
Extracellular ATP is known to function as a neurotransmitter and/or neuromodulator in the CNS and to modulate various physiological functions of microglia18. We previously reported a relationship between chronic pain and ATP in animal models7, and ATP has been implicated in the chronic pain mechanism19. We thus compared ATP-evoked responses of iMG cells from healthy volunteers and patients with fibromyalgia. Interestingly, gene expression of TNF-α is significantly higher in iMG cells from patients with fibromyalgia (Fig. 1b). In addition, TNF-α protein levels are also significantly higher in the culture supernatant of iMG cells from patients with fibromyalgia (Fig. 1c). In contrast, there are no statistically significant differences in the gene expression of IL-1β (Supplementary Fig. 1a,b). However, protein levels of IL-1β are significantly higher in the culture supernatant of iMG cells from patients with fibromyalgia (Supplementary Fig. 1c). In addition, There are no significant differences between two groups in secretion of IL-6 and IL-8, pro-inflammatory cytokines and IL-10, an anti-inflammatory cytokine (Supplementary Fig. 1d–f). At basal level, TNF-α gene expression levels are significantl lower in iMG cells from patients with fibromyalgia (Fig. 1d). On the other hand, there are no significant differences in phagocytic activity and gene expression of TNF-α and IL-1β during phagocytosis between healthy volunteers and patients with fibromyalgia (Supplementary Fig. 2). These results suggest that microglia in patients with fibromyalgia are hyperresponsive to ATP stimulation, which may result in increasing the release of TNF-α in the CNS.
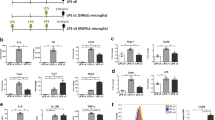
We conducted further correlational analyses between TNF-α gene expression in ATP-stimulated iMG cells and various clinical parameters including the severity of pain (Supplementary Table 2). Interestingly, we show a moderately positive correlation between TNF-α expression level and subjective pain intensity assessed by the visual analog scale (VAS) (Fig. 2a). Pain intensity and pain interference are also moderately positively correlated with TNF-α expression level (Fig. 2b,c). Fibromyalgia is a disease that is often comorbid with psychiatric disorders20. In particular, a relationship between fibromyalgia and psychiatric symptoms such as depression and anxiety has been reported in a previous study21. Here, we show a moderate positive correlation between TNF-α expression level and severity of both anxiety and depression (Fig. 2d,e). In contrast, IL-1β is not significantly correlated with any clinical scores (Supplementary Fig. 3). On the other hand, a moderate negative correlation is observed between TNF-α expression level and QOL (Fig. 2f). These findings suggest the possibility that the degree of subjective pain, psychiatric symptoms (depression and anxiety) and QOL in patients with fibromyalgia is controlled by the levels of microglia-derived TNF-α. Indeed, rodent studies have shown that TNF-α is an important factor in neuropathic pain22,23, and we previously demonstrated that TNF-α exhibits pain-related behaviors when administered into the lateral ventricle in rats24. TNF-α is also considered to play an important role in psychiatric disorders such as depression25,26. In addition, modulating microglia-derived TNF-α is suggested to be one of the therapeutic targets for psychiatric disorders14,27,28. Fibromyalgia is often comorbid with psychiatric disorders especially depression possibly due to shared pathological traits20.
Correlation analyses between TNF-α expression and subjective clinical scores. Correlation between the natural log of fold-increase in TNF-α expression with iMG cells during ATP stimulation and several subjective clinical scores (a) SF-MPQ-VAS; (b) BPI-pain intensity; (c) BPI-pain interference; (d) HAD (anxiety); (e) HAD (depression); (f) EQ-5D (QOL). As a result of Shapiro-Wilk normality test, correlations were analyzed by the Spearman rank correlation test using the data of all subjects (healthy participants and patients with fibromyalgia). r indicates the correlation coefficient.
This is the first study to suggest abnormal activation of microglia in fibromyalgia at the human cellular level. The present data indicate the positive relationship between microglial abnormality and clinical symptoms of fibromyalgia. Interestingly, iMG cells of fibromyalgia showed hyperresponsiveness to extracellular ATP and increases in the proinflammatory cytokine TNF-α. IL-1β is an important proinflammatory cytokine in the process of microglial activation via inflammasome signaling29. In the present study, gene expression of IL-1β (mRNA) is not changed, while the protein level of IL-1β in the supernatant is increased. These data suggest that microglial IL-1β is activated in earlier stages compared to TNF-α. Similar to iMG cells, PBMC-derived macrophages enhanced TNF-α expression by ATP stimulation. Therefore, enhanced TNF-α expression of patients’ iMG cells by ATP may not be specific to iMG cells, and further investigation should be conducted to compare the responses to ATP between iMG cells and PBMC-derived macrophages. On the other hand, fractalkine (CX3CL1) is also reported to be involved in chronic pain as well as ATP30,31. Thus, not only ATP but also fractalkine may also modulate iMG cells, and further studies are needed to clarify how fractalkine modulates iMG cells in patients with fibromyalgia. In addition, further investigations should clarify whether inflammasome related molecules such as IL-18 and surface expressions such as HLA-DR, CD80 and CD86 are modulated in iMG cells from patients with fibromyalgia. Of particular note, statistically moderate correlations are observed between ATP-induced TNF-α expression and various subjective parameters of pain, depression, anxiety, and QOL. In conclusion, microglia-derived TNF-α may be a possible key modulating factor of fibromyalgia, and future translational research is needed to establish a novel diagnostic system and therapeutic strategies against fibromyalgia. We could not validate the cause-effect relationship in the present study design, thus prospective studies are needed. For example, time-course assay at different severity levels of pain in the same patient can reveal whether iMG analysis can be utilized as a severity assessment tool. We believe that iMG technique sheds new light on clarifying dynamic molecular pathologies of microglia and on developing objective assessment tools in a variety of non-organic brain diseases, and further translational studies are warranted.
Materials and Methods
Subjects
The present study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the Graduate School of Medical Sciences, Kyushu University. We recruited female adult patients who were diagnosed with fibromyalgia based on the 1990 American College of Rheumatology (ACR) classification criteria32 Female adult healthy volunteers were also recruited (Supplementary table 1). Healthy volunteers (as control group participants) had no history of fibromyalgia. There were no significant differences in age between the experimental and control groups. All participants provided written informed consent, after which we drew blood samples and administered several pain scales and other psychometric assessments.
Study participants completed the following four self-administered questionnaires. Sensory and affective components of pain were assessed via the short-form McGill Pain Questionnaire (SF-MPQ). Overall pain intensity was measured using the Visual Analog Scale (VAS) of the SF-MPQ33,34 and the Brief Pain Inventory (BPI). The BPI Interference Scale is a 7-item self-report measure that assesses the extent to which pain interferes with various components of functioning such as activity, mood, and sleep35. The Pain Catastrophizing Scale (PCS) assesses three aspects of catastrophic thinking regarding the pain experience: rumination, magnification, and helplessness36,37. The Hospital Anxiety and Depression Scale (HADS) is a widely used measure of anxiety and depression38. The EuroQol-5 Dimensions (EQ-5D) consists of 5 quality-of-life (QOL) domains: mobility, self-care, usual activities, pain or discomfort, and anxiety or depression. Scores on the EQ-5D range from 0.594 to 1.00 with a higher score indicating better health-related quality of life39,40.
Induction of induced microglia-like (iMG) cells from human peripheral blood
Peripheral blood was collected using a heparinized tube from healthy volunteers and patients with fibromyalgia. Peripheral blood mononuclear cells (PBMCs) were isolated by Histopaque-1077 (Sigma Chemical Co., St. Louis, MO, USA) density gradient centrifugation. PBMCs were resuspended with RPMI-1640 (Nacalai Tesque, Kyoto, Japan), 10% heat-inactivated fetal bovine serum (FBS; Japan Bio Serum, Hiroshima, Japan), and 1% antibiotic/antimycotic (Invitrogen, Carlsbad, CA, USA). PBMCs were plated onto culture chambers at a density of 4 × 105 cells/ml and cultured overnight in standard culture conditions (37 °C, 5% CO2). After overnight incubation, culture supernatant and non-adherent cells were removed. Adherent cells (monocytes) were cultured with RPMI-1640 Glutamax (Invitrogen) supplemented with 1% antibiotic/antimycotic and recombinant human GM-CSF (10 ng/ml; R&D Systems, Minneapolis, MN, USA) and recombinant human IL-34 (100 ng/ml; R&D Systems) for 14 days to develop the iMG cells14,15.
Quantitative real time-polymerase chain reaction (qRT-PCR)
To assess gene expression patterns in iMG cells after treatment with adenosine triphosphate (ATP) or during phagocytosis, we performed qRT-PCR using a LightCycler 480 system (Roche Diagnostics, Mannheim, Germany). ATP (50 µg/ml; Sigma) or latex beads-FITC solution (Cayman Chemical) was added to the iMG cells and stimulated in standard culture conditions. After the stimulation by ATP (one hour) or beads (24 hours), the iMG cells were washed respectively, and total RNA was extracted using a High Pure RNA Isolation kit (Roche Diagnostics) according to the manufacturer’s protocol, and subjected to cDNA synthesis using a Transcriptor First Strand cDNA Synthesis kit (Roche Diagnostics). qRT-PCR for TNF-α and IL-β was performed using their respective primers. Primer sequences were as follows: TNF-α, F: 5′-CAGCCTCTTCTCCTTCCTGAT-3′, R: 5′-GCCAGAGGGCTFATTAGAGA-3′; IL-1β, F: 5′-TACCTGTCCTGCGTGTTGAA-3′, R: 5′-TCTTTGGGTAATTTTTGGGATCT-3′. Beta 2-microglobulin of the Universal ProbeLibrary (Roche Diagnostics) was used as a housekeeping control gene.
Cytokine measurement
Cytokine (TNF-α, IL-1β, IL-6, IL-8 and IL-10) concentrations of blood plasma and culture supernatant of iMG cells during ATP stimulation were measured by the Cytometric Beads Array System (CBA; BD Biosciences, Franklin Lakes, NJ, USA) according to the manufacturer’s protocol. After incubation with ATP for one hour, culture supernatants were centrifuged at 10000 x g for 10 minutes and kept frozen at −80 °C until assayed. Plasma was also kept frozen at −80 °C until assayed. The culture supernatant and plasma were incubated with cytokine capture beads and PE-conjugated detection antibodies according to each protocol. After incubation, capture beads were washed and measurement data were acquired using a FACS ARIA™ flow cytometer (BD Biosciences). Data analysis was performed using FCAP Array software (BD Biosciences).
Phagocytosis
Phagocytosis was examined via image-base cytometer (TALI, Invitrogen) using a Phagocytosis Assay Kit (Cayman Chemical, Ann Arbor, MI, USA) according to the manufacturer’s protocol. iMG cells were cultured in 24-well plates (Corning, Corning, NY, USA) at a density of 4 × 105 cells/ml. We added 50 μl of the latex beads-FITC solution to each well, and incubated the cells in standard culture conditions for 24 hours. After harvesting the cells, we measured the fluorescence intensity of FITC using an image-base cytometer.
Statistical analyses
Normality was assessed by Shapiro-Wilk normality tests. In the case of normal distribution, statistical differences between groups (healthy participants versus patients with fibromyalgia) were analyzed by Student’s t-test (two-tailed). In the case of non-normal distribution, statistical differences between groups were analyzed by Mann-Whitney U test (two-tailed). Correlations were analyzed by the Spearman rank correlation test using the data of all subjects (healthy participants and patients with fibromyalgia). In the correlation analysis, we used natural log of fold-increase in TNF-α expression with iMG cells during ATP stimulation. All statistical analyses were performed using GraphPad Prism 6 (GraphPad Software, Inc., La Jolla, CA, USA). P < 0.05 was considered statistically significant.
References
Wolfe, F. et al. The American College of Rheumatology Preliminary Diagnostic Criteria for Fibromyalgia and Measurement of Symptom Severity. Arthrit Care Res 62, 600–610, doi:10.1002/acr.20140 (2010).
Gracely, R. H., Petzke, F., Wolf, J. M. & Clauw, D. J. Functional magnetic resonance imaging evidence of augmented pain processing in fibromyalgia. Arthritis Rheum-Us 46, 1333–1343, doi:10.1002/art.10225 (2002).
Bradley, L. A. Pathophysiology of fibromyalgia. Am J Med 122, S22–30, doi:10.1016/j.amjmed.2009.09.008 (2009).
Uceyler, N., Hauser, W. & Sommer, C. Systematic review with meta-analysis: cytokines in fibromyalgia syndrome. BMC musculoskeletal disorders 12, 245, doi:10.1186/1471-2474-12-245 (2011).
Littlejohn, G. Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nature reviews. Rheumatology 11, 639–648, doi:10.1038/nrrheum.2015.100 (2015).
Block, M. L., Zecca, L. & Hong, J. S. Microglia-mediated neurotoxicity: uncovering the molecular mechanisms. Nat Rev Neurosci 8, 57–69, doi:10.1038/nrn2038 (2007).
Tsuda, M. et al. P2X(4) receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424, 778–783, doi:10.1038/nature01786 (2003).
Masuda, T. et al. Transcription factor IRF5 drives P2X4R(+)-reactive microglia gating neuropathic pain. Nat Commun 5, doi:10.1038/ncomms4771 (2014).
Kato, T. A., Ohgidani, M., Sagata, N. & Kanba, S. Directly Induced Glia/Neuronal Cells from Human Peripheral Tissues: A Novel Translational Research Tool for Neuropsychiatric Disorders. Advances in Neuroimmune Biology 6, 95–105, doi:10.3233/NIB-160109 (2016).
Muffat, J. et al. Efficient derivation of microglia-like cells from human pluripotent stem cells. Nat Med 22, 1358–1367, doi:10.1038/nm.4189 (2016).
Abud, E. M. et al. iPSC-Derived Human Microglia-like Cells to Study Neurological Diseases. Neuron 94, 278–293, doi:10.1016/j.neuron.2017.03.042 (2017).
Pandya, H. et al. Differentiation of human and murine induced pluripotent stem cells to microglia-like cells. Nat Neurosci 20, 753–759, doi:10.1038/nn.4534 (2017).
Avior, Y., Sagi, I. & Benvenisty, N. Pluripotent stem cells in disease modelling and drug discovery. Nat Rev Mol Cell Bio 17, 170–182, doi:10.1038/nrm.2015.27 (2016).
Ohgidani, M. et al. Direct induction of ramified microglia-like cells from human monocytes: Dynamic microglial dysfunction in Nasu-Hakola disease. Scientific reports 4, 4957, doi:10.1038/srep04957 (2014).
Ohgidani, M., Kato, T. A. & Kanba, S. Introducing directly induced microglia-like (iMG) cells from fresh human monocytes: a novel translational research tool for psychiatric disorders. Front Cell Neurosci 9, doi:10.3389/fncel.2015.00184 (2015).
Ohgidani, M. et al. Microglial CD206 Gene Has Potential as a State Marker of Bipolar Disorder. Front Immunol 7, doi:10.3389/fimmu.2016.00676 (2017).
Sellgren, C. M. et al. Patient-specific models of microglia-mediated engulfment of synapses and neural progenitors. Mol Psychiatr 22, 170–177, doi:10.1038/mp.2016.220 (2017).
Illes, P. & Ribeiro, J. A. Molecular physiology of P2 receptors in the central nervous system. Eur J Pharmacol 483, 5–17, doi:10.1016/j.ejphar.2003.10.030 (2004).
Milligan, E. D. & Watkins, L. R. Pathological and protective roles of glia in chronic pain. Nat Rev Neurosci 10, 23–36, doi:10.1038/nrn2533 (2009).
Mease, P. Fibromyalgia syndrome: Review of clinical presentation, pathogenesis, outcome measures, and treatment. J Rheumatol 32, 6–21 (2005).
Arnold, L. M. Management of fibromyalgia and comorbid psychiatric disorders. J Clin Psychiat 69, 14–19 (2008).
Ignatowski, T. A. et al. Brain-derived TNF alpha mediates neuropathic pain. Brain Res 841, 70–77, doi:10.1016/S0006-8993(99)01782-5 (1999).
Ren, W. J. et al. Peripheral Nerve Injury Leads to Working Memory Deficits and Dysfunction of the Hippocampus by Upregulation of TNF-alpha in Rodents. Neuropsychopharmacol 36, 979–992, doi:10.1038/npp.2010.236 (2011).
Oka, T., Wakugawa, Y., Hosoi, M., Oka, K. & Hori, T. Intracerebroventricular injection of tumor necrosis factor-alpha induces thermal hyperalgesia in rats. Neuroimmunomodulat 3, 135–140, doi:10.1159/000097238 (1996).
Dowlati, Y. et al. A Meta-Analysis of Cytokines in Major Depression. Biol Psychiat 67, 446–457, doi:10.1016/j.biopsych.2009.09.033 (2010).
Fasick, V., Spengler, R. N., Samankan, S., Nader, N. D. & Ignatowski, T. A. The hippocampus and TNF: Common links between chronic pain and depression. Neurosci Biobehav R 53, 139–159, doi:10.1016/j.neubiorev.2015.03.014 (2015).
Kato, T. A. et al. Neurotransmitters, psychotropic drugs and microglia: clinical implications for psychiatry. Current medicinal chemistry 20, 331–344, doi:10.2174/0929867311320030003 (2013).
Sato-Kasai, M. et al. Aripiprazole inhibits polyI: C-induced microglial activation possibly via TRPM7. Schizophr Res 178, 35–43, doi:10.1016/j.schres.2016.08.022 (2016).
Iwata, M., Ota, K. T. & Duman, R. S. The inflammasome: Pathways linking psychological stress, depression, and systemic illnesses. Brain Behav Immun 31, 105–114, doi:10.1016/j.bbi.2012.12.008 (2013).
Lindia, J. A., McGowan, E., Jochnowitz, N. & Abbadie, C. Induction of CX3CL1 expression in astrocytes and CX3CR1 in microglia in the spinal cord of a rat model of neuropathic pain. J Pain 6, 434–438, doi:10.1016/j.jpain.2005.02.001 (2005).
Sun, J. L. et al. CX3CL1/CX3CR1 regulates nerve injury-induced pain hypersensitivity through the ERK5 signaling pathway. J Neurosci Res 91, 545–553, doi:10.1002/jnr.23168 (2013).
Wolfe, F. et al. The American-College-of-Rheumatology 1990 Criteria for the Classification of Fibromyalgia - Report of the Multicenter Criteria Committee. Arthritis Rheum-Us 33, 160–172, doi:10.1002/art.1780330203 (1990).
Melzack, R. The Short-Form Mcgill Pain Questionnaire. Pain 30, 191–197, doi:10.1016/0304-3959(87)91074-8 (1987).
Arimura, T. et al. Pain Questionnaire Development Focusing on Cross-Cultural Equivalence to the Original Questionnaire: The Japanese Version of the Short-Form McGill Pain Questionnaire. Pain Med 13, 541–551, doi:10.1111/j.1526-4637.2012.01333.x (2012).
Uki, J., Mendoza, T., Cleeland, C. S., Nakamura, Y. & Takeda, F. A brief cancer pain assessment tool in Japanese: The utility of the Japanese Brief Pain Inventory - BPI-J. J Pain Symptom Manag 16, 364–373, doi:10.1016/S0885-3924(98)00098-0 (1998).
Mastsuoka, H. & Sakano, Y. Assessment of Cognitive Aspect of Pain: Development, Reliability, and Validation of Japanese Version of Pain Catastrophizing Scale. JPN J Psychosom Med 47, 95–102 (2007).
Iwaki, R. et al. Global Catastrophizing vs Catastrophizing Subdomains: Assessment and Associations with Patient Functioning. Pain Med 13, 677–687, doi:10.1111/j.1526-4637.2012.01353.x (2012).
Zigmond, A. S. & Snaith, R. P. The Hospital Anxiety and Depression Scale. Acta Psychiat Scand 67, 361–370, doi:10.1111/j.1600-0447.1983.tb09716.x (1983).
Williams, A. Euroqol - a New Facility for the Measurement of Health-Related Quality-of-Life. Health Policy 16, 199–208 (1990).
Nishimura, S., Tsuchiya, A., Hisashige, A., Ikegami, N. & Ikeda, S. The development of the Japanese EuroQol Instrument. Iryo To Shakai 8, 109–123, doi:10.4091/iken1991.8.1_109 (1998).
Acknowledgements
The authors would like to thank Ms Miwa Irie, Ms Aya Yamada, Mr. Shogo Inamine, Ms Yuka Matsushita and Dr. Mao Shibata for their technical assistance. This work was supported by Grant-in-Aid for Scientific Research on (1) Research Project on Elucidation of Chronic Pain from the Japan Agency for Medical Research and Development (AMED) (to M.H. & T.A.K.), (2) KAKENHI - the Japan Society for the Promotion of Science (26713039 to T.A.K. and 17K19915 to M.O.), (3) Innovative Areas “Glia Assembly” of The Ministry of Education, Culture, Sports, Science, and Technology, Japan (25117011 to S.K.), (4) Young Principal Investigators’ Research Grant of Innovation Center for Medical Redox Navigation, Kyushu University (to T.A.K.), and (5) SENSHIN Medical Research Foundation (to T.A.K., M.O., and S.K.). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to the scientific process leading up to the writing of the present manuscript. T.A.K., the principal investigator of the present research, M.O., the first author and M.H., the third author created the conception and design of the project and wrote the protocol. Clinical recruitment was performed by M.H., C.H., R.I., and N.Su. The performance of experiments and data analyses/interpretation were performed by M.O., K.H., R.I., C.H., T.A.K., M.H., and M.T. M.O. wrote the first draft of the manuscript. Critical revisions of the manuscript were made by T.A.K., R.H., K.I., N.Sa. and S.K. All authors approved this submission in its current form.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ohgidani, M., Kato, T.A., Hosoi, M. et al. Fibromyalgia and microglial TNF-α: Translational research using human blood induced microglia-like cells. Sci Rep 7, 11882 (2017). https://doi.org/10.1038/s41598-017-11506-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-11506-4
This article is cited by
-
Ameliorative effects of Fingolimod (FTY720) on microglial activation and psychosis-related behavior in short term cuprizone exposed mice
Molecular Brain (2023)
-
Microglia polarization in nociplastic pain: mechanisms and perspectives
Inflammopharmacology (2023)
-
Serum proteome profiles revealed dysregulated proteins and mechanisms associated with fibromyalgia syndrome in women
Scientific Reports (2020)
-
Protective Effect of Esculetin, Natural Coumarin in Mice Model of Fibromyalgia: Targeting Pro-Inflammatory Cytokines and MAO-A
Neurochemical Research (2020)
-
The intensity of joint pain in relation to changes in serum TNFα during therapy with anti-TNFα inhibitors
Inflammopharmacology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.