Abstract
Behaviour problems are common in companion felines, and problematic behaviour may be a sign of chronic stress. In laboratory animals, early weaning increases the risk for aggression, anxiety, and stereotypic behaviour. However, very few studies have focused on early weaning in one of the world’s most popular pets, the domestic cat, although weaning soon after the critical period of socialisation is common practice. To study the effects of early weaning (<12 weeks) on behaviour, a large data set (N = 5726, 40 breeds) was collected from home-living domestic cats through a questionnaire survey. The results show that weaning before 8 weeks of age increases the risk for aggression, but not fearful behaviour. Moreover, cats weaned after 14 weeks of age have a lower probability for aggression towards strangers than early weaned cats and a lower probability for stereotypic behaviour (excessive grooming) than cats weaned at 12 weeks. The effect of weaning age on stereotypic behaviour is partially explained by the effects on aggression. These findings indicate that early weaning has a detrimental effect on behaviour, and suggest delayed weaning as a simple and inexpensive approach to significantly improve the welfare of millions of domestic cats.
Similar content being viewed by others
Introduction
Early weaning, defined as permanent separation from the mother before the time it would occur in nature1, has several impacts on health2,3,4,5 and behaviour of animals. Early weaning may lead to neurobiological changes, for example, alter the neuroendocrine stress response6, but this effect has not been found in all studies7. Furthermore, early weaning can impair memory8 and cognition, such as extinction learning9, and it seems to especially blunt social learning10. Behavioural changes linked to early weaning are extensive: early weaning may lead to impairments in social behaviour as well as increased anxiety11, 12 and aggression7, 12, and these behavioural changes may persist for a long time13. However, moderate levels of early life stress may, on the other hand, increase resilience towards stressors, displayed by diminished behavioural signs of anxiety and levels of blood cortisol14, and the behavioural changes caused by early life stress may be adaptive15. Furthermore, many factors may influence the impact of early weaning. For example, the effects of early life stress and early weaning can differ between sexes13, 16,17,18, and the impacts of early weaning may arise only when combined with post-weaning social isolation18. Importantly, the effects of early life stress depend on the developmental timing of the stressor19, 20 and therefore early weaning after the critical period of socialisation may not induce behavioural changes.
Besides affecting normal behavioural repertoire, early weaning may impact the expression of abnormal behaviours, such as stereotypies1, which may be caused by central nervous system dysfunction21. Stereotypies are invariable and repeated behaviours that are often expressed when animals experience adverse or frustrating situations22. Examples of stereotypic behaviour patterns include rocking and hair plucking in rhesus macaques23, crib-biting in horses24, and pacing in tigers and other carnivores25. Stereotypies are commonly seen in captive animals, both wild and domesticated, but seem to be absent in nature22. Many environmental and genetic factors may induce the development of stereotypies. The occurrence of stereotypies is, at least to some extent, heritable26 and it may be correlated with personality27,28,29,30. Furthermore, early weaning has been shown to increase stereotypic behaviour in captive and laboratory animals1, for example, tail-biting in mink31 and wire-gnawing in mice32. In domestic dog, early weaning and poor maternal care has been associated with stereotypic tail chasing30. However, early weaning does not necessarily produce stereotypic animals, especially when combined with environmental enrichment33. Furthermore, sometimes the increase in stereotypic behaviour can be temporary7.
One companion animal in which early weaning is very common is the domestic cat (Felis catus Linnaeus 1758), which may be the most popular companion animal in the world, with close to 100 million domestic cats living in Europe alone34. Feral cats wean their kittens at four to eight weeks of age, but kittens usually stay with their mother for the first four months of their lives35. In cats, the critical period of socialisation occurs between 2 to 8 weeks of age36. Consequently, separation from the mother and littermates at an age of eight weeks is common. For example, The Royal Society for the Prevention of Cruelty to Animals37 recommends a minimum separation age of eight weeks. Moreover, The American Veterinary Medical Association considers 7–9 weeks of age to be the ideal time to move to a new home38, but the effects of weaning at this life stage have not been evaluated. In a previous study, laboratory kittens reared in isolation in a brooder showed deficits in social behaviour, behaved anxiously, and showed difficulties in habituation to novel objects, displayed aggression towards other kittens and showed more random movement than kittens reared with the mother and littermates39. Similarly, laboratory cats separated from their mother and siblings at two weeks of age behaved anxiously in novel environments, showed aggression towards other cats and people, and displayed random movement40. However, laboratory cats separated at 6 weeks of age did not display higher levels of aggression or random movement than cats weaned at 12 weeks of age40, indicating that, at least in laboratory settings, the timing of weaning is important. Stereotypic behaviour in cats is proposed to be in part caused or worsened by early weaning1. In a recent study, early weaning was indeed correlated with stereotypic wool sucking in Birman cats41.
The aim of this study was to examine the effects of early weaning on the behaviour of domestic cats using the large population-based survey data from our feline health and behaviour questionnaire42. Here, we have focused on the effects of early weaning by examining the behavioural differences between early and late weaned conspecifics in a sample of 5726 home-living domestic cats in 40 breeds using logistic regression. In our study, cats separated from the mother before 12 weeks of age are considered early weaned, following the recommendation of The Humane Society of the United States43. Accordingly, we considered late weaned cats to be weaned after this 12-week recommended age, with our recommended weaning age group consisting of cats weaned at 12–13 weeks of age and late weaned at 14 weeks of age or older. As early weaning is common in farm and companion animals1, research on the topic is important since it may identify possible neural mechanisms and intervention approaches that could significantly improve the welfare and health of millions of animals, including cats, which are popular pets34.
Results
Using logistic regression, we studied the effects of early weaning on behaviour in 5726 home-living domestic cats in 40 breeds. Our study revealed that behavioural problems are common in cats42. For example, 41% of cats were at least slightly aggressive towards other cats and nearly 32% displayed at least one bout of wool sucking during their life. We found that several environmental factors affected behaviour (Table 1; Supplementary Table S1; Supplementary Figures S1–S37). We also discovered that our behavioural traits (besides stereotypic behaviour) grouped into three personality factors: extraversion, aggression, and shyness, and these personality factors influenced behaviour. In this study, we focused on the effects of early weaning. As personality factors may influence the effect of weaning age on behaviour, we first ran all the models without any personality traits and then with the relevant personality traits (as defined by model selection) included. We report the former models when the removal of personality traits alters the effect of early weaning significantly.
Effects of early weaning on social behaviour
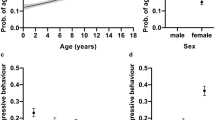
In the logistic regression analyses (Table 1), cats weaned before 8 weeks of age were significantly more likely to behave aggressively towards strangers than cats weaned at 12–13 weeks of age (Table 2; Fig. 1). Moreover, cats weaned in adulthood or not weaned at all were significantly less likely to show aggression towards other cats, family members, and strangers than other weaning age group cats. Furthermore, cats weaned at 14–15 weeks of age were significantly less likely to display aggression towards strangers than early weaned cats.
The effect of weaning age on social behaviour in logistic regression analyses. Grey circles are the groups considered early weaned. (a) Cats weaned in adulthood were less aggressive towards family members than other cats. (b) Cats weaned before 8 weeks of age were more likely aggressive towards strangers than cats weaned at 12–13 weeks of age. Cats weaned at 14–15 weeks of age were less aggressive than early weaned cats. Furthermore, cats weaned in adulthood were less aggressive than other cats. (c) Cats weaned in adulthood were less likely to display aggression towards other cats. (d) Weaning age did not affect shyness towards strangers. (e) Weaning age did not affect probability for decreased contact. Error bars indicate 95% confidence limits. N = 5726.
Effects of early weaning on non-social behaviour
In logistic regression analysis (Table 1), cats weaned in adulthood or not weaned at all were significantly less likely to display shyness towards novel objects than other cats (Fig. 2; Table 2). Weaning age affected the probability to display stereotypic behaviour as well. When personality traits were left out of the final models, cats weaned at 14–15 weeks of age were less likely to groom excessively than cats weaned at 12–13 weeks of age (χ2 = 7.14, DF = 1, P = 0.0075). Cats weaned as adults were less likely to perform stereotypic wool sucking (χ2 = 15.19, DF = 1, P < 0.0001) and excessive grooming (χ2 = 4.78, DF = 1, P = 0.0288) than other cats. However, the probability of stereotypic behaviour increased with increasing scores in shyness and aggression, and wool sucking increased with increasing extraversion score as well. Despite not detecting significant multicollinearity, when these personality traits were included in the models, as favoured by model selection, the differences between these weaning age groups decreased slightly (Fig. 2; Table 2). The occurrence of stereotypic behaviour seemed to be correlated, as some cats had co-occurring wool sucking and excessive grooming (polychoric r = 0.294, P < 0.001). Cats weaned before 8 weeks of age were significantly more likely to have an owner-evaluated behaviour problem than cats weaned at 12–13 weeks of age (χ2 = 18.73, DF = 1, P < 0.0001), but similarly, this probability slightly decreased when including personality traits in the model.
The effect of weaning age on non-social behaviour in logistic regression analyses. Grey circles are the groups considered early weaned. (a) Cats weaned in adulthood or not weaned at all had a lower probability for shyness towards novel objects than other cats. (b) Cats weaned in adulthood or not weaned at all were less likely to perform wool sucking than other cats. (c) Cats weaned at 14–15 weeks of age were less likely to groom excessively than cats weaned 12–13 weeks of age. (d) Cats weaned before 8 weeks of age were more likely to have an owner-evaluated behaviour problem than cats weaned at 12–13 weeks of age. Error bars indicate 95% confidence limits. N = 4925 (wool sucking), N = 5683 (excessive grooming), N = 5550 (owner-evaluated behaviour problem), and N = 5726 (shyness).
Discussion
This study showed that early weaning can have detrimental effects on cat behaviour: we found a predisposition for aggression in early weaned cats, and discovered that the late weaned cats were less likely to behave aggressively and display stereotypic behaviour. The lower probability of stereotypic behaviour in the late weaned cats was partially explained by a lower probability of aggression, which in turn was correlated with stereotypic behaviour. These results suggest delayed weaning as a simple means to improve the quality of life in domestic cats. Given that the cat is one of the most popular pets34, understanding the genetic and environmental factors that affect the welfare of cats is important. A high prevalence of behavioural issues was observed in our study cohort, indicating a need for improving welfare.
We utilised a citizen science approach by involving thousands of cat owners and breeders in the data collection, and collected a large behavioural data of home-living cats in multiple weaning age groups. However, the questionnaire approach has a few limitations. Questionnaires have a subjective component, but the reliability of questionnaires has been good in previous studies and the answers have strongly correlated with the behaviour of the animals44,45,46. Furthermore, sometimes questionnaires can detect some aspects of behaviour other methods may not detect47. As we used an online questionnaire, our behavioural data is a convenience sample. Enthusiastic cat people and people using social media were, therefore, more likely to respond to the questionnaire. However, this is not expected to affect our results, but this effect is likely seen in the high proportion of pedigree cats and low proportion of early weaned cats. Finally, our study is cross-sectional, rather than experimental. Therefore, based on our results, we cannot conclude that early weaning is the cause of the behaviour changes seen in this study. However, our results are in agreement with previous experimental studies on cats39, 40 and other animals1, 7, 12.
We discovered that early weaning increased the probability for aggression. In earlier studies conducted on laboratory cats, early weaned cats have showed elevated aggression towards both people and other cats39, 40. However, these cats were separated from their mothers much younger than separation usually occurs in companion cats. Furthermore, in one study, no differences in aggression were found between cats weaned at 6 weeks of age and at 12 weeks of age40. Moreover, the home environment is more environmentally and socially enriched than laboratory settings, which could reduce48 or even reverse some effects of early life stress49. However, abnormal aggression caused by severe early life stress seems to be quite resilient to enrichment50, which could explain why early weaned cats in our study show elevated aggression towards strangers even in a home environment.
Our results showed that delayed weaning decreased the probability of stereotypic behaviour. Supporting our findings, it has been previously noted that some forms of stereotypic behaviour are nursing behaviours that would normally be directed towards the mother51. Similarly, wool sucking in cats is thought to arise from the motivation to suckle52. A link between stereotypic wool sucking and early weaning was discovered in an earlier study41. Other studies have reported a similar effect in other animals, with early weaning increasing stereotypic behaviour motivated by nursing53 as well as general stereotypic behaviour31, 32, 54. Stereotypic behaviour was also affected by personality in our study. Cats scoring high in aggression and shyness displayed more stereotypic behaviour, as discovered earlier in rhesus macaques29, 55. Interestingly, the effect of weaning age on stereotypic behaviour decreased when the personality factors were included in the models. As weaning age influenced aggression as well, this indicates that weaning age affects stereotypic behaviour partially indirectly by influencing levels of aggression which, in turn, influences the occurrence of stereotypic behaviour. Furthermore, owners were more likely to report a behaviour problem in cats weaned before 8 weeks of age, indicating that subjectively cat owners regard the behaviour of these early weaned cats as problematic. The difference in the probability of behaviour problem between cats weaned before 8 weeks of age and between 12–13 weeks of age was large (probabilities being 18% and 7.9%, respectively), indicating that either early weaned cats differ from late weaned cats in some other behavioural traits than the ones studied here, or that our questionnaire does not accurately reflect the severity of the negative behavioural traits studied.
Based on our results and previous direct neurobiological studies in other species, early weaning may cause changes in brain function. We hypothesise that the behavioural changes in early weaned cats may be caused by dysfunction of cortical-basal ganglia circuits56. Socially deprived animals display decreased behavioural extinction as well as increased perseveration and behavioural inflexibility9, 40, and the same has been observed in stereotypic57,58,59 and aggressive57 animals. Neurobiologically, socially deprived animals have been discovered, for example, to be more sensitive to dopamine agonists56, 60 and have higher dopamine and lower serotonin levels in nucleus accumbens56, indicating changes in basal ganglia function. This hypothesis could be investigated further with, for example, non-invasive extinction tests59, a dopamine antagonist trial, or future metabolomics tools.
Early weaning may threaten the welfare of cats. Aggression is often defensive in nature or, in other words, induced by fear61. Furthermore, aggression arises in the hypothalamus and induces the activation of the HPA-axis62 and dopaminergic system63, which are also involved in stress. Therefore, it is likely that aggressive cats suffer from stress, either acute or chronic in nature. Stress can, in turn, impair health64,65,66,67. Furthermore, the occurrence of stereotypic behaviour can also threaten welfare. Cats that groom excessively may pull out patches of hair, leading to wounds that can become infected68. Similarly, symptoms of wool sucking include sucking and chewing textiles and plastics. Cats may swallow pieces of these materials, which can lead to intestinal blockages and even premature death. Moreover, aggression is a common reason for relinquishment in cats69 and aggressive cats may also be more likely to be euthanized. We unexpectedly discovered that cats weaned in adulthood or not weaned at all had a lower probability for aggression, shyness, and abnormal behaviour than other weaning age groups. Even more surprisingly, cats weaned at 14–15 weeks of age had a lower probability for excessive grooming than cats weaned at 12–13 weeks of age. Cat behaviour was previously thought to be relatively stable after the critical period of socialisation, which ends at 8 weeks of age36. We hypothesise that extended maternal care is the cause of this decrease in aggressive and abnormal behaviour, as many mothers may nurse and care for their kittens well into adulthood.
In the future, we plan to collect more data and extend our questionnaire to include more environmental factors. Many early life environmental factors, such as maternal care, living conditions before weaning, socialisation, and post-weaning living conditions, may affect behaviour and interact with weaning age, but as our questionnaire did not include these questions, these effects could not be addressed. Therefore, we aim to collect more data to study the interaction of weaning age with other environmental factors and to examine, whether breeds are differentially influenced by weaning age.
Our findings suggest that early weaning can lead to decreased mental welfare. As cats weaned at 14–15 weeks of age were at a lower risk for stereotypic behaviour, the welfare of home living cats may be improved by pushing the recommended weaning age to 14 weeks. This would be a simple and inexpensive way to improve companion feline welfare.
Methods
Questionnaire
A multiple choice online questionnaire was designed to collect extensive information on the health, living conditions and behaviour of Finnish domestic cats. Owners defined their cat’s activity level, tendency to seek human contact (later labelled as ‘contact with people’), aggressiveness towards family members, strangers, and other cats, as well as shyness towards strangers and novel stimuli, ranging from ‘not at all’ to ‘very much’ on a 5-point Likert-type scale. The owners also defined how much the cat licks, bites, and sucks itself using the same Likert-type scale. The owners were asked whether the cat bites, sucks, or eats wool or plastic (labelled as ‘wool sucking’). Here, the options were ‘never’, ‘1–3 times in the cat’s lifetime’, ‘1–12 times per year’, ‘1–4 times per month’, ‘1–3 times per week’, ‘daily’, ‘many times per day’, and ‘most of the day’. Furthermore, the owners were asked whether they thought that their cat had a behaviour problem with the answering options being ‘no’, ‘yes, self-evaluated’, and ‘yes, diagnosed by a veterinarian’. This subjective question helped us understand, which behaviours owners may find problematic. The questionnaire also included demographic questions, such as age, sex, breed, current presence of other cats in the household, and access to outdoors. Furthermore, cat owners could report the weaning age of the cat using nine categorical options: before 8 weeks of age, at 8–9 weeks of age, at 10–11 weeks of age, at 12–13 weeks of age, at 14–15 weeks of age, between 16 weeks and 1 year of age, in adulthood, not weaned at all, or weaning age unknown.
Informed consent was obtained from all participants. Cat owners were informed that the questionnaire answers would be used for research. We emphasized that all information given by the owners would remain strictly confidential and that individual cats or owners could not be identified from the published results.
The questionnaire was opened December 2012 and advertised on Facebook and via cat breed organisations. The questionnaire is still open and can be answered on the website http://www.kissangeenit.fi.
Statistical analyses
Some new variables were created before analyses. Firstly, due to the large number of breeds with few individuals, some cat breeds were grouped together (Supplementary Table S1). These grouped breeds, such as Abyssinian and Somali, have a strong genetic relationship70, 71. Breeds with only a few respondents were combined under ‘other’ breed group. Secondly, non-intact cats were either castrated or spayed, had a hormonal implant inserted (males only), or were receiving contraceptive oral pills (females only); all of these methods inhibit fertility and change the hormonal status of the cat. Therefore, a new binomial variable called ‘hormonal status’ was created, in which cats were regarded as either intact or sterilized. The data initially consisted of 7397 cats. Cats with missing values in personality traits (N = 352) were removed. Furthermore, cats with missing information on weaning age (N = 60) or weaning age unknown (N = 936) were removed, as were cats with missing values in other explanatory variables (N = 302). After careful data examination, some cats had a clearly false age and they were removed as well (N = 21). At this point, the data consisted of 5726 individuals (4925 in wool sucking due to omitting the mildest wool sucking cases, 5683 individuals in grooming and 5550 in behaviour problem due to missing data).
Personality factors were used as covariates in the analyses, as behavioural traits may be intercorrelated. Therefore, we wanted to control for these variables, to understand whether early weaning impacts behavioural traits indirectly, by increasing another trait affecting the studied trait, rather than directly. To obtain these personality factors, principal component analysis was utilised. As the personality traits were coded on a Likert-type scale, polychoric correlations instead of Pearson correlations were used in the principal component analysis. Number of factors to be extracted was based on Kaiser’s stopping rule, scree test, and parallel analysis. Kaiser’s stopping rule suggested a three-factor solution, with eigenvalues of factors 3 and 4 being 1.11 and 0.62, respectively. Scree plot and parallel analysis confirmed this. Since the intercorrelations of the factors were low, orthogonal Varimax rotation was requested. The first factor consisted of all aggression traits and was labelled as ‘Aggression’, and the second factor consisted of shyness traits and was therefore labelled ‘Shyness’. The third factor was composed of activity level and contact with people and was hence dubbed ‘Extraversion’ (Supplementary Table S2).
Logistic regression was used to study the effects of early weaning on behaviour. Nine response variables (and therefore nine analyses) were used: behaviour problem; contact with people; aggression towards family members, strangers, and other cats; shyness towards novel objects and strangers; wool sucking; and excessive grooming. All the response variables were reduced to binomial categories. In all aggression and shyness traits, the event was levels 2–5. In grooming, the event consisted of levels 4–5, since some owners considered 1 as a normal level of grooming, whereas some considered the moderate level (3) as normal. In contact with people, low levels of contact were considered interesting and therefore the event consisted of levels 1–3, as most cats were reported to have a high level of contact to people. In wool sucking, ‘never’ was considered a non-event and occurrence of wool sucking of ‘1–4 times per month’ or higher constituted the event, as we wanted to compare the cats that never display wool sucking to cats that have displayed this behaviour frequently. In behaviour problem, the owner and veterinary diagnoses were grouped together and considered an occurrence of owner-evaluated behaviour problem.
Explanatory variables were selected based on previous literature and forward stepwise model selection by AIC values was used. The model selection was initiated with a starter model of weaning age, sex, and age, as weaning age was the variable of primary interest and different age groups and sexes tend to behave differently72,73,74,75,76. The model selection process favoured the inclusion of several additional variables in the model. In the tested models, we also included some interactions between the weaning age and other variables (Supplementary Table S3), but these interactions were not included in the final models as they decreased model fit. The final models and AIC model selection are shown in Supplementary Table S3.
Since early weaning was compared to the recommended weaning age of 12 weeks, contrasts between the early weaned categories (weaned under 8 weeks of age, at 8–9 weeks of age, and at 10–11 weeks of age) and the category weaned at 12–13 weeks of age were requested, as well as a contrast between early weaned cats and cats weaned at 14–15 weeks of age. Furthermore, a contrast between cats weaned in adulthood or not weaned at all and all the other cats, as well as a contrast between cats weaned at 12–13 weeks of age and 14–15 weeks of age were requested. As the number of pairwise comparisons was high due to several categorical variables, all P values, including the overall effect of variables, were controlled for false discovery rate to decrease the probability of type I error. Contrasts between the differentially weaned groups described above were chosen a priori, and thus the P values of these contrasts were not adjusted. Significance cut-off P value was set to P < 0.05.
Multicollinearity was tested by requesting the generalised variance inflation factor. All the variables fulfilled this assumption, with the variance inflation factor being under 2.5 in all the variables. Furthermore, Pearson goodness-of-fit test for each analysis was examined and all models had a non-significant P value. All statistical analyses were conducted using SAS software version 9.4 (SAS Institute Inc. 2002–2014), except for multicollinearity testing which was done with package car77 in R78.
Data Availability
Data is available from the corresponding author on request, as the data is from privately owned family cats, and therefore there is a possibility to identify an individual cat from the data.
References
Latham, N. R. & Mason, G. J. Maternal deprivation and the development of stereotypic behaviour. Appl. Anim. Behav. Sci. 110, 84–108 (2008).
McLamb, B. L. et al. Early Weaning Stress in Pigs Impairs Innate Mucosal Immune Responses to Enterotoxigenic E. coli Challenge and Exacerbates Intestinal Injury and Clinical Disease. PLOS ONE 8, e59838 (2013).
Lackeyram, D., Yang, C., Archbold, T., Swanson, K. C. & Fan, M. Z. Early weaning reduces small intestinal alkaline phosphatase expression in pigs. J. Nutr. 140, 461–468 (2010).
LaBarba, R. C. & White, J. L. Maternal deprivation and the response to Erlich carcinoma in BALB/c mice. Psychosom. Med. 33, 458–461 (1971).
Ader, R. & Friedman, S. Social factors affecting emotionality and resistance to disease in animals. V. Early separation from the mother and response to a transplanted tumor in the rat. Psychosom. Med. 27, 119–122 (1965).
Sánchez, M. M., Ladd, C. O. & Plotsky, P. M. Early adverse experience as a developmental risk factor for later psychopathology: evidence from rodent and primate models. Dev. Psychopathol. 13, 419–449 (2001).
Jarvis, S. et al. Effects of weaning age on the behavioural and neuroendocrine development of piglets. Appl. Anim. Behav. Sci. 110, 166–181 (2008).
Wang, A. et al. Epigenetic Upregulation of Corticotrophin-Releasing Hormone Mediates Postnatal Maternal Separation-Induced Memory Deficiency. PLOS ONE 9, e94394 (2014).
Gluck, J. P. & Sackett, G. P. Extinction Deficits in Socially Isolated Rhesus Monkeys (Macaca mulatta). Dev. Psychol. 12, 173–174 (1976).
Lévy, F., Melo, A. I., Galef, B. G., Madden, M. & Fleming, A. S. Complete maternal deprivation affects social, but not spatial, learning in adult rats. Dev. Psychobiol. 43, 177–191 (2003).
Kanari, K., Kikusui, T., Takeuchi, Y. & Mori, Y. Multidimensional structure of anxiety-related behavior in early-weaned rats. Behav. Brain Res. 156, 45–52 (2005).
Kikusui, T., Takeuchi, Y. & Mori, Y. Early weaning induces anxiety and aggression in adult mice. Physiol. Behav. 81, 37–42 (2004).
Kikusui, T., Kiyokawa, Y. & Mori, Y. Deprivation of mother-pup interaction by early weaning alters myelin formation in male, but not female, ICR mice. Brain Res. 1133, 115–122 (2007).
Lyons, D. M., Parker, K. J. & Schatzberg, A. F. Animal Models of Early Life Stress: Implications for Understanding Resilience. Dev. Psychobiol. 52, 616–624 (2010).
Sih, A. Effects of early stress on behavioral syndromes: An integrated adaptive perspective. Neurosci. Biobehav. Rev. 35, 1452–1465 (2011).
Elfwing, M. et al. Early stress causes sex-specific, life-long changes in behaviour, levels of gonadal hormones, and gene expression in chickens. PLOS ONE 10, e0125808 (2015).
Kikusui, T. & Mori, Y. Behavioural and neurochemical consequences of early weaning in rodents. J. Neuroendocrinol. 21, 427–431 (2009).
Ferdman, N., Murmu, R. P., Bock, J., Braun, K. & Leshem, M. Weaning age, social isolation, and gender, interact to determine adult explorative and social behavior, and dendritic and spine morphology in prefrontal cortex of rats. Behav. Brain Res. 180, 174–182 (2007).
Gee, D. G. & Casey, B. J. The impact of developmental timing for stress and recovery. Neurobiol. Stress 1, 184–194 (2015).
Roque, S., Mesquita, A. R., Palha, J. A., Sousa, N. & Correia-Neves, M. The behavioral and immunological impact of maternal separation: a matter of timing. Front. Behav. Neurosci. 8, 192 (2014).
Mason, G., Clubb, R., Latham, N. & Vickery, S. Why and how should we use environmental enrichment to tackle stereotypic behaviour? Appl. Anim. Behav. Sci. 102, 163–188 (2007).
Mason, G. J. Stereotypies: a critical review. Anim. Behav. 41, 1015–1037 (1991).
Pomerantz, O., Paukner, A. & Terkel, J. Some stereotypic behaviors in rhesus macaques (Macaca mulatta) are correlated with both perseveration and the ability to cope with acute stressors. Behav. Brain Res. 230, 274–280 (2012).
Bachmann, I., Bernasconi, P., Herrmann, R., Weishaupt, M. A. & Stauffacher, M. Behavioural and physiological responses to an acute stressor in crib-biting and control horses. Appl. Anim. Behav. Sci. 82, 297–311 (2003).
Mohapatra, R. K., Panda, S. & Acharya, U. R. Study on activity pattern and incidence of stereotypic behavior in captive tigers. J. Vet. Behav. Clin. Appl. Res. 9, 172–176 (2014).
Jeppesen, L. L., Heller, K. E. & Bildsøe, M. Stereotypies in female farm mink (Mustela vison) may be genetically transmitted and associated with higher fertility due to effects on body weight. Appl. Anim. Behav. Sci. 86, 137–143 (2004).
Svendsen, P. M. et al. Selection against stereotypic behaviour may have contradictory consequences for the welfare of farm mink (Mustela vison). Appl. Anim. Behav. Sci. 107, 110–119 (2007).
Cussen, V. A. & Mench, J. A. The Relationship between Personality Dimensions and Resiliency to Environmental Stress in Orange-Winged Amazon Parrots (Amazona amazonica), as Indicated by the Development of Abnormal Behaviors. PLOS ONE 10, e0126170 (2015).
Vandeleest, J. J., McCowan, B. & Capitanio, J. P. Early rearing interacts with temperament and housing to influence the risk for motor stereotypy in rhesus monkeys (Macaca mulatta). Appl. Anim. Behav. Sci. 132, 81–89 (2011).
Tiira, K. et al. Environmental Effects on Compulsive Tail Chasing in Dogs. PLOS ONE 7, e41684 (2012).
Mason, G. J. Tail-biting in mink (Mustela vison) is influenced by age at removal from the mother. Anim. Welf. 3, 305–311 (1994).
Würbel, H. & Stauffacher, M. Physical condition at weaning affects exploratory behaviour and stereotypy development in laboratory mice. Behav. Processes 43, 61–69 (1998).
Jeppesen, L. L., Heller, K. E. & Dalsgaard, T. Effects of early weaning and housing conditions on the development of stereotypies in farmed mink. Appl. Anim. Behav. Sci. 68, 85–92 (2000).
FEDIAF. Facts & figures. (2014). http://www.fediaf.org/who-we-are/facts-and-figures.html (2014). (Date of access: 12/01/2017).
Bradshaw, J. W. S., Casey, R. & Brown, S. The Behaviour of the Domestic Cat. (CABI, 2012).
Bernstein, P. The human-cat relationship in The Welfare of Cats (ed. Rochlitz, I.) 47–89 (Springer, 2007).
RSPCA. How old should a kitten be before they are adopted/purchased? RSPCA Australia knowledgebase http://kb.rspca.org.au/How-old-should-a-kitten-be-before-they-are-adoptedpurchased_313.html (2009). (Date of access: 21/10/2016).
AVMA. Selecting a Pet Cat. https://www.avma.org/public/PetCare/Pages/Selecting-a-Pet-Cat.aspx (Date of access: 21/10/2016).
Guyot, G. W., Bennett, T. L. & Cross, H. A. The effects of social isolation on the behavior of juvenile domestic cats. Dev. Psychobiol. 13, 317–329 (1980).
Seitz, P. F. D. Infantile Experience and Adult Behavior in Animal Subjects: II. Age of Separation from the Mother and Adult Behavior in the Cat. Psychosom. Med. 21, 353–378 (1959).
Borns-Weil, S. et al. A case-control study of compulsive wool-sucking in Siamese and Birman cats (n = 204). J. Vet. Behav. Clin. Appl. Res. 10, 543–548 (2015).
Vapalahti, K. et al. Health and Behavioral Survey of over 8000 Finnish Cats. Front. Vet. Sci. 3, 70 (2016).
The Humane Society of the United States. Kitten Behavior Basics. http://www.humanesociety.org/animals/cats/tips/kitten_behavior_basics.html (2014). (Date of access: 21/10/2016).
Feaver, J., Mendl, M. & Bateson, P. A method for rating the individual distinctiveness of domestic cats. Anim. Behav. 34, 1016–1025 (1986).
Momozawa, Y. et al. Assessment of equine temperament by a questionnaire survey to caretakers and evaluation of its reliability by simultaneous behavior test. Appl. Anim. Behav. Sci. 84, 127–138 (2003).
Wilsson, E. & Sinn, D. L. Are there differences between behavioral measurement methods? A comparison of the predictive validity of two ratings methods in a working dog program. Appl. Anim. Behav. Sci. 141, 158–172 (2012).
Bennett, S. L., Litster, A., Weng, H.-Y., Walker, S. L. & Luescher, A. U. Investigating behavior assessment instruments to predict aggression in dogs. Appl. Anim. Behav. Sci. 141, 139–148 (2012).
Iwata, E., Kikusui, T., Takeuchi, Y. & Mori, Y. Fostering and environmental enrichment ameliorate anxious behavior induced by early weaning in Balb/c mice. Physiol. Behav. 91, 318–324 (2007).
Francis, D. D., Diorio, J., Plotsky, P. M. & Meaney, M. J. Environmental Enrichment Reverses the Effects of Maternal Separation on Stress Reactivity. J. Neurosci. 22, 7840–7843 (2002).
Tulogdi, Á. et al. Effects of resocialization on post-weaning social isolation-induced abnormal aggression and social deficits in rats. Dev. Psychobiol. 56, 49–57 (2014).
Berkson, G. Development of abnormal stereotyped behaviors. Dev. Psychobiol. 1, 118–132 (1968).
Mackie, M. Wool Sucking in Cats. http://www.usask.ca/wcvm/herdmed/applied-ethology/behaviourproblems/woolcat.html (2001). (Date of access: 21/10/2016).
Sharman, D. F., Mann, S. P., Fry, J. P., Banns, H. & Stephens, D. B. Cerebral dopamine metabolism and stereotyped behaviour in early-weaned piglets. Neuroscience 7, 1937–1944 (1982).
Jones, M. A., Mason, G. & Pillay, N. Early social experience influences the development of stereotypic behaviour in captive-born striped mice. Rhabdomys. Appl. Anim. Behav. Sci. 123, 70–75 (2010).
Gottlieb, D. H., Capitanio, J. P. & McCowan, B. Risk factors for stereotypic behavior and self-biting in rhesus macaques (Macaca mulatta): Animal’s history, current environment, and personality. Am. J. Primatol. 75, 995–1008 (2013).
Hall, F. S. et al. Isolation Rearing in Rats: Pre- and Postsynaptic Changes in Striatal Dopaminergic Systems. Pharmacol. Biochem. Behav. 59, 859–872 (1998).
Ijichi, C. L., Collins, L. M. & Elwood, R. W. Evidence for the role of personality in stereotypy predisposition. Anim. Behav. 85, 1145–1151 (2013).
Garner, J. P. & Mason, G. J. Evidence for a relationship between cage stereotypies and behavioural disinhibition in laboratory rodents. Behav. Brain Res. 136, 83–92 (2002).
Protopopova, A., Hall, N. J. & Wynne, C. D. L. Association between increased behavioral persistence and stereotypy in the pet dog. Behav. Processes 106, 77–81 (2014).
Lewis, M. H., Gluck, J. P., Beauchamp, A. J., Keresztury, M. F. & Mailman, R. B. Long-term effects of early social isolation in Macaca mulatta: changes in dopamine receptor function following apomorphine challenge. Brain Res. 513, 67–73 (1990).
Duffy, D. L., Hsu, Y. & Serpell, J. A. Breed differences in canine aggression. Appl. Anim. Behav. Sci. 114, 441–460 (2008).
Kruk, M. R., Halász, J., Meelis, W. & Haller, J. Fast positive feedback between the adrenocortical stress response and a brain mechanism involved in aggressive behavior. Behav. Neurosci. 118, 1062–1070 (2004).
de Almeida, R. M. M., Ferrari, P. F., Parmigiani, S. & Miczek, K. A. Escalated aggressive behavior: Dopamine, serotonin and GABA. Eur. J. Pharmacol. 526, 51–64 (2005).
Glaser, R. & Kiecolt-Glaser, J. K. Science and society: Stress-induced immune dysfunction: implications for health. Nat. Rev. Immunol. 5, 243–251 (2005).
Cavigelli, S. A. & McClintock, M. K. Fear of novelty in infant rats predicts adult corticosterone dynamics and an early death. Proc. Natl. Acad. Sci. USA 100, 16131–16136 (2003).
Schneiderman, N., Ironson, G. & Siegel, S. D. Stress and health: psychological, behavioral, and biological determinants. Annu. Rev. Clin. Psychol. 1, 607–628 (2005).
Cohen, S., Janicki-Deverts, D., Miller, G. E. Psychological Stress and Disease. JAMA 298, 1685–1687 (2007).
Reinhardt, V. Hair pulling: a review. Lab. Anim. 39, 361–369 (2005).
Casey, R. A., Vandenbussche, S., Bradshaw, J. W. S. & Roberts, M. A. Reasons for relinquishment and return of domestic cats (Felis silvestris catus) to rescue shelters in the UK. Anthrozoös 22, 347–358 (2009).
Lipinski, M. J. et al. The ascent of cat breeds: genetic evaluations of breeds and worldwide random-bred populations. Genomics 91, 12–21 (2008).
O’Brien, S. J. et al. State of cat genomics. Trends Genet. 24, 268–279 (2008).
Gartner, M. C. & Weiss, A. Scottish wildcat (Felis silvestris grampia) personality and subjective well-being: Implications for captive management. Appl. Anim. Behav. Sci. 147, 261–267 (2013).
Gartner, M. C., Powell, D. M. & Weiss, A. Personality Structure in the Domestic Cat (Felis silvestris catus), Scottish Wildcat (Felis silvestris grampia), Clouded Leopard (Neofelis nebulosa), Snow Leopard (Panthera uncia), and African Lion (Panthera leo): A Comparative Study. J. Comp. Psychol. 128, 414–426 (2014).
Gosling, S. D. Personality Dimensions in Spotted Hyenas (Crocuta crocuta). J. Comp. Psychol. 112, 107–118 (1998).
Lindell, E. M., Erb, H. N. & Houpt, K. A. Intercat aggression: A retrospective study examining types of aggression, sexes of fighting pairs, and effectiveness of treatment. Appl. Anim. Behav. Sci. 55, 153–162 (1997).
Hart, B. L. & Hart, L. A. Your ideal cat: insights into breed and gender differences in cat behavior. (Purdue University Press, 2013).
Fox, J. & Weisberg, S. An R Companion to Applied Regression. (SAGE Publications, 2011).
R Core Team (2013). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. www.R-project.org/.
Acknowledgements
We thank all cat owners who participated in the study. This study was partially funded by the Finnish Cat Association, the Jane and Aatos Erkko Foundation, and ERANET-NEURON.
Author information
Authors and Affiliations
Contributions
M.A. conceived and designed the study with help from H.L. and K.V. M.A conducted the statistical analyses with help from K.V. M.A. and H.L. drafted the manuscript with help from K.V.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ahola, M.K., Vapalahti, K. & Lohi, H. Early weaning increases aggression and stereotypic behaviour in cats. Sci Rep 7, 10412 (2017). https://doi.org/10.1038/s41598-017-11173-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-11173-5
This article is cited by
-
Aggressiveness, ADHD-like behaviour, and environment influence repetitive behaviour in dogs
Scientific Reports (2022)
-
The Early-Life «Programming» of Anxiety-Driven Behaviours in Adulthood as a Product of Predator-Driven Evolution
Evolutionary Biology (2022)
-
Breed differences of heritable behaviour traits in cats
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.