Abstract
This study aimed to investigate application of Onodera prognostic nutrition index (OPNI) and neutrophil-to-lymphocyte ratio (NLR) in evaluating risk of postoperative complications in Crohn’s disease (CD). Clinical data of 108 postoperative CD patients in 9 years were respectively reviewed. OPNI and NLR were within 1 week preoperatively. Average OPNI was 38.8 ± 8.2 and significantly lower in patients with: CD type B3; lymphopenia; decreased haemoglobin, prealbumin, and albumin; and daily enteral nutrition <500 kcal/d. Average NLR was 5.9 ± 12.1 and significantly higher in patients with: CD type B3, neutrophilia, lymphopenia, decreased prealbumin, and enteral nutrition <500 kcal/d. Youden index was maximal at OPNI 39.8 and NLR 4.1, patients were divided into two groups by OPNI 39.8 and NLR 4.1; Low OPNI (≤39.8) group had significantly greater incidence of type B3, lymphopenia, decreased haemoglobin, prealbumin and albumin, and enteral nutrition <500 kcal/day, more likely to have intra-abdominal bleeding. High NLR group (≥4.1) had significantly greater incidence of type B3, neutrophilia, and lymphopenia, more likely to develop lung infection. OPNI and NLR were significantly negatively correlated. Smoking within 1 year preoperatively, OPNI <39.8, NLR ≥ 4.1 were independent risk factors for postoperative complications in CD.
Similar content being viewed by others
Introduction
The Onodera prognostic nutrition index (OPNI), established by Onodera after the analysis of 200 gastrointestinal surgery patients in 1984, is an index used to evaluate the nutritional condition and predict surgical risk of gastrointestinal surgery patients1, 2. Many studies have investigated the relationship between the OPNI and gastrointestinal diseases3, 4. Another parameter used for prognostic evaluation in gastrointestinal diseases is the neutrophil-to-lymphocyte ratio (NLR), which is an index used to measure the severity of systemic inflammation5, 6. Crohn’s disease (CD) is an inflammatory gastrointestinal disease that can cause systemic inflammation and severe malnutrition, which influences prognosis and postoperative recovery. Although several studies have reported the relationship between OPNI and postoperative complications, NLR and Crohn’s disease severity in Crohn’s disease patients, no studies, until now, have yet investigated whether OPNI and NLR could be used for evaluation of postoperative complications in Crohn’s disease patients at same time. In the present study, we investigated the application of the OPNI and the NLR in risk evaluation of postoperative complications in patients with CD.
Results
Distribution of OPNI and NLR According to Clinical Features
The average OPNI and NLR of all patients were 38.8 ± 8.2 and 5.9 ± 12.1, respectively. The distribution of OPNI and NLR according to clinical features, such as sex, age, smoking history, blood type, primary lesion location, disease type, and preoperative BMI are shown in Tables 1 and 2.
The mean OPNI of the control group was higher than that of the patients with disease type B3 (t = 2.247, P = 0.027), with preoperative lymphopenia (t = 5.880, P = 0.000), lower than normal haemoglobin (t = 4.984, P = 0.000), albumin (t = 11.178, P = 0.000), and prealbumin (t = 4.987, P = 0.000), and patients whose enteral nutritional intake was less than 500 kcal/day within 2 weeks preoperatively (t = 5.027, P = 0.000).
The mean NLR of the control group was lower than that of the patients with disease type B3 (P = 0.009), preoperative neutrophilia (P = 0.000), preoperative lymphopenia (P = 0.000), lower than normal prealbumin (P = 0.028), and an enteral nutritional intake of less than 500 kcal/day within 2 weeks preoperatively (P = 0.043).
ROC Curves for OPNI and NLR, and a Comparison of Clinical Features After Grouping According to the OPNI and NLR Cutoff Values
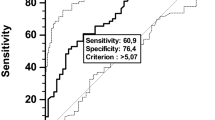
We drew the ROC curve of the OPNI according to whether postoperative complications occurred (Fig. 1).
The area under the curve was 0.699. When the OPNI was 39.8, the Youden index was maximal, with a sensitivity of 70% and specificity of 68%, and the prediction of risk of postoperative complications was most accurate. Taking 39.8 as the cutoff value, patients were divided into the high OPNI group (OPNI ≥ 39.8, n = 53) and the low OPNI group (OPNI < 39.8, n = 55). The differences in clinical features between the two groups were then analysed (Table 3).
Compared with the high OPNI group, the low OPNI group had a greater incidence of: disease type B3 (X2 = 5.354, P = 0.021); preoperative lymphopenia (X2 = 9.864, P = 0.002); decreased preoperative haemoglobin (X2 = 17.341, P = 0.000), prealbumin (X2 = 10.166, P = 0.001), and albumin (X2 = 75.363, P = 0.000); and enteral nutritional intake less than 500 kcal/day within 2 weeks preoperatively (X2 = 20.298, P = 0.000).
In order to find what specific types of complications can be caused by low OPNI, we calculated the incidence of seven complications in the low and high OPNI group (Table 4) and each complication was listed as fourfold table according to the incidence of complications and the level of OPNI (Table 5). we found that when OPNI was below cutoff value (39.8), Crohn’s disease patients were more likely to have intra-abdominal bleeding (14.55%, P = 0.018).
We drew the ROC curve of NLR according to whether postoperative complications occurred (Fig. 2).
The area under the curve was 0.675. When the NLR was 4.1, the Youden index was maximal, with a sensitivity of 70% and specificity of 56.4%, and the prediction of the risk of postoperative complications was most accurate. Taking 4.1 as the cutoff value, patients were divided into the high NLR group (NLR ≥ 4.1, n = 55) and the low NLR group (NLR < 4.1, n = 53). The differences in clinical features between the two groups were then analysed (Table 6).
Compared with the low NLR group, the high NLR group had a significantly greater incidence of: disease type B3 (X2 = 5.354, P = 0.021), preoperative neutrophilia (X2 = 5.560, P = 0.018), and preoperative lymphopenia (X2 = 25.961, P = 0.000).
In order to find what specific types of complications can be caused by high NLR, we calculated the incidence of seven complications in the low and high NLR group (Table 7) and each complication was listed as fourfold table according to the incidence of complications and the level of NLR (Table 8). we found that when NLR was higher than cut-off value (4.1), Crohn’s disease patients were more likely to develop lung infection (7.27%, P = 0.046).
Correlation Between OPNI and NLR and Independent Risk Factors for Postoperative Complications
The OPNI and NLR were significantly negatively correlated (r = −0.420, P = 0.000, Table 9).
The OPNI and NLR were then combined to calculate the incidence of postoperative complications (Table 10). The incidence of postoperative complications in patients with concurrent low OPNI and high NLR was 48.6%, which was significantly higher than in patients with other OPNI and NLR combinations (X2 = 12.255, P = 0.007).
The OPNI and NLR were included into univariate analysis of risk factors for postoperative complications, then we screened meaningful variables (Table 11).
Univariate analysis showed that the following indicators were statistically significant: history of smoking within 1 year preoperatively (X2 = 5.999, P = 0.014); enteral nutritional intake within 2 weeks preoperatively was less than 500 kcal/day (X2 = 7.308, P = 0.007); lower than normal preoperative lymphocyte count (X2 = 5.745, P = 0.017) and prealbumin (X2 = 4.398, P = 0.036); low OPNI (X2 = 8.345, P = 0.004), and high NLR (X2 = 6.047, P = 0.014).
The above results were further analysed by logistic regression (Table 12). The independent risk factors for postoperative complications were: history of smoking within 1 year preoperatively (OR 3.006, 95% CI 1.170–7.727, P = 0.022), OPNI (OR 2.727, 95% CI 1.024–7.262, P = 0.045) and NLR (OR 2.782, 95% CI 1.042–7.425, P = 0.041).
Discussion
CD is a chronic non-specific intestinal inflammatory disease that tends to recur throughout the patient’s life. The incidence of CD shows significant distributional differences according to race and region. A 2013 study on the geographic variation and environmental risk factors for CD showed that the incidence of CD is higher in Europe and America, with an average annual prevalence of 13.7–198.5/105 and a morbidity of 3.74–14.6/105 7. The prevalence and morbidity of CD in China are increasing each year; the prevalence and morbidity of CD were 2.29/105 and 1.21/105, respectively, from 2003 to 2007, which was increased compared with that from 1950 to 2002 (1.38/105 and 0.28/105, respectively)8.
The treatment of CD is mainly based on internal medicine; however, surgical intervention is still needed. In the natural course of CD, about 80% of patients need at least one surgical treatment in their lifetime9. The high incidence of postoperative complications in CD presents a challenge for surgeons. The incidence of postoperative complications in CD patients is significantly higher than after other intestinal resection surgery and is considered very satisfactory if the incidence is 10%10, 11. The incidence of postoperative complications in CD patients in China is 9.3–38%12. In the present study, postoperative complications occurred in 30 patients, and the overall incidence of postoperative complications was 27.8%, which is consistent with the literature. Death occurred in six patients. Therefore, it is very important to evaluate the risk factors of postoperative complications in CD patients.
OPNI is a tool based on nutritional and immune status to assess patients’ nutritional and inflammatory status and predict surgical risk. Albumin is synthesized by the liver, and albumin levels decline if nutritional intake is inadequate for a long period; hence, albumin can be used as an index of chronic protein malnutrition to reflect the general nutritional status of patients. The lymphocyte count reflects both the nutritional status and the immune function of patients. A decline in lymphocyte count can be caused by either malnutrition or poor cellular immune function13. Many scholars reported that the OPNI plays an important role in the prognosis evaluation of digestive system neoplasms.
CD is a chronic inflammatory and autoimmune disease that occurs as a result of abnormal immune function of T cells14. Most CD patients already have a malnutritional status preoperatively, which seriously affects postoperative recovery15. We designed this study to determine whether the OPNI could be used as a predictor of CD postoperative complications. In order to increase the scientific accuracy of the present study, we used the cutoff value calculated by the ROC curve to divide patients into the high and low OPNI groups, instead of using an empirical value (45) like in many previous studies.
Our study shows that the OPNI is an independent predictor of postoperative complications. Low OPNI indicates that the patient status was one of malnutrition and poor immunization. We also found that the high OPNI group had a greater incidence of non-B3 type CD than B3 type. According to clinical experience, the nutritional status of most B3 type patients is indeed worse than that of non-B3 patients, which suggests that disease type was a possible factor affecting the OPNI.
The enteral nutritional intake of most patients in the high OPNI group was more than 500 kcal/day within 2 weeks preoperatively, which suggests that the administration of enteral nutrition preoperatively was a possible factor affecting the OPNI. We can improve the prognosis of CD patients by increasing the OPNI by reasonable application of enteral nutrition. Compared with parenteral nutrition, enteral nutrition has its unique advantages in treatment of CD, such as regulating the intestinal tract flora, maintaining remission state, and promoting intestinal mucous rehabilitation16. CD recurrence can be effectively prevented if the energy provided by the elemental diet is more than 900 kcal/day17. This is further enhanced by the addition of specific nutrients, such as glutamine, omega-3 polyunsaturated fatty acids, and probiotics.
The early stage of inflammation is a proinflammatory state mediated by inflammatory factors such as TNF-α, interleukin-1, and interleukin-6, which are released by neutrophils, macrophages, and monocytes. This systemic inflammatory response is associated with the inhibition of neutrophil apoptosis, but it can also cause tissue damage18. Lymphocyte apoptosis in the spleen and thymus also increases correspondingly, causing immunosuppression, multiple organ dysfunction, and death19. Therefore, the NLR is an index for measuring systemic inflammation20. The influence of CD on inflammation, the application of immunosuppressive agents, and preoperative severe malnutrition result in an increase in neutrophils and a decrease in lymphocytes in CD patients. This increase in lymphocytes, decrease in neutrophils, and improvement in the systemic inflammatory response happen simultaneously21. More serious complications may occur if neutrophils rise and lymphocyte decline continuously for 1 week. Thus, the NLR can reflect the severity of the disease. In 2011, it was reported that the NLR is superior to white blood cell count in predicting the prognosis of acute pancreatitis22. In 2012, it was reported that the NLR is associated with the severity of non-alcoholic hepatic adipose infiltration23. In 2013, it was reported that the NLR could be used as a predictive index of the severity of ulcerative colitis24. However, the relationship between the NLR and CD has not been reported until now. Our findings suggest that the NLR of non-B3 patients is lower than that of B3 patients. Most non-B3 patients belonged to the low NLR group. According to clinical experience, the severity of B3 patients is indeed worse than that of non-B3 patients. Therefore, we consider that the NLR can predict the severity of CD, which is closely linked with prognosis. Univariate and multivariate analysis showed that when the NLR was ≥4.1, the risk of postoperative complications increased by 2.782 times.
The OPNI is a predictive index based on nutrition and immunity, and the NLR is an indicator for measuring systemic inflammation. We found that OPNI and NLR in CD patients were significantly negatively correlated. More severe systemic inflammation results in a worse nutrition status, and an increased risk of postoperative complications. The incidence of postoperative complications in patients with concurrent low OPNI and high NLR was 48.6%, which was much higher than the other groups.
Our study shows that history of smoking within 1 year preoperatively was an independent risk factor for postoperative complications. The incidence of postoperative complications in CD patients with a smoking history was three times higher than that in patients without a smoking history. Other studies have also suggested that smoking is an important risk factor25, 26 for postoperative complications such as respiratory27 and circulatory issues28, 29, wound infection30, and delayed healing31. In contrast, a recent study reported the opposite32. Further research is required to determine whether these diametrically opposite conclusions were associated with racial differences, the amount of nicotine, and the body’s tolerance to nicotine.
The albumin level and neutrophil and lymphocyte counts that are needed for the calculation of OPNI and NLR are included in preoperative routine blood testing, which is simple, fast, inexpensive, and available even in the most basic hospital. The above indices can increase the accuracy of prediction of postoperative complications in CD patients, while not increasing the patient’s economic burden and physical pain. The combined application of OPNI and NLR has better predictive value and is worth further promotion in clinical practice.
Limitations
As a retrospective study, there are many shortcomings in this research, such as limited sample size, and bias caused by single center analysis, which all can cause some interference to the results of the study. Therefore, the conclusion of this study needs to be verified by a larger sample of retrospective studies or prospective studies.
Materials and Methods
General Information
We reviewed the clinical records of 108 CD patients who underwent bowel resection in Peking Union Medical College Hospital between 2004 and 2013. There were 67 (62%) males and 41 (38%) females, and the male-to-female ratio was 1.6:1. Average patient age was 37.6 ± 13.1 years (range 13–70 years). Average duration of hospitalization was 45.9 ± 24.9 days (range 2–124 days). Average preoperative BMI was 18.0 ± 3.4 kg/m2 (range 11.7–30.42 kg/m2). Thirty-eight patients (35.2%) had a history of smoking within 1 year preoperatively, and 25 (25.9%) had a history of appendectomy. No patients had a family history of CD. Postoperative complications occurred in 30 cases, with an overall incidence rate of 27.8%; intraperitoneal haemorrhage occurred in 9/30 cases (30%), wound infection in 6/30 cases (20%), intestinal perforation or fistula in 5/30 cases (16.7%), respiratory infection in 4/30 cases (13.3%), incomplete intestinal obstruction in 3/30 cases (10%), abdominal infection in 2/30 cases (6.7%), and intestinal perforation in 1/30 cases (3.3%) in the first 17 days postoperatively and rebleeding on the first day after reoperation. The case with intestinal perforation died due to respiratory failure at last.
As a retrospective research, all experimental protocols and all methods performed were approved by Ethics Review Board of Chinese Academy of Medical Sciences and Peking Union Medical College Hospital (CAMS& PUMCH), and Ethical Review Number is S-K302.
All patients in this study were treated and followed-up in our department. We got their permission to use their blood test results during treatment for research. All the information/image(s) in this article can be in an online open-access publication.
Inclusion and Exclusion Criteria
Inclusion criteria:
-
No history of bowel resection
-
Bowel resection performed for CD
-
Postoperative pathological diagnosis of CD
Exclusion criteria:
-
History of previous bowel resection
-
Decreased neutrophils, lymphocytes and albumin in peripheral blood within 1 week preoperatively
-
No follow-up record
A final total of 108 cases were included.
Calculation of OPNI and NLR
According to routine blood and biochemical testing conducted within 1 week preoperatively, the OPNI and the NLR were calculated as follows:
-
(1)
OPNI = albumin (g/L) +5× lymphocyte count (109/L)
-
(2)
NLR = neutrophil count (109/L)/lymphocyte count (109/L)
Grouping Criteria
There were 18 parameters investigated, including sex, age, history of smoking within 1 year preoperatively, history of appendectomy, preoperative disease duration, primary lesion, disease type (B3 or not), extraintestinal manifestations, perianal lesions, emergency surgery, preoperative body mass index (BMI), blood type, preoperative haemoglobin, preoperative neutrophil count, preoperative lymphocyte count, preoperative albumin, preoperative prealbumin, and preoperative intake of enteral nutrition. The classification method of patient age, disease location and disease type was in accordance with the CD Montreal standards formulated at the International Congress of Gastroenterology in 2005 [7]. Age was divided into three groups: A1 (<17 years), A2 (17–40 years) and A3 (>40 years). Disease location was divided into four groups: type L1 (ileum), type L2 (colon), type L3 (ileocolon), and type L4 (upper gastrointestinal tract). The cases in which the lesions involved both the upper gastrointestinal tract and L1–L3 were classified as L1–L3. The disease type was divided into three groups: type B1 (non-obstruction, non-perforation), type B2 (obstruction), and type B3 (perforation). All patients were divided into the non-enteral nutrition group or the enteral nutrition group, with an enteral nutritional intake of 500 kcal/day within 2 weeks preoperatively used as the cut-off.
Statistical analysis
The data were analysed using statistical software SPSS version 19(SPSS, Solutions Statistical Package for the Social Sciences, manufacturer’s name: International Business Machines Corporation, IBM. Armonk, New York, U.S.A). Enumeration data were analysed using Pearson’s chi-squared test or Fisher’s exact test. Measurement data were analysed by the independent samples t-test, Mann-Whitney U test and Kruskal-Wallis test.
The Youden index = sensitivity − (1− specificity), and was calculated by the receiver operating characteristic (ROC) curve.
The OPNI and NLR values with optimal sensitivity and specificity were regarded as the cutoff values according to which all patients were divided into high and low OPNI and NLR groups. We then analysed the distribution of characteristics and correlation between OPNI and NLR. We performed logistic regression analysis to investigate the relationship between OPNI, NLR, and occurrence of complications in CD patients.
Data availability statement
The datasets analyzed during the current study are not publicly available due to intellectual property protection but are available from the corresponding author on reasonable request.
References
Onodera, T., Goseki, N. & Kosaki, G. Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients. Nihon Geka Gakkai Zasshi 85(9), 1001–1005 (1984).
Sagawa, M. et al. The significance of Onodera’s prognostic nutritional index for the treatment of gastrointestinal cancer. Gan To Kagaku Ryoho 35, 2253–2255 (2008).
Nozoe, T. et al. Prognostic nutritional index:a tool to predict the biological aggressiveness of gastric carcinoma. Surg Today 40(5), 440–443 (2010).
Sachlova, M., Majek, O. & Tucek, S. Prognostic value of scores based on malnutrition or systemic inflammatory response in patients with metastatic or recurrent gastric cancer. Nutrition and cancer 66(8), 1362–70 (2013).
Absenger, G. et al. Preoperative neutrophilto-lymphocyte ratio predicts clinical outcome in patients with stage II and III colon cancer. Anticancer Res 33(10), 4591–4 (2013).
Li, Y. et al. Preoperative NLR for predicting survival rate after radical resection combined with adjuvant immunotherapy with CIK and postoperative chemotherapy in gastric cancer. J Cancer Res Clin Oncol. doi:10.1007/s00432-016-2330-1 (2017).
Galeone, C. et al. Crohn’s disease in Italy: A critical review of the literature using different data sources. Dig Liver Dis. doi:10.1016/j.dld.2016.12.033 (2017).
Zheng, J. J. et al. Comparison of the incidence and prevalence of Crohn’s disease in different years in China. Chin J Intern Med 50, 597–600 (2011).
Ha, F. J., Thong, L. & Khalil, H. Quality of Life after Intestinal Resection in Patients with Crohn Disease: A Systematic Review. Dig Surg.. doi:10.1159/000453590 (2017).
Iesalnieks, I., Dederichs, F., Kilger, A., Schlitt, H. J. & Agha, A. Postoperative morbidity after bowel resections in patients with Crohn’s disease: risk, managementstrategies, prevention. Z Gastroenterol 50, 595–600 (2012).
Brouquet, A. et al. Surgery for intestinal Crohn’s disease recurrence. Surgery 148(5), 936–946 (2010).
Niu, L. Y. et al. Application of nutritional support in perioperative period of Crohn’s disease. Chin J Surg 47(4), 275–278 (2009).
Leandro-Merhi, V. A., Bráz, V. N. & Aquino, J. L. Is total lymphocyte count related to nutritional markers in hospitalized older adults? Arq Gastroenterol 54(1), 79–82 (2017).
Stange, E. F. & Wehkamp, J. Recent advances in understanding and managing Crohn’s disease. F1000Res 21(5), 2896 (2016).
Forbes, A. et al. ESPEN guideline: Clinical nutrition in inflammatory bowel disease. Clin Nutr, 1–27 (2016).
Triantafillidis, J. K., Vagianos, C. & Papalois, A. E. The role of enteral nutrition in patients with inflammatory bowel disease: current aspects. Biomed Res Int 2015, 197167 (2015).
Osamu, W. et al. Enteral nutrition decreases hospitalization rate in patients with Crohn’s disease. J Gastroenterol Hepatol 25(1), 134–137 (2010).
Jimenez, M. F. et al. Dysregulated expression of neutrophil apoptosis in the systemic inflammatory response syndrome. Arch Surg 132, 1263–70 (1997).
Wesche, D. E. et al. Leukocyte apoptosis and its significance in sepsis and shock. J Leukoc Biol 78, 325–37 (2005).
Farah, R. et al. Neutrophil lymphocyte ratio better addition to CRP than CD64 index as marker for infection in COPD. Panminerva Med. (2017).
Zahorec, R. Ratio of neutrophil to lymphocyte counts-rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy 102(1), 5–14 (2001).
Azab, B. et al. Neutrophil-lymphocyte ratio as a predictor of adverse outcomes of acute pancreatitis. Pancreatology 11(4), 445–52 (2011).
Alkhouri, N. et al. Neutrophil to lymphocyte ratio: A new marker for predicting steatohepatitis and fibrosis in patients with non-alcoholic fatty liver disease. Liver Int 32(2), 297–302 (2012).
Celikbilek, M. et al. Neutrophil–Lymphocyte Ratio as a Predictor of Disease Severity in Ulcerative Colitis. J Clin Lab Anal 27(1), 72–6 (2013).
Qiu, F. et al. Impacts of cigarette smoking on immune responsiveness: Up and down or upside down? Oncotarget 8(1), 268–284 (2017).
Choi, S. H. & Stommel, M. Impact of Age at Smoking Initiation on Smoking-Related Morbidity and All-Cause Mortality. Am J Prev Med. (2017).
Simsek, E., Karaman, Y., Gonullu, M., Tekgul, Z. & Cakmak, M. The effect of passive exposure to tobacco smoke on perioperative respiratory complications and the duration of recovery. Braz J Anesthesiol 66(5), 492–498 (2016).
Purvis, T. E. et al. Impact of smoking on postoperative complications after anterior cervical discectomy and fusion. J Clin Neurosci (2017).
Babayan, R. K. Re: Smoking and the risk of mortality and vascular and respiratory events in patients undergoing major surgery. J Urol 192(2), 451 (2014).
Goltsman, D., Munabi, N. C. & Ascherman, J. A. The Association between Smoking and Plastic Surgery Outcomes in 40,465 Patients: An Analysis of the American College of Surgeons National Surgical Quality Improvement Program Data Sets. Plast Reconstr Surg 139(2), 503–511 (2017).
Martin, C. T., Gao, Y., Duchman, K. R. & Pugely, A. J. The Impact of Current Smoking and Smoking Cessation on Short-Term Morbidity Risk After Lumbar. Spine Surgery. Spine (Phila Pa 1976) 41(7), 577 (2016).
Joyce, M. R., Hannaway, C. D., Strong, S. A., Fazio, V. W. & Kiran, R. P. Impact of smoking on disease phenotype and postoperative outcomes for Crohn’s disease patients undergoing surger. y. Langenbecks Arch Surg 398(1), 39–45 (2013).
Acknowledgements
This study was supported by The capital health research and development of special fund number: 2014-3-4014.
Author information
Authors and Affiliations
Contributions
Jian-chun Yu and Wei-ming Kang designed this research. Jian-chun Yu and Wei-ming Kang supervised this research. Chang-zhen Zhu and Xiao-xu Yang wrote this article. Chang-zhen Zhu analysed the data and Zhi-qiang Ma, Xin Ye, Kang Li and Dong Liu collected materials. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kang, WM., Zhu, CZ., Yang, XX. et al. Application of the Onodera prognostic nutrition index and neutrophil-to-lymphocyte ratio in risk evaluation of postoperative complications in Crohn’s disease. Sci Rep 7, 8481 (2017). https://doi.org/10.1038/s41598-017-09265-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-09265-3
This article is cited by
-
The predictive value of perioperative circulating markers on surgical complications in patients undergoing robotic-assisted radical prostatectomy
World Journal of Surgical Oncology (2023)
-
Prognostic value of Onodera’s nutritional index for intermediate- and high-risk gastrointestinal stromal tumors treated with or without tyrosine kinase inhibitors
World Journal of Surgical Oncology (2021)
-
Low psoas muscle area is associated with postoperative complications in Crohn’s disease
International Journal of Colorectal Disease (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.