Abstract
This study examined the association of sleep with inflammation and oxidative stress biomarkers, and with functional outcomes, after stroke rehabilitation. The rehabilitation effects on biomarkers and functional outcomes were also evaluated. Twenty subacute stroke survivors received 4 weeks of upper limb rehabilitation. Baseline inflammatory (i.e., soluble intercellular adhesion molecule-1, sICAM-1) and oxidative stress biomarkers (i.e., glutathione peroxidase, GPx and malondialdehyde, MDA) were assessed, as were sleep outcomes. Positive correlations were observed between baseline level of sICAM-1 and number of awakenings at post-treatment (ρ = 0.51, p < 0.05) as well as between baseline level of MDA and post-performance time of the Wolf Motor Function Test (WMFT-time) (ρ = 0.46, p < 0.05). In addition, MDA levels were significantly decreased, and functional outcomes of the modified Rankin Scale (mRS), functional ability scale of the WMFT, and Stroke Impact Scale (SIS-total, and SIS-physical function) were improved after the rehabilitation. This pilot study emphasizes the relationship among biomarkers, sleep, and functional outcomes after stroke rehabilitation. Oxidative stress markers may be useful predictors of functional outcomes in subacute stroke survivors.
Similar content being viewed by others
Introduction
Stroke, a leading cause of long-term disability, produces immense health and economic burdens globally1. Over the last two decades, the relationship between stroke and sleep disorders has been of growing interest to researchers and practitioners2, 3. Sleep disorders are found in up to 78% of stroke survivors4. These sleep disorders, including sleep apnea, insomnia, hypersomnia, and parasomnia, interfere with normal physical, mental, social, and emotional functioning5. Sleep is associated with a patient’s ability to achieve the full potential for stroke recovery6; therefore, untreated sleep disorders may diminish rehabilitation efforts and result in poor functional prognosis.
Evidence suggests that sleep disorders are an independent factor that predicts functional disability and longer hospitalization following stroke7. Specifically, Kaneko et al. found that stroke survivors with sleep apnea had lower functional ability, as measured by the Functional Independence Measure (FIM), than did those without sleep apnea7. A more recent study also suggests that sleep disturbances in stroke survivors can detrimentally affect functional status, such as scores on the modified Barthel Index and grip strength8.
The existing literature describing the relationships between sleep disorders and biomarkers, such as inflammatory and oxidative stress pathways, is extensive9, 10. A meta-analysis of 51 studies found that, compared to control subjects, patients with obstructive sleep apnea (OSA) have higher levels of inflammatory markers such as C-Reactive protein (CRP), tumor necrosis factor alpha (TNF-α), and intercellular adhesion molecule (ICAM)10. These levels are positively correlated to the level of OSA severity10. On the other hand, previous studies have suggested that elevated inflammatory and oxidative markers may be associated with the pathogenesis of cardiovascular and cerebrovascular diseases10,11,12. The underlying mechanisms may involve endothelial dysfunction, systemic inflammation, oxidative stress, and metabolic dysregulation resulting from hypoxemia, reoxygenation, and intrathoracic pressure13. Increased production of pro-inflammatory cytokines has been observed in stroke survivors and may be associated with poor stroke outcomes. Specifically, elevated cytokines, such as interleukin-6, presented in the early stroke period are closely associated with worse stroke outcomes assessed by the Barthel Index and modified Rankin scale14. Another study showed that reduced levels of inflammation are negatively correlated with the Barthel Index, suggesting that the decrease of inflammatory markers was beneficial for stroke recovery15. In addition, our previous work demonstrated that the changes of patients in oxidative stress level, measured by 8-hydroxy-2′-deoxyguanosine, were significantly correlated with the upper limb motor function, muscle power, and perception of pain16. Disturbed sleep is associated with an increased level of inflammation10. There is also evidence suggesting that systemic inflammation results in a disruption of sleep17, 18. Chronic inflammation may reduce sleep continuity and depth and induce sleep disorder, such as insomnia, suggesting a relationship between disturbed sleep and inflammation19. Therefore, examining the associations among level of inflammation, level of oxidative stress, and sleep outcomes is particularly relevant after stroke, given that changes in any one of these factors are proposed to be related to functional prognosis and the effectiveness of rehabilitation treatment post-stroke20, 21.
Recent work by Chen and colleagues22 demonstrated that the severity of OSA is positively associated with the total antioxidant capacity (TAC) and CRP in ischemic stroke subjects. Specifically, the results of the study showed that the levels of CRP and TAC were positively correlated with the oxyhemoglobin desaturation index; the TAC levels were negatively correlated with mean arterial oxygen saturation in stroke survivors with severe OSA22. However, what has been lacking to date is examination of the relationships among the biomarkers, sleep, and functional and health-related outcomes after stroke rehabilitation.
Stroke rehabilitation programs, such as robot-assisted therapy23, 24 and task-oriented training25, 26, may improve functional outcomes in stroke survivors. Research supports the efficacy of upper limb robot-assisted therapy for improving motor and functional outcomes in stroke patients23, 24. Moreover, task-oriented training with stroke survivors improves functional outcomes and overall health-related quality of life25, 26. A better understanding of the changes of the inflammation, oxidative stress, and sleep indicators following stroke rehabilitation is crucial not only for predicting treatment-related outcomes but also for monitoring clinical response and adverse effects.
The pilot study aimed to: (1) characterize the relationships among baseline biomarkers (i.e., inflammation and oxidative stress), sleep, and functional and health-related outcomes at post-treatment in subacute stroke patients, and (2) investigate the changes in levels of biomarkers, sleep, and functional and health-related outcomes after 4 weeks of rehabilitation intervention. We hypothesized that (1) higher levels of inflammation and oxidative stress at baseline would be associated with poor sleep quality and functional and health-related outcomes post-treatment, and (2) the levels of inflammation and oxidative stress would decrease, and the functional and health-related outcomes would improve, after the intervention.
Results
Baseline characteristics
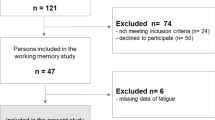
The study enrolled a total of 20 patients with subacute stroke. Table 1 summarizes the patients’ demographic and baseline clinical characteristics. The mean age was 48.17 years, and the ratio of males to females was 1:1. Functional impairment and level of disability, as measured by self-report and performance-based measures, were generally moderate. Individuals with and without sleep disturbances, based on the global Pittsburg Sleep Quality Index (PSQI) score, and the patient characteristics are also included. Among these patients, fifteen were characterized as bad sleepers (i.e., global PSQI score > 5). The ratio of good sleepers to bad sleepers was 1:3.
Association between inflammation, oxidative stress markers, sleep and functional outcomes
The Spearman rank correlation coefficient revealed that the pre-sICAM-1 was positively correlated with the post-number of awakenings (ρ = 0.506, p < 0.05) (Table 2), indicating that patients with higher baseline levels of inflammation had higher numbers of awakenings at post-treatment. However, the pre-sICAM-1 level had no statistically significant correlations with other sleep and functional outcomes (all p’s > 0.05). In addition, there was a significant positive correlation between pre-MDA and post-WMFT-time (ρ = 0.462, p < 0.05), and an almost significant correlation between pre-MDA and post-mRS (ρ = 0.421, p = 0.065). However, pre-GPx was not correlated with any post-sleep or post-functional outcomes (all p’s > 0.05).
Changes in biomarkers, sleep, and functional outcomes from pre-treatment to post-treatment
The pre- and post-treatment levels of biomarkers, sleep outcomes, and functional outcomes are reported in Table 3. For the inflammation and oxidative stress markers, only the MDA demonstrated a significant decrease from pre- to post-treatment (z = 2.54, p < 0.05), indicating lower oxidant levels of the patients after treatment. No significant pre- or post-treatment changes were noted for the PSQI total score, sleep percentage, and number of awakenings (all p’s > 0.05). However, there was a trend of improvement for biomarkers and sleep outcomes from pre-treatment to post-treatment. Furthermore, after treatment, patients improved significantly on most of the functional outcomes. Specifically, significant changes from pre- to post-treatment were found on the mRS (z = 3.45, p = 0.001), WMFT-FAS (z = −3.72, p < 0.001), SIS-total (z = −2.98, p = 0.003), and SIS-physical function (z = −3.38, p = 0.001).
Discussion
This study aimed to characterize the relationships of baseline inflammation and oxidative stress biomarkers to sleep and to functional and health-related outcomes after 4 weeks of rehabilitation intervention in subacute stroke patients. First of all, a positive association was observed between the baseline level of inflammation and the number of awakenings at post-treatment. We also found a positive correlation between the baseline oxidant level and post-treatment upper limb motor function (i.e., the post-WMFT-time). A similar trend was found for baseline oxidant level and post-treatment level of stroke disability (i.e., the post-mRS). In addition, significant effects were decreased oxidant levels and improved motor function and health-related outcomes after the rehabilitation. Although not statistically significant, there was a trend suggesting promising and beneficial rehabilitation intervention effects on the biomarkers and sleep outcomes.
The present study expands on previous findings by showing the association between inflammation markers and objectively measured sleep outcomes in stroke survivors. Elevated levels of inflammation have been found to predict an increased risk for adverse health outcomes such as myocardial infarction and diabetes in healthy populations27, 28. In this study, increases in the baseline level of the inflammatory sICAM-1 were found to be associated with higher numbers of awakenings at post-treatment. This result may support the hypothesis that high inflammation at baseline is related to poor sleep quality via the worsening of comorbidities associated with inflammation. This finding is partly consistent with previous findings that higher levels of inflammation reduce non-rapid-eye-movement sleep due to diminished sleep-promoting effects, and thereby increase wakefulness29. Many studies have found that a sleep deficiency induces systematic inflammation, which may be associated with metabolic changes and immunological deregulation29,30,31. This study, on the other hand, suggests a reciprocal regulation between inflammatory response and sleep loss. Our finding of a link between higher baseline inflammation and poor sleep quality echoes previous work suggests that pro-inflammatory cytokines induce sickness symptoms such as sleepiness, fatigue, and poor cognition32.
Oxidative stress has been found to play an important role in acute ischemic stroke pathogenesis. Ozkul et al. investigated serum MDA levels in patients with acute stroke and found a negative correlation with clinical outcomes measured by the Canadian Neurological Scale33. Although elevated oxidative stress has been reported in stroke patients, few studies have described the correlation between this biomarker and functional outcomes after stroke rehabilitation. Hsieh et al. demonstrated that urinary 8-hydroxy-2′-deoxyguanosine (8-OHdG), a biomarker of oxidative DNA damage, is a valid predictor of functional outcomes in chronic stroke survivors. Specifically, they showed that the baseline 8-OHdG content was significantly correlated with post-treatment functional outcomes such as the Fugl-Meyer Assessment (FMA) and Medical Research Council scale (MRC)16. In the present study, we measured the MDA level as a marker of oxidative stress and observed that baseline serum MDA levels were significantly correlated with post-treatment motor function measured by the WMFT-time. This relationship may not be attributed to the influence of treatment because WMFT-time scores did not change significantly from pre- to post-treatment. A higher level of baseline MDA content was significantly associated with less favorable functional outcomes (i.e., WMFT-time) after rehabilitation. This finding indicated that the oxidative stress markers might be valid in predicting functional outcomes in stroke survivors. Our results also found a similar trend for baseline oxidant level and post-treatment level of disability measured by the mRS. However, we did not find any significant correlations between antioxidants (i.e., GPx) and post-treatment functional outcomes.
Another key finding is that the 4 weeks of rehabilitation produced significant effects on oxidative stress levels, indicating that the intervention, did not produce oxidative stress in the stroke survivors, but rather alleviated it. In addition, some of the functional and health-related outcomes were significantly improved after the intervention as compared with the baseline. Rehabilitative intervention such as robot-assisted therapy is considered to be an intervention approach for restoring upper limb function and has been examined in large clinical trials in stroke patients34, 35. Regular rehabilitative exercise including functional and movement training has been shown to promote upregulation of antioxidant capacity and attenuate oxidative stress in stroke patients36. Although our results showed a trend of increasing antioxidant capacity, the significantly decreased oxidative stress may suggest the possibility of the reduced occurrence of oxidative damage, which may partially contribute to improving functional and health-related outcomes.
Sleep disorders, which are common in stroke patients, are associated with poorer stroke recovery outcomes and increased cerebrovascular morbidity. It has been shown that, in stroke survivors, better sleep quality is associated with better functional status8 and cognitive recovery37. Although not designed for this purpose, we noted that stroke survivors with good sleep quality at baseline had lower oxidant levels and greater health-related outcomes (i.e., the SIS total score) after the treatment. All participants had improved motor function and physical function, but only the bad sleepers showed decreased level of disability following the treatment. Individuals with sleep disturbance had lower level of disability at baseline and may have more room for improvement. Of note, a limitation of the study was the relatively small sample size, especially the good sleepers. Follow-up study using a larger sample for study is needed to validate the findings. In addition, we used a subjective measure, the PSQI, to quantify sleep quality and differentiated between good and bad sleepers. Further research employing an objective sleep monitoring system, such as actigraphy, to record the quality, quantity, and rhythm of sleep may provide better understanding of the relationship of sleep outcomes to functional recovery after stroke. Also, future work could include other potential inflammatory and oxidative stress biomarkers, such as CRP or TAC, to establish a strong correlation between biomarkers of oxidative stress and functional outcomes. Finally, future work could also examine different rehabilitative approaches that activate the biochemical regulations most likely to promote neuronal plasticity and recovery following stroke.
Methods
Participants
Patients with subacute stroke were recruited if they met the following criteria: (1) unilateral stroke with onset of less than 6 months; (2) upper extremity score of the Fugl-Meyer Assessment (FMA) > 10; (3) no excessive spasticity in any joints of the affected arm (modified Ashworth Scale ≤ 3); (4) ability to follow study instructions and perform tasks (Mini-Mental State Examination Score ≥ 22); (5) no other neurologic or major orthopedic diseases. All participants signed informed consent forms approved by the institutional review boards. Ethical approval was provided by the Institutional Review Board for Human Studies of Chang Gung Memorial Hospital. All methods were performed in accordance with the relevant guidelines and regulations.
Stroke rehabilitation intervention
The stroke rehabilitation programs included robot-assisted therapy and task-oriented approach training. All patients received one 90-minute therapy session each day, 5 days per week, for 4 weeks. The training sessions were delivered by certified occupational therapists, and the daily logs of therapeutic activities were recorded.
Robot-assisted therapy was carried out using the Bi-Manu-Track device (Reha-Stim Co, Berlin, Germany) for bilateral arm symmetrical training24, 38 . This robotic device delivers forearm pronation/supination and wrist flexion/extension movements. Each movement pattern has 3 modes: (1) passive-passive, (2) active-passive, and (3) active-active. In the passive-passive mode, the robot controls movements of both arms. In the active-passive mode, either the affected or non-affected arm moves the robot handle while the other robot handle moves the non-affected or affected arm at the same movement speed and amplitude. In the active-active mode, both arms perform movements by overcoming an initial isometric resistance. The movement speed, amount of resistance, and range of movement can be adjusted individually. In this study, each participant performed an average of 1,200 to 1,600 movement repetitions for each session.
During the task-oriented training, participants practiced functional activities in accordance with their functional levels and individual goals. Examples of the functional activities are as follows: (1) walk to a water fountain, fill a glass with water, and drink; (2) wring out a wet rag and wipe a Table 3 fold towels and walk to a drawer and put the towels in it. The therapist used task-oriented approach principles and promoted motor function by increasing the complexity of the task over time. The therapist encouraged the patient to use the affected arm to execute tasks and then provided feedback to facilitate performance during training.
Outcome measures
All measurements were obtained at baseline (pre-treatment) and 4 weeks after treatment (post-treatment).
Sleep outcomes
Self-reported sleep disturbances were evaluated using the Pittsburgh Sleep Quality Index (PSQI), which includes 7 components: subjective sleep quality, latency, duration, efficiency, sleep disturbances, use of sleep medication, and daytime somnolence. The component scores are summed to produce a global score (ranging from 0 to 21). Higher scores indicate worse sleep quality. A global PSQI score greater than 5 indicates poor sleepers39. In addition to the subjective measurement of sleep outcome, each participant wore an accelerometer to detect and record arm movements. The data collected from the accelerometers were used as a proxy for assessment of sleep and wake. The sleep percentage (total sleep time divided by total wearing time) and the total number of awakenings for 3 consecutive days at baseline and 4 weeks post-treatment (3 valid days were required for analysis) were collected using the MicroMini-Motionlogger activity monitor (Ambulatory Monitoring, New York, NY, USA). The accelerometer was set to record hand movement in 60-sec epochs using the Proportional Integrating Mode (PIM) that captures the amplitude of arm movement. The data were further configured using ACT-Millennium software (AMI) and processed in Action4 software. The accelerometer was worn on the non-affected wrist. The accelerometer was removed during the 90-minute therapy session; therefore, the average total wearing time for each participant was approximately 22 to 22.5 hours per day. While wearing the accelerometer, participants completed daily monitoring logs to document the activities they performed (e.g., walking, watching TV, reading…etc.), the time they went to bed, and periods when the accelerometer was removed. The accelerometer daily monitoring log was used to identify any discrepancy between self-reported functional use of the non-affected arm and the accelerometric data.
Inflammation and oxidative stress biomarkers
We evaluated the level of the soluble intercellular adhesion molecule-1 (sICAM-1) as the pro-inflammatory marker. Elevated concentrations of sICAM-1 are associated with local or systemic inflammation40. The evaluated oxidative stress indicators included glutathione peroxidase (GPx) and malondialdehyde (MDA). GPx is an antioxidant enzyme that can be used to evaluate the body’s defense mechanism against oxidative stress indicators. MDA is a biomarker for tissue damage and reperfusion41. A higher level of GPx indicates a higher antioxidant level, whereas a higher level of MDA reflects a higher oxidant level. Standard operating procedures consistent with expert consensus recommendations were used to collect, process, and store blood samples42. We performed the measurements using colorimetric enzyme-linked immunosorbent assay (ELISA) following the manufacturer’s instructions for sICAM-1 (BioLegend, San Diego, CA, USA) and MDA (MDA-586, OxisResearch, Portland, OR, USA). Level of GPx was evaluated using the Glutathione Peroxidase assay kit (Cayman Chemical Company, Ann Arbor, MI, USA). The units of measurement are expressed as ng/ml for sICAM-1, nmol/min/ml for GPx, and μM for MDA.
Functional and health-related outcomes
We used the modified Rankin Scale (mRS), Wolf Motor Function Test (WMFT), and Stroke Impact Scale (SIS) to evaluate level of stroke disability, motor function, and health outcomes, respectively. Level of stroke disability related to activity performance and participation was assessed using the mRS. The mRS is a six-point ordinal scale commonly used in stroke patients43. The mRS score ranges from 0 to 5, with 0 representing “no symptoms” and 5 representing “severe disability.” A change of one or more points indicates meaningful improvement or decline in functional independence over time44. Its validity and inter-rater reliability are well documented45. The WMFT is a laboratory-based measurement for assessing upper-extremity motor function46. The WMFT comprises 17 tasks with 15 function-based and 2 strength-based tasks. We calculated two different scores: the WMFT-time (time required to complete the tasks, with a maximum of 120 s) and WMFT-FAS (Functional Ability Scale, which is used to assess the functional capacity of each task; 0 indicates that the patient cannot attempt the task, and 5 indicates that the movement appears normal). The psychometric properties of the WMFT have been validated in patients with stroke47, 48. The SIS is a stroke-specific, self-report, and health status measure that assesses multidimensional stroke outcomes49. Items are scored on a Likert-type scale of 1 (inability to complete the item) to 5 (no difficulty at all). We collected data from all domains and generated a global score (SIS-total). The subdomains in physical function (SIS-physical function) and ratings on overall stroke recovery (SIS-stroke recovery) were also reported.
Statistical analysis
We used the Spearman rank correlation coefficient to quantify the correlations among the pre-treatment biomarkers (e.g., inflammation and oxidative stress), sleep, and functional and health-related outcomes at post-treatment. We also performed a Wilcoxon signed rank test to examine the statistical significance of changes from pre-treatment to post-treatment in all variables. Statistical analysis was performed in SPSS version 19.0. The level of significance was set at p < 0.05.
References
Mozaffarian, D. et al. Heart disease and stroke statistics–2015 Update: a report from the American Heart Association. Circulation 131, doi:10.1161/CIR.0000000000000152 (2015).
Ferre, A. et al. Strokes and their relationship with sleep and sleep disorders. Neurol (English Ed) 28, 103–118 (2013).
Ramar, K. & Surani, S. The relationship between sleep disorders and stroke. Postgrad Med 122, 145–153, doi:10.3810/pgm.2010.11.2232 (2010).
Pasic, Z., Smajlovic, D., Dostovic, Z., Kojic, B. & Selmanovic, S. Incidence and types of sleep disorders in patients with stroke. Med Arh 65, 225–227 (2011).
Colten, H., Altevogt, B. B. M. & Colten., H. Sleep disorders and sleep deprivation: an unmet public health problem. Committee on Sleep Medicine and Research, doi:10.1097/01.CHI.0000270812.55636.3b (2006).
Baglioni, C. et al. Polysomnographic characteristics of sleep in stroke: a Systematic review and meta- analysis. PLoS One 11, 1–23, doi:10.1371/journal.pone.0148496 (2016).
Kaneko, Y., Hajek, V. E., Zivanovic, V., Raboud, J. & Bradley, T. D. Relationship of sleep apnea to functional capacity and length of hospitalization following stroke. Sleep 26, 293–297 (2003).
Kim, S. A., Kim, J., Kim, Y., Yang, K. I. & Kim, D.-E. The relationship between sleep disturbance and functional status in mild stroke patients. Ann Rehabil Med Orig Artic Ann Rehabil Med 39, 545–552, doi:10.5535/arm.2015.39.4.545 (2015).
Mancuso, M. et al. Oxidative stress biomarkers in patients with untreated obstructive sleep apnea syndrome. Sleep Med 13, 632–636, doi:10.1016/j.sleep.2011.10.030 (2012).
Nadeem, R. et al. Serum inflammatory markers in obstructive sleep apnea: a meta-analysis. J Clin Sleep Med 9, 1003–1012, doi:10.5664/jcsm.3070 (2013).
Calvin, A. D., Albuquerque, F. N., Lopez-Jimenez, F. & Somers, V. K. Obstructive sleep apnea, inflammation, and the metabolic syndrome. Metab Syndr Relat Disord 7, 271–278, doi:10.1089/met.2008.0093 (2009).
Jean-Louis, G. et al. Obstructive sleep apnea and cardiovascular disease: evidence and underlying mechanisms. Minerva Pneumol 48, 277–293 (2009).
Somers, V. K. et al. Sleep apnea and cardiovascular disease: an American Heart Association/American College of Cardiology Foundation scientific statement. Circulation 118, 1080–1111, doi:10.1161/CIRCULATIONAHA.107.189420 (2008).
Basic Kes, V., Simundic, A. M., Nikolac, N., Topic, E. & Demarin, V. Pro-inflammatory and anti-inflammatory cytokines in acute ischemic stroke and their relation to early neurological deficit and stroke outcome. Clin. Biochem. 41, 1330–1334 (2008).
Manolescu, B. N. et al. Dynamics of inflammatory markers in post-acute stroke patients undergoing rehabilitation. Inflammation 34, 551–558 (2011).
Hsieh, Y. W. et al. The reliability and predictive ability of a biomarker of oxidative DNA damage on functional outcomes after stroke rehabilitation. Int. J Mol Sci 15, 6504–6516, doi:10.3390/ijms15046504 (2014).
Mills, P. J. et al. Inflammation and Sleep in Healthy Individuals. Sleep 30, 729–735 (2007).
Motivala, S. J., Sarfatti, A., Olmos, L. & Irwin, M. R. Inflammatory markers and sleep disturbance in major depression. Psychosom Med 67, 187–194 (2005).
Raison, C. L. et al. Chronic interferon-alpha administration disrupts sleep continuity and depth in patients with hepatitis C: association with fatigue, motor slowing, and increased evening cortisol. Biol. Psychiatry 68, 942–949 (2010).
Whiteley, W. et al. Inflammatory markers and poor outcome after stroke: a prospective cohort study and systematic review of interleukin-6. PLoS Med 6, e1000145, doi:10.1371/journal.pmed.1000145 (2009).
Whiteley, W., Chong, W. L., Sengupta, A. & Sandercock, P. Blood markers for the prognosis of ischemic stroke: a systematic review. Stroke 40, e380–e389, doi:10.1161/STROKEAHA.108.528752 (2009).
Chen, C. Y. et al. Association of inflammation and oxidative stress with obstructive sleep apnea in ischemic stroke patients. Sleep Med 16, 113–118, doi:10.1016/j.sleep.2014.07.027 (2015).
Kwakkel, G., Kollen, B. J. & Krebs, H. I. Effects of robot-assisted therapy on upper limb recovery after stroke: a systematic review. Neurorehabil. Neural Repair 22, 111–21 (2008).
Hsieh, Y. W. et al. Dose-response relationship of robot-assisted stroke motor rehabilitation: the impact of initial motor status. Stroke 43, 2729–2734 (2012).
Rensink, M., Schuurmans, M., Lindeman, E. & Hafsteinsdóttir, T. Task-oriented training in rehabilitation after stroke. J. Adv. Nurs. 65, 737–754 (2009).
Lin, K., Chen, Y., Chen, C., Wu, C. & Chang, Y. The effects of bilateral arm training on motor control and functional performance in chronic stroke: a randomized controlled study. Neurorehabil. Neural Repair 24, 42–51 (2010).
Ridker, P. M., Rifai, N., Stampfer, M. J. & Hennekens, C. H. Plasma concentration of interleukin-6 and the risk of future myocardial infarction among apparently healthy men. Circulation 101, 1767–1772 (2000).
Pradhan, A. D., Manson, J. E., Rifai, N., Buring, J. E. & Ridker, P. M. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. Jama 286, 327–334 (2001).
Miller, M. & Cappuccio, F. P. Biomarkers of cardiovascular risk in sleep-deprived people. J Hum Hypertens 27, 583–5888, doi:10.1038/jhh.2013.27 (2013).
Dowd, J. B., Goldman, N. & Weinstein, M. Sleep duration, sleep quality, and biomarkers of inflammation in a Taiwanese population. Ann Epidemiol 21, 799–806, doi:10.1016/j.annepidem.2011.07.004 (2011).
Hurtado-Alvarado, G. et al. Sleep loss as a factor to induce cellular and molecular inflammatory variations. Clin Dev Immunol 2013, doi:10.1155/2013/801341 (2013).
Dantzer, R. Cytokine-induced sickness behavior: where do we stand? Brain Behav Immun 15, 7–24 (2001).
Ozkul, A. et al. Oxidative stress in acute ischemic stroke. J Clin Neurosci 14, 1062–1066 (2007).
Cho, K. H. & Song, W. Robot-assisted reach training for improving upper extremity function of chronic stroke. Tohoku J Exp Med 149–155, doi:10.1620/tjem.237.149 (2015).
Prange, G. B., Jannink, M. J. A., Groothuis-Oudshoorn, C. G. M., Hermens, H. J. & Ijzerman, M. J. Systematic review of the effect of robot-aided therapy on recovery of the hemiparetic arm after stroke. J Rehabil Res Dev 43, 171–184 (2006).
Kihoin, N., Tanaka, K., Okuno, M. & Okamoto, T. Exercise attenuates oxidative stress in patients with stroke. Neurol Asia 21, 7–16 (2016).
Duss, S. B. et al. The role of sleep in recovery following ischemic stroke: a review of human and animal data. Neurobiol Sleep Circadian Rhythm 0–1, doi:10.1016/j.nbscr.2016.11.003 (2016).
Hesse, S., Schmidt, H., Werner, C. & Bardeleben, A. Upper and lower extremity robotic devices for rehabilitation and for studying motor control. Curr Opin Neurol 16, 705–10 (2003).
Buysse, D. J., Reynolds, C. F., Monk, T. H., Berman, S. R. & Kupfer, D. J. The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res 28, 193–213 (1989).
deLemos, J. A., Hennekens, C. H. & Ridker, P. M. Plasma concentration of soluble vascular cell adhesion molecule-1 and subsequent cardiovascular risk. J Am Coll Cardiol 36, 423–426 (2000).
Pham-Huy, L. A., He, H. & Pham-Huy, C. Free radicals, antioxidants in disease and health. Int J Biomed Sci 4, 89–96 (2008).
Tuck, M. K. et al. Standard operating procedures for serum and plasma collection: early detection research network consensus statement. J Proteome Res 8, 113–117, doi:10.1021/pr800545q (2009).
van Swieten, J. C., Koudstaal, P. J., Visser, M. C., Schouten, H. J. & vanGijn, J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke 19, 604–607 (1988).
Weimar, C. et al. Assessment of functioning and disability after ischemic stroke. Stroke 33, 2053–2059 (2002).
Meyer, B. C., Hemmen, T. M., Jackson, C. M. & Lyden, P. D. Modified National Institutes of Health Stroke Scale for use in stroke clinical trials: prospective reliability and validity. Stroke 33, 1261–1266 (2002).
Wolf, S. L., Lecraw, D. E., Barton, L. A. & Jann, B. B. Forced use of hemiplegic upper extremities to reverse the effect of learned nonuse among chronic stroke and head-injured patients. Exp Neurol 104, 125–132 (1989).
Morris, D. M., Uswatte, G., Crago, J. E., Cook, E. W. & Taub, E. The reliability of the wolf motor function test for assessing upper extremity function after stroke. Arch Phys Med Rehabil 82, 750–755 (2001).
Whitall, J., Savin, D. N., Harris-Love, M. & Waller, S. M. Psychometric properties of a modified wolf motor function test for people with mild and moderate upper-extremity hemiparesis. Arch Phys Med Rehabil 87, 656–660 (2006).
Duncan, P. W., Bode, R. K., Min Lai, S. & Perera, S. Rasch analysis of a new stroke-specific outcome scale: the stroke impact scale. Arch Phys Med Rehabi 84, 950–963 (2003).
Acknowledgements
This study was partly supported by the National Health Research Institutes (NHRI-EX106-10403PI), the Healthy Ageing Research Center at Chang Gung University (EMRPD1F0321, CRRPD1B0013), and Chang Gung Memorial Hospital (BMRP553 and BMRPD25) in Taiwan.
Author information
Authors and Affiliations
Contributions
T.T.Y., Y.W.H., K.C.L., and C.Y.W. contributed to study design, grant and project management, and manuscript revisions. T.T.Y. and Y.W.H. analyzed the data, interpreted the data, and drafted the manuscript. Y.W.H. was involved in data collection. C.L.C. provided medical consultation. J.S.W. was involved in biomarker analysis and provided consultation. All authors have given final approval of the version to be published.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yeh, Tt., Hsieh, Yw., Wu, Cy. et al. A Preliminary Investigation of the Association of Sleep With Inflammation and Oxidative Stress Biomarkers and Functional Outcomes After Stroke Rehabilitation. Sci Rep 7, 8634 (2017). https://doi.org/10.1038/s41598-017-08931-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08931-w
This article is cited by
-
Plasma glutathione as a risk marker for the severity and functional outcome of acute atherothrombotic and cardioembolic stroke
The Egyptian Journal of Neurology, Psychiatry and Neurosurgery (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.