Abstract
Two dimensional (2D) molecular crystals have attracted considerable attention because of their promising potential in electrical device applications, such as high-performance field-effect transistors (FETs). However, such devices demand high voltages, thereby considerably increasing power consumption. This study demonstrates the fabrication of organic FETs based on 2D crystalline films as semiconducting channels. The application of high-κ oxide dielectrics allows the transistors run under a low operating voltage (−4 V). The devices exhibited a high electrical performance with a carrier mobility up to 9.8 cm2 V−1 s−1. Further results show that the AlOx layer is beneficial to the charge transport at the conducting channels of FETs. Thus, the device strategy presented in this work is favorable for 2D molecular crystal-based transistors that can operate under low voltages.
Similar content being viewed by others
Introduction
Two-dimensional (2D) molecular crystals assembled through weak van der Waals forces are a promising class of materials for molecular packing and charge transport and exhibit significant potential for electronic applications1,2,3,4,5,6,7. Efforts have been devoted to the fabrications of organic field-effect transistors (OFETs) and p-n junctions that use ultrathin crystalline films grown on 2D atomic crystals, such as graphene, boron nitride, and MoS2. Recently, deposition of 2D molecular crystals on amorphous silicon oxide (SiO2) has been performed successfully through solution-based processes, which is compatible with current semiconductor manufacturing8,9,10,11,12,13,14,15,16,17. These OFETs also yielded high carrier mobility that are comparable to polycrystalline silicon. However, a high voltage (i.e., normally over 30 V) is necessary to operate such devices, which results in extra power consumption, because of the low dielectric constant of SiO2. Therefore, lowering the operating voltage is greatly important, particularly for portable and wearable electronics. An effective approach is to employ high-κ materials for the dielectric layers in transistor architectures, such that a low voltage can generate an adequate charge density in the conducting channel18,19,20,21. Therefore, producing low-operating-voltage and high-performance OFETs based on 2D molecular crystals using high-κ oxides as the dielectric layer, is noteworthy.
Herein, we demonstrate a low-voltage bottom-gate top-contact (BGTC) OFET that utilizes 2D molecular crystals as the conducting channel and AlOx as the dielectric. The molecular crystals were solution-processed through a floating-coffee-ring-driven assembly according to the methods presented in our previous work17. Our devices can operate at a low gate bias of −4 V and exhibit a high carrier mobility (μ FET) up to 9.8 cm2 V−1 s−1, a large on/off ratio of 105, and a small subthreshold swing of 160 mV dec−1. The results demonstrate that the proposed strategy has significant potential in fabricating low-voltage and high-performance OFETs employing high-κ dielectrics and 2D molecular crystals.
Results
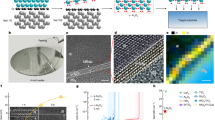
The p-type small-molecule semiconductor of dioctylbenzothienobenzothiophene (C8-BTBT) exhibits a considerably high carrier mobility and thus has been used to fabricate the OFETs22,23,24. The highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) of C8-BTBT are −5.39 eV and −1.55 eV, respectively25. Figure 1a illustrates the BGTC structure of the transistor device that adopts thermally deposited AlOx with a thickness of ~18 nm as the dielectric layer. Figure 1b shows the atomic force microscopy (AFM) image of the AlOx dielectric, which illustrates a particularly smooth surface with a root-mean-square (RMS) roughness of 2.23 Å. The hydrophilicity of the AlOx dielectric layer is enhanced through UV–ozone treatment. Improving the hydrophilicity is conducive to the subsequent solution-based process for molecular crystal growth. Besides, the surface roughness of UV-ozone-treated AlOx is 2.75 Å (Supplementary Fig. S1). Given that the capacitance and gate leakage current are both critical to the gate dielectric in low-voltage OFETs, we employ an Au/AlOx/Si capacitor structure. The measured capacitance and dielectric constant are 0.37 μF/cm2 and ~9.0, respectively (measured at the voltage frequency of 10 Hz, Fig. 1c). Moreover, the AlOx capacitance decreases from 0.37 μF/cm2 to 0.25 μF/cm2 when the frequency of the applied voltage increases from 10 Hz to 1 MHz. This effect is mainly due to the interfacial traps produced during the UV–ozone treatment. The dielectric capacitance of AlOx exhibits negligible change when the applied voltage increases from −6 V to 6 V (Supplementary Fig. S2), and its leakage current is 10−7 A/cm2 when the applied voltage is −4 V (Fig. 1d). Therefore, the thermally deposited AlOx can be used as a gate insulating layer because of its superior performance as a dielectric material. The coffee-ring-driven method (Supplementary Fig. S3) is utilized for the deposition of 2D films where C8-BTBT molecules assemble into 2D crystalline films with a large size of ~200 μm on the AlOx surface (Fig. 2a)17. Supplementary Fig. S4 shows a deposited bilayer C8-BTBT film with a large size of several millimeters. The obtained molecular films consist of different C8-BTBT layers with step-and-terrace structures. Figure 2b and c show the AFM images of the two steps as marked by dotted squares in Fig. 2a, where the thicknesses are 2.99 and 5.21 nm, respectively. The molecules in the third layer are nearly perpendicular to the substrates with regard to the C8-BTBT molecular length. The previous results show that C8-BTBT molecules in first layer are more tilted to the substrate than that in upper layers, because the weak van der Waals interactions among the small molecules decrease rapidly from the dielectric surface to the upper molecular layers6, 26. The schematic illustration of C8-BTBT molecular packing is shown in Supplementary Fig. S5. Furthermore, the bilayer C8-BTBT exhibits uniform thin films with atomic smoothness (RMS roughness: 1.22 Å, Fig. 2d). The crystalline properties of the bilayer C8-BTBT are characterized through high-resolution AFM (Fig. 2e). More than 10 points are selected randomly for scanning by high-resolution AFM (Supplementary Fig. S6). The AFM images show nearly identical lattice constants, namely, a = 6.21 ± 0.16 Å, b = 8.12 ± 0.12 Å, and θ = 88.1 ± 1.4°. These results indicate that the bilayer C8-BTBT films each contain a crystalline phase with highly morphologic uniformity over a large area. Similar crystalline characteristics are also observed in the C8-BTBT trilayer (Supplementary Fig. S7). The crystalline properties of our molecular crystals are summarized in Supplementary Table S1. Therefore, the AlOx layer formed through thermal evaporation facilitates the deposition of high-quality 2D C8-BTBT crystalline films.
(a) Optical microscopy image of the 2D C8-BTBT crystalline films. The bilayer and trilayer are clearly observed. Scale bar, 20 μm. (b) AFM morphology image of the step between bilayer and trilayer films (red square in (a)). Scale bar, 500 nm. (c) AFM morphology image of the step between bilayer films and the substrate (blue square in (a)). Scale bar, 500 nm. (d) AFM morphology image of uniform bilayer films (brown square in (a)). Scale bar, 500 nm. (e) High-resolution AFM image of the bilayer C8-BTBT molecules on the AlOx/Si substrate. Scale bar, 1 nm. The inset shows the corresponding fast Fourier transforms of the AFM image with lattice indices.
Discussion
The bilayer crystalline films and AlOx are used as conducting channels and gate dielectrics, respectively, to fabricate the planar transistors. Figure 3a and b show the typical transfer and output characteristics of a bilayer C8-BTBT-based FET, respectively. These properties indicate that a low operating voltage (−4 V) is sufficient to operate the device properly with an evident field effect. The drain current in the output curves reaches the saturation region even at a drain voltage of −4 V. A nearly linear increase in the drain current is also observed in the small range of the drain voltage, indicating a nearly ohmic contact with an efficient charge injection from the metal contact to the conducting channel (inset of Fig. 3b). Moreover, the device exhibits a high electrical performance and yields a carrier mobility of up to 6.5 cm2 V−1 s−1, near-zero threshold voltage of −0.7 V, small subthreshold swing of 160 mV dec−1, and large on/off ratio of >105. The carrier mobility value was calculated in the saturation region using the equation:
where W and L are the channel width and length, respectively, C i is the gate dielectric capacitance, V T is the threshold voltage. Besides, 2D crystalline films with only several layers can greatly enhance the charge injection process by significantly decreasing the access resistance related to the charge injection from the metal/semiconductor interface to the active channel27, 28. And the width-normalized contact resistance in our device is estimated to be ~360 Ω cm by using the Y-function method; the value is among the lowest ones for the contact resistance of organic transistors (Supplementary Figs S8 and S9). For comparison, we also fabricated BGTC FETs based on a five-molecular-layer C8-BTBT crystal. The threshold voltage and mobility are −2.7 V and 0.98 cm2 V−1 s−1, respectively (Supplementary Fig. S10). Besides, the width-normalized contact resistance is 7600 Ω cm, which is much larger than that in FETs based on bilayer crystal (Supplementary Fig. S11). We observed a negligible hysteresis from the transfer curve as the applied gate voltage sweeps backwards. We also prepared 20 devices with AlOx dielectrics, obtaining an average mobility of 4.7 ± 1.9 cm2 V−1 s−1 (Fig. 3c). Furthermore, the highest mobility obtained is 9.8 cm2 V−1 s−1 (Supplementary Fig. S12). To the best of our knowledge, our device exhibits a record-high value of the carrier mobility for low-voltage OFETs (Supplementary Table S2). Typical OFETs based on C8-BTBT reported in literature are summarized in Supplementary Table S3.
(a) Transfer characteristics at a drain voltage of −4 V. The inset shows the optical microscope image of the measured device. Scale bar, 20 μm. (b) Output characteristics at different gate voltages. Inset shows a linear increase in the drain currents in a small range of the drain voltage, indicating a nearly ideal ohmic contact in our devices. (c) Mobility distribution from 20 devices.
Apart from low-voltage operation and high performance, suitable bias-stress stability is also significant for practical applications. Figure 4a shows the bias-stress characteristics of a bilayer C8-BTBT-based FET. Even when tested in the ambient condition, the drain current of the device shows nearly negligible change under a prolonged operation of 104 s. Apart from the high-quality bilayer C8-BTBT crystalline films, low-voltage operation generates limited heat during the electrical measurements, hence also contributes to the stability of our transistor device. Figure 4b shows that the shapes of the transfer curves before and after the bias-stress test exhibit a small shift to a more negative gate voltage. There is a negligible change in the threshold voltage during the test (~0.1 V) (Supplementary Fig. S13). The estimated carrier mobility slightly decreases from 6.3 cm2 V−1 s−1 to 5.7 cm2 V−1 s−1. We also evaluated the stability by testing a device maintained in ambient condition for up to 30 days. Figure 4c shows that both the drain current and carrier mobility only decrease slightly.
Consequently, the presented results prove the promising features of thermally evaporated AlOx as a gate dielectric for low-voltage OFETs with bilayer molecular crystals as conducting channels. For comparison, we prepared the FET samples that utilized SiO2 and HfO2 (Fig. 5). A large operating voltage of −20 V is necessary to operate the SiO2-based device, and the estimated carrier mobility is 4.8 ± 2.1 cm2 V−1 s−1. The operating voltage can be properly lowered to −4 V, whereas the carrier mobility can be as low as 0.4 ± 0.3 cm2 V−1 s−1 when applying HfO2 as the gate insulator. The decreased mobility in the device with HfO2 is mainly due to the strong interaction at the interface between the conducting channel and high-κ dielectric29, 30. This interaction results in the increased localization of the charge carriers6, 31, 32, which is consistent with the result that OFETs based SiO2 exhibit a higher carrier mobility than that based on AlOx. Further studies on the charge carrier properties in our ultrathin molecular crystals is of great interest. Thus, to develop a technique that allows for the fabrication of 2D-crystalline-film-based transistors that employ vacuum as the dielectric layer is necessary29.
Electrical characteristics of the C8-BTBT-based OFETs on SiO2/Si and HfO2/Si. (a) Transfer characteristics at a drain voltage of −20 V. (b) Output characteristics at different gate voltages of a device using SiO2. (c) Transfer characteristics at a drain voltage of −4 V. (d) Output characteristics at different gate voltages of a device using HfO2.
Note that AlOx also has a high dielectric constant, whereas the obtained carrier mobility is similar to that of the device with SiO2, and much higher than those of the HfO2-based devices. The maximum density of interfacial traps (N trap) is estimated from the values of the subthreshold swing (SS) to examine the performance, especially the carrier mobility, exhibited in the device that uses AlOx:
where q is the electronic charge, SS is the subthreshold swing, e is Euler’s number, k is the Boltzmann’s constant, T is the absolute temperature, and C i is the gate dielectric capacitance. The trap density of AlOx is ~3.9 × 1012 cm−2, which is in the same range as those of SiO2 and HfO2. The intrinsic charge transport behavior is determined by performing the temperature-dependent measurement on the electrical performance of the FETs with different oxide dielectrics (Fig. 6a). The carrier mobilities calculated from the transfer curves can all be fitted to linear lines in the plots of ln(μ FET) versus 1/T (Fig. 6b), which indicates that the hopping transport dominates in all devices. Furthermore, the activation energy (E a) can be estimated through the Arrhenius equation:
where μ 0 is the trap-free mobility. Temperature-dependent measurements for the devices with different dielectric layers are summarized in Table 1. The device that utilizes AlOx exhibits the lowest E a value of 30.8 meV and a high μ 0 of 12 cm2 V−1 s−1. And E a is considered to be related to the width of the distribution of trap states33,34,35. Therefore, the high carrier mobility obtained in the AlOx-based device is attributed to a low energetic disorder, a narrow width for the density of trap states in the dielectric interface, and a close packing among the C8-BTBT molecules36,37,38. The results reveal that AlOx can provide a beneficial interface for the transport of charge carriers. Besides, despite a good structural quality of our bilayer C8-BTBT crystals and high electrical performance obtained from the AlOx-based transistors, the charge transport exhibits as a hopping-like rather than a band-like behavior. Similar results were also reported in literature, which imply that the property of the dielectric layer can affect the charge transport in the conducting channel29, 38,39,40,41. Besides, our recent results also reveal that the charge transport behavior can be greatly influenced by the contact resistance42,43,44.
In conclusion, we fabricated low-voltage and high-performance OFETs that employ solution-processed bilayer molecular crystals and high-κ material of AlOx as the conducting channels and the gate dielectrics, respectively. The devices can operate under a low applied voltage of −4 V and exhibit excellent electrical performance with the highest carrier mobility of up to 9.8 cm2 V−1 s−1. Moreover, further studies indicated that the AlOx application in FET devices is favorable to the interfaces among the 2D molecular crystals, in which the charge carrier transport has small activation energy. The results demonstrated the advantages of the proposed strategy to attain low-voltage and high-performance OFETs.
Methods
Fabrication of the AlOx layer: The Si substrate was sequentially cleaned by sonication in acetone and isopropanol for 10 min each. The oxide dielectric of AlOx with a thickness of ~18 nm was thermally evaporated under a deposition speed of 0.1 Å s−1 with a base pressure of 10−5 Torr. The AlOx was then treated by UV-ozone for 15 min.
Deposition of the 2D C8-BTBT Crystals: The p-type organic semiconductor C8-BTBT was supplied by Nippon Kayaku Co. and was adopted without further purification. C8-BTBT (1.0 wt%) was dissolved in a mixture of anisole and p-anisaldehyde (0.5 wt%) which were the good solvent and the antisolvent, respectively. The UV-ozone-treated AlOx was sequentially cleaned in acetone and isopropanol. Before the droplet was casted onto the AlOx substrate, the solution was shaken for ~30 s to deposit from a homogeneous solution. A mechanical pump was then employed to vent the air through a pipe positioned ~1 mm from the droplet (Supplementary Fig. S3).
Characterizations of the C8-BTBT Crystals: An Olympus BX51 was used to obtain the optical microscopy images. Two AFM types were performed in this work. The characterizations were performed on a Veeco Multimode 8 under the ambient conditions for the regular AFM. The experiments were then performed on an Asylum Cypher under ambient conditions utilizing Asylum ARROW UHF AFM tips for the high-resolution AFM.
Fabrication and Electrical Measurements of FETs: Few-layered C8-BTBT was deposited onto the AlOx substrates for the OFET fabrication, as shown in Fig. 1. Patterned Au films with a thickness of 100 nm and Au pads with dimensions of 30 μm × 100 μm were thermally evaporated under a deposition speed of 0.2 Å s−1. The two Au pads were subsequently transferred to the top of the C8-BTBT crystal to form the source and drain electrodes (Supplementary Fig. S14). Electrical measurements were performed utilizing an Agilent B1500 semiconductor parameter analyzer in a closed-cycle cryogenic probe station with a base pressure of 10−5 Torr.
References
Cai, S.-L. et al. The Organic Flatland-Recent Advances in Synthetic 2D Organic Layers. Adv. Mater. 27, 5762–5770 (2015).
Huang, J., Sun, J. & Katz, H. E. Monolayer‐Dimensional 5,5′‐Bis(4‐hexylphenyl)‐2,2′‐bithiophene Transistors and Chemically Responsive Heterostructures. Adv. Mater. 20, 2567–2572 (2008).
Wu, B. et al. Precise, Self-Limited Epitaxy of Ultrathin Organic Semiconductors and Heterojunctions Tailored by van der Waals Interactions. Nano Lett. 16, 3754–3759 (2016).
Mannebach, E. M. et al. High Hole Mobility and Thickness-Dependent Crystal Structure in α,ω-Dihexylsexithiophene Single-Monolayer Field-Effect Transistors. Adv. Funct. Mater. 23, 554–564 (2012).
Park, B.-N., Seo, S. & Evans, P. G. Channel formation in single-monolayer pentacene thin film transistors. J. Phys. D: Appl. Phys. 40, 3506–3511 (2007).
Zhang, Y. et al. Probing Carrier Transport and Structure-Property Relationship of Highly Ordered Organic Semiconductors at the Two-Dimensional Limit. Phys. Rev. Lett. 116, 016602–6 (2016).
Asadi, K., Wu, Y., Gholamrezaie, F., Rudolf, P. & Blom, P. W. M. Single-Layer Pentacene Field-Effect Transistors Using Electrodes Modified With Self-assembled Monolayers. Adv. Mater. 21, 4109–4114 (2009).
Chen, H. et al. Solution-Processable, Low-Voltage, and High-Performance Monolayer Field-Effect Transistors with Aqueous Stability and High Sensitivity. Adv. Mater. 27, 2113–2120 (2015).
Solution-Processable Septithiophene Monolayer Transistor. 1–6, doi:10.1002/adma201103522 (2012).
Hutchins, D. O. et al. Spin cast self-assembled monolayer field effect transistors. Organic Electronics 13, 464–468 (2012).
Shan, L. et al. Monolayer Field-Effect Transistors of Nonplanar Organic Semiconductors with Brickwork Arrangement. Adv. Mater. 27, 3418–3423 (2015).
Wang, B. et al. Addressable growth of oriented organic semiconductor ultra-thin films on hydrophobic surface by direct dip-coating. Organic Electronics 24, 170–175 (2015).
Li, L. et al. Controllable Growth and Field-Effect Property of Monolayer to Multilayer Microstripes of an Organic Semiconductor. J. Am. Chem. Soc. 132, 8807–8809 (2010).
Jiang, L. et al. Millimeter-Sized Molecular Monolayer Two-Dimensional Crystals. Adv. Mater. 23, 2059–2063 (2011).
Mottaghi, M. et al. Low-Operating-Voltage Organic Transistors Made of Bifunctional Self-Assembled Monolayers. Adv. Funct. Mater. 17, 597–604 (2007).
Ringk, A. et al. n-Type self-assembled monolayer field-effect transistors for flexible organic electronics. Organic Electronics 14, 1297–1304 (2013).
Wang, Q. et al. 2D Single-Crystalline Molecular Semiconductors with Precise Layer Definition Achieved by Floating-Coffee-Ring-Driven Assembly. Adv. Funct. Mater. 26, 3191–3198 (2016).
Klauk, H., Zschieschang, U., Pflaum, J. & Halik, M. Ultralow-power organic complementary circuits. Nature 445, 745–748 (2007).
Kaltenbrunner, M. et al. An ultra-lightweight design for imperceptible plastic electronics. Nature Publishing Group 499, 458–463 (2013).
Majewski, L. A., Schroeder, R. & Grell, M. Low-Voltage, High-Performance Organic Field-Effect Transistors with an Ultra-Thin TiO2 Layer as Gate Insulator. Adv. Funct. Mater. 15, 1017–1022 (2005).
Tiwari, S. P., Zhang, X.-H., Potscavage, W. J. Jr. & Kippelen, B. Low-voltage solution-processed n-channel organic field-effect transistors with high-k HfO2 gate dielectrics grown by atomic layer deposition. Appl. Phys. Lett. 95, 223303–4 (2009).
Yuan, Y. et al. Ultra-high mobility transparent organic thin film transistors grown by an off-centre spin-coating method. Nature Communications 5, 1–9 (2014).
Minemawari, H. et al. Inkjet printing of single-crystal films. Nature Publishing Group 475, 364–367 (2011).
Ebata, H. et al. Highly Soluble [1]Benzothieno[3,2- b]benzothiophene (BTBT) Derivatives for High-Performance, Solution-Processed Organic Field-Effect Transistors. J. Am. Chem. Soc. 129, 15732–15733 (2007).
Kobayashi, H. et al. Hopping and band mobilities of pentacene, rubrene, and 2,7-dioctyl[1]benzothieno[3,2- b][1]benzothiophene (C 8-BTBT) from first principle calculations. The Journal of Chemical Physics 139, 014707–8 (2013).
He, D. et al. Two-dimensional quasi-freestanding molecular crystals for high-performance organic field-effect transistors. Nature Communications 5, 1–7 (2017).
Wang, S. D., Yan, Y. & Tsukagoshi, K. Understanding contact behavior in organic thin film transistors. Appl. Phys. Lett. 97, 063307–3 (2010).
Minari, T., Miyadera, T., Tsukagoshi, K., Aoyagi, Y. & Ito, H. Charge injection process in organic field-effect transistors. Appl. Phys. Lett. 91, 053508–3 (2007).
Hulea, I. N. et al. Tunable Fröhlich polarons in organic single-crystal transistors. Nat Mater 5, 982–986 (2006).
Acton, B. O. et al. Dielectric Surface-Controlled Low-Voltage Organic Transistors via n-Alkyl Phosphonic Acid Self-Assembled Monolayers on High- kMetal Oxide. ACS Appl. Mater. Interfaces 2, 511–520 (2010).
Liu, Q. et al. Low-voltage organic field-effect transistors based on novel high- κorganometallic lanthanide complex for gate insulating materials. AIP Advances 4, 087140–6 (2014).
Su, Y. R. et al. A low-temperature, solution-processed high- kdielectric for low-voltage, high-performance organic field-effect transistors (OFETs). J. Phys. D: Appl. Phys. 46, 095105–9 (2013).
Kim, S. H., Jang, M., Yang, H., Anthony, J. E. & Park, C. E. Physicochemically Stable Polymer-Coupled Oxide Dielectrics for Multipurpose Organic Electronic Applications. Adv. Funct. Mater. 21, 2198–2207 (2011).
Jung, S., Ji, T. & Varadan, V. K. Temperature sensor using thermal transport properties in the subthreshold regime of an organic thin film transistor. Appl. Phys. Lett. 90, 062105–3 (2007).
Knipp, D., Street, R. A., V lkel, A. & Ho, J. Pentacene thin film transistors on inorganic dielectrics: Morphology, structural properties, and electronic transport. Journal of Applied Physics 93, 347–355 (2003).
Pivrikas, A., Ullah, M., Sitter, H. & Sariciftci, N. S. Electric field dependent activation energy of electron transport in fullerene diodes and field effect transistors: Gill’s law. Appl. Phys. Lett. 98, 092114–4 (2011).
Kronemeijer, A. J. et al. Two-Dimensional Carrier Distribution in Top-Gate Polymer Field-Effect Transistors: Correlation between Width of Density of Localized States and Urbach Energy. Adv. Mater. 26, 728–733 (2013).
Xie, W. et al. Temperature-Independent Transport in High-Mobility Dinaphtho-Thieno-Thiophene (DNTT) Single Crystal Transistors. Adv. Mater. 25, 3478–3484 (2013).
Konezny, S. J., Bussac, M. N. & Zuppiroli, L. Hopping and trapping mechanisms in organic field-effect transistors. Phys. Rev. B 81, 045313–12 (2010).
Hamaguchi, A. et al. Single-Crystal-Like Organic Thin-Film Transistors Fabricated from Dinaphtho[2,3- b:2′,3′- f]thieno[3,2- b]thiophene (DNTT) Precursor-Polystyrene Blends. Adv. Mater. 27, 6606–6611 (2015).
Darmawan, P. et al. Optimal Structure for High-Performance and Low-Contact-Resistance Organic Field-Effect Transistors Using Contact-Doped Coplanar and Pseudo-Staggered Device Architectures. Adv. Funct. Mater. 22, 4577–4583 (2012).
Xu, Y. et al. Joule’s law for organic transistors exploration: Case of contact resistance. Journal of Applied Physics 113, 064507–6 (2013).
Xu, Y. et al. Contacts between Two- and Three-Dimensional Materials: Ohmic, Schottky, and p– nHeterojunctions. ACS Nano 10, 4895–4919 (2016).
Liu, C., Xu, Y., Li, Y., Scheideler, W. & Minari, T. Critical Impact of Gate Dielectric Interfaces on the Contact Resistance of High-Performance Organic Field-Effect Transistors. J. Phys. Chem. C 117, 12337–12345 (2013).
Acknowledgements
This study was supported partially by 973 projects under Grant No. 2013CBA01600 and 2013CB932900, NSFC under Grant No. 61574074, NSFJS under Grant No. BK20170075, and Open Partnership Joint Projects of NSFC-JSPS Bilateral Joint Research Projects under Grant No. 61511140098.
Author information
Authors and Affiliations
Contributions
Q.J.W., Y.L., and Y.S. conceived and designed the experiments; Q.J.W., S.J., J.Q., Y.W., and L.Z. carried out the experiments; L.S., Y.J.Z., and Y.H.Z. performed the AFM characterizations; Y.Li and Q. Wang wrote the manuscript text and prepared the figures. X.R.W. and Y.D.Z. joined the discussion on the results and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Q., Jiang, S., Qian, J. et al. Low-voltage, High-performance Organic Field-Effect Transistors Based on 2D Crystalline Molecular Semiconductors. Sci Rep 7, 7830 (2017). https://doi.org/10.1038/s41598-017-08280-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08280-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.