Abstract
Limited evidence revealed conflicting results on relationship between phthalate exposure and clinical pregnancy loss (gestational weeks >6). A prospective cohort study in Chinese pregnant women (n = 3220) was conducted to investigate the association between urinary phthalate metabolites and clinical pregnancy loss (gestational weeks 6 to 27; n = 109). Morning urine samples during gestational weeks 5 to 14 (mean 10.42) were collected to measure monomethyl phthalate (MMP), monoethyl phthalate (MEP), monobutyl phthalate (MBP), monobenzyl phthalate (MBzP), mono (2-ethylhexyl) phthalate (MEHP), mono (2-ethyl-5-oxohexyl) phthalate (MEOHP) and mono (2-ethyl-5-hydroxyhexyl) phthalate (MEHHP). The concentrations of low- and high-molecular weight phthalate metabolites (ΣLMWP <250 Da and ΣHMWP >250 Da) were calculated. Adjusted logistic regression models showed increased risks of clinical pregnancy loss in women with higher creatinine- normalized concentrations of MEP, MBP, MEOHP, MEHHP, ΣLMWP and ΣHMWP. Stratified analysis by gestational weeks (10 weeks) of miscarriage indicated positive associations of MEP, MEOHP, MEHHP and ΣHMWP with embryonic loss (during gestational weeks 6 to 10). The only association of foetal loss (during gestational weeks 11 to 27) was observed with MEHHP. Our findings suggested that Chinese women who were exposed to phthalates during early pregnancy had an increased risk of clinical pregnancy loss, especially embryonic loss.
Similar content being viewed by others
Introduction
Phthalates are produced on a large scale and extensively used worldwide, causing ubiquitous contamination of the environment1,2,3,4. General population is continually exposed to phthalates in daily life5. Humans are exposed to phthalates through ingestion, inhalation, dermal absorption, and medical treatment6,7,8,9. These chemicals are quickly metabolized into corresponding monoesters and oxidized monoesters, and are excreted in urine10, 11. Urine is the matrix of choice for biomonitoring nonpersistent chemicals including phthalates12. The detection rate of phthalates in urine samples collected from pregnant women is nearly 100%, reported by international studies13,14,15,16,17,18. Some phthalates even penetrate placenta19 and can be detected in placental tissue19, meconium20, amniotic fluid20,21,22 and cord blood23. These findings suggest that human exposure to phthalates may begin in utero22. Therefore, unborn foetuses of pregnant women are regarded as a high-risk group for the potential adverse health effects of phthalate exposure24.
In China, clinical pregnancy loss is defined as a miscarriage that a clinically-recognized pregnancy (gestational weeks >6) involuntarily ends before 28 complete weeks25. The incidences were reported 6–14% in China, while over 20% in other countries26. A recent Chinese study revealed that the incidence of pregnancy loss was 9.04% in rural and 3.75% in urban areas27. Pregnancy loss is one of the most common adverse pregnancy outcomes worldwide and causes physical and psychological traumas for pregnant women and their families. Besides, economic burden is heavy for patients themselves and their countries. Pregnancy loss is a multi-factorial disorder linked to interactions between genetic and environmental factors, such as maternal age, smoking, caffeine and alcohol consumption, and exposure to environmental pollutants28. The variety of environmental exposures linked to miscarriage continue to increase. Much attention focuses on the environmental endocrine disruptors and phthalates are among them.
Phthalates can interfere with the development of multiple body systems, including reproductive system. Epidemiological evidences show that phthalate exposure may cause adverse pregnancy consequences (e.g. increased blood pressure and decreased blood glucose) and abnormal foetal growth (e.g. low birth weight, preterm birth, decreased gestational age and birth length)20, 22, 29,30,31,32. To our knowledge, the effect of phthalate exposure on pregnancy loss is still unclear due to insufficient epidemiological studies. Three cohort studies based on small sample sizes showed inconsistent associations between phthalate exposure and pregnancy loss in two countries33,34,35. A cohort study of 128 Danish women found that increased level of mono (2-ethylhexyl) phthalate (MEHP) was associated with higher risk of subclinical embryonic loss (within 6 gestational weeks), and lower risk of clinical pregnancy loss (conception lasting >6 weeks)33. A cohort study of 303 American women undergoing medical assisted reproduction revealed significant relationships between di (2-ethylhexyl) phthalate (DEHP) metabolites and biochemical and total pregnancy loss34. However, another cohort study of 221 American women had opposite findings that mono (2-ethyl-5-hydroxyhexyl) phthalate (MEOHP) and DEHP were related to decreased risk of subclinical embryonic loss, and insignificant relationship between any analyzed compounds and clinical pregnancy loss35. In China, all current conclusions on associations between phthalates and miscarriage are solely based on case-control studies36,37,38. A case-control study (132 clinical pregnancy loss cases and 172 healthy controls) revealed that an increased risk of clinical pregnancy loss was associated with monoethyl phthalate (MEP) and mono-n-butyl phthalate (MBP)36. Another study suggested that exposure to monoethyl phthalate (MMP) and metabolites of DEHP was associated with missed miscarriage37. Peng et al.38 found that mono-iso-butyl phthalate (MiBP) and MBP concentrations in women with unexplained recurrent spontaneous abortion were remarkably higher than those in control group. Differences in study design, population characteristics, sample size, and miscarriage type are likely to contribute to the conflicting results. Therefore, prospective cohort studies are urgently needed to further identify the associations between phthalates and miscarriage in China.
This prospective cohort study based on large sample size was conducted in China. The aim was to investigate the association between phthalate exposure during early pregnancy and clinical pregnancy loss.
Results
Demographic characteristics
Demographic characteristics of women who were included (n = 3 220) and excluded (n = 254) in the present study were shown in Supplementary material, Table S1. The BMI was 20.88 ± 2.85 in the enrolled group, significantly lower than that (21.54 ± 3.25) in the excluded group. Besides, the educational level was lower (middle school or below; 19.60% vs. 34.65%) and the proportion of alcohol-drinking was higher (8.32% vs. 3.15%) in the included participants. No significant differences were found between the included and excluded individuals in terms of age, smoking and parity. The detailed information for study participants was summarized in Table 1. Compared to women with single live births, there were significant differences of maternal age, parity and gestational weeks of urine collection in women with clinical pregnancy loss, but no difference was observed regarding prepregnancy BMI, smoking, drinking and education. In terms of stratification descriptions, embryonic loss was significantly associated with gestational weeks of urine collection, while foetal loss was significantly associated with maternal age, prepregnancy BMI, parity and gestational weeks of urine collection.
Phthalate metabolite concentrations
The overall creatinine-normalized concentrations of phthalate metabolites were identical to our previous study39, as participants were from the MABC. The detectable frequencies of most phthalate metabolites were over 99%, except for MBzP (64%).
Phthalates and clinical pregnancy loss
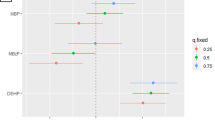
The results of logistic regression analyses were shown in Table 2. After adjustments for maternal age, education, parity and gestational weeks of urine collection, concentrations of MEP, MBP, MEOHP, MEHHP, ΣLMWP, and ΣHMWP were significantly associated with an increased risk of clinical pregnancy loss.
Stratification analyses
Stratification analyses (Table 3) revealed that increased risk of embryonic loss was associated to higher concentrations of MEP, MEOHP, MEHHP and ΣHMWP. For the risk of foetal loss, the only significant association observed was with the higher level of MEHHP.
Discussion
In the present study, we observed that both ΣLMWP (including MEP and MBP) and ΣHMWP (MEOHP and MEHHP) during early pregnancy were associated with clinical pregnancy loss, especially with embryonic loss, among Chinese women in the study. According to the results of previous related studies33,34,35,36,37,38, the magnitude of associations and types of phthalate metabolites with pregnancy loss remain inconsistent. We hypothesized that the discrepancy between the present study and the previous studies was attributed partially to the differences in maternal characteristics, study design, sample size and miscarriage type and partially to co-linearity among phthalate metabolites. For instance, previous studies were from three different countries (Denmark, America and China) and both cohort33,34,35 and case control study designs36,37,38 were employed. All studies had small sample sizes which would create large variations among studies. For example, cohort studies conducted by Toft et al.33 and Jukic et al.35 included only 128 and 221 pregnant women, while Messerlian et al.34 recruited 303 pregnant women undergoing medically assisted reproduction. Among case-conrol studies, Mu et al.36 enrolled 132 clinical pregnancy loss cases and 172 healthy controls; Yi et al.37 included 150 women with missed miscarriages and 150 healthy controls; Peng et al.38 enrolled only a total of 30 cases with unexplained recurrent spontaneous abortion and 30 controls. In addition, these three case-control studies recruited individuals with different miscarriage types. As indicated above, phthalate metabolites are usually co-existing agents thus highly correlated with each other. Those were found significant in the analyses could be just surrogates for other co-existing ones. This was why most studies indicated that exposure to this family of compounds during pregnancy increased the risks of miscarriage, although which members were related to adverse pregnancy outcomes is still debatable.
The unfavorable effect of phthalates in population is similar to observations from experimental studies. Evidences from rat and mouse models showed chemicals such as DEHP, di-n-butyl phthalate (DBP), diisobutyl phthalate, and diethyl phthalate induced decreased numbers and sizes of litters, reduced live-born offspring or embryo survival, and increased midpregnancy abortions, postimplantation loss, and intrauterine absorption40,41,42,43,44,45,46,47.
Our results indicated embryonic loss was more sensitive to phthalate exposure. The Danish Prospective Cohort Study of Fecundity (1992–1994)33 found that a relationship between MEHP and subclinical pregnancy loss [adjusted odds ratio (ORadj) = 10.83 and 40.67, 95% confidence interval (95%CI) = 1.16−101.48 and 4.48−369.50 for the second tertile (T2) and T3, respectively, compared with T1), whereas an inverse association between MEHP and clinical pregnancy loss was unexpectedly identified (ORadj = 0.17, 95%CI = 0.03−0.95 for T2 compared with T1). The Environment and Reproductive Health Study (2004–2014)34 suggested that DEHP metabolites were associated with both biochemical and total pregnancy loss. Therefore, it was suggested that the pregnancy loss was more susceptible to phthalate exposure when the gestational age was shorter. However, the North Carolina Early Pregnancy Study (1982–1986)35 showed that exposure to MEOHP and DEHP reduced the risk of subclinical embryonic loss (p = 0.04 and 0.001, respectively; ORs and 95%CIs were not shown in the article), but none of the analyzed compounds were related to clinical pregnancy loss. Additional human studies are warranted to verify these associations.
Phthalates are known as environmental endocrine disrupters. Some phthalates [i.e., DEHP, DBP, butyl benzyl phthalate (BBzP), and their metabolites] reportedly reduced the production of oestradiol and progesterone in the ovaries through a receptor-mediated signalling pathway (cAMP and peroxisome proliferator-activated receptors)46, 48, 49. Their adverse effects on endocrine functioning is likely to change the circulating levels of hormones responsible for maintaining pregnancy. In addition, phthalates were found associated with increments of inflammation biomarkers (i.e., IL-6 and IL-10) and oxidative stress (i.e., 8-hydroxyguanosine and 8-isoprostane)13, 50, 51. Some investigations suggested pregnancy failure might also be mediated by the recruitment of a highly activated uterine natural killer cell52. Stress-triggered immune and endocrine events during pregnancy could induce miscarriage52. Another hypothesis suggested that angiogenic factors (placental growth factor and soluble fms-like tyrosine kinase-1) and placenta log interspersed nuclear elements-1 methylation were decreased by phthalates29, 53. Therefore, phthalates could disrupt placental development and function during gestation. This is particularly concerning, because the placenta establishes close contact between the mother and foetus and enables the exchange of gases, nutrients, and waste products54, 55. Notably, Mose et al.19 determined that short-chained phthalate monoesters could cross the placenta by slow transfer, but long-chained monoesters exhibited no placental transfer. These evidences biologically support the hypothesis that some phthalates could have adverse effects on the clinical pregnancy loss.
To our knowledge, this study is the first prospective cohort study in China that has investigated the association of phthalate exposure with clinical pregnancy loss, embryonic loss, and foetal loss. However, there are some limitations that should be addressed. Firstly, the proportion of pregnancy losses in the present study is substantially low. We hypothesized several explanations. One is that miscarriage occurred prior to clinical detection or first prenatal visits. Another is that our participants had higher socioeconomic status (SES), since miscarriage risk was increased with lower SES status27. As the MABC is a hospital-based study, the third possible explanation is that the community derived data were not taken into account in this study, thereby causing a lower incidence. In this situation, it was likely that we underestimated the relationship of miscarriage with phthalate metabolites. Secondly, we only used a single-spot urine sample for assessing the phthalate exposure in early pregnancy. It has been reported that a single measurement could be used as a typical measurement for a period ranging from weeks to months, when sources or exposure patterns are consistent17. Therefore, it is unlikely to affect our results. Finally, we did not differentiate the two isomers of MnBP and MiBP; they were combined and designated as ‘MBP’ in our study.
In conclusion, exposures to MEP, MBP, MEOHP, MEEHP, ΣLMWP and ΣHMWP in early pregnancy (5 to 14 weeks’ gestation) are independent risk factors for clinical pregnancy loss, especially for embryonic loss, in Chinese pregnant women. These findings should be verified by additional scientific evidences.
Methods
Study population
The present study was conducted on the basis of the Ma’anshan Birth Cohort (MABC) study. From May 2013 to September 2014, the MABC consecutively recruited 3,474 pregnant women at their first visits (gestational weeks 5 to 14) in Ma’anshan Maternal and Child Health Centre in China56. Meanwhile, questionnaires regarding demographic characteristics were obtained and first-morning urine samples were collected for measuring phthalate metabolites. Information on pregnancy/birth outcomes, such as single live birth, clinical pregnancy loss, and stillbirth, was obtained from medical records.
Of the 3,474 women, 81 women with ectopic pregnancy (n = 2), stillbirth (n = 10), therapeutic abortion (n = 30), or multiple gestations (n = 39) were excluded. 173 women (162 women with live births and 11 women with miscarriages) whose urine specimens were unavailable were also excluded from the analysis. The remaining 3,220 women, consisting of 3,111 women with single live birth and 109 women with clinical pregnancy loss, were included in the current study. Of the 109 women, 64 women underwent embryonic loss (gestational weeks 6 to 10) and 45 women underwent foetal loss (gestational weeks 11 to 27)57.
All the participants provided written informed consents. The recruitment and subsequent follow-up protocols (including the urinary analysis of phthalates) were both approved by the ethics committee of Anhui Medical University (approval No. 2013119). All the study methods were performed in accordance with the relevant guidelines.
Covariates
A series of potential confounding covariates were selected, including maternal age (continuous), prepregnancy body mass index (BMI; continuous), current cigarette consumption (yes or no), alcohol consumption (yes or no), educational level (middle school or below, high school, junior college, university or above) and parity (0 or >1).
Exposure assessment
Seven urinary phthalate metabolites including MMP, MEP, MBP, monobenzyl phthalate (MBzP), MEHP, MEOHP, and mono (2-ethyl-5-hydroxyhexyl) phthalate (MEHHP) were measured through high-performance liquid chromatography-tandem mass spectrometry (6410LC-MS, Agilent Technologies Co., Santa Clara, CA, USA), using a previously reported methodology with modifications58, 59. Concentrations below the limit of detection (LOD) were assigned with LOD/√2 for calculations. Urine creatinine was measured using a creatinine assay kit based on the picric kinetic method (Jiancheng Bioengineering Institute, Nanjing, China).
Statistical analyses
Phthalate metabolites were categorized into the low- (ΣLMWP; <250 Da) and high-molecular weight phthalates (ΣHMWP; >250 Da)60, which were calculated according to the following equations:
The phthalate metabolite concentrations were creatinine-normalized to account for the impact of maternal urine dilution.
The creatinine-normalized concentrations of phthalate metabolites had a skewed distribution. Therefore, phthalate metabolite concentrations were natural log transformed. Women were grouped into tertiles according to the concentrations (T1 < P 1/3, P 1/3 < T2 < P 2/3 and T3 > P 2/3 ). Logistic regressions were used to analyze the odds ratios (ORs) and 95% confidence intervals (95% CIs) for the risks of clinical pregnancy loss. Each phthalate was included in one model, because they were all correlated with each other with Spearman r ranging from 0.04 to 0.61 (all p values < 0.05). Covariates which were significantly associated with clinical pregnancy loss, embryonic loss and foetal loss (p < 0.05) were adjusted for corresponding models. Education level is reportedly played an important role in pregnancy loss risk, therefore it was included as a confounder27. Stratification analyses were also performed by types of miscarriage.
All statistical analyses were performed using the SPSS software (Version 10.0), and p < 0.05 was considered statistically significant.
References
Dodson, R. E. et al. Endocrine disruptors and asthma-associated chemicals in consumer products. Environ Health Perspect 120, 935–943 (2012).
Hernández-Díaz, S., Mitchell, A. A., Kelley, K. E., Calafat, A. M. & Hauser, R. Medications as a potential source of exposure to phthalates in the U.S. population. Environ Health Perspect 117, 185–189 (2009).
Just, A. C. et al. Urinary and air phthalate concentrations and self-reported use of personal care products among minority pregnant women in New York city. J Expo Sci Environ Epidemiol 20, 625–633 (2010).
Schettler, T. Human exposure to phthalates via consumer products. Int J Androl 29, 134–139, discussion 181–135 (2006).
Wittassek, M. et al. Internal phthalate exposure over the last two decades–a retrospective human biomonitoring study. Int J Hyg Environ Health 210, 319–333 (2007).
Andrady, A. L. & Neal, M. A. Applications and societal benefits of plastics. Philos Trans R Soc Lond B Biol Sci 364, 1977–1984 (2009).
Bekö, G. et al. Children’s phthalate intakes and resultant cumulative exposures estimated from urine compared with estimates from dust ingestion, inhalation and dermal absorption in their homes and daycare centers. PLoS One 8, e62442 (2013).
Gaspar, F. W. et al. Phthalate exposure and risk assessment in California child care facilities. Environ Sci Technol 48, 7593–7601 (2014).
Koch, H. M. et al. Identifying sources of phthalate exposure with human biomonitoring: results of a 48 h fasting study with urine collection and personal activity patterns. Int J Hyg Environ Health 216, 672–681 (2013).
Frederiksen, H., Skakkebaek, N. E. & Andersson, A. M. Metabolism of phthalates in humans. Mol Nutr Food Res 51, 899–911 (2007).
Wittassek, M. & Angerer, J. Phthalates: metabolism and exposure. Int J Androl 31, 131–138 (2008).
Koch, H. M. & Calafat, A. M. Human body burdens of chemicals used in plastic manufacture. Philos Trans R Soc Lond B Biol Sci 364, 2063–2078 (2009).
Ferguson, K. K. et al. Urinary phthalate metabolite associations with biomarkers of inflammation and oxidative stress across pregnancy in Puerto Rico. Environ Sci Technol 48, 7018–7025 (2014).
Kim, Y. et al. Prenatal exposure to phthalates and infant development at 6 months: prospective Mothers and Children’s Environmental Health (MOCEH) study. Environ Health Perspect 119, 1495–1500 (2011).
Kobrosly, R. W. et al. Prenatal phthalate exposures and neurobehavioral development scores in boys and girls at 6–10 years of age. Environ Health Perspect 122, 521–528 (2014).
Lien, Y. J. et al. Prenatal exposure to phthalate esters and behavioral syndromes in children at 8 years of age: Taiwan Maternal and Infant Cohort Study. Environ Health Perspect 123, 95–100 (2015).
Suzuki, Y. et al. Exposure assessment of phthalate esters in Japanese pregnant women by using urinary metabolite analysis. Environ Health Prev Med 14, 180–187 (2009).
Téllez-Rojo, M. M. et al. Prenatal urinary phthalate metabolites levels and neurodevelopment in children at two and three years of age. Sci Total Environ 461–462, 386–390 (2013).
Mose, T., Knudsen, L. E., Hedegaard, M. & Mortensen, G. K. Transplacental Transfer of Monomethyl Phthalate and Mono(2-ethylhexyl) Phthalate in a Human Placenta Perfusion System. Int J Toxicol 26, 221–229 (2007).
Zhang, Y. et al. Phthalate levels and low birth weight: a nested case-control study of Chinese newborns. J Pediatr 155, 500–504 (2009).
Jensen, M. S. et al. Phthalates and perfluorooctanesulfonic acid in human amniotic fluid: temporal trends and timing of amniocentesis in pregnancy. Environ Health Perspect 120, 897–903 (2012).
Latini, G. et al. In utero exposure to di-(2-ethylhexyl)phthalate and duration of human pregnancy. Environ Health Perspect 111, 1783–1785 (2003).
Yan, X. et al. Phthalates Biomarker Identification and Exposure Estimates in a Population of Pregnant Women. Hum Ecol Risk Assess 15, 565–578 (2009).
Wittassek, M., Koch, H. M., Angerer, J. & Brüning, T. Assessing exposure to phthalates - the human biomonitoring approach. Mol Nutr Food Res 55, 7–31 (2011).
Wang, X. et al. Conception, early pregnancy loss, and time to clinical pregnancy: a population-based prospective study. Fertil Steril 79, 577–584 (2003).
Lewington, S. et al. Temporal trends of main reproductive characteristics in ten urban and rural regions of China: the China Kadoorie Biobank study of 300 000 women. Int J Epidemiol 43, 1252–1262 (2014).
Zheng, D., Li, C., Wu, T. & Tang, K. Factors associated with spontaneous abortion: a cross-sectional study of Chinese populations. Reprod Health 14, 33 (2017).
Oliver, A. & Overton, C. Diagnosis and management of miscarriage. Practitioner 258, 25–28 (2014).
Zhao, Y. et al. Prenatal phthalate exposure, infant growth, and global DNA methylation of human placenta. Environ Mol Mutagen 56, 286–292 (2015).
Huang, P. C., Kuo, P. L., Chou, Y. Y., Lin, S. J. & Lee, C. C. Association between prenatal exposure to phthalates and the health of newborns. Environ Int 35, 14–20 (2009).
Meeker, J. D. et al. Urinary phthalate metabolites in relation to preterm birth in Mexico city. Environ Health Perspect 117, 1587–1592 (2009).
Wolff, M. S. et al. Prenatal phenol and phthalate exposures and birth outcomes. Environ Health Perspect 116, 1092–1097 (2008).
Toft, G. et al. Association between pregnancy loss and urinary phthalate levels around the time of conception. Environ Health Perspect 120, 458–463 (2012).
Messerlian, C. et al. Urinary Concentrations of Phthalate Metabolites and Pregnancy Loss Among Women Conceiving with Medically Assisted Reproduction. Epidemiology 27, 879–888 (2016).
Jukic, A. M. et al. Urinary Concentrations of Phthalate Metabolites and Bisphenol A and Associations with Follicular-Phase Length, Luteal-Phase Length, Fecundability, Early Pregnancy Loss. Environ Health Perspect 124, 321–328 (2016).
Mu, D. et al. Levels of Phthalate Metabolites in Urine of Pregnant Women and Risk of Clinical Pregnancy Loss. Environ Sci Technol 49, 10651–10657 (2015).
Yi, H. et al. A pilot study on association between phthalate exposure and missed miscarriage. Eur Rev Med Pharmacol Sci 20, 1894–1902 (2016).
Peng, F. et al. A study on phthalate metabolites, bisphenol A and nonylphenol in the urine of Chinese women and unexplained recurrent spontabeous abortion. Environ Res 150, 622–628 (2016).
Gao, H. et al. Season-dependent concentrations of urinary phthalate metabolites among Chinese pregnant women: Repeated measures analysis. Environ Int S0160-4120, 30878–30879 (2017).
Ema, M., Kurosaka, R., Amano, H. & Ogawa, Y. Developmental toxicity evaluation of mono-n-butyl phthalate in rats. Toxicol Lett 78, 101–106 (1995).
Gray, L. E. Jr., Laskey., J. & Ostby, J. Chronic di-n-butyl phthalate exposure in rats reduces fertility and alters ovarian function during pregnancy in female Long Evans hooded rats. Toxicol Sci 93, 189–195 (2006).
Jarfelt, K. et al. Antiandrogenic effects in male rats perinatally exposed to a mixture of di(2-ethyl hexyl) phthalate and di(2-ethylhexyl) adipate. Reprod Toxicol 19, 505–515 (2005).
Lamb, J. C. 4th, Chapin, R. E., Teague, J., Lawton, A. D. & Reel, J. R. Reproductive effects of four phthalic acid esters in the mouse. Toxicol Appl Pharmacol 88, 255–269 (1987).
Mankidy, R., Wiseman, S., Ma, H. & Giesy, J. P. Biological impact of phthalates. Toxicol Lett 217, 50–58 (2013).
Saillenfait, A. M., Sabaté, J. P. & Gallissot, F. Development toxic effects of diisobutyl phthalate, the methyl-branched analogue of di-n-butyl phthalate, administered by gavage to rats. Toxicol Lett 165, 39–46 (2006).
Schmidt, J. S., Schaedlich, K., Fiandanese, N., Pocar, P. & Fischer, B. Effects of di(2-ethylhexyl) phthalate (DEHP) on female fertility and adipogenesis in C3H/N mice. Environ Health Perspect 120, 1123–1129 (2012).
Wine, R. N., Li, L. H., Barnes, L. H., Gulati, D. K. & Chapin, R. E. Reproductive roxicity of di-n-butyl phthalate in a continuous breeding protocol in Sprague-Dawley rats. Environ Health Perspect 105, 102–107 (1997).
Lovekamp-Swan, T. & Davis, B. J. Mechanisms of phthalate ester toxicity in the female reproductive system. Environ Health Perspect 111, 139–145 (2003).
Treinen, K. A., Dodson, W. C. & Heindel, J. J. Inhibition of FSH-stimulated cAMP accumulation and progesterone production by mono(2-ethylhexyl) phthalate in rat granulosa cell cultures. Toxicol Appl Pharmacol 106, 334–340 (1990).
Ferguson, K. K., Loch-Caruso, R. & Meeker, J. D. Urinary phthalate metabolites in relation to biomarkers of inflammation and oxidative stress: NHANES 1999–2006. Environ Res 111, 718–726 (2011).
Ferguson, K. K., McElrath, T. F., Chen, Y. H., Mukherjee, B. & Meeker, J. D. Urinary phthalate metabolites and biomarkers of oxidative stress in pregnant women: a repeated measures analysis. Environ Health Perspect 123, 210–216 (2015).
Kwak-Kim, J., Bao, S., Lee, S. K., Kim, J. W. & Gilman-Sachs, A. Immunological modes of pregnancy loss: inflammation, immune effectors, and stress. Am J Reprod Immunol 72, 129–140 (2014).
Ferguson, K. K., McElrath, T. F., Cantonwine, D. E., Mukherjee, B. & Meeker, J. D. Phthalate metabolites and bisphenol-A in association with circulating angiogenic biomarkers across pregnancy. Placenta 36, 699–703 (2015).
Nelissen, E. C., van Montfoort, A. P., Dumoulin, J. C. & Evers, J. L. Epigenetics and the placenta. Hum Reprod Update 17, 397–417 (2011).
Rossant, J. & Cross, J. C. Placental development: lessons from mouse mutants. Nat Rev Genet 2, 538–548 (2001).
Ge, X. et al. Maternal Snoring May Predict Adverse Pregnancy Outcomes: A Cohort Study in China. PLoS One 11, e0148732 (2016).
Porter, T. F. & Scott, J. R. Evidence-based care of recurrent miscarriage. Best Pract Res Clin Obstet Gynaecol 19, 85–101 (2005).
Wang, H. X. et al. Rapid sensitive analysis of phthalate metabolites, bisphenol A, and endogenous steroid hormones in human urine by mixed-mode solid-phase extraction, dansylation, and ultra-performance liquid chromatography coupled with triple quadruple mass spectrometry. Anal Bioanal Chem 405, 4313–4319 (2013).
Gao, H. et al. Determination of seven phthalate metabolites in human urine by high performance liquid chromatography-tandem mass spectrometry. Se Pu 33, 622–627 (2015).
Engel, S. M. et al. Prenatal phthalate exposure is associated with childhood behavior and executive functioning. Environ Health Perspect 118, 565–571 (2010).
Acknowledgements
Funding was provided by the National Natural Science Foundation of China (No. 81330068).
Author information
Authors and Affiliations
Contributions
H.G. wrote the main manuscript text. J.H.H., P.Z. and Y.Q.X. conceptualized this project. F.B.T. supervised and administered this project. K.H., S.Q.Y., L.J.M., X.G. and H.G. conducted the field surveys. Y.Y.X., Z.X.J. and J.S. designed the experiments. H.G. and Y.W.Z. performed the experiments. X.G.T. corrected the language and ensured the proper gramma and meaning of sentences. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, H., Zhang, Yw., Huang, K. et al. Urinary concentrations of phthalate metabolites in early pregnancy associated with clinical pregnancy loss in Chinese women. Sci Rep 7, 6800 (2017). https://doi.org/10.1038/s41598-017-06450-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-06450-2
This article is cited by
-
Individual and joint effects of phthalates exposure on the risk of early miscarriage
Journal of Exposure Science & Environmental Epidemiology (2023)
-
Exposure characteristics of phthalate metabolites among the Zunyi cohort of pregnant women in Southwest China
Environmental Science and Pollution Research (2022)
-
Maternal urine phthalate metabolite exposure and miscarriage risk: a nested case–control study of the Zunyi Birth Cohort
Environmental Science and Pollution Research (2022)
-
Association between prenatal phthalate exposure and gestational metabolic syndrome parameters: a systematic review of epidemiological studies
Environmental Science and Pollution Research (2021)
-
Sex-specific difference in placental inflammatory transcriptional biomarkers of maternal phthalate exposure: a prospective cohort study
Journal of Exposure Science & Environmental Epidemiology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.