Abstract
Multiple factors and conditions can lead to impaired wound healing. Chronic non-healing wounds are a common problem among the elderly. To identify microRNAs negatively impacting the wound repair, global miRNA profiling of wounds collected from young and old mice was performed. A subset of miRNAs that exhibited an age-dependent expression pattern during wound closure was identified, including miR-31 and miR-200c. The expression of miR-200 family members was markedly downregulated upon wounding in both young and aged mice, with an exception of acute upregulation of miR-200c at the early phase of wound healing in aged skin. In unwounded aged skin (versus unwounded younger skin), the level of miR-200c was also found elevated in both human and mice. Overexpression of miR-200c in human ex vivo wounds delayed re-epithelialisation and inhibited cell proliferation in the wound epithelium. Modulation of miR-200c expression in both human and mouse keratinocytes in vitro revealed inhibitory effects of miR-200c on migration, but not proliferation. Accelerated wound closure in vitro induced by anti-miR-200c was associated with upregulation of genes controlling cell migration. Thus, our study identified miR-200c as a critical determinant that inhibits cell migration during skin repair after injury and may contribute to age-associated alterations in wound repair.
Similar content being viewed by others
Introduction
Wound healing is a complex process that aims to repair skin integrity and functions, and can be divided into four phases: haemostasis, inflammation, proliferation and remodelling. Each phase overlaps with another and is characterised by coordinated activation and interplay of the cutaneous residential and migratory cells regulated by an array of signalling pathways, including platelet-derived growth factor (PDGF), transforming growth factor beta (TGF-β), vascular endothelial growth factor (VEGF), stromal cell-derived factor 1 (SDF-1) and fibroblast growth factors (FGF)1,2,3,4,5. Multiple factors and conditions can lead to impaired wound healing. Chronic wound healing disorders are a common problem among the elderly6, 7. In healthy older individuals, acute wound healing is temporally delayed as well. Ageing affects multiple processes, including DNA repair, mitochondrial function, cell cycle, proteolysis and cellular metabolism8, 9. Ageing-related impairment in wound repair is associated with alterations in all major components of healing process7, 10.
A class of non-coding RNA molecules that have recently emerged as critical factors in wound healing are the microRNAs (miRNAs)11,12,13. miRNAs are about 22-25 nucleotides in length and affect a wide range of cellular processes. miRNAs function by inhibiting mRNA translation or by targeting mRNA for degradation14,15,16,17. Increasing evidence suggest that miRNAs regulate gene expression program and outcome during wound healing. For example, cutaneous wound repair is compromised in both keratinocyte-specific and endothelial-specific Dicer-ablated mice, which asserts that miRNAs are implicated in keratinocyte differentiation and angiogenesis in the healing process18, 19. The possible involvement of miRNAs in skin wound healing has been illuminated by several expression profiling studies that have found differential expression of many miRNAs in wounded skin18, 20,21,22,23,24,25,26,27. Some notable examples include miR-130a, miR-132, miR-155, miR-198, miR-21, miR-31 and miR-378a13, 23, 24, 26, 28,29,30. miR-155 acts as an important player in controlling the inflammatory response during skin repair; genetic deletion of miR-155 in mice leads to accelerated healing associated with elevated numbers of macrophages and increased type-1 collagen deposition in wounded tissue30. TGF-β inducible miR-132 and miR-31 were found to be upregulated during the transition from the inflammatory to the proliferative phase in human skin promoting keratinocyte proliferation23, 28. In contrast, miR-378a negatively affects the remodelling phase, delaying the healing of mouse skin wounds by downregulating β3 integrin and vimentin29.
The Tomic-Canic lab identified a candidate set of microRNAs that contributes to the chronic non-healing wounds13. They observed enhanced levels of miR-21 and miR-130 in venous ulcers patients, which delay healing of human wounds by targeting leptin receptor (LepR)13.
Although miRNAs have emerged as key players in skin repair, their contribution to the aged-associated changes in the skin and impairment in wound healing remains unknown. The objective of the current study was to identify expressional changes of miRNAs during wound healing in aged versus young skin using mouse model, and to define the role for distinct miRNAs in the control of keratinocyte proliferation, migration and differentiation that might contribute to the age-associated alterations in cutaneous wound healing.
Results and Discussion
Age-dependent changes in miRNA expression fluctuation during cutaneous wound healing
In order to identify the candidate miRNAs that might compromise wound healing and contribute to the age-associated delay in wound repair, global miRNA profiling was performed in mouse back telogen skin of young (8-week-old) and aged (2-year-old) animals at distinct time points after wounding (Supplementary Table 1a,b). Bioinformatics analysis revealed 37 miRNAs that exhibited opposite changes with at least 2-fold differences in their levels at days 3 and 5 (D3 and D5) post wounding compared to unwounded skin (day 0; D0) (Fig. 1a, Table 1). Such age-dependent differential expression of selected miRNAs might suggest their involvement in ageing-associated changes in cutaneous wound repair. Interestingly, microarray validation by RT-qPCR confirmed the contrasting expression of miR-31. Significantly increased levels of miR-31 in unwounded aged versus (vs) young skin were decreased on days 3 and 5 after injury, in contrast to increase in miR-31 expression in young mice on days 3 and 5 post-wounding (Fig. 1b, Supplementary Tables 1 and 2). The observed changes in the dynamics of miR-31 levels in aged skin during wound healing suggest that miR-31 may compromise wound repair in aged skin. miR-31 is known to be highly expressed in the activated keratinocytes under hyperproliferative conditions, including anagen phase of the hair cycle, psoriasis, and cutaneous squamous cell carcinoma31,32,33,34,35. Moreover, our finding reconciles well with the previous reports about the positive impact of miR-31 on re-epithelialisation during acute wound healing28. miR-31 expression has been shown to be gradually increased in the epithelial tongue and promotes keratinocyte proliferation and migration during wound repair28. Therefore, diminished expression of miR-31 post-wounding in aged skin may suppress keratinocyte proliferation. In addition, the observed decreased miR-31 levels in aged skin may also contribute to the changes in the inflammatory response, as this miRNA has been found to positively regulate inflammatory cytokine and chemokine production in primary human keratinocytes35.
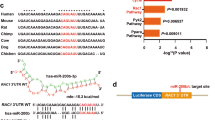
A differentially expressed subset of miRNAs in young versus aged skin during wound healing. (a) Microarray: heat map represents miRNAs that exhibited opposite dynamic of their expression (with at least 2-fold differences in fold changes) in young versus aged mouse wounds at days 3 and 5 (d3 and d5) post wounding compared to unwounded skin (d0). (Green: high expression; red: low expression); (b) RT-qPCR: increased miR-31 expression in 2-year-old versus 8-week-old intact mouse back skin, and increase in miR-31 levels during wound healing in the skin of 8-week-old mice in contrast to the skin of 2-year-old mice; (c) RT-qPCR: decreased expression of all miR-200 family members on day 3, 5, and 7 after wounding in the skin of 8-week-old mice; (d) RT-qPCR: decreased expression of miR-200 family after wounding, except for upregulation of miR-200c expression on day 3 after wounding in the skin of 2-year-old mice; (e) RT-qPCR: markedly elevated levels of miR-200c in intact telogen skin of 2-year-old versus 8-week-old mice; (f) RT-qPCR: prominent elevation in miR-200c expression in the epidermis of a healthy 72-year-old donor versus 50-year-old individual.
We also observed that the expression of all members of miR-200 family was markedly downregulated in both young and aged mouse wounds (Fig. 1c,d, Supplementary Table 1a,b), with the exception of acute upregulation of miR-200c observed at day 3 of wound healing in aged skin (Fig. 1d, Table 1). Interestingly, it was previously reported that the first phases of healing are delayed in aged wounds with significant decline in the rate of re-epithelialisation that takes place at day 3 after wounding7, 36.
In addition to the transient upregulation of miR-200c during wound healing in aged mouse skin, miR-200c expression was significantly increased in the intact unwounded skin of 2-year-old mice in contrast to 8 week-old mice (Fig. 1e, Supplementary Table 2). Similarly, miR-200c levels are higher in the human aged epidermis (Fig. 1f). Due to the altered levels of miR-200c in aged skin and during early stages of wound healing in old mice we selected this miRNA for further investigation.
There are multiple reasons to suggest that miR-200c may be involved in wound healing. The miR-200 family consists of epithelial-specific miRNAs that are known to function as negative regulators of epithelial to mesenchymal transition (EMT) by targeting E-cadherin transcriptional repressors, zinc finger E-box-binding homeobox 1 (ZEB1) and ZEB237. EMT is an important process that also occurs in wound healing, and is required for keratinocyte activation and their migration across the wound bed38. This fact makes miR-200 family members potential candidates for the regulation of re-epithelialisation during wound healing. Our data suggest that the increased expression of miR-200c in aged skin could contribute to the impaired skin repair associated with ageing and might be implicated in the pathogenesis of chronic wounds.
miR-200c regulates keratinocyte migration and differentiation
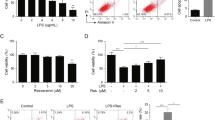
Keratinocyte migration, proliferation and differentiation are all critical components of successful re-epithelialisation during wound healing4, 5, 39. To begin to elucidate the potential effects of miR-200c on keratinocyte activity during wound healing, in vitro “scratch” assay was performed using primary mouse and human keratinocytes. Similar to in vivo observations, miR-200c expression was downregulated in both primary mouse and human keratinocytes during closure of scratch-induced wounds (Fig. 2a,b). To investigate the functional significance of miR-200c in keratinocytes, loss and gain of function experiments were employed. Possible effects of miR-200c on keratinocyte proliferation were evaluated by transfecting primary mouse and human epidermal keratinocytes with miR-200c inhibitor for 48 hours followed by quantitative analysis of bromodeoxyuridine (BrdU) positive cells. No significant difference in the proliferation rate was detected in anti-miR-200c treated and corresponding control groups in both mouse and human keratinocytes (Fig. 2c). Consistently, quantification of Ki-67 positive cells in the primary mouse keratinocytes 24 hours after transfection with miR-200c mimic also revealed no effect of miR-200c on keratinocyte proliferation (Supplementary Figure 1a,b). Moreover, fluorescence-activated cell sorting (FACS) analysis of human immortalised keratinocytes, HaCaT cells, transfected with miR-200c mimic did not show any changes in proliferation in response to the increased levels of miR-200c (Supplementary Figure 1c).
miR-200c exerts negative effects on keratinocyte migration and positive effect on keratinocyte differentiation. (a) RT-qPCR: dramatic decrease in miR-200c expression in primary mouse keratinocytes during scratch wound healing assay in vitro; (b) RT-qPCR: downregulation of miR-200c expression in primary human keratinocytes during scratch wound healing assay in vitro; (c) Lack of difference in the number of BrdU+ cells in both mouse (PMK) and human (PHK) keratinocytes transfected with miR-200c inhibitor versus scrambled RNA control; (d) miR-200c mimic suppresses migration of both human and mouse keratinocytes. Transwell migration assay: primary mouse or human keratinocytes were transfected with miR-200c mimic or negative control RNA and allowed to migrate for 48 hours (n = 3; mean ± SE; **p < 0.001; Wilcoxon rank sum test); (e) Scratch assay: primary mouse keratinocytes transfected with miR-200c inhibitor exhibited accelerated migration compared to the negative control (n = 3; mean ± SD; *p < 0.05; Student’s t test); (f) RT-qPCR: Accelerated wound closure induced by anti-miR-200c is associated with upregulation of genes controlling cell migration, including Zeb1, Srf, Clic4, Rac1, Met in mouse keratinocytes treated with miR-200c inhibitor (n = 3; mean ± SD; *p < 0.05; Student’s t test); (g) Ca2+-induced differentiation in mouse primary keratinocytes; increase in the expression of miR-200c 48 hours after Ca2+ treatment (n = 3, mean ± SD; **p < 0.01; Student’s t test); (h) Ca2+-induced differentiation in human keratinocytes; increase in the expression of miR-200c at 48 h and 72 h after Ca2+ treatment (n = 3, mean ± SD; **p < 0.01; Student’s t test); (i) Ca2+-induced differentiation in mouse keratinocytes; downregulation of Loricrin and Involucrin expression in primary mouse keratinocytes transfected with miR-200c inhibitor 48 hours after Ca2+ treatment (n = 3, mean ± SD; *p < 0.05; Student’s t test).
However, miR-200c mimic significantly reduced primary mouse and human keratinocyte migration in transwell assay (Fig. 2d), while inhibition of miR-200c in keratinocytes resulted in significant acceleration of their migration in scratch assay (Fig. 2e). Moreover, accelerated wound closure induced by anti-miR-200c in scratch assay was associated with upregulation of Zeb1, serum response factor (Srf), chloride intracellular channel 4 (Clic4), RAS-related C3 botulinum toxin substrate 1 (Rac1) and hepatocyte growth factor receptor (Met) (Fig. 2f). All these genes have previously been shown to be involved in the control of cell migration during cutaneous wound healing40,41,42,43 and, more importantly, are potential target genes of miR-200c identified by the TargetScan software44.
Next, we examined the involvement of miR-200c in keratinocyte differentiation. Keratinocyte differentiation was induced in both primary mouse and human keratinocytes in vitro by their exposure to high calcium medium45; it was associated with significant upregulation in the expression of miR-200c (Fig. 2g,h). Transfection of mouse keratinocytes with miR-200c inhibitor resulted in decreased expression of differentiation-associated genes, such as Krt1, Lor and Ivl, while the expression of Krt14, a marker of undifferentiated keratinocytes, was not affected by miR-200c inhibition (Fig. 2g). Thus, miR-200c can exert stimulatory effects on epidermal differentiation that is also known to be essential for proper wound healing. Indeed, it has been demonstrated that there is incomplete activation and deregulated differentiation of keratinocytes in human chronic wounds46, 47. Specifically, the expression of early differentiation markers is suppressed, whereas late differentiation markers such as involucrin and transglutaminase 1 are upregulated in venous ulcers, when compared to healthy skin46. Therefore, the aberrant expression of miR-200c in the epithelial edges of chronic wounds (Fig. 1g) could have a negatively impact not only on keratinocyte migration, but may also interfere with their differentiation.
Taken together, these data suggest that miR-200c can be involved in the regulation of different aspects of wound healing, sustaining keratinocyte differentiation and inhibiting their migration.
miR-200c compromises wound healing in human ex vivo skin
To further demonstrate the inhibitory effects of miR-200c on skin repair induced by injury, a human ex vivo skin wound healing model was used as described before48. Excisional wounds were treated with either miR-200c mimic or a scrambled control for 5 consecutive days (Fig. 3a). Histomorphological analysis of wound epithelium revealed that the area of the hyper-proliferative epithelium and the epithelial tongue length were significantly reduced in miR-200c treated biopsies compared to the controls (Fig. 3b,c). This was associated with significantly decreased proliferation in the regenerating epithelium, as was determined by quantitative analysis of Ki-67 positive cells (Fig. 3d,e). The wound epithelium treated with miR-200c mimic exhibited reduced expression of Keratin 16, Keratin 17 and CD49f (Integrin, alpha 6), markers of keratinocyte migration (Fig. 3f,g,h). The expression of Keratins 16 and 17 is normally induced in response to injury and stimulates the epithelialisation potential of keratinocytes49, 50. CD49f or laminin-binding integrin alpha6 also contributes to the successful re-epithelialisation by stimulating keratinocyte migration51,52,53. Therefore, this experiment confirms that aberrant levels of miR-200c may indeed compromise wound healing by suppressing the process of re-epithelialisation.
miR-200c delays wound healing in human skin ex vivo. (a) Representative images of wound histology of miR-200c mimic and negative control RNA treated skins 5 days post-wounding (H&E staining, WE – wound epithelium, scale bar 50 µm); (b) Significantly reduced area of wound epithelium in miR-200c mimic treated wound on day 5 after wounding versus the control (n = 5; mean ± SE, ***p < 0.0001, Student’s t-test); (c) Significantly reduced wound epithelial tongue length in miR-200c mimic treated wounds on day 5 post-wounding (n = 5; mean ± SE, ***p < 0.0001, Student’s t-test); (d–e) Ki-67+ proliferating keratinocytes seen in the wound epithelial tongue of ex vivo human wounds following 5 days of miR-200c mimic treatment (scale bars 50 μm); significantly decreased percentage of Ki-67+ proliferating cells in the wound epithelium after 5 days of miR-200c mimic treatment compared to the control (n = 4; mean ± SE; *p < 0.05; Student’s t test). (f) Reduced expression of Keratin 16 in miR-200c mimic treated wounds compared to the control (arrows; scale bar 50 µm); (g) Decreased expression of Keratin 17 in miR-200c mimic treated wounds compared to the control (arrows; scale bar 50 µm); (h) The expression of CD49f is suppressed in miR-200c mimic treated wounds compared to the control (arrows; scale bar 50 µm).
In conclusion, our study for the first time 1) reports differences in the expression of miRNAs in young and aged mouse skin wounds that suggest involvement of various miRNAs in age-associated impairment in wound healing; 2) provides evidence about contribution of miR-31 to the delay in wound healing in aged skin; 3) identifies miR-200c as an important player in successful re-epithelialisation during cutaneous wound healing that can exert positive and negative effects on keratinocyte differentiation and migration, respectively. Elevated levels of miR-200c in the skin could contribute to the age-associated delay in wound healing and compromised skin repair in chronic wounds.
Material and Methods
Animals and tissue collection
Animal studies were performed under protocols approved by UK Home Office Project License. A full-thickness 3 mm wound was introduced by punch biopsy onto back skin of 8-week-old and 2-year-old mice at the telogen stage of the hair cycle54, 55. Skin samples were collected on days 0, 3, 5, and 7 after wounding and were snap-frozen in liquid nitrogen54, 55.
Human wound healing organ culture assay
Human skin was obtained with inform consent from elective plastic surgery cases (rhytidectomy) of healthy donors, and was used for experiments that were approved by the Ethics committee of the University of Bradford, and under the auspices of the Human Tissue Act UK (2006). To create a partial thickness cutaneous wound, two parallel incisions were made in the skin 1 mm apart extending to the mid-dermis and the central strip was sharply excised using dissecting scissors48. Punch biopsies with the linear partial thickness wound in the centre were excised and transferred to six-well plates containing Dulbecco’s modified Eagle medium (DMEM), supplemented with 10% foetal bovine serum, sodium pyruvate and antibiotics48. Acute wounds were topically treated at the time of wounding with 50 μM of miR-200c mimic and corresponding scrambled control (Dharmacon) dissolved in 30% pluronic F-127 gel (Sigma)13.
Microarray and RT-qPCR analysis
Total RNA was isolated from homogenised tissue or cultured cells using the manufacturer’s protocol of TRI Reagent or Direct-zol RNA MiniPrep kit (Zymo Research). For eliminating genomic DNA, RNA samples were treated with DNase I (6 u/ul) (Zymo Research).
miRNA microarray profiling of young mouse wounds was performed using miRCURY LNA microRNA Array (7th Gen) (Exiqon, Vedbaek, Denmark). miRNA microarray profiling of aged mouse wounds was performed by LC Sciences (Houston, TX USA). Microarray data have been deposited to the Gene Expression Omnibus (GSE97034).
RT-qPCR for miR-31 and the members of miR-200 family was performed using corresponding TaqMan Real Time PCR Assays (Applied Biosystems) as described before32, 56, 57. For mRNA detection, total RNA was converted into complementary DNA using Reverse Transcription System (Promega, UK). RT-qPCR was performed on Applied Biosystems StepOne Plus system (Applied Biosystems) using Fast SYBR Green Master Mix (Applied Biosystems) and the corresponding primers (Table 2). Relative gene expression was calculated using the Genex software (Bio-Rad) based on the Ct (ΔΔCt) equitation method and normalised to U6 or Gapdh. Statistical analysis of RT-qPCR data was performed using Wilcoxon rank sum test.
Cell culture and transfections
Primary mouse epidermal keratinocytes (PMKs) were prepared from newborn mice and were grown in Eagle’s minimal essential medium EMEM (Lonza, UK) supplemented with 4% chelated foetal bovine serum as previously described56, 58. Primary human keratinocytes (PHKs) were isolated as previously described59 and grown in Keratinocyte Growth Medium 2 (PromoCell, Germany). HaCaT keratinocytes were grown in Dulbecco’s modified Eagle’s medium (Invitrogen) supplemented with heat-inactivated 10% foetal bovine serum and 1% penicillin-streptomycin at 37 °C, 5% CO2. To induce keratinocyte differentiation, cells were maintained in high-calcium medium (1.8 mM) for up to 72 hours. Transfections of the cells were performed using Lipofectamine RNAiMAX (ThermoFisher Scientific) using 100 nM miR-200c mimic (ThermoFisher Scientific), 200 nM inhibitor (Dharmacon) and corresponding negative controls according to the manufacturers’ protocol. All transfection and treatments were performed in triplicate.
Transwell migration assay
Transwell assay was performed as previously described54. Twenty-four hours after cell seeding to the membrane in the upper chamber of the transwell insert, keratinocytes were transfected with miR-200c mimic or negative control RNA as described above. Keratinocytes were allowed to migrate over 48 hours through the insert membrane, after which the cells attached to the top surface of the membrane were removed with a cotton swab. Cells that had migrated to the bottom surface were fixed in 4% paraformaldehyde and counterstained with DAPI (Vector Laboratories, USA). The number of DAPI positive nuclei of migrated keratinocytes per microscopic field was counted. Statistical analysis was performed using Wilcoxon rank sum test.
Scratch assay
Scratch assay was performed as previously described54, 57. A scratch was made in the monolayer of transfected keratinocytes using a P10 pipette tip. Mitomycin C (2 mg/ml; VWR) was included in the migration assay to block cell proliferation. The distance between the leading edges of the migrating keratinocytes was measured using ImageJ software (National Institutes of Health, Bethesda) and normalised to 0 hours. Statistical analysis was performed using unpaired Student’s t-test.
Quantitative wound histomorphometry
Ex vivo wound samples (n = 5) were processed for hematoxylin and eosin (H&E) staining, and analysed using VisiCam (VWR International, UK) software. The epithelial tongue area (µm2) and length (µm) were measured and compared at day 5 post-wounding. To assess cell proliferation, the number of Ki-67+ and DAPI+ cells was counted along the basal layer of the wound epithelial tongue using ImageJ software (National Institutes of Health, Bethesda) and converted to a percentage, as previously described55. Statistical analysis was performed using unpaired Student’s t-test.
Immunofluorescence
Formalin-fixed cryosections (10 µM-thickness) were incubated with primary antibodies against Ki-67 (Abcam; 1:100), Keratin 16 (Abcam; 1:250), Keratin 17 (Abcam; 1:100) and CD49f (BD Pharmingen; 1:100) overnight followed by application of the corresponding Alexa-546 or Alexa-555-labeled antibodies (Invitrogen, UK) for 45 min at 37 °C. Cell nuclei were counterstained with DAPI (Vector Labs, UK). Image analysis was performed using a fluorescent microscope in combination with DS-C1 digital camera and ACT-2U image analysis software (Nikon).
For BrdU analysis, the keratinocytes were seeded on collagen-coated sterile glass coverslips in a 6-well cell culture dish and transfected with miR-200c inhibitor or negative control RNA. 48 h after treatment, cells were treated with 10 µM BrdU (Sigma; 2 hours; 37 °C). Next, the cells were fixed with 4% paraformaldehyde (30 min, RT) followed by denaturation in 2 M HCl (30 min, 37 °C) and neutralisation in 0.1 M sodium borate. The cells were stained with FITC-conjugated Anti-BrdU (BD Biosciences; 30 min). Cell nuclei were counterstained with DAPI (Vector Labs, UK). Fluorescent microscopy images from 10 randomly selected fields per coverslip were taken, and the numbers of DAPI+ nuclei and FITC+ nuclei were counted using ImageJ software (NIH, Bethesda, MD USA). Statistical analysis was performed using Wilcoxon rank sum test.
Flow cytometry
To assess proliferation rate, fluorescence-activated cell sorting (FACS) was performed with HaCaT cells stained with 20 ug/ml of 7-aminoactinomycin D (7-AAD) (VWR, UK; 15 min, RT) as previously described32. The percentage of cells at distinct phases of the cell cycle was analysed with a Beckman Coulter – CyAn2 ADP analyser (Beckman Coulter, UK).
References
Barrientos, S., Stojadinovic, O., Golinko, M. S., Brem, H. & Tomic-Canic, M. Growth factors and cytokines in wound healing. Wound Repair Regen 16, 585–601, doi:WRR410 (2008).
Falanga, V. Growth factors and wound healing. The Journal of dermatologic surgery and oncology 19, 711–714 (1993).
Schafer, M. & Werner, S. Transcriptional control of wound repair. Annu Rev Cell Dev Biol 23, 69–92, doi:10.1146/annurev.cellbio.23.090506.123609 (2007).
Shaw, T. J. & Martin, P. Wound repair at a glance. J Cell Sci 122, 3209–3213, doi:10.1242/jcs.031187 (2009).
Pastar, I. et al. Epithelialization in Wound Healing: A Comprehensive Review. Advances in wound care 3, 445–464, doi:10.1089/wound.2013.0473 (2014).
Jaul, E. Non-healing wounds: the geriatric approach. Arch Gerontol Geriatr 49, 224–226, doi:10.1016/j.archger.2008.08.005 (2009).
Sgonc, R. & Gruber, J. Age-related aspects of cutaneous wound healing: a mini-review. Gerontology 59, 159–164, doi:10.1159/000342344 (2013).
Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217, doi:10.1016/j.cell.2013.05.039 (2013).
Makrantonaki, E. & Zouboulis, C. C. Molecular mechanisms of skin aging: state of the art. Ann N Y Acad Sci 1119, 40–50, doi:10.1196/annals.1404.027 (2007).
Bentov, I. & Reed, M. J. Anesthesia, microcirculation, and wound repair in aging. Anesthesiology 120, 760–772, doi:10.1097/ALN.0000000000000036 (2014).
Lai, W. F. & Siu, P. M. MicroRNAs as regulators of cutaneous wound healing. J Biosci 39, 519–524 (2014).
Moura, J., Borsheim, E. & Carvalho, E. The Role of MicroRNAs in Diabetic Complications-Special Emphasis on Wound Healing. Genes (Basel) 5, 926–956, doi:10.3390/genes5040926 (2014).
Pastar, I. et al. Induction of specific microRNAs inhibits cutaneous wound healing. J Biol Chem 287, 29324–29335, doi:10.1074/jbc.M112.382135 (2012).
Ambros, V. The functions of animal microRNAs. Nature 431, 350–355, doi:10.1038/nature02871 (2004).
Andl, T. & Botchkareva, N. V. MicroRNAs (miRNAs) in the control of HF development and cycling: the next frontiers in hair research. Exp Dermatol 24, 821–826, doi:10.1111/exd.12785 (2015).
Filipowicz, W., Bhattacharyya, S. N. & Sonenberg, N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nature reviews. Genetics 9, 102–114, doi:10.1038/nrg2290 (2008).
Botchkareva, N. V. MicroRNA/mRNA regulatory networks in the control of skin development and regeneration. Cell Cycle 11, 468–474, doi:10.4161/cc.11.3.19058 (2012).
Ghatak, S. et al. Barrier Function of the Repaired Skin Is Disrupted Following Arrest of Dicer in Keratinocytes. Mol Ther 23, 1201–1210, doi:10.1038/mt201565 (2015).
Suarez, Y. et al. Dicer-dependent endothelial microRNAs are necessary for postnatal angiogenesis. Proc Natl Acad Sci USA 105, 14082–14087, doi:10.1073/pnas.0804597105 (2008).
Jin, Y. et al. MicroRNA-99 family targets AKT/mTOR signaling pathway in dermal wound healing. PloS one 8, e64434, doi:10.1371/journal.pone.0064434 (2013).
Liang, L. et al. Integrative analysis of miRNA and mRNA paired expression profiling of primary fibroblast derived from diabetic foot ulcers reveals multiple impaired cellular functions. Wound Repair Regen 24, 943–953, doi:10.1111/wrr.12470 (2016).
Liang, P. et al. MicroRNA profiling in denatured dermis of deep burn patients. Burns 38, 534–540, doi:10.1016/j.burns.2011.10.014 (2012).
Li, D. et al. MicroRNA-132 enhances transition from inflammation to proliferation during wound healing. J Clin Invest 125, 3008–3026, doi:10.1172/JCI79052 (2015).
Wang, T. et al. miR-21 regulates skin wound healing by targeting multiple aspects of the healing process. Am J Pathol 181, 1911–1920, doi:10.1016/j.ajpath.2012.08.022 (2012).
Liu, Y., Yang, D., Xiao, Z. & Zhang, M. miRNA expression profiles in keloid tissue and corresponding normal skin tissue. Aesthetic Plast Surg 36, 193–201, doi:10.1007/s00266-011-9773-1 (2012).
Sundaram, G. M. et al. ‘See-saw’ expression of microRNA-198 and FSTL1 from a single transcript in wound healing. Nature 495, 103–106, doi:10.1038/nature11890 (2013).
Yin, S. Y. et al. The Phytochemical Shikonin Stimulates Epithelial-Mesenchymal Transition (EMT) in Skin Wound Healing. Evid Based Complement Alternat Med 2013, 262796, doi:10.1155/2013/262796 (2013).
Li, D. et al. MicroRNA-31 Promotes Skin Wound Healing by Enhancing Keratinocyte Proliferation and Migration. The Journal of investigative dermatology 135, 1676–1685, doi:10.1038/jid.2015.48 (2015).
Li, H. et al. Anti-microRNA-378a enhances wound healing process by upregulating integrin beta-3 and vimentin. Mol Ther 22, 1839–1850, doi:10.1038/mt.2014.115 (2014).
van Solingen, C., Araldi, E., Chamorro-Jorganes, A., Fernandez-Hernando, C. & Suarez, Y. Improved repair of dermal wounds in mice lacking microRNA-155. J Cell Mol Med 18, 1104–1112, doi:10.1111/jcmm.12255 (2014).
Joyce, C. E. et al. Deep sequencing of small RNAs from human skin reveals major alterations in the psoriasis miRNAome. Human molecular genetics 20, 4025–4040, doi:10.1093/hmg/ddr331 (2011).
Mardaryev, A. N. et al. Micro-RNA-31 controls hair cycle-associated changes in gene expression programs of the skin and hair follicle. FASEB J 24, 3869–3881, doi:10.1096/fj.10-160663 (2010).
Wang, A. et al. MicroRNA-31 is overexpressed in cutaneous squamous cell carcinoma and regulates cell motility and colony formation ability of tumor cells. PloS one 9, e103206, doi:10.1371/journal.pone.0103206 (2014).
Yan, S. et al. NF-kappaB-induced microRNA-31 promotes epidermal hyperplasia by repressing protein phosphatase 6 in psoriasis. Nature communications 6, 7652, doi:10.1038/ncomms8652 (2015).
Xu, N. et al. MicroRNA-31 is overexpressed in psoriasis and modulates inflammatory cytokine and chemokine production in keratinocytes via targeting serine/threonine kinase 40. Journal of immunology 190, 678–688, doi:10.4049/jimmunol.1202695 (2013).
Ashcroft, G. S., Horan, M. A. & Ferguson, M. W. Aging is associated with reduced deposition of specific extracellular matrix components, an upregulation of angiogenesis, and an altered inflammatory response in a murine incisional wound healing model. The Journal of investigative dermatology 108, 430–437 (1997).
Gregory, P. A., Bracken, C. P., Bert, A. G. & Goodall, G. J. MicroRNAs as regulators of epithelial-mesenchymal transition. Cell Cycle 7, 3112–3118, doi:10.4161/cc.7.20.6851 (2008).
Stone, R. C. et al. Epithelial-mesenchymal transition in tissue repair and fibrosis. Cell Tissue Res 365, 495–506, doi:10.1007/s00441-016-2464-0 (2016).
Lewis, C. J., Mardaryev, A. N., Sharov, A. A., Fessing, M. Y. & Botchkarev, V. A. The Epigenetic Regulation of Wound Healing. Advances in wound care 3, 468–475, doi:10.1089/wound.2014.0522 (2014).
Chmielowiec, J. et al. c-Met is essential for wound healing in the skin. J Cell Biol 177, 151–162, doi:10.1083/jcb.200701086 (2007).
Davis, J., Burr, A. R., Davis, G. F., Birnbaumer, L. & Molkentin, J. D. A TRPC6-dependent pathway for myofibroblast transdifferentiation and wound healing in vivo. Dev Cell 23, 705–715, doi:10.1016/j.devcel.2012.08.017 (2012).
Koegel, H. et al. Loss of serum response factor in keratinocytes results in hyperproliferative skin disease in mice. J Clin Invest 119, 899–910, doi:10.1172/JCI37771 (2009).
Tscharntke, M. et al. Impaired epidermal wound healing in vivo upon inhibition or deletion of Rac1. J Cell Sci 120, 1480–1490, doi:10.1242/jcs.03426 (2007).
Agarwal, V., Bell, G. W., Nam, J. W. & Bartel, D. P. Predicting effective microRNA target sites in mammalian mRNAs. Elife 4, doi:10.7554/eLife.05005 (2015).
Calautti, E., Missero, C., Stein, P. L., Ezzell, R. M. & Dotto, G. P. fyn tyrosine kinase is involved in keratinocyte differentiation control. Genes Dev 9, 2279–2291 (1995).
Stojadinovic, O. et al. Deregulation of keratinocyte differentiation and activation: a hallmark of venous ulcers. J Cell Mol Med 12, 2675–2690, doi:10.1111/j.1582-4934.2008.00321.x (2008).
Usui, M. L., Mansbridge, J. N., Carter, W. G., Fujita, M. & Olerud, J. E. Keratinocyte migration, proliferation, and differentiation in chronic ulcers from patients with diabetes and normal wounds. J Histochem Cytochem 56, 687–696, doi:10.1369/jhc.2008.951194 (2008).
Lewis, C. J., Mardaryev, A. N., Sharpe, D. T. & Botchkareva, N. V. Inhibition of bone morphogenetic protein signalling promotes wound healing in a human ex vivo model. Eur J Plast Surg 38, 1–12 (2015).
Wawersik, M. J., Mazzalupo, S., Nguyen, D. & Coulombe, P. A. Increased levels of keratin 16 alter epithelialization potential of mouse skin keratinocytes in vivo and ex vivo. Molecular biology of the cell 12, 3439–3450 (2001).
Patel, G. K., Wilson, C. H., Harding, K. G., Finlay, A. Y. & Bowden, P. E. Numerous keratinocyte subtypes involved in wound re-epithelialization. The Journal of investigative dermatology 126, 497–502, doi:10.1038/sj.jid.5700101 (2006).
Kligys, K. R. et al. alpha6beta4 integrin, a master regulator of expression of integrins in human keratinocytes. J Biol Chem 287, 17975–17984, doi:10.1074/jbc.M111.310458 (2012).
Lotz, M. M. et al. Intestinal epithelial restitution. Involvement of specific laminin isoforms and integrin laminin receptors in wound closure of a transformed model epithelium. Am J Pathol 150, 747–760 (1997).
Lotz, M. M., Rabinovitz, I. & Mercurio, A. M. Intestinal restitution: progression of actin cytoskeleton rearrangements and integrin function in a model of epithelial wound healing. Am J Pathol 156, 985–996, doi:10.1016/S0002-9440(10)64966-8 (2000).
Lewis, C. J. et al. Bone morphogenetic protein signaling suppresses wound-induced skin repair by inhibiting keratinocyte proliferation and migration. The Journal of investigative dermatology 134, 827–837, doi:10.1038/jid.2013.419 (2014).
Mardaryev, A. N. et al. Lhx2 differentially regulates Sox9, Tcf4 and Lgr5 in hair follicle stem cells to promote epidermal regeneration after injury. Development 138, 4843–4852, doi:10.1242/dev.070284 (2011).
Ahmed, M. I. et al. MicroRNA-214 controls skin and hair follicle development by modulating the activity of the Wnt pathway. J Cell Biol 207, 549–567, doi:10.1083/jcb.201404001 (2014).
Ahmed, M. I., Mardaryev, A. N., Lewis, C. J., Sharov, A. A. & Botchkareva, N. V. MicroRNA-21 is an important downstream component of BMP signalling in epidermal keratinocytes. J Cell Sci 124, 3399–3404, doi:10.1242/jcs.086710 (2011).
Lichti, U., Anders, J. & Yuspa, S. H. Isolation and short-term culture of primary keratinocytes, hair follicle populations and dermal cells from newborn mice and keratinocytes from adult mice for in vitro analysis and for grafting to immunodeficient mice. Nat Protoc 3, 799–810, doi:10.1038/nprot.2008.50 (2008).
Alase, A. A. et al. IFNlambda Stimulates MxA Production in Human Dermal Fibroblasts via a MAPK-Dependent STAT1-Independent Mechanism. The Journal of investigative dermatology 135, 2935–2943, doi:10.1038/jid.2015.317 (2015).
Acknowledgements
This work was supported by the grant from the Medical Research Council UK (MR/K011324/1) to N.V. Botchkareva.
Author information
Authors and Affiliations
Contributions
E.A. and D.B. performed experiments and analysed data; E.A. wrote the manuscript; M.I.A. gave technical support; A.N.M. contributed to data interpretation; N.V.B. conceived the study, supervised its analysis and edited the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aunin, E., Broadley, D., Ahmed, M.I. et al. Exploring a Role for Regulatory miRNAs In Wound Healing during Ageing:Involvement of miR-200c in wound repair. Sci Rep 7, 3257 (2017). https://doi.org/10.1038/s41598-017-03331-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-03331-6
This article is cited by
-
Bridging tissue repair and epithelial carcinogenesis: epigenetic memory and field cancerization
Cell Death & Differentiation (2024)
-
Young fibroblast-derived exosomal microRNA-125b transfers beneficial effects on aged cutaneous wound healing
Journal of Nanobiotechnology (2022)
-
miRNA-200c-3p targets talin-1 to regulate integrin-mediated cell adhesion
Scientific Reports (2021)
-
A database on differentially expressed microRNAs during rodent bladder healing
Scientific Reports (2021)
-
A survey of miRNAs involved in biomineralization and shell repair in the freshwater gastropod Lymnaea stagnalis
Discover Materials (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.