Abstract
Cryotherapy is commonly used in the treatment of skeletal muscle injuries. However, the data to support the use of cryotherapy is inconclusive, and the biochemical etiology of cryotherapy in human skeletal muscle remains largely unknown. We therefore sought to determine how a clinically-relevant dose of cryotherapy would impact the transcriptome and metabolome of skeletal muscle. Eight healthy male subjects (age 24.7 ± 4.5 years, BMI 22.2 ± 1.6) received a 15 minute bout of local cryotherapy, delivered via ice cup massage over the anterolateral thigh. This resulted in an 85% decrease in skin temperature and a predicted 27% reduction in intramuscular temperature. The contralateral side served as a non-treated control. Two hours after cryotherapy, muscle biopsies were obtained to analyze changes in the transcriptome, metabolome, and activation of p38 MAPK, ERK1/2, Akt, and p70S6K proteins. No changes were detected in the transcriptome between control and cooled muscles. Cryotherapy reduced levels of hexose sugars and hypoxanthine by 1.3%, but no statistically different changes were observed in 60 additional metabolites. Overall, no differences in phosphorylated p38 MAPK, ERK1/2, Akt, and p70S6K were observed. A clinically relevant dose of cryotherapy produced negligible acute biochemical and molecular changes in the skeletal muscle of human subjects.
Similar content being viewed by others
Introduction
Skeletal muscle injuries are among the most prevalent types of injuries observed in the sports medicine setting1. Cryotherapy is commonly used to treat skeletal muscle injuries, and is thought to work by reducing inflammation, decreasing metabolic demands of tissue and reactive oxygen species production, and promoting the general regenerative response of tissues1,2,3. Cryotherapy is frequently administered in either a static form, such as an ice bag, for a period of 15 to 30 minutes, or in an active form, such as ice cup massage, for a period of 10 to 15 minutes1,2,3,4. These doses of cryotherapy typically result in a 3–8 °C decrease in intramuscular (IM) temperature that persist up to an hour or more after application3, 5,6,7, and are thought to alter levels of small molecule metabolites in tissue8. Despite the frequent use of topical cryotherapy, there is inconclusive evidence to support its use in the treatment of skeletal muscle injuries, and the precise biological mechanisms of action of cryotherapy are not well understood9, 10.
Numerous animal and in vitro cell culture studies have evaluated the therapeutic use of cold in experimental models of muscle injury and regeneration, and have reported mixed results of cryotherapy on outcomes associated with improved muscle regeneration11,12,13,14,15. Two limiting factors in many of these studies are that the extent of cooling is either much greater than what is typically used clinically, or that the cooling is administered via immersion of large portions of the body, resulting in alterations in core temperature and potential systemic effects that are not observed in local treatments. Numerous studies across different types of organisms and tissues have demonstrated that prolonged exposure to cold is also known to induce the expression of so-called “cold shock” genes, CIRBP, CSDE1, RBM3 and YBX1, which encode proteins important for RNA processing and stability16. These cold shock genes are also upregulated during skeletal muscle hypertrophy and may play a role in promoting muscle growth and protection of muscle fiber nuclei against apoptosis17, 18, but it was unknown whether these genes are impacted by a clinically relevant dose of cryotherapy.
As the evidence to support the use of cryotherapy is inconclusive, and the biological effects of topical cooling have not been well studied in humans, we sought to conduct a basic science study to better define the physiological effects of cryotherapy in human muscle. To accomplish this, we measured changes in the transcriptome, metabolome, and in the activation of signaling pathways involved in muscle growth following a single bout of cryotherapy. Based on the proposed mechanisms of action of cryotherapy and previous studies in animal models, and the involvement of cold shock genes in muscle hypertrophy, we hypothesized that a single therapeutic application of cold would result in wide-spread changes in the metabolome and transcriptome of muscle tissue, and activate the Akt and p70S6K signaling pathways. To test this hypothesis, we used a paired design in healthy subjects where we administered a clinically relevant dose of cryotherapy to one leg, while the other leg served as a control. Two hours after cryotherapy, biopsies were obtained from the treated and untreated muscles, and subjected to biochemical and molecular analyses.
Results
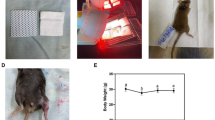
Results are presented as mean ± SD. The age of subjects in the study was 24.7 ± 4.5 years, BMI was 22.2 ± 1.6, and Tegner physical activity score was 7.3 ± 1.2. An overview of the ice cup mold, sham mold, and treatment size and area are presented in Fig. 1A–C. Using ultrasound, subjects had a subcutaneous fat thickness of 3.5 ± 2.8 mm in the region of biopsy. The measured skin temperature values are shown in Fig. 1D, and the predicted IM temperatures are shown in Fig. 1E. From the start to the end of the cooling, skin temperature decreased by 85%, resulting in a 27% predicted decrease in muscle temperature. By the time of biopsy, skin temperature remained 4% colder than the starting value, and the predicted IM temperature was 12% lower. Core temperature was 36.3 ± 0.2 °C, and for all subjects varied less than 0.2 °C throughout the study, indicating that local cryotherapy did not change core temperature.
Overview of treatment area and temperature measurements. (A) Ice cup (left) and sham mold (right). (B) Template used to create an outline of the area to apply the cryocup or sham mold. (C) Demonstration of the area of the template and biopsy location on a subject. (D) Measured skin temperature values and (E) predicted intramuscular (IM) temperature measurements. Values are mean ± SD.
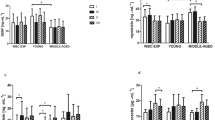
For gene expression measurements, microarray analysis failed to identify any genes that were significantly different from each other. This is also shown in the heat map of the microarray data (Fig. 2A). While no differences were detected using microarrays, using qPCR we sought to measure changes in the cold shock genes, CIRBP, CSDE1, RBM3, and YBX1, which are reported to be induced after prolonged exposure to low temperatures. No differences were detected in relative levels of cold shock genes (Fig. 2B–E).
Heat map and gene expression. (A) Heatmap of microarray data from control and cooled muscles. Targeted gene expression measurements of the cold shock genes, (B) CIRBP, (C) CSDE1, (D) RBM3 and (E) YBX1, from control and cooled muscles. The expression of each gene was normalized to the stable housekeeping gene B2M. Differences were tested using paired t-tests, and p-values are indicated in each panel. N = 8 subjects.
We then measured differences in metabolites involved in glycolytic, oxidative and amino acid metabolism (Figs 3 and 4). Of the 62 analytes measured, only hexoses (six carbon monosaccharides, which include glucose, galactose and fructose) and hypoxanthine were significantly different, both of which displayed an approximate 1.3% decrease in cooled tissue compared to controls (Fig. 4).
Metabolites without available isotopic standards. Relative abundance of 45 analytes measured in control and cooled muscles. Data were log2 transformed prior to analysis. Differences were tested using paired t-tests, and p-values are indicated in each panel. Only hexoses and hypoxanthine were significantly different between control and cooled samples. N = 8 subjects.
Finally, we measured changes in phosphorylation of proteins important in metabolism, protein synthesis, and mechanotransduction in muscle. No differences in p38 MAPK, ERK1/2, Akt, or p70S6K phosphorylation were observed between control and cooled muscles (Fig. 5A–E).
Immunoblots. Quantitative band densitometry analysis of immunoblots for (A) p-p38 MAPK, (B) p-ERK1/2, (C) p-Akt and (D) p-p70S6K from control and cooled muscles. Differences were tested using paired t-tests, and p-values are indicated in each panel. (E) Actual immunoblots of analyzed tissue. A Coomassie stained membrane is shown as a control for protein loading. N = 4 subjects.
Discussion
Cryotherapy is among the most common therapeutic modalities used to treat skeletal muscle injuries, but there is a lack of biological and epidemiological evidence to support its widespread use1, 3, 10. We hypothesized that a single therapeutic application of cold would result in wide-spread changes in the metabolome and transcriptome of muscle tissue. However, to our surprise, we found cryotherapy induced very few changes in the metabolome, and no changes in the transcriptome or in the activation of p38 MAPK, ERK1/2, Akt, or p70S6K in muscle. The combined results from this work indicate that a single dose of cryotherapy, administered following standard of care guidelines, has a negligible acute impact on gene expression, cellular metabolism, and signal transduction pathways important in muscle growth and metabolism.
There are generally two different types of cryotherapy used in sports medicine. Local cryotherapy is where ice or cold packs are administered to a specific region of a limb or defined area on the trunk or head. This type of cryotherapy, which is typically used to treat a local injury, changes the temperature of the tissue in the immediate area but does not change core temperature5, 19. Less common than topical use, cryotherapy can also be administered systemically to large portions of the body, either through immersing an entire limb or large region of the body in cold water, or exposing the area to extremely cold air. For this second type of cryotherapy, which is often used in an attempt to enhance recovery following a bout of exercise or improve training and conditioning, IM temperature can decrease by 6 °C and core temperature up to 2 °C19, 20. As core temperature is impacted by systemically administered cryotherapy, and changes in circulating levels of catecholamines and cytokines are observed in this form of cryotherapy20, 21, the mechanism of action between local and systemic cryotherapy are likely different. As such, we will largely focus our discussion on work evaluating local cryotherapy.
Previous studies have indicated that local cryotherapy, achieved through the application of an ice bag for 30 minutes to the anterior thigh, can result in a reduction of skin temperature by 80% and IM temperature, measured 2 cm deep to the subcutaneous fat layer, by 23%5, 22. Two hours after the delivery of cryotherapy in these studies, IM temperature still had not returned to pre-treatment levels. Several other studies have determined that topical cryotherapy across different anatomical sites can cool the temperature of muscle at the level which we obtained biopsies3, 5,6,7, 22. Although we did not directly measure IM temperature in this study, the skin temperature measurements and predicted IM measurements are consistent with the findings of Jutte and Merrick5, 22. Additionally, to ensure more uniform and rapid cooling of tissue, we used an ice cup approach instead of ice bags, as a previous study demonstrated ice cup therapy was 37% more effective than ice bags in reducing IM temperature in the gastrocnemius muscle23.
Much of our understanding of the basic biology of cryotherapy in skeletal muscle physiology comes from in vitro or animal studies. Decreasing the temperature of cultured muscle stem cells by 5 °C over normal culture conditions reduced their proliferation rate, but also decreased apoptosis13. When rats were subjected to an ischemia/reperfusion muscle injury, three hours of cryotherapy applied after the injury reduced neutrophil accumulation and reactive oxygen species formation, and enhanced the metabolic function of mitochondria14. In rats, following cryolesion-injury, repeated bouts of topical cryotherapy resulted in reduction of markers of reactive oxygen species formation24. In a rat muscle crush-injury model, however, 20 minutes of ice applied immediately after injury resulted in reduced inflammation in the acute phase, but in the long term delayed muscle regeneration15. Six hours of topical cryotherapy improved microcirculation and reduced immune cell infiltration after a contusion injury in rats11. Although these studies have provided useful information regarding mechanism, the dose and extent of cryotherapy is generally greater than what is typically used clinically2, and rodent muscle displays different metabolic properties and responses to environmental stimuli than human skeletal muscle25, which is why we sought to explore a clinically relevant dose of cryotherapy in human subjects.
For human skeletal muscle, locally reducing temperature generally reduces the ability of muscle to generate active tension26, 27, but little is known about how local tissue cooling affects biochemical and molecular processes in human muscle tissue28. One published model predicts a decrease of 0.17 mM of ATP and 0.54 mM of phosphocreatine per 1 °C reduction in IM temperature8, however local cooling of the quadriceps muscle group with a cuff perfused with 0 °C water for 1 hour does not change the levels of ATP, creatine or phosphocreatine29. Topical cooling also did not change ATP or phosphocreatine levels in hand muscles30. These experimental findings are consistent with the observed results in the current manuscript. Although the mechanism is not understood, hypoxanthine is released from cultured human neuronal progenitor cells in response to a hypothermic challenge31, and this might explain why hypoxanthine levels were lower in cooled muscles. Increased glucose oxidation helps to protect cultured cardiomyocytes from hypothermia-induced apoptosis32, and elevated glucose oxidation in response to cryotherapy may explain the observed decrease in hexose levels observed in the current study. In a wrist flexor eccentric injury study, topical cryotherapy increased the circulating levels of markers of muscle fiber damage, creatine kinase and myoglobin, but decreased the levels of pro-inflammatory cytokines, IL-6, IL-12, and TNF-α33. No genes related to inflammation were differentially regulated in this study. We are unaware of any studies which have looked at changes in the transcriptome of skeletal muscle in the context of cryotherapy, but lowering incubator temperature from 37 °C to 33 °C for 2 hours changed less than 1% of the transcriptome of cultured human umbilical endothelial cells34.
Cold shock genes and proteins are thought to play a role in muscle hypertrophy17, 18, and because of this, we sought to evaluate if cryotherapy induced the activation of signaling pathways involved in muscle growth. In terms of modulating signaling pathways, chronic cooling of human lung fibroblasts by adjusting the culture temperature from 37 °C to 25 °C did not impact Akt phosphorylation, but did reduce ERK1/2 phosphorylation by 50% or more35. Cooling also increased the levels of the cold shock proteins CIRBP and RBM3 in these cells by 2-fold or greater35. These cold shock proteins are also induced during skeletal muscle hypertrophy17, 18, but were not affected by cryotherapy in the current study. Overall, while there is a paucity of data regarding the therapeutic use of ice on human muscle in vivo, our findings are generally consistent with what data is available from the literature, and suggest that topical cryotherapy does not have a profound impact on the biochemistry or molecular biology of skeletal muscle.
There are several limitations to this study. We only used a single dose of cryotherapy and measured changes at one time point. We think that two hours is an appropriate time window, as other perturbations such as exercise can induce widespread transcriptional changes to skeletal muscle within two hours36. We evaluated the transcriptome and phosphorylation of selected proteins, but we did not directly measure changes in total protein abundance. We also selected p38 MAPK, ERK1/2, Akt and p70S6K as signaling proteins to evaluate based on their role in regulating numerous physiological processes in skeletal muscle37 and the correlation between cold shock genes and muscle hypertrophy16, but it is possible cryotherapy affects the activation of other signaling pathways that were not measured. We did not directly assess IM temperature for reasons discussed above, but substantial changes in skin temperature were observed in response to ice cup treatment, and several studies have shown that local cryotherapy is able to cool muscle by 3–8 °C at a depth of 2 cm3, 5,6,7. Prior to evaluating effects in injured skeletal muscle, we sought to determine how cryotherapy impacted otherwise healthy muscle. It is possible that cryotherapy could have a more profound effect on the biological processes of injured muscles. Despite these limitations, we think that this study provided an important contribution to our understanding of the mechanism of action of the therapeutic use of ice in humans.
Cryotherapy is a staple in the treatment of skeletal muscle and other soft tissue injuries1. Despite the widespread use of this modality, there is a surprising lack of evidence to support its use. This is the first work, to our knowledge, that provided a comprehensive biological evaluation of the acute effects of cryotherapy on the skeletal muscle. We observed that a clinically utilized dose of cryotherapy does not have a substantial impact on the transcriptome or metabolome of healthy muscle tissue. This largely agrees with epidemiological studies and meta-analyses which have failed to demonstrate a positive impact of cryotherapy on the treatment of skeletal muscle injuries9, 10. Given the high rates of skeletal muscle injuries in the physically active population, further work which explores the effect of cryotherapy on the cellular and molecular processes that regulate muscle repair after injury in humans is necessary to further refine the therapeutic use of cold in the sports medicine setting.
Methods
Abbreviations
A list of abbreviations is provided in Table 1.
Human Subjects
This study was approved by the University of Michigan Medical School IRB (HUM00114172) and conformed to the Declaration of Helsinki. Informed written consent was obtained from subjects prior to participation in the study. Physically active subjects who were 18–40 years of age were eligible for inclusion. Subjects were excluded if they use tobacco products, have a history of cold intolerance or urticaria, lower extremity injury, or any major medical illness, disease, myopathy, or rheumatism. A total of 8 subjects were recruited, all male.
Study Design
Subjects reported to the lab in the morning following an overnight fast, and were instructed to avoid any physical activity other than activities of daily living for 48 hours prior to testing. The height and weight of subjects were measured, and subjects then completed the Tegner physical activity survey38. Each leg of the subject was randomized to either undergo cryotherapy or serve as the sham control, so that each subject acted as their own control. A template was placed on the anterolateral thigh of the subject, and a surgical pen was used to outline the area of cooling or sham treatment. An ultrasound system (Logiq Book XP, GE Healthcare, Chicago, IL) was used to measure subcutaneous fat thickness in the outlined area, and plan for the muscle biopsy procedure.
After resting comfortably on a treatment table for 20 minutes, cryotherapy was administered to one leg through the use of a 5 cm2 reusable ice cup mold (Cryocup, Cryo Therapy, Monticello, MN). The ice cup mold had been filled with tap water and frozen overnight in a −20 °C freezer. The area of cryotherapy, as outlined by the template, was 45 cm2 and designed to accommodate two-widths of a 5 cm2 ice cup. The ice cup was swept from one end of the template to the other over a one second interval. The total time of delivery was 15 minutes. Skin temperature was measured prior to beginning cryotherapy, and every three minutes after beginning cryotherapy using an infrared dual laser thermometer (model eT650D, ennoLogic, Eugene, OR). After administering cryotherapy, temperature measurements were performed at 1 to 5 minute intervals for the next two hours. The contralateral limb was treated for 15 minutes with a sham poly-(methyl methacrylate) mold that was the same size as the ice cup. The sham mold was maintained at room temperature (24 °C), and delivered under the same conditions as the ice cup. Pilot studies determined that the sham mold did not induce a change in the temperature of skin, and surface temperature measurements therefore did not need to be performed at regular intervals on the sham side. Core temperature measurements were obtained from a Thermoscan5 tympanic membrane thermometer (Braun, Cincinnati, OH) prior to beginning cryotherapy, at the end of cryotherapy, and again at the time of biopsy.
Two hours following the administration of the ice cup or the sham therapy, a biopsy was obtained from the vastus lateralis muscle in the middle of the treatment area. The skin overlying the area was scrubbed with ChloraPrep (CareFusion, San Diego, CA) and the biopsy area was infiltrated with 3–5 mL of 1% lidocaine into the subcutaneous tissue. A 1 cm incision was made in the skin and fascia using a scalpel blade, and using ultrasound guidance a 4 g UCH muscle biopsy needle (Dixons Surgical Instruments, Wickford, UK) was placed into the muscle such that the cutting window of the needle was 2 cm deep to the subcutaneous fascia. Suction was applied, and the needle was passed to obtain a biopsy. This was repeated three times to obtain three biopsy samples. In order to be used for biochemical or molecular analyses, biopsies had to be at least 50 mg in mass. This resulted in complete sets of biopsies for all 8 subjects for metabolomics and gene expression measurements, and 4 for protein measurements. Biopsies were rapidly cleaned, weighed, snap frozen in liquid nitrogen, and stored at −80 °C. After the biopsy, the skin was closed using Dermabond (J&J, New Brunswick, NJ) reinforced with Steri-Strips (3 M, Saint Paul, MN).
Estimation of Intramuscular Temperature
The muscle fibers of the vastus lateralis are relatively long, approximately 8 cm in length39, and to ensure that the placement of a temperature probe into the muscle did not impact observed changes in the transcriptome or metabolome of muscle, we relied on surface temperature measurements to predict IM temperature values. Skin temperature measurements were obtained as described above. We then used this data to estimate IM temperature changes based on the measurements of Jutte and colleagues5, who measured skin temperature and IM temperature 2 cm deep to the subcutaneous fat layer of the anterolateral thigh of subjects who were receiving ice bag cryotherapy.
Gene Expression Measurements
RNA was isolated from muscle biopsies and gene expression was performed as previously described40, 41. Biopsies were homogenized in QIAzol (Qiagen, Valencia, CA), and RNA was isolated using a miRNeasy kit (Qiagen) supplemented with the use of DNase I (Qiagen). RNA quality was assessed using a BioAnalyzer system (Agilent, Santa Clara, CA) All samples had A260/A280 ratios greater than 1.8 and RNA integrity numbers greater than 8.0. After reverse transcription of 200 ng of RNA using iScript supermix (Bio-Rad, Hercules, CA), quantitative PCR (qPCR) was conducted in a CFX96 real time thermal cycler using iTaq SYBR green supermix reagents (Bio-Rad). A list of RNA transcripts and primer sequences is provided in Supplementary Table S1. The 2−ΔCt technique was used to normalize the expression of mRNA transcripts to the stable housekeeping gene β2-microglobulin (B2M), and differences between cooled and sham muscles were tested using paired t-tests (α = 0.05) in Prism version 7.0 (GraphPad Software, La Jolla, CA).
Microarray measurements were performed by the University of Michigan DNA Sequencing Core following manufacturer recommendations as previously described40. A total of 100 ng of RNA from cooled and control muscle biopsies of 6 subjects were analyzed. RNA was prepared for microarray analysis using a Pico WTA system (NuGen, San Carlos, CA) and hybridized to Human Gene ST 2.1 strips (Affymetrix). Expression values were calculated using a robust multi-array average, and data were log2 transformed. Differences between control and cooled samples were calculated using paired t-tests (α = 0.05), and p-values were adjusted for multiple comparisons using a Benjamini-Hochberg false discovery rate of 0.05. ArrayStar version 13 (DNASTAR, Madison, WI) was used generate a heat map using hierarchical clustering with Euclidean clustering and centroid linkage. The microarray dataset has been uploaded to the NIH GEO database (accession ID GSE89097).
Metabolomics
Metabolomics measurements were performed by the University of Michigan Metabolomics Core as previously described42. Muscle biopsies were homogenized and metabolites extracted using an 8:1:1 mixture of methanol, chloroform and water containing isotope-labeled internal standards. Liquid chromatography-mass spectrometry analysis was performed in an Agilent system consisting of a 1260 UPLC module coupled with a 6520 quadrupole-time-of-flight mass spectrometer. Metabolites were separated using a 150 × 1 mm Luna NH2 hydrophillic interaction chromatography column (Phenomenex, Torrance, CA). The mass spectrometer was operated in electrospray ionization mode, and data were processed using MassHunter software (Agilent). Metabolites were either normalized to the nearest isotope-labeled internal standard and quantitated using two replicated injections of five standards to create a linear calibration curve with accuracy better than 80% for each standard, or normalized to the nearest internal standard, and the peak areas were used for differential analysis. MetaboAnalyst 3.0 software (McGill University, Montreal, QC) was then used to log2-transform data and perform paired t-tests (α = 0.05) on the analyzed data. Because isotopic standards were available for some but not all of the metabolites, the data were log2 transformed and analyzed separately between the two groups, and presented in two figures.
Immunoblots
Immunoblots were performed as previously described40, 43. Muscle biopsies were homogenized in ice cold RIPA Lysis and Extraction Buffer (ThermoFisher Scientific, Grand Island, NY) supplemented with a 1:100 protease and phosphatase inhibitor cocktail (ThermoFisher Scientific). After homogenization and sonication, lysates were spun at 13,000 × g, and the supernatants were collected. Protein concentration of samples was measured using a BCA Protein Assay Kit (ThermoFisher Scientific). Samples were then diluted in Laemmli sample buffer (Bio-Rad), placed in boiling water for 2 minutes, and 20 µg of protein was separated on AnyKD gels (Bio-Rad). Proteins were transferred to either nitrocellulose (p38 MAPK and Akt) or PVDF (ERK1/2 and p70S6K) membranes using the Trans-Blot SD semi-dry transfer apparatus (Bio-Rad), blocked with 5% bovine serum albumin, and incubated with rabbit primary antibodies (1:1000) from Cell Signaling Technology (Danvers, MA) against phosphorylated Akt (T308, catalog # 13808), phosphorylated ERK1/2 (T202/Y204, catalog # 4511), phosphorylated p38 MAPK (T180/Y182, catalog # 9211), phosphorylated p70S6K (T389, catalog # 9430). Primary antibodies were detected using a goat anti-rabbit HRPO conjugated antibody (catalog # 7074, Cell Signaling Technology) at a concentration of 1:10000, along with Clarity enhanced chemiluminescent detection reagents (Bio-Rad). Membranes were imaged using a ChemiDoc imaging system (Bio-Rad), and band densitometry was calculated in relative units (RU). Following detection, membranes were stained with Coomassie Brilliant Blue to verify equal protein loading.
References
Järvinen, T. A. H., Järvinen, T. L. N., Kääriäinen, M., Kalimo, H. & Järvinen, M. Muscle injuries: biology and treatment. Am J Sports Med 33, 745–764, doi:10.1177/0363546505274714 (2005).
Nolan, T. P. & Michlovitz, S. L. Modalities for therapeutic intervention. (F.A. Davis Co, 2005).
Dykstra, J. H. et al. Comparisons of cubed ice, crushed ice, and wetted ice on intramuscular and surface temperature changes. J Athl Train 44, 136–141, doi:10.4085/1062-6050-44.2.136 (2009).
MacAuley, D. Do textbooks agree on their advice on ice? Clin J Sport Med 11, 67–72, doi:10.1097/00042752-200104000-00001 (2001).
Jutte, L. S., Merrick, M. A., Ingersoll, C. D. & Edwards, J. E. The relationship between intramuscular temperature, skin temperature, and adipose thickness during cryotherapy and rewarming. Archives of physical medicine and rehabilitation 82, 845–850, doi:10.1053/apmr.2001.23195 (2001).
Rupp, K. A., Herman, D. C., Hertel, J. & Saliba, S. A. Intramuscular temperature changes during and after 2 different cryotherapy interventions in healthy individuals. J Orthop Sports Phys Ther 42, 731–737, doi:10.2519/jospt.2012.4200 (2012).
Hunter, E. J., Ostrowski, J., Donahue, M., Crowley, C. & Herzog, V. Effect of Salted Ice Bags on Surface and Intramuscular Tissue Cooling and Rewarming Rates. J Sport Rehabil 25, 70–76, doi:10.1123/jsr.2014-0289 (2016).
Binzoni, T., Hiltbrand, E., Terrier, F., Cerretelli, P. & Delpy, D. Temperature dependence of human gastrocnemius pH and high-energy phosphate concentration by noninvasive techniques. Magn Reson Med 43, 611–614, doi:10.1002/(ISSN)1522-2594 (2000).
Hubbard, T. J., Aronson, S. L. & Denegar, C. R. Does Cryotherapy Hasten Return to Participation? A Systematic Review. J Athl Train 39, 88–94 (2004).
Torres, R., Ribeiro, F., Alberto Duarte, J. & Cabri, J. M. H. Evidence of the physiotherapeutic interventions used currently after exercise-induced muscle damage: systematic review and meta-analysis. Phys Ther Sport 13, 101–114, doi:10.1016/j.ptsp.2011.07.005 (2012).
Schaser, K.-D. et al. Prolonged superficial local cryotherapy attenuates microcirculatory impairment, regional inflammation, and muscle necrosis after closed soft tissue injury in rats. Am J Sports Med 35, 93–102, doi:10.1177/0363546506294569 (2007).
Vieira Ramos, G. et al. Cryotherapy Reduces Inflammatory Response Without Altering Muscle Regeneration Process and Extracellular Matrix Remodeling of Rat Muscle. Sci Rep 6, 18525, doi:10.1038/srep18525 (2016).
Harding, R. L. et al. The effect of temperature on apoptosis and adipogenesis on skeletal muscle satellite cells derived from different muscle types. Physiol Rep 3, e12539, doi:10.14814/phy2.12539 (2015).
Puntel, G. O. et al. Cryotherapy reduces skeletal muscle damage after ischemia/reperfusion in rats. J Anat 222, 223–230, doi:10.1111/joa.2013.222.issue-2 (2013).
Takagi, R. et al. Influence of icing on muscle regeneration after crush injury to skeletal muscles in rats. J Appl Physiol 110, 382–388, doi:10.1152/japplphysiol.01187.2010 (2011).
Mihailovich, M., Militti, C., Gabaldón, T. & Gebauer, F. Eukaryotic cold shock domain proteins: highly versatile regulators of gene expression. Bioessays 32, 109–118, doi:10.1002/bies.200900122 (2010).
Dupont-Versteegden, E. E. et al. Identification of cold-shock protein RBM3 as a possible regulator of skeletal muscle size through expression profiling. Am J Physiol Regul Integr Comp Physiol 295, R1263–73, doi:10.1152/ajpregu.90455.2008 (2008).
Ferry, A. L., Vanderklish, P. W. & Dupont-Versteegden, E. E. Enhanced survival of skeletal muscle myoblasts in response to overexpression of cold shock protein RBM3. AJP - Cell Physiology 301, C392–402, doi:10.1152/ajpcell.00098.2011 (2011).
White, G. E. & Wells, G. D. Cold-water immersion and other forms of cryotherapy: physiological changes potentially affecting recovery from high-intensity exercise. Extrem Physiol Med 2, 26, doi:10.1186/2046-7648-2-26 (2013).
Hausswirth, C. et al. Parasympathetic activity and blood catecholamine responses following a single partial-body cryostimulation and a whole-body cryostimulation. PLoS ONE 8, e72658, doi:10.1371/journal.pone.0072658 (2013).
Roberts, L. A., Nosaka, K., Coombes, J. S. & Peake, J. M. Cold water immersion enhances recovery of submaximal muscle function after resistance exercise. Am J Physiol Regul Integr Comp Physiol 307, R998–R1008, doi:10.1152/ajpregu.00180.2014 (2014).
Merrick, M. A., Jutte, L. S. & Smith, M. E. Cold Modalities With Different Thermodynamic Properties Produce Different Surface and Intramuscular Temperatures. J Athl Train 38, 28–33 (2003).
Zemke, J. E., Andersen, J. C., Guion, W. K., McMillan, J. & Joyner, A. B. Intramuscular temperature responses in the human leg to two forms of cryotherapy: ice massage and ice bag. J Orthop Sports Phys Ther 27, 301–307, doi:10.2519/jospt.1998.27.4.301 (1998).
Siqueira, A. F. et al. Multiple cryotherapy applications attenuate oxidative stress following skeletal muscle injury. Redox Rep. 1–7, doi:10.1080/13510002.2016.1239880 (2016).
Kowalski, G. M. & Bruce, C. R. The regulation of glucose metabolism: implications and considerations for the assessment of glucose homeostasis in rodents. Am J Physiol Endocrinol Metab 307, E859–71, doi:10.1152/ajpendo.00165.2014 (2014).
Davies, C. T. & Young, K. Effect of temperature on the contractile properties and muscle power of triceps surae in humans. Journal of applied physiology: respiratory, environmental and exercise physiology 55, 191–195 (1983).
Ranatunga, K. W., Sharpe, B. & Turnbull, B. Contractions of a human skeletal muscle at different temperatures. J Physiol (Lond) 390, 383–395, doi:10.1113/jphysiol.1987.sp016707 (1987).
Binzoni, T. & Delpy, D. Local temperature changes and human skeletal muscle metabolism. J Physiol Anthropol Appl Human Sci 20, 159–174, doi:10.2114/jpa.20.159 (2001).
Starkie, R. L., Hargreaves, M., Lambert, D. L., Proietto, J. & Febbraio, M. A. Effect of temperature on muscle metabolism during submaximal exercise in humans. Exp Physiol 84, 775–784, doi:10.1111/eph.1999.84.issue-4 (1999).
Wade, A. J. et al. Influence of muscle temperature during fatiguing work with the first dorsal interosseous muscle in man: a 31P-NMR spectroscopy study. Eur J Appl Physiol 81, 203–209, doi:10.1007/s004210050031 (2000).
Dalen, M. L., Frøyland, E., Saugstad, O. D., Mollnes, T. E. & Rootwelt, T. Post-hypoxic hypothermia is protective in human NT2-N neurons regardless of oxygen concentration during reoxygenation. Brain Res. 1259, 80–89, doi:10.1016/j.brainres.2008.12.055 (2009).
Wheeler, T. J., Wiegand, C. B. & Chien, S. Fructose-1,6-bisphosphate enhances hypothermic preservation of cardiac myocytes. J. Heart Lung Transplant. 24, 1378–1384, doi:10.1016/j.healun.2004.08.013 (2005).
Tseng, C.-Y. et al. Topical cooling (icing) delays recovery from eccentric exercise-induced muscle damage. J Strength Cond Res 27, 1354–1361, doi:10.1519/JSC.0b013e318267a22c (2013).
Yang, D. et al. Transcriptomic analysis of mild hypothermia-dependent alterations during endothelial reperfusion injury. Cell Physiol Biochem 25, 605–614, doi:10.1159/000315079 (2010).
Neutelings, T., Lambert, C. A., Nusgens, B. V. & Colige, A. C. Effects of mild cold shock (25 °C) followed by warming up at 37 °C on the cellular stress response. PLoS ONE 8, e69687, doi:10.1371/journal.pone.0069687 (2013).
Louis, E., Raue, U., Yang, Y., Jemiolo, B. & Trappe, S. Time course of proteolytic, cytokine, and myostatin gene expression after acute exercise in human skeletal muscle. J Appl Physiol 103, 1744–1751, doi:10.1152/japplphysiol.00679.2007 (2007).
Gumucio, J. P., Sugg, K. B. & Mendias, C. L. TGF-β Superfamily Signaling in Muscle and Tendon Adaptation to Resistance Exercise. Exerc Sport Sci Rev 43, 93–99, doi:10.1249/JES.0000000000000041 (2015).
Tegner, Y. & Lysholm, J. Rating systems in the evaluation of knee ligament injuries. Clin Orthop Relat Res 43–49, 10.1097/00003086-198509000-00007 (1985).
Friederich, J. & Brand, R. Muscle fiber architecture in the human lower limb. J Biomech 23, 91–95, doi:10.1016/0021-9290(90)90373-B (1990).
Hudgens, J. L. et al. Platelet-Rich Plasma Activates Proinflammatory Signaling Pathways and Induces Oxidative Stress in Tendon Fibroblasts. Am J Sports Med 44, 1931–1940, doi:10.1177/0363546516637176 (2016).
Mendias, C. L. et al. Changes in skeletal muscle and tendon structure and function following genetic inactivation of myostatin in rats. J Physiol (Lond) 593, 2037–2052, doi:10.1113/jphysiol.2014.287144 (2015).
Harfmann, B. D. et al. Muscle-specific loss of Bmal1 leads to disrupted tissue glucose metabolism and systemic glucose homeostasis. Skelet Muscle 6, 12, doi:10.1186/s13395-016-0082-x (2016).
Gumucio, J. P., Flood, M. D., Phan, A. C., Brooks, S. V. & Mendias, C. L. Targeted inhibition of TGF-β results in an initial improvement but long-term deficit in force production after contraction-induced skeletal muscle injury. J Appl Physiol 115, 539–545, doi:10.1152/japplphysiol.00374.2013 (2013).
Acknowledgements
This work was supported by NIH grants F32-AR067086 and U24-DK097153. We would like to thank Professor Charles Burant for helpful discussion regarding the analysis of metabolomics data.
Author information
Authors and Affiliations
Contributions
D.S., K.S., T.A. and C.M. designed the study and wrote the manuscript. D.S., K.S., N.D., E.S.E., T.A., and C.M. performed experiments. D.S., K.S., N.D. and C.M. performed data analysis. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sarver, D.C., Sugg, K.B., Disser, N.P. et al. Local cryotherapy minimally impacts the metabolome and transcriptome of human skeletal muscle. Sci Rep 7, 2423 (2017). https://doi.org/10.1038/s41598-017-02754-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02754-5
This article is cited by
-
A reference single-cell transcriptomic atlas of human skeletal muscle tissue reveals bifurcated muscle stem cell populations
Skeletal Muscle (2020)
-
Thermoelectric Heat Patch for Clinical and Self-Management: Melanoma Excision Wound Care
Annals of Biomedical Engineering (2019)
-
Multiple Cold-Water Immersions Attenuate Muscle Damage but not Alter Systemic Inflammation and Muscle Function Recovery: A Parallel Randomized Controlled Trial
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.