Abstract
Glucokinase-mediated glucose signaling induces insulin secretion, proliferation, and apoptosis in pancreatic β-cells. However, the precise molecular mechanisms underlying these processes are not clearly understood. Here, we demonstrated that glucokinase activation using a glucokinase activator (GKA) significantly upregulated the expression of Fibulin-5 (Fbln5), a matricellular protein involved in matrix-cell signaling, in isolated mouse islets. The islet Fbln5 expression was induced by ambient glucose in a time- and dose-dependent manner and further enhanced by high-fat diet or the deletion of insulin receptor substrate 2 (IRS-2), whereas the GKA-induced increase in Fbln5 expression was diminished in Irs-2-deficient islets. GKA-induced Fbln5 upregulation in the islets was blunted by a glucokinase inhibitor, KATP channel opener, Ca2+ channel blocker and calcineurin inhibitor, while it was augmented by harmine, a dual-specificity tyrosine phosphorylation-regulated kinase (DYRK) 1 A inhibitor. Although deletion of Fbln5 in mice had no significant effects on the glucose tolerance or β-cell functions, adenovirus-mediated Fbln5 overexpression increased glucose-stimulated insulin secretion in INS-1 rat insulinoma cells. Since the islet Fbln5 expression is regulated through a glucokinase/KATP channel/calcineurin/nuclear factor of activated T cells (NFAT) pathway crucial for the maintenance of β-cell functions, further investigation of Fbln5 functions in the islets is warranted.
Similar content being viewed by others
Introduction
Glucose metabolism plays an important role in normal β-cell functions such as insulin production and insulin secretion, and also in β-cell growth and survival1, 2. Glucose signaling in the pancreatic β-cells has also been shown to be involved in β-cell proliferation in both humans and rodents3,4,5,6. Glucokinase, a member of the hexokinase family, is the predominant enzyme catalyzing the phosphorylation of glucose in the pancreatic β-cells and the liver. Glucokinase acts as a glucose sensor for insulin secretion from the pancreatic β-cells7 and is required for the effects of glucose signaling on β-cell proliferation8. Heterozygous inactivating mutations of glucokinase cause type 2 maturity onset diabetes of the young (MODY2), and homozygous or compound heterozygous inactivating glucokinase mutations cause a more severe phenotype known as permanent neonatal diabetes mellitus (PNDM), which manifests at birth9. On the other hand, heterozygous activating glucokinase mutations cause persistent hyperinsulinemic hypoglycemia (PHHI)10, associated with increased β-cell mass and β-cell proliferation11. We have shown previously that glucokinase activation ameliorates endoplasmic reticulum (ER) stress-mediated apoptosis of the pancreatic β-cells12, while another report revealed that genetic activation of β-cell glucokinase causes cell apoptosis associated with DNA double-strand breaks and activation of the tumor suppressor protein p5313. Thus, glucokinase appears to play important roles in β-cell function, replication, and survival. These findings inspired the development of a therapeutic strategy for diabetes by targeting glucokinase. Glucokinase activators (GKAs) increase the glucose affinity and maximum velocity (Vmax) of glucokinase, leading to enhanced glucose-induced insulin secretion from the islets and enhanced hepatic glucose uptake14. This ability suggests a potential pharmacological role of GKAs in the treatment of diabetes. However, further investigation is needed to determine the efficacy and safety of GKAs; for example, downstream targets of glucose metabolism in the β-cells have not yet been clearly revealed.
Fibulin-5 (Fbln5; also known as EVEC or DANCE), a matricellular protein, is essential for elastic fiber assembly15, 16. Fbln5 is secreted by various cell types, including vascular smooth muscle cells (SMCs), fibroblasts and endothelial cells. Fbln5 expression is usually downregulated after birth, but reactivated upon tissue injury17, 18. Fbln5 has several non-elastogenic functions, for example, regulation of proteases via its integrin-binding domain19,20,21,22. Fbln5 has also been shown to bind to the α5β1 fibronectin receptor and the β1 integrin21, 23. Indeed, Fbln5 plays critical roles in cell proliferation, migration and invasion of certain tumors and smooth muscle cells24, 25. Mice lacking in Fbln5 exhibit systemic elastic fiber defects, including loose skin, tortuous aorta, emphysematous lungs, and genital prolapse16, 26. However, the precise nature of the involvement of Fbln5 in metabolism remains unknown.
In this study, we found that treatment with a GKA induced Fbln5 gene expression in mouse pancreatic islets. Although it has been reported that interaction of the islets with some specific extracellular matrix molecules is important for islet/β-cell survival27, 28, the precise expression levels and roles of these molecules in the pancreatic islets and β-cell functions remain obscure. In this study, we focused on the regulation of Fbln5 expression in the pancreatic β-cells.
Results
Glucokinase activation induced Fbln5 expression in the pancreatic islets
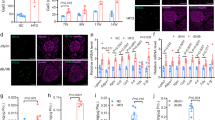
At first, we identified by gene expression microarray analysis (GSE41248), that stimulation of mouse pancreatic islets with a GKA for 24 hours induced Fbln5 expression in the islets (12.6-fold enhanced expression as compared to that in the vehicle control; p = 0.0043)12. To validate this upregulation of Fbln5 expression by treatment with a GKA in mouse pancreatic islets, we investigated Fbln5 mRNA expression in isolated islets from C57BL/6 J mice. Consistent with the results of the microarray analysis, the Fbln5 mRNA expression in the isolated islets was significantly increased, in a time-dependent manner, by treatment with a GKA (Fig. 1a). Ambient glucose also induced Fbln5 expression in the islets in a concentration-dependent manner (Fig. 1b). We detected FBLN5 protein expression in the wild-type mouse islets, as well as in INS-1 rat insulinoma cell line (Fig. 1c and d) but not in the Fbln5-deficient (Fbln5 −/−) islets (Fig. 1c). The treatment with a GKA also increased FBLN5 protein expression levels in INS-1 cells (Fig. 1d). Moreover, in glucokinase hetero-deficient (Gck +/−) mouse islets, GKA-stimulated Fbln5 mRNA expression levels were reduced as compared to those in the islets from wild-type mice (Fig. 1e). No difference was detected in Fbln5 mRNA expression levels between vehicle-treated Gck +/− islets and the wild-type islets (p = 0.357) (Fig. 1e). These results suggest that Fbln5 expression is induced by glucokinase activation in the pancreatic islets. Furthermore, the GKA-induced increase in Fbln5 expression was more pronounced in the islets of mice reared on a high-fat diet for 20 weeks than in the islets of standard chow-fed mice (Fig. 1f), although there were no significant differences between the vehicle-treated islets from standard chow-fed and high-fat diet-fed mice (p = 0.24), consistent with the report that glucokinase-mediated signaling in the β-cells is activated by a high-fat diet8, 29. In contrast, in insulin receptor substrate 2 (IRS-2)-deficient (Irs-2 −/−) mouse islets, basal Fbln5 expression was significantly increased compared with those of wild-type mice (Fig. 1g). However, the response of Fbln5 induction to GKA was almost abolished in Irs-2 −/− mouse islets (Fig. 1g). It may also explain the more pronounced upregulation of islet Fbln5 expression in high-fat diet-fed mice than in normal chow-fed mice, as GKA is known to induce IRS-2 expression in the β-cells of mice reared on a high-fat diet8. The lack of Fbln5 induction in Irs-2 −/− islets suggests that IRS-2 is involved in the GKA-induced upregulation of islet Fbln5 expression. Moreover, we found that Fbln5 was strongly expressed in the islets of 2-week-old pre-weaning mice, the expression level decreasing by 6 or 12 weeks of age (Fig. 1h). This expression pattern of Fbln5 is consistent with the expression of the proliferation marker Ki67 in the islets (Fig. 1i).
Glucokinase activation was associated with upregulation of Fbln5 gene expression in pancreatic islets. (a) Fbln5 mRNA expression levels in isolated islets from C57BL/6 J mice incubated for 2, 6, 12 and 24 hours with 30 μmol/L of GKA CpdA or vehicle (DMSO) (n = 4). (b) Fbln5 mRNA expression levels in isolated islets from C57BL/6 J mice (n = 4) after 24 hours’ incubation with 2.8, 5.6, 11.1 or 22.2 mmol/L of glucose. (c) Immunoblotting for FBLN5 in islets isolated from wild-type or Fbln5 −/− mice. Full-length blots are presented in Supplementary Fig. 4. (d) Immunoblotting for FBLN5 in INS-1 cells treated with vehicle, 22.2 mmol/L of glucose, or 30 μmol/L of GKA CpdA for 24 hours (n = 4). Full-length blots are presented in Supplementary Fig. 4. (e) Fbln5 mRNA expression levels in islets from 12-week-old Gck +/− mice and their wild-type littermates after incubation with 30 μmol/L of GKA CpdA for 24 hours (n = 4). (f) Fbln5 gene expression levels in islets isolated from 12-week old standard chow-fed mice (SC) and 20-week of high-fat diet (HFD)-fed mice incubated with or without 30 μmol/L of GKA CpdA (vehicle; DMSO) for 24 hours (n = 4). (g) Fbln5 mRNA expression levels in islets from 12-week-old Irs-2 −/− mice and their wild-type littermates after incubation with 30 μmol/L of GKA CpdA for 24 hours (n = 4). (h–i) mRNA expression levels of Fbln5 (h) or Ki67 (i) in isolated islets from 2-, 6- and 12-week-old C57BL/6 J mice (n = 3–4). Data are represented as means ± SEM. *p < 0.05, **p < 0.01.
Glucokinase/KATP channel/calcineurin/NFAT signaling is required for glucose-mediated Fbln5 expression in islets
We next assessed the signaling pathways underlying the GKA-induced upregulation of Fbln5 in the pancreatic islets. Treatment with D-mannoheptulose, a specific inhibitor of glucokinase, completely abolished the GKA-induced upregulation of Fbln5 in the pancreatic islets (Fig. 2a). In addition, treatment with diazoxide, a KATP channel (ATP-sensitive potassium channel) opener, also suppressed the GKA-induced elevation of Fbln5 expression in the islets (Fig. 2b). Treatment with OSI-906, a dual insulin and IGF-1 receptor inhibitor, did not reduce the Fbln5 induction by GKA, but enhanced it (Fig. 2c). These results imply that an influx of Ca2+ into the β-cells via depolarization of the plasma membrane accompanied by the closure of KATP channel, and not the autocrine action of insulin, is involved in the GKA-induced upregulation of Fbln5 in the pancreatic islets.
Glucose-signal induced Fbln5 upregulation via the glucokinase/KATP channel/calcineurin/NFAT signaling pathway in pancreatic islets. (a–f) Fbln5 mRNA expression. Islets from C57BL/6 J mice were incubated with 10 nmol/L of D-mannoheptulose (MH) (n = 4) (a), 200 μmol/L of diazoxide (n = 4) (b), 200 nmol/L of OSI-906 (n = 4) (c), 50 μmol/L of nifedipine (ND), 10 μmol/L of FK506 (n = 3) (d), 10 μmol/L of harmine (n = 3) (e), or a combination of 10 μmol/L of FK506 and 10 μmol/L of harmine (n = 4) (f) for 24 hours in the presence or absence of 30 μmol/L of GKA CpdA (vehicle; DMSO). Data are represented as means ± SEM. *p < 0.05, **p < 0.01.
Calcineurin is activated in an intracellular Ca2+-dependent manner30, leading to NFAT activation by dephosphorylation and subsequent translocation of NFAT from the cytosol to the nucleus31. Glucose-induced regulation of Irs-2 expression has been reported to be mediated via this Ca2+/calcineurin/NFAT signaling in the pancreatic β-cells32. Hence, we evaluated the effects of a Ca2+ channel blocker, a calcineurin inhibitor, and a DYRK1A inhibitor on the upregulation of Fbln5 in the islets treated with a GKA. Blockade of the L-type voltage-dependent Ca2+ channels (L-type VDCCs) with nifedipine in isolated mouse islets abrogated the GKA-induced increase in Fbln5 expression in the islets (Fig. 2d). Moreover, treatment with FK506, which specifically inhibits calcineurin activity, also almost completely abolished the GKA-induced increase in Fbln5 expression in the pancreatic islets (Fig. 2d). Dual-specificity tyrosine phosphorylation-regulated kinases (DYRKs), including DYRK1A, inactivate the NFAT1 proteins by phosphorylating its SP-3 motif33. Notably, harmine, a DYRK1A inhibitor, enhanced the Fbln5 expression induced by treatment with a GKA (Fig. 2e). The effect of harmine on the increment in Fbln5 expression in islets was blunted in the presence of FK506 (Fig. 2f). These results suggest that the transcriptional regulation of Fbln5 in the islets is mediated by glucose signaling and downstream Ca2+/calcineurin/NFAT signaling.
Fbln5 −/− mice exhibited normal glucose tolerance and normal glucose-stimulated insulin secretion and β-cell proliferation evoked by GKA
To investigate the role of Fbln5 in glucose metabolism and insulin secretion, we used 8- to 12-week-old Fbln5 knockout (Fbln5 −/−) mice16 to evaluate whether Fbln5 deletion may influence glucose homeostasis in vivo. Fbln5 −/− mice showed normal glucose tolerance and comparable insulin secretion during an oral glucose tolerance test (Fig. 3a and b). No significant difference in glucose-stimulated insulin secretion was observed between islets isolated from Fbln5 −/− mice and wild-type mice (Fig. 3c). These results imply that Fbln5 has no effect on insulin secretion in healthy young adult mice.
Fbln5 deficiency in mice had no effect on glucose tolerance, glucose-stimulated insulin secretion or β-cell proliferation. (a) Blood glucose levels during an oral glucose tolerance test (OGTT) in 8-week-old Fbln5 −/− mice and wild-type mice (1.5 g/kg body weight, n = 11–13). (b) Serum insulin levels during the OGTT (n = 11–13). (c) Glucose-stimulated insulin secretion in ten isolated islets from 8-week-old Fbln5 −/− mice and wild-type mice (n = 6–12). (d,e) β-cell mass in 8-week-old Fbln5 −/− mice and wild-type mice (n = 5). Representative images of the pancreas showing brown staining of insulin (d) and the ratio of the β-cell mass relative to the area of the whole pancreas (e) (n = 5). The scale bar represents 100 µm in the pancreas images. (f,g) EdU incorporation in islets isolated from 8-week-old C57BL/6 J mice. Representative images of pancreatic islets after 48 hours of incubation with EdU in the presence or absence of 30 μmol/L of GKA CpdA (f). Insulin is stained red, nuclei are stained blue with 4′, 6-diamidino-2-phenylindole [DAPI], and EdU-positive nuclei are stained green. The white arrows indicate EdU-positive β-cells. The scale bar represents 50 µm. The ratio of EdU-positive β-cells to the total count of insulin-positive β-cells is shown (g). More than 3000 cells were counted in each group (n = 2). Data are represented as means ± SEM. *p < 0.05.
We next assessed the β-cell mass in the 8- to 12-week-old Fbln5 −/− mice. No significant differences in the islet morphology and the β-cells area relative to the total pancreatic area were observed between the wild-type mice and Fbln5 −/− mice (Fig. 3d and e). Furthermore, we evaluated the GKA-induced β-cell proliferation activity in Fbln5 −/− and wild-type islets. Treatment with GKA for 48 hours markedly increased the EdU-incorporated proliferating insulin-positive β-cells to a similar extent in the islets isolated from both genotypes of mice (Fig. 3f and g). On the other hand, the fluorescent intensity of insulin was significantly increased in the GKA-treated islets compared with the vehicle-treated islets in wild-type mice, but not in Fbln5 −/− mice (see Supplementary Fig. S1a). This result in islets from wild-type mice is consistent with the observation that glucokinase activation enhances insulin gene expression and insulin secretion in β-cells12, 34. However, GKA-induced insulin secretion was not decreased in Fbln5 −/− islets compared with wild-type islets (see Supplementary Fig. S1b). Insulin content in GKA-treated Fbln5 −/− islets showed a tendency to be decreased compared to wild-type islets, but it did not reach statistical significance (see Supplementary Fig. S1b). Thus, Fbln5 is not required for the development and maintenance of β-cell function or proliferation.
In immunohistochemical analysis of paraffin-embedded endocrine pancreatic tissue from 8-week-old wild-type mice and Fbln5 −/− mice, FBLN5 was seemed to be around CD34 (endothelial marker) -positive interstitial tissue, but not in β-cells, α-cells, or δ-cells in the islets (see Supplementary Fig. S2). Next, fetal pancreatic tissue paraffin sections from wild-type mice at the age of embryonic day 15 were immunostained for FBLN5. The area and intensity of the FBLN5 signal in the islets seemed to be more abundant compared with those in adult mice (see Supplementary Fig. S3a). Then, we used non-paraffinized cultured islets from 8-week-old wild-type mice for immunostaining. Notably, FBLN5-positive β-cells were detectable in non-paraffinized adult wild-type islets (see Supplementary Fig. S3b). In addition, FBLN5 is observed at cytoplasmic granular structures in INS-1 cells, as shown in Supplementary Fig. S3c, suggesting that FBLN5 is also expressed in β-cells.
Adenovirus-mediated Fbln5 overexpression increased glucose-stimulated insulin secretion and inhibited cell proliferation in INS-1 cells
Next, we evaluated the properties of Fbln5 by forced expression of Fbln5 gene expression using an adenovirus vector (Ad-Fbln5) in INS-1 cells. Following adenovirus-mediated infection of Ad-Fbln5 in INS-1 cells, overexpression of Fbln5 was confirmed by measuring the mRNA and protein expression levels (Fig. 4a and b). The cells overexpressing Fbln5 showed enhanced insulin secretion in the presence of 11.1 mmol/L glucose as compared to the control cells (1.6-fold, p = 0.017), although basal insulin secretion was not significantly different between the Ad-Fbln5- and Ad-GFP- infected INS-1 cells (Fig. 4c). The effects of Fbln5 overexpression on the cell proliferation activity was evaluated by measuring the EdU incorporation and Ki67 expression in Ad-Fbln5-infected INS-1 cells. We were almost able to ignore the GFP-signals when we adjusted the gain of signals according to the fluorescent intensity of EdU. (Fig. 4d left panel). The ratio of EdU-incorporated proliferating INS-1 cells to the total count of INS-1 cells tended to be decreased in the Ad-Fbln5-infected cells as compared with that in the control cells (Fig. 4d). In addition, Ad-Fbln5-infected INS-1 cells showed significant reduction in the Ki67 expression (Fig. 4e). These results indicate that overexpression of Fbln5 enhances insulin secretion whereas decreases cell proliferation in β-cells.
Fbln5 overexpression in INS-1 cells enhanced glucose-stimulated insulin secretion, but inhibited β-cell proliferation. (a) Fbln5 mRNA levels in INS-1 cells after infection with Ad-GFP or Ad-Fbln5 for 48 hours (n = 2). (b) Immunoblotting for FBLN5 in INS-1 cells infected with Ad-GFP or Ad-Fbln5. Full-length blots are presented in Supplementary Fig. 4. (c) Glucose-stimulated insulin secretion by INS-1 cells infected with Ad-GFP or Ad-Fbln5 (n = 6–12). (d) EdU incorporation in INS-1 cells infected with Ad-GFP or Ad-Fbln5 for 48 hours. Cells were incubated for 3 hours with 10 µM of EdU after adenovirus infection. left: Representative images of Ad-GFP infected INS-1 cells or Ad-Fbln5 infected INS-1 cells stained for EdU. Ad-GFP infected cells incubated without EdU are shown as control. Insulin is stained red, nuclei are stained blue with DAPI, and EdU-positive nuclei are stained green. The scale bar represents 50 µm. right: The ratio of EdU-positive cells to the total count of insulin-positive cells is shown. More than 6000 cells were counted in each group (n = 4). (e) Ki67 mRNA expression in INS-1 cells infected with Ad-Fbln5 or Ad-GFP for 48 hours (n = 4). Data are represented as means ± SEM. *p < 0.05.
Discussion
In this study, we showed the glucose signaling-induced transcriptional regulation of Fbln5 in pancreatic islets, which is mediated by glucose metabolism via glucokinase and downstream Ca2+/calcineurin/NFAT signaling pathway (Fig. 5).
A schematic representation of glucokinase-mediated Fbln5 expression in pancreatic islets. Fbln5 gene expression was induced by glucokinase activation through ambient high glucose concentrations or GKA in pancreatic islets. Depolarization of the membrane accompanied by the closure of KATP channels, Ca2+ influx and calcineurin activation are required for this Fbln5 upregulation. A DYRK1A inhibitor, harmine, enhanced Fbln5 expression in the islets induced by glucokinase activation, possibly via NFAT signaling. DYRK1A; dual-specificity tyrosine phosphorylation-regulated kinase 1 A, GKA; glucokinase activator, KATP channel; ATP-sensitive potassium channel, NFAT; nuclear factor of activated T cells, VDCC; voltage-dependent Ca2+ channels.
Because β-cells are exposed to high ambient glucose concentrations under the diabetic condition, glucokinase, which acts as a glucose sensor, transmits the impact of the hyperglycemia to the β-cells. In the pancreatic islets, glucokinase is mainly expressed in the β-cells, with very low levels of expression observed in the α-cells (unpublished data). We confirmed GKA-induced increase in FBLN5 expression in INS-1 cells. Immunohistochemical analysis of INS-1 cells also supported that FBLN5 is expressed in the β-cells. However, further investigation using more specific antibody is needed to clarify the localization of FBLN5 since we observed FBLN5 signal not only in cytoplasm but also in the nucleus in INS-1 cells. FBLN5 immunofluorescence from paraffin-embedded tissue was mainly detected in non-β-cells tissue in the islets. FBLN5 is reported to be deposited on microfibrils during the development of mature elastic fiber26. Co-staining FBLN5 with CD34 in islets from paraffin-embedded specimens indicated that FBLN5 is strongly expressed in endothelial cells or small vessels in islets, consistent with the previous reports, which showed FBLN5 secretion from vascular smooth muscle cells or endothelial cells35, 36. FBLN5 was also detectable in β-cells in the non-paraffinized cultured islets. It is therefore possible that FBLN5 in β-cells was lost or masked in the process of paraffin embedding or deparaffinization. Since dual inhibition of insulin and IGF-1 receptor with OSI-906 did not abrogate GKA-induced Fbln5 upregulation in the islets, it is unlikely that this upregulation is mediated by an autocrine action of insulin on the insulin receptor. Because FBLN5 is a secreted protein, islet-derived FBLN5 might be deposited outside of the islets and play a role in non-islet tissue functions.
Our data showed that GKA-induced upregulation of Fbln5 was more pronounced in islets isolated from obese mice reared on a high-fat diet than in the islets of control mice fed normal chow. This could be explained by the involvement of glucokinase in the compensatory β-cell hyperplasia induced by a high-fat diet8, 29. Consistent with this notion, we found that Irs-2 deletion increased basal Fbln5 expression and attenuated the GKA-induced upregulation of Fbln5 in the isolated islets. Chronic hyperglycemia in Irs-2 −/− mouse may cause the elevation in Fbln5 in the islets at the basal state. Other factors that are related to insulin resistance with Irs-2 deletion in mice can possibly be involved in this basal elevation. Glucose-induced transcriptional regulation of Irs-2 gene expression in the β-cells is mediated by the Ca2+/calcineurin/NFAT pathway32, which is involved in β-cell proliferation in mice and humans37, 38. In addition, the DYRK1A inhibitor has been demonstrated to enhance β-cell proliferation in mice39, 40. It is also reported that GKA-induced increase of the mRNA expressions of Nfatc1 and its downstream genes are involved in β-cell maturation and β-cell proliferation in neonatal islets38. A recent study showed that glucose-induced mouse pancreatic β-cell proliferation is mediated via IRS-2, MTOR and cyclin D2, but not by the insulin receptor41. We also found that the Fbln5 expression was higher in the islets harvested from pre-weaning mice, which showed robust β-cell proliferation as confirmed by the high Ki67 expression. Fbln5 is strongly expressed during embryogenesis and plays a role in tissue remodeling25. Therefore, Fbln5 could be a predictor for compensatory β-cell proliferation and remodeling of β-cell mass induced by activation of IRS-2 expression.
How does NFAT signaling regulate the transcriptional activity of Fbln5 ? Fbln5 expression is positively regulated via transforming growth factor β1 (TGF-β1) in fibroblasts or epithelial cells25. Calcineurin inhibitors induce the TGF-β receptor-triggered signaling cascade in the mesangial cells or kidney42. Hypoxia-inducible factor-1α (HIF-1α) is also a Fbln5-inducible factor in the endothelial cells43. HIF-1α expression is also reportedly regulated through calcineurin activity or dephosphorylation of RACK1 in mast cells44, 45. We have identified NFAT consensus sequences in the 5-upstream region of the mouse Fbln5 gene at: −698 to −693 (AGGAAA), +386 to +391 (TGGAAA), +428 to +433 (TGGAAA), +591 to +596 (TGGAAA), and 4 other sites from the first transcription initiation site. Further analysis, including of the TGF-β and HIF-1α pathways, are needed to clarify the precise mechanism of Fbln5 transcription via NFAT in pancreatic islets.
Loss of systemic Fbln5 expression had no significant effects on the insulin secretion from the pancreatic islets or β-cell proliferation/expansion in young adult mice, suggesting that Fbln5 does not seem to be involved in β-cell development or functions at this stage of life. However, the effects of Fbln5 on pancreatic β-cell functions under diabetic- or insulin-resistant conditions remain unclear. In addition, we showed that Fbln5 expression is abundant in the islets from fetal or pre-weaning mice. Testing juvenile mice, therefore, is required for further investigation into the physiological role of Fbln5 in the context of developmental process. Fbln5 overexpression in INS-1 cells revealed that Fbln5 could positively regulate glucose-stimulated insulin secretion from the pancreatic β-cells. By contrast, Fbln5 overexpression possibly suppress cell proliferation in the INS-1 cells. In fact, Fbln5 overexpression decreased Ki67 expression in INS-1 cells, although Fbln5 and Ki67 expression were increased in proliferating juvenile islets. Fbln5 is reported to promote cell proliferation or tumor growth in mouse 3T3-L1 fibroblasts or human HT1080 fibrosarcoma cells25, mouse pancreatic ductal adenocarcinoma46, and human gastric cancer MGC-803 cells47. On the other hand, several previous studies have demonstrated inhibition of cell proliferation by Fbln5 overexpression in mouse vascular smooth muscle cells18, human breast cancer cells48, mink lu Mv1Lu epithelial cells25, primary human saphenous vein endothelial cells49, and rat retinal pigment epithelial cells50. Thus, further investigation of the pathway that mediate Fbln5 action on β-cell proliferation is required. These effects of Fbln5 on β-cell functions and β-cell proliferation might be explained by the distinct proliferative and functional state of the β-cells. A previous study showed that a high rate of insulin production suppressed β-cell proliferation because of increased ER stress, in a cell-autonomous manner51. On the other hand, genes involved in β-cell functions were suppressed when proliferation-related genes were upregulated in replicating β-cells52.
There is a report in the literature which suggests that another matricellular protein, SPARC, which is expressed in stromal cells within the islets, can regulate β-cell growth and survival by inhibiting growth factor responses53. Thus, the interactions between Fbln5 and pancreatic β-cell functions, which are still poorly understood, may represent novel molecular mechanisms involved in glucose metabolism and provide new insights for the treatment in diabetes.
In summary, we demonstrated that expression of the matricellular protein Fbln5 is upregulated by high ambient glucose concentrations in the pancreatic islets though glucokinase-dependent glucose and downstream Ca2+/calcineurin/NFAT signaling. Further study of the regulation of islet Fbln5 expression is warranted, especially in relation to glucose signaling and proliferation of β-cells.
Methods
Animals and Animal Care
All the animal procedures were performed in accordance with the guidelines of the Animal Care Committee of Yokohama City University. The protocol was approved by the Yokohama City University Institutional Animal Care and Use Committee (IACUC) (Permit Number: F-A-16-026). C57BL/6 J mice were purchased from Jackson. We backcrossed Fbln5 knockout (Fbln5 −/−) mice16, 19 with C57BL/6 J mice more than 10 times. Both Fbln5 −/− mice and wild-type littermates were fed a standard chow (MF, Oriental Yeast, Tokyo, Japan) or a high-fat diet (Clea Japan, Tokyo, Japan). All the experiments were conducted on male littermates. Animal housing rooms were maintained at a constant room temperature (25 °C) and a 12-hour light (7:00 a.m.) /dark (7:00 p.m.) cycle.
Adenovirus
Fbln5-overexpressing recombinant adenovirus (Ad-Fbln5)18 and GFP-expressing control adenovirus (Ad-GFP) were used for the experiments at a multiplicity of infection of 50 viruses per cell. In brief, the FLAG-tagged full-length rat Fbln5 was inserted in an adenoviral vector (pACCMVpLpA(−) loxP-SSP). Viruses were generated by transfection into the Human Embryo Kidney 293 (HEK293) cell line.
Islet isolation and culture
Isolation of islets from mice was conducted using collagenase, as described in a previous report54. The isolated islets were cultured in RPMI 1640 medium (Wako Pure Chemical Industries) containing 5.6 mmol/L glucose supplemented with 10% FCS, 100 units/mL of penicillin, and 100 μg/mL of streptomycin. The islets were treated with 30 μmol/L of GKA Cpd A, 50 μmol/L of nifedipine, 10 μmol/L of FK506, 10 μmol/L of D-mannoheptulose (Toronto Research Chemicals), 200 μmol/L of diazoxide (Wako Pure Chemical Industries), 200 nmol/L of OSI-906 (Selleck Chemicals). All the reagents were added concomitantly to the medium in each experiment.
Oral glucose tolerance test
All the mice were denied access to food for 14–16 hours before the oral glucose tolerance test (OGTT) and then orally loaded with glucose at 1.5 mg/g body weight. Blood glucose levels and serum insulin levels were determined using Glutest Neo Super (Sanwa Chemical Co. Kanagawa, Japan) and an insulin ELISA kit (Morinaga Institute of Biological Science, Yokohama, Japan), respectively.
Glucose-stimulated insulin secretion in isolated islets and INS-1 cells
Ten islets isolated from Fbln5 −/− mice and wild-type mice were incubated at 37 °C for 1.5 hours in Krebs-Ringer bicarbonate buffer containing 2.8, 11.1 or 22.2 mmol/L of glucose. When examining the effect of Fbln5 deficiency on GKA-induced insulin secretion, islets were incubated at 37 °C for 1.5 hours in Krebs-Ringer bicarbonate buffer containing 2.8 mmol/L glucose with or without 30 µmol/L of GKA CpdA, or 11.1 mmol/L glucose without GKA CpdA. For measuring insulin content, islets were extracted with acid ethanol. INS-1 cells were infected with adenovirus (Ad-GFP or Ad-Fbln5) and cultured for 48 hours. Subsequently, the cells were incubated at 37 °C for 2 hours in Krebs-Ringer bicarbonate buffer containing 2.8, 11.1 or 22.2 mmol/L of glucose. Then, the insulin concentration in the assay buffer and insulin content was measured with an insulin ELISA kit.
Cell culture
INS-1 (832/13) cells55 were cultured in RPMI 1640 containing 10 mmol/L of HEPES, 11.1 mmol/L of glucose, 10% FBS, 1 mmol/L of sodium pyruvate, 2 mmol/L of L-glutamine, 50 μmol/L of 2-mercaptoethanol, 100 units/mL of penicillin and 100 μg/mL of streptomycin. The cells were maintained at 37 °C in humidified air containing 5% CO2. Before the experiments, the INS-1 cells were starved by incubation in RPMI1640 medium containing 2.8 mmol/L of glucose, 100 units/mL of penicillin, and 100 μg/mL of streptomycin for 16 hours.
Histological analysis
Pancreatic tissue sections from embryonic day15 and 8-week-old Fbln5 −/− mice and wild-type mice were analyzed after formalin fixation and paraffin embedding. For non-paraffinized tissue staining, isolated islets from 8-week-old wild-type mice attached to 0.1%-gelatin-coated coverslips (Falcon) were analyzed after fixation with paraformaldehyde. Pancreatic islets isolated from 8-week-old wild-type mice were analyzed after fixation without paraffin embedding. The sections or attached islets on coverslips were immunostained with antibody directed against insulin (Santa Cruz Biotechnology), glucagon (Abcam), somatostatin (GeneTex), CD34 (Santa Cruz Biotechnology), or rabbit polyclonal anti-fibulin-5 (BSYN 1923; 1:100)16. FBLN5 signal was enhanced by tyramide signal amplification, using a TSA Fluorescein System (Perkin Elmer, NEL741001KT), in paraffin-embedding sections. Biotinylated secondary antibodies, a VECTASTAIN Elite ABC Kit, and a DAB Substrate Kit (Vector Laboratories) were used to examine the sections using bright-field microscopy to determine the β-cell mass, and Alexa Fluor 488-, 555- and 647-conjugated secondary antibodies (Invitrogen) were used for the fluorescence microscopy. Images were acquired using a BZ-9000 microscope (Keyence) or the FluoView FV1000-D confocal laser scanning microscope (Olympus). The proportion of the area of the pancreatic tissue occupied by β-cells was calculated using BIOREVO software (Keyence), as described previously56. The fluorescence levels of insulin in GKA-treated wild-type and Fbln5 −/− islets were determined using Image J software. All images, which were acquired under the same condition, were converted to gray scale. Then, we randomly selected 5 regions of separate islets in each group and measured fluorescence levels. The fluorescent intensity were normalized by the mean background fluorescence levels.
Proliferation Assay
Isolated islets from Fbln5 −/− mice and wild-type mice were incubated with a modified thymidine analog, EdU (5-ethynyl-2’-deoxyuridine; Click-iT EdU; Invitrogen Cat. No. C10637) in the presence or absence of 30 μmol/L of GKA. After the treatment for 48 hours, the islets were fixed and sections were prepared of the embedded islets in 1% agarose-gel.
INS-1 cells were infected with an adenovirus vector (Ad-GFP or Ad-Fbln5) and cultured for 48 hours. Then, the cells were incubated with 10 µM of EdU for 3 hours and fixed. EdU incorporation and detection were performed as described in the manufacturer’s protocol. The images were taken using the FluoView FV1000-D confocal laser scanning microscope. We counted EdU-positive proliferative cells after adjusting the gain of fluorescence. At that condition there were no significant changes in the fluorescent intensity between GFP-positive cells and GFP-negative cells.
Real-time PCR
Total RNA was isolated from the pancreatic islets using an RNase-free DNase and RNeasy Kit (Qiagen, Valencia, CA). cDNA was prepared using High Capacity cDNA Reverse-Transcription Kits (Applied Biosystems). Quantitative PCR was performed by using TaqMan Gene Expression Assays (7900 Real-Time PCR System; Applied Biosystems) with the THUNDERBIRD qPCR Master Mix (TOYOBO). All the probes were purchased from Applied Biosystems (mouse Fbln5; Mm00488601_m1, mouse β-actin; Mm00607939_s1, mouse Ki67; Mm01278617_m1, rat Fbln5; Rn0069712_m1, rat β-actin; Rn00667869_m1, rat Ki67; Rn01451446_m1). Data were normalized to the expression level of β-actin.
Immunoblotting
For immunoblotting, isolated mouse islets and INS-1 cells were lysed in ice-cold RIPA buffer with protease and phosphatase inhibitor cocktail. The islets and cell extracts were subjected to immunoblotting. The primary antibodies used were rabbit anti-FBLN5 (BSYN1923) at the dilution of 1:100, or Anti-Fibulin-5-Antibody (Millipore) at the dilution of 1:5000, and β-actin (Sigma-Aldrich). Densitometry was performed using Image J software.
Statistical analysis
All the data are expressed as the means ± SEM, and were analyzed using the Student’s t test or ANOVA. Differences between two groups were analyzed by Student’s t test (Figs 1a, 3b-c and e, 4, Supplementary Fig. S1b-c). For comparisons among more than two groups, we used the one-way ANOVA followed by the Tukey HSD post hoc test (Figs 1b and d–i, 2a and d, 3g, Supplementary Fig. S1a). When the data had unequal variance, we used Welch’s one-way ANOVA followed by the Games-Howell post hoc test (Fig. 2b,c,e,f). Differences were considered significant if the p value was <0.05 (*) or <0.01 (**).
References
Otonkoski, T., Andersson, S., Knip, M. & Simell, O. Maturation of insulin response to glucose during human fetal and neonatal development. Studies with perifusion of pancreatic isletlike cell clusters. Diabetes 37, 286–291 (1988).
Soleimanpour, S. A. et al. Calcineurin signaling regulates human islet {beta}-cell survival. J Biol Chem 285, 40050–40059, doi:10.1074/jbc.M110.154955 (2010).
Alonso, L. C. et al. Glucose infusion in mice: a new model to induce beta-cell replication. Diabetes 56, 1792–1801, doi:10.2337/db06-1513 (2007).
Bonner-Weir, S., Deery, D., Leahy, J. L. & Weir, G. C. Compensatory growth of pancreatic beta-cells in adult rats after short-term glucose infusion. Diabetes 38, 49–53, doi:10.2337/diab.38.1.49 (1989).
Kwon, G., Marshall, C. A., Pappan, K. L., Remedi, M. S. & McDaniel, M. L. Signaling elements involved in the metabolic regulation of mTOR by nutrients, incretins, and growth factors in islets. Diabetes 53(Suppl 3), S225–232, doi:10.2337/diabetes.53.suppl_3.S225 (2004).
Porat, S. et al. Control of pancreatic beta cell regeneration by glucose metabolism. Cell Metab 13, 440–449, doi:10.1016/j.cmet.2011.02.012 (2011).
Matschinsky, F. M. Banting Lecture 1995. A lesson in metabolic regulation inspired by the glucokinase glucose sensor paradigm. Diabetes 45, 223–241, doi:10.2337/diab.45.2.223 (1996).
Terauchi, Y. et al. Glucokinase and IRS-2 are required for compensatory beta cell hyperplasia in response to high-fat diet-induced insulin resistance. J Clin Invest 117, 246–257, doi:10.1172/jci17645 (2007).
Njolstad, P. R. et al. Neonatal diabetes mellitus due to complete glucokinase deficiency. The New England journal of medicine 344, 1588–1592, doi:10.1056/nejm200105243442104 (2001).
Osbak, K. K. et al. Update on mutations in glucokinase (GCK), which cause maturity-onset diabetes of the young, permanent neonatal diabetes, and hyperinsulinemic hypoglycemia. Human mutation 30, 1512–1526, doi:10.1002/humu.21110 (2009).
Kassem, S. et al. Large islets, beta-cell proliferation, and a glucokinase mutation. The New England journal of medicine 362, 1348–1350, doi:10.1056/NEJMc0909845 (2010).
Shirakawa, J. et al. Glucokinase activation ameliorates ER stress-induced apoptosis in pancreatic beta-cells. Diabetes 62, 3448–3458, doi:10.2337/db13-0052 (2013).
Tornovsky-Babeay, S. et al. Type 2 diabetes and congenital hyperinsulinism cause DNA double-strand breaks and p53 activity in beta cells. Cell metabolism 19, 109–121, doi:10.1016/j.cmet.2013.11.007 (2014).
Grimsby, J. et al. Allosteric activators of glucokinase: potential role in diabetes therapy. Science (New York, N.Y.) 301, 370–373, doi:10.1126/science.1084073 (2003).
Nakamura, T. et al. Fibulin-5/DANCE is essential for elastogenesis in vivo. Nature 415, 171–175, doi:10.1038/415171a (2002).
Yanagisawa, H. et al. Fibulin-5 is an elastin-binding protein essential for elastic fibre development in vivo. Nature 415, 168–171, doi:10.1038/415168a (2002).
Kowal, R. C., Richardson, J. A., Miano, J. M. & Olson, E. N. EVEC, a novel epidermal growth factor-like repeat-containing protein upregulated in embryonic and diseased adult vasculature. Circ Res 84, 1166–1176, doi:10.1161/01.RES.84.10.1166 (1999).
Spencer, J. A. et al. Altered vascular remodeling in fibulin-5-deficient mice reveals a role of fibulin-5 in smooth muscle cell proliferation and migration. Proc Natl Acad Sci USA 102, 2946–2951, doi:10.1073/pnas.0500058102 (2005).
Budatha, M. et al. Extracellular matrix proteases contribute to progression of pelvic organ prolapse in mice and humans. J Clin Invest 121, 2048–2059, doi:10.1172/JCI45636 (2011).
Kapustin, A. et al. Fibulin-5 binds urokinase-type plasminogen activator and mediates urokinase-stimulated beta1-integrin-dependent cell migration. Biochem J 443, 491–503, doi:10.1042/BJ20110348 (2012).
Schluterman, M. K. et al. Loss of fibulin-5 binding to beta1 integrins inhibits tumor growth by increasing the level of ROS. Dis Model Mech 3, 333–342, doi:10.1242/dmm.003707 (2010).
Yue, W. et al. Fibulin-5 suppresses lung cancer invasion by inhibiting matrix metalloproteinase-7 expression. Cancer research 69, 6339–6346, doi:10.1158/0008-5472.can-09-0398 (2009).
Lomas, A. C. et al. Fibulin-5 binds human smooth-muscle cells through alpha5beta1 and alpha4beta1 integrins, but does not support receptor activation. Biochem J 405, 417–428, doi:10.1042/BJ20070400 (2007).
Tang, J. C., Xie, A. Y. & Cai, X. J. [Diverse functions of fibulin-5 in tumors]. Molekuliarnaia biologiia 48, 875–880 (2014).
Schiemann, W. P., Blobe, G. C., Kalume, D. E., Pandey, A. & Lodish, H. F. Context-specific effects of fibulin-5 (DANCE/EVEC) on cell proliferation, motility, and invasion. Fibulin-5 is induced by transforming growth factor-beta and affects protein kinase cascades. J Biol Chem 277, 27367–27377, doi:10.1074/jbc.M200148200 (2002).
Hirai, M. et al. Fibulin-5/DANCE has an elastogenic organizer activity that is abrogated by proteolytic cleavage in vivo. J Cell Biol 176, 1061–1071, doi:10.1083/jcb.200611026 (2007).
Hammar, E. et al. Extracellular matrix protects pancreatic beta-cells against apoptosis: role of short- and long-term signaling pathways. Diabetes 53, 2034–2041, doi:10.2337/diabetes.53.8.2034 (2004).
Weber, L. M., Hayda, K. N. & Anseth, K. S. Cell-matrix interactions improve beta-cell survival and insulin secretion in three-dimensional culture. Tissue engineering. Part A 14, 1959–1968, doi:10.1089/ten.tea.2007.0238 (2008).
Takamoto, I. et al. Crucial role of insulin receptor substrate-2 in compensatory beta-cell hyperplasia in response to high fat diet-induced insulin resistance. Diabetes, obesity & metabolism 10(Suppl 4), 147–156, doi:10.1111/j.1463-1326.2008.00951.x (2008).
Rusnak, F. & Mertz, P. Calcineurin: form and function. Physiological reviews 80, 1483–1521 (2000).
Hogan, P. G., Chen, L., Nardone, J. & Rao, A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes & development 17, 2205–2232, doi:10.1101/gad.1102703 (2003).
Demozay, D., Tsunekawa, S., Briaud, I., Shah, R. & Rhodes, C. J. Specific glucose-induced control of insulin receptor substrate-2 expression is mediated via Ca2+-dependent calcineurin/NFAT signaling in primary pancreatic islet beta-cells. Diabetes 60, 2892–2902, doi:10.2337/db11-0341 (2011).
Gwack, Y. et al. A genome-wide Drosophila RNAi screen identifies DYRK-family kinases as regulators of NFAT. Nature 441, 646–650, doi:10.1038/nature04631 (2006).
Lu, M. et al. Characterization of a novel glucokinase activator in rat and mouse models. PLoS One 9, e88431, doi:10.1371/journal.pone.0088431 (2014).
Williamson, M. R., Shuttleworth, A., Canfield, A. E., Black, R. A. & Kielty, C. M. The role of endothelial cell attachment to elastic fibre molecules in the enhancement of monolayer formation and retention, and the inhibition of smooth muscle cell recruitment. Biomaterials 28, 5307–5318, doi:10.1016/j.biomaterials.2007.08.019 (2007).
Yanagisawa, H., Schluterman, M. K. & Brekken, R. A. Fibulin-5, an integrin-binding matricellular protein: its function in development and disease. J Cell Commun Signal 3, 337–347, doi:10.1007/s12079-009-0065-3 (2009).
Heit, J. J. et al. Calcineurin/NFAT signalling regulates pancreatic beta-cell growth and function. Nature 443, 345–349, doi:10.1038/nature05097 (2006).
Goodyer, W. R. et al. Neonatal beta cell development in mice and humans is regulated by calcineurin/NFAT. Developmental cell 23, 21–34, doi:10.1016/j.devcel.2012.05.014 (2012).
Dirice, E. et al. Inhibition of DYRK1A Stimulates Human beta-Cell Proliferation. Diabetes 65, 1660–1671, doi:10.2337/db15-1127 (2016).
Wang, P. et al. A high-throughput chemical screen reveals that harmine-mediated inhibition of DYRK1A increases human pancreatic beta cell replication. Nat Med 21, 383–388, doi:10.1038/nm.3820 (2015).
Stamateris, R. E. et al. Glucose Induces Mouse beta-Cell Proliferation via IRS2, MTOR, and Cyclin D2 but Not the Insulin Receptor. Diabetes 65, 981–995, doi:10.2337/db15-0529 (2016).
Akool el, S. et al. Molecular mechanisms of TGF beta receptor-triggered signaling cascades rapidly induced by the calcineurin inhibitors cyclosporin A and FK506. Journal of immunology (Baltimore, Md.: 1950) 181, 2831-2845 (2008).
Guadall, A. et al. Fibulin-5 is up-regulated by hypoxia in endothelial cells through a hypoxia-inducible factor-1 (HIF-1alpha)-dependent mechanism. J Biol Chem 286, 7093–7103, doi:10.1074/jbc.M110.162917 (2011).
Liu, Y. V. et al. Calcineurin promotes hypoxia-inducible factor 1alpha expression by dephosphorylating RACK1 and blocking RACK1 dimerization. J Biol Chem 282, 37064–37073, doi:10.1074/jbc.M705015200 (2007).
Walczak-Drzewiecka, A., Ratajewski, M., Wagner, W. & Dastych, J. HIF-1alpha is up-regulated in activated mast cells by a process that involves calcineurin and NFAT. Journal of immunology (Baltimore, Md.: 1950) 181, 1665–1672, doi:10.4049/jimmunol.181.3.1665 (2008).
Topalovski, M., Hagopian, M., Wang, M. & Brekken, R. A. Hypoxia and Transforming Growth Factor beta Cooperate to Induce Fibulin-5 Expression in Pancreatic Cancer. J Biol Chem 291, 22244–22252, doi:10.1074/jbc.M116.730945 (2016).
Shi, X. Y. et al. Effect of Fibulin-5 on cell proliferation and invasion in human gastric cancer patients. Asian Pacific journal of tropical medicine 7, 787–791, doi:10.1016/s1995-7645(14)60137-1 (2014).
Mohamedi, Y. et al. Fibulin-5 downregulates Ki-67 and inhibits proliferation and invasion of breast cancer cells. International journal of oncology 48, 1447–1456, doi:10.3892/ijo.2016.3394 (2016).
Preis, M. et al. Effects of fibulin-5 on attachment, adhesion, and proliferation of primary human endothelial cells. Biochem Biophys Res Commun 348, 1024–1033, doi:10.1016/j.bbrc.2006.07.156 (2006).
Li, F., Xu, H., Zeng, Y. & Yin, Z. Q. Overexpression of fibulin-5 in retinal pigment epithelial cells inhibits cell proliferation and migration and downregulates VEGF, CXCR4, and TGFB1 expression in cocultured choroidal endothelial cells. Current eye research 37, 540–548, doi:10.3109/02713683.2012.665561 (2012).
Szabat, M. et al. Reduced Insulin Production Relieves Endoplasmic Reticulum Stress and Induces beta Cell Proliferation. Cell Metab 23, 179–193, doi:10.1016/j.cmet.2015.10.016 (2016).
Klochendler, A. et al. The Genetic Program of Pancreatic beta-Cell Replication In Vivo. Diabetes 65, 2081–2093, doi:10.2337/db16-0003 (2016).
Ryall, C. L. et al. Novel role for matricellular proteins in the regulation of islet beta cell survival: the effect of SPARC on survival, proliferation, and signaling. J Biol Chem 289, 30614–30624, doi:10.1074/jbc.M114.573980 (2014).
Shirakawa, J. et al. Protective effects of dipeptidyl peptidase-4 (DPP-4) inhibitor against increased beta cell apoptosis induced by dietary sucrose and linoleic acid in mice with diabetes. J Biol Chem 286, 25467–25476, doi:10.1074/jbc.M110.217216 (2011).
Hohmeier, H. E. et al. Isolation of INS-1-derived cell lines with robust ATP-sensitive K+ channel-dependent and -independent glucose-stimulated insulin secretion. Diabetes 49, 424–430, doi:10.2337/diabetes.49.3.424 (2000).
Shirakawa, J. et al. Effects of liraglutide on beta-cell-specific glucokinase-deficient neonatal mice. Endocrinology 153, 3066–3075, doi:10.1210/en.2012-1165 (2012).
Acknowledgements
This work was supported in part by Grants-in-Aid for Scientific Research (B) 16H05329s from the Ministry of Education, Culture, Sports, Science and Technology of Japan (to Y. Te.). We thank Dr. Christopher Newgard (Duke University) for providing us with the INS-1 cells (832/13 cells). We also thank Mitsuyo Kaji and Eri Sakamoto (Yokohama City University) for technical assistance, and Misa Katayama for secretarial assistance.
Author information
Authors and Affiliations
Contributions
T.O., J.S., and Y. Te. designed the study. T.O. and J.S. performed the experiments, analyzed the data and wrote the manuscript. H.Y. provided us the Fbln5KO mice and contributed to the discussion. M.K., S.Y., K.T., and Y. To. assisted in the experiments. All authors gave final approval of the version to be published.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Okuyama, T., Shirakawa, J., Yanagisawa, H. et al. Identification of the matricellular protein Fibulin-5 as a target molecule of glucokinase-mediated calcineurin/NFAT signaling in pancreatic islets. Sci Rep 7, 2364 (2017). https://doi.org/10.1038/s41598-017-02535-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02535-0
This article is cited by
-
Translational research on human pancreatic β-cell mass expansion for the treatment of diabetes
Diabetology International (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.