Abstract
The purpose of the current study was to compare the measurements between a new optical biometer based on swept-source optical coherence tomography (SS-OCT), the OA-2000 (Tomey, Japan), and an optical biometer based on optical low coherence reflectometry (OLCR), the Lenstar (Haag-Streit, Switzerland). Ninety-nine eyes of 99 healthy subjects were included. The axial length (AL), central corneal thickness (CCT), anterior chamber depth (ACD), aqueous depth (AD), lens thickness (LT), keratometry (K) readings, including flat K (Kf), steep K (Ks), mean K (Km), astigmatism vectors J0, J45 at diameters of 2.5 and 3.0 mm, and white-to-white diameter (WTW) were measured three times each using both biometer in normal eyes by random sequence. Bland-Altman analysis showed good agreement between the SS-OCT and OLCR devices for AL, AD, ACD, LT, with narrow 95% LoA (−0.05 to 0.07 mm, −0.09 to 0.10 mm, −0.10 to 0.09 mm, and −0.06 to 0.22 mm, respectively), and the P values of ACD were both >0.05. The CCT, Kf, Ks, Km, J0, J45 and WTW values provided by the OA-2000 were in good agreement with the Lenstar, and statistically significant differences were detected for some of them but not clinical differences. The agreement was excellent especially for AL.
Similar content being viewed by others
Introduction
Accurate and precise measurements of ocular biometric parameters are crucial for many clinical and research applications in ophthalmology. Axial length (AL) is a fundamental parameter for calculating the most suitable intraocular lens (IOL) in cataract surgery and refractive lens exchange. A previous study reported that AL reading plays an important role in predicting postoperative refractive error and attributes to 54% of the actual refractive error1. Accurate measurements of anterior chamber depth (ACD), aqueous depth (AD) and keratometry play a major role in the calculation of standard as well as phakic IOLs2, 3. ACD is also used to screen the primary angle closure glaucoma and monitor the changes of the anterior eye segment during accommodation4,5,6. Central corneal thickness (CCT) is used to screen candidates for refractive surgery, in order to reduce the risk of postoperative ectasia7. CCT readings are also crucial to detect contact lens-induced edema, and help to diagnose corneal diseases and glaucoma8,9,10,11.
Ultrasound biometry has been the common technique for a long time to measure ACD, lens thickness (LT) and AL because of its cost-effectiveness and ease of use8, 9. Nevertheless, it has some shortcomings, such as corneal-probe contact with possible corneal epithelial lesions, the risk of infections and the need for topical anesthesia12. Since the advent of the IOLMaster (Carl Zeiss Meditec, Jena, Germany), optical biometry has been widely used and regarded as the gold standard for ocular biometric parameters13, 14. The Lenstar (Haag-Streit, Bem, Switzerland), which is based on optical low coherence reflectometry (OLCR), was the first competitor of the IOLMaster. A single non-contact measurement can simultaneously provide nine biometric parameters, including AL, ACD, CCT, LT, keratometry, retinal thickness, white-to-white distance (WTW) and eccentricity of the visual optical line15. Previous studies have demonstrated excellent intraoperator repeatability, interoperator reproducibility and accuracy of the Lenstar16,17,18. The OA-2000 (Tomey, Japan) is a newly introduced optical biometer, based on swept-source optical coherence tomography (SS-OCT). Its measurements were shown to be repeatable19, 20. A previous study used a functional prototype which based on OLCR might have provided different results19. However, to the best of our knowledge, comparison of ocular biometric parameters between the commercially available SS-OCT version of the OA-2000 and the commonly used biometer Lenstar has never been performed.
The purpose of this study was to prospectively evaluate agreement between the AL, ACD, AD, keratometry, CCT, LT and WTW measurements derived from the OA-2000 based SS-OCT and the Lenstar in normal eyes for the first time in normal eyes.
Results
Ninety-nine right eyes of 99 healthy volunteers (58 women), with a mean age of 29.4 ± 9.6 years (range: 19 to 64 years), were enrolled in the study. The mean spherical equivalent refraction was −3.36 ± 2.46 diopters (D) (range: −8.875 to 1.375 D).
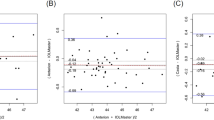
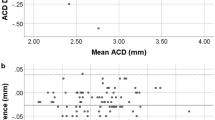
Table 1 exhibits the mean, minimum and maximum values of the parameters obtained by the OA-2000 and Lenstar. The comparison and agreement data are reported in Table 2. The AL, CCT, LT and WTW values showed statistically significant differences (P < 0.01), but relatively narrow 95% LoA. The 95% LoA of AL was the narrowest with a range from −0.05 to 0.07 mm (Figs 1–4). The ACD and AD values by the two biometers were similar with a maximum absolute 95% LoA of 0.10 mm (P > 0.05) (Figs 5 and 6). The 95% LoA of corneal topography results including Kf, Ks, Km, J0 and J45 were in a narrow range with maximum values of −0.32 to 0.36 D, although some of them showed significant differences (P < 0.05) (Figs 7–16).
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring central corneal thickness. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring lens thickness. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring white-to-white. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring anterior chamber depth. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring flat keratometry (Kf) at 2.5 mm. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring steep keratometry (Ks) at 2.5 mm. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring mean keratometry (Km) at 2.5 mm. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring vectors J0 at 2.5 mm. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring vectors J45 at 2.5 mm. Solid lines represent the bias between both devices and dotted lines represent the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring flat keratometry (Kf) at 3.0 mm. Solid lines represent the bias between both devices and dotted lines represent at the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring steep keratometry (Ks) at 3.0 mm. Solid lines represent the bias between both devices and dotted lines represent at the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring mean keratometry (Km) at 3.0 mm. Solid lines represent the bias between both devices and dotted lines represent at the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring vectors J0 at 3.0mm. Solid lines represent the bias between both devices and dotted lines represent at the 95% confidence interval for the difference.
Bland-Altman plots show the agreement between OA-2000 swept-source optical coherence tomography and Lenstar optical low coherence reflectometry for measuring vectors J45 at 3.0 mm. Solid lines represent the bias between both devices and dotted lines represent at the 95% confidence interval for the difference.
Discussion
As a newly available optical biometer, the OA-2000 should be compared with the currently established biometers in clinical use19. The Lenstar was commercially available after the IOLMaster but provides more comprehensive parameters with different measuring principles, and its accuracy is well established17. An agreement analysis, based on the definitions adopted by the British Standards Institution as recommended by Bland and Altman, such as Bland-Altman plots and LoA, has been applied in our previous study but not been applied between the OA-2000 and Lenstar20. Thus, it is necessary to study the agreements and differences between the two devices, and detect and confirm the value and application prospect of the new device21,22,23. In the current study, according to our data, most parameters showed good agreement, especially for the AL, ACD and K values.
Minor difference was found between these two biometers for measuring AL, with a mean value of 0.01 mm. A 0.08 mm error in AL would result in an error of 0.20 to 0.34 D according to the SRK/T formula and a 0.10 mm measuring error may be equivalent to about 0.25 to 0.3 D in the spectacle plane. Thus, the maximum 95% LoA of 0.07 mm between OA-2000 and Lenstar was thought to be well acceptable in clinic2, 17. Our results were similar to those of Cruysberg et al. with a 95% LoA range of 0.07 to −0.01 mm between Lenstar and IOLMaster17. As compared to the results of Goebels et al. whose mean AL difference between the functional prototype and Lenstar in cataract, IOLMaster was −0.03 ± 0.09 mm and −0.02 ± 0.03 mm, respectively, mean difference was 0.03 mm with a large range of −0.48 to 0.67 mm, but our result was smaller and indicated excellent AL agreement19. Agreement in the current study was also better than that in Kongsap’s study, which reported a mean difference of −0.02 ± 0.03 mm between OA-2000 and IOLMaster 500 (Carl Zeiss Meditec, Dublin, CA, USA)24.
In the current study, the CCT provided by OA-2000 and Lenstar was 523.97 ± 31.59 μm and 536.80 ± 32.38 μm, respectively, the former also provided lower CCT values than Lenstar in a previous study19. This slight difference may be due to the different algorithms and analysis programs of the two devices for boundary determination. A comparison between IOLMaster 700 (Carl Zeiss Meditec AG), which adopted SS-OCT technology and Lenstar showed good agreement with 95% LoA of −8.69 to 8.99 μm25. The Lenstar also showed good agreement with the ultrasound (95% LoA was −6.8 to 9.6 μm in unoperated eyes) and RTVue OCT (RTVue-100; Optovue Inc., Fremont, CA, USA) (95% LoA was −3.1 to 11.8 μm), which were a little narrower than our results26, 27. But the CCT values showed a relatively similar range of 95% LoA between Lenstar and Visante AS-OCT (Carl Zeiss Meditec, Dublin, California, USA) in the study by Cruysberg et al. with a maximum value of 24.1 μm, which was slightly larger than in our study17. Similar results were found in the comparison between Sirius (Costruzione Strumenti Oftalmici, Florence, Italy) and ultrasound, which also showed good agreement for CCT measurements28. When the CCT values were compared between SP-3000P (NCSM; Topcon Corp, Tokyo, Japan), EM-3000 (Tomey, Nagoya, Japan), SP-02 (Costruzione Strumenti Oftalmici, Italy) and ultrasound pachymetry (Tomey Inc., Nagoya, Japan), the 95% LoA ranges were broader than ours (the minimum range was −29.3 to 1.7 for comparison between SP-3000P and EM-3000, the maximum range was −62.1 to 8.7 for comparison between SP-3000P and SP-02)29. As the CCT vary up to 20 μm over a day in individuals and every 25 μm deviation would lead to about 1 mmHg correction, the current result was clinically acceptable and showed moderate agreement25, 30.
The ACD and LT values may be affected by accommodation, but the former only changed by −0.09 to −0.14 mm with accommodation in myopia eyes31,32,33. The ACD did not show significant difference with accommodation in emmetropic subjects in the study by Dominguez-Vicent et al.34, 35. The OA-2000 and Lenstar use similar light spot for focusing and automatically measure along the visual axis, which reduces the accommodation influence. The differences between them for AD, ACD and LT values were low to 0.01 ± 0.05 mm, 0.00 ± 0.05 mm, 0.08 ± 0.07 mm within a clinically acceptable narrow 95% LoA range in the current study. Agreement was higher with respect to comparisons among the functional prototype of OA-2000 and Lenstar, IOLMaster 500, and AL-Scan (Nidek Co, Aichi, Japan) in previous studies19, 24, 36. It is surprising to compare agreement between OA-2000 and Lenstar for ACD measurements in the present study and in the study by Goebel et al.: in our sample the mean value was the same for both devices (3.57 mm), whereas in the above-mentioned paper the OA-2000 provided a remarkably higher mean value (3.71 vs 3.09 mm). It is likely that such a discrepancy depends on the fact that Goebel et al. investigated a functional prototype based on OLCR, while we analyzed the commercially available machine based on SS-OCT. Technical development of the OA-2000 from the prototype to the final version may explain these differences. The good performance and agreement suggested a potential application in the IOL power formulae of new generation, which use ACD and LT as additional variables2, 37.
In this study, the K values from OA-2000 were simultaneously assessed at 2.5 mm and 3.0 mm diameters, while central corneal topography provided by the Lenstar was at 1.65 mm and 2.3 mm. As J0 and J45 based on Fourier transformation were shown to be useful in previous studies for astigmatism evaluation, we additionally calculated these results38. All the results showed good agreement with a maximum 95% LoA of 0.36D although the differences of K values at 3.0 mm were slightly larger than those at 2.5 mm, but relatively better than that between AL-Scan and Lenstar36. The total measurement errors of Lenstar were 0.18D and 0.24D for anterior steep and flat keratometry, respectively, and they were close to that of Placido39. Comparison of keratometry obtained by Lenstar and Topolyzer (WaveLight Technologie AG, Erlangen, Germany), Galilei G6 (Ziemer, Port, Switzerland) showed acceptable 95% LoA range but not as narrow as that in our current study40, 41. Although Goebels et al. had shown the R1 and R2 values of OA-2000 and Lenstar, they were mainly comparing the AL and ACD. This was the first time that the differences of keratometry between OA-2000 and Lenstar were comprehensively analyzed, and good agreements were achieved19.
The agreement of WTW measurement was not always good in prior studies because of the edge recognition technology. The Lenstar showed good agreement with IOLMaster in natural pupils but not in cycloplegic subjects42. The measurements were also similar between Lenstar with AL-Scan but not with Sirius36, 43. Currently, better agreement between OA-2000 and Lenstar than that between AL-Scan and Lenstar (95% LoA ranged from −0.69 to 0.34 mm), the OA-2000 and IOLMaster 500 (95% LoA ranged from −1.85 to 1.42 mm) was found with narrower 95% LoA range of −0.50 to 0.24 mm24, 36.
There were some limitations in this study. We only included healthy volunteers, and their ocular condition was easily captured due to their good cooperation and transparent refractive media. We confirmed that the OA-2000 has a significant advantage when measuring AL in cataract patients with respect to the IOLMaster and Aladdin optical biometer (Topcon, Japan)44. But the success rate of OA-2000 in patients with other ocular diseases still needs further analysis. Besides, as a type of SS biometer, comparisons with other SS-OCT should be performed in the future.
In summary, most parameters acquired by OA-2000 were comparable to those obtained by Lenstar including AL, CCT, AD, ACD, LT, K values and WTW, with relatively narrow 95% LoA range. The AL values showed the best agreement among the available parameters.
Subjects and Methods
This prospective study recruited 99 normal subjects from the Eye Hospital of Wenzhou Medical University. The research protocol was in accordance with the tenets of the Declaration of Helsinki, and was approved by the Office of Research Ethics, Eye Hospital of Wenzhou Medical University. All subjects signed informed consent after understanding the purpose of the research.
The exclusion criteria were recent contact lens wear (rigid contact lens within four weeks and soft contact lens within two weeks), any active ocular pathology, history of ocular surgery and trauma, intraocular pressure >21 mmHg, fundus disease and systemic diseases with ocular symptoms.
The Lenstar applies an OLCR principle to measure AL, CCT, ACD, and LT with the wavelength of 820 nm superluminescent diode laser. It also employs 950 nm light to measure WTW, pupil diameter (PD), corneal curvature and pupil by image analysis. The flattest meridian K and steepest meridian K values are analyzed by the position of 32 projected light reflections at two rings with diameters of 1.65 and 2.30 mm. Keratometry is calculated by transforming the corneal curvature radius into diopters (D) using the 1.3375 refractive index. WTW is measured using the image of the corneal radius and the iris derived from keratometry.
The OA-2000 uses the Placido ring cone and optical interference principle to measure the axial dimensions of the ocular structure. The Fourier domain optical interference is applied to measure the AL, CCT, ACD and LT parameters using the wavelength of 1060 nm swept source laser. The 515 nm and 750 nm light-emitting diodes (LEDs) are used to determine the K values, WTW and PD, respectively. It also employs the Placido ring cone to simultaneously measure corneal shape and the radius of corneal curvature at 2.5 mm and 3.0 mm positions. Each Placido ring is 256 points. It totally captures 2,304 points by nine rings.
We analyzed AD and ACD, respectively. The former is the distance from the corneal endothelium to the anterior surface of the lens capsule, while the latter represents the distance from the corneal epithelium to the lens45. Astigmatism was analyzed using J0 and J45 vectors according to the following formulae38:
All subjects were requested to sit with their foreheads against the headrest, chins on a chin-rest, open both eyes and fixate on an internal target within each device. They were instructed to completely blink twice in order to spread an optically smooth tear film on the cornea before each measurement. After each scan, the device was moved backward and realigned for the next measurement. Each subject was measured until three valid results were obtained by each device. The sequence order measurement of the Lenstar and the OA-2000 was confirmed by the MedCalc Statistical Software (version 13.0, MedCalc Software Inc., Belgium). In order to avoid the effects of diurnal variation in corneal shape and ocular structure, the entire examination was accomplished in less than 15 minutes46. Only one eye of each subject was chosen to be measured in order to avoid structural similarities between fellow eyes47.
Statistical Analysis
All data were analyzed using SPSS software for Windows version 21 (IBM corporation, USA), MedCalc statistical software (version 14.1, MedCalc Software Inc, Belgium) and Microsoft Office Excel 2010 (Microsoft Corp, WA, USA). P < 0.05 was considered as statistically significant. Kolmogorov-Smirnov test was applied to confirm that the data were normally distributed. The results were expressed as the means ± standard deviations (SD).
Paired t-tests were used to compare the differences in ocular biometry measurements (AL, ACD, AD, CCT, LT, WTW and keratometry) between the Lenstar and the OA-2000. Bland-Altman plots were used to assess the agreement between the two devices. The 95% limits of agreement (LoA) were expressed as the mean difference ± 1.96 SD of difference, which indicated an interval within which 95% of the differences between measurements are expected to lie21, 48.
References
Olsen, T. Sources of error in intraocular lens power calculation. J Cataract Refract Surg 18, 125–129, doi:10.1016/S0886-3350(13)80917-0 (1992).
Olsen, T. Calculation of intraocular lens power: a review. Acta Ophthalmol Scand 85, 472–485, doi:10.1111/j.1600-0420.2007.00879.x (2007).
Lee, A. C., Qazi, M. A. & Pepose, J. S. Biometry and intraocular lens power calculation. Curr Opin Ophthalmol 19, 13–17, doi:10.1097/ICU.0b013e3282f1c5ad (2008).
Devereux, J. G. et al. Anterior chamber depth measurement as a screening tool for primary angle-closure glaucoma in an East Asian population. Arch Ophthalmol 118, 257–263, doi:10.1001/archopht.118.2.257 (2000).
Langenbucher, A. et al. Measurement of accommodation after implantation of an accommodating posterior chamber intraocular lens. J Cataract Refract Surg 29, 677–685, doi:10.1016/S0886-3350(02)01893-X (2003).
Tsorbatzoglou, A., Nemeth, G., Szell, N., Biro, Z. & Berta, A. Anterior segment changes with age and during accommodation measured with partial coherence interferometry. J Cataract Refract Surg 33, 1597–1601, doi:10.1016/j.jcrs.2007.05.021 (2007).
Wang, Z., Chen, J. & Yang, B. Posterior corneal surface topographic changes after laser in situ keratomileusis are related to residual corneal bed thickness. Ophthalmology 106, 406–409; discussion 409–410, doi:10.1016/S0161-6420(99)90083-0 (1999).
Swarbrick, H. A., Wong, G. & O’Leary, D. J. Corneal response to orthokeratology. Optom Vis Sci 75, 791–799, doi:10.1097/00006324-199811000-00019 (1998).
Doughty, M. J. & Zaman, M. L. Human corneal thickness and its impact on intraocular pressure measures: a review and meta-analysis approach. Surv Ophthalmol 44, 367–408, doi:10.1016/S0039-6257(00)00110-7 (2000).
Auffarth, G. U., Wang, L. & Volcker, H. E. Keratoconus evaluation using the Orbscan Topography System. J Cataract Refract Surg 26, 222–228, doi:10.1016/S0886-3350(99)00355-7 (2000).
Ucakhan, O. O., Ozkan, M. & Kanpolat, A. Corneal thickness measurements in normal and keratoconic eyes: Pentacam comprehensive eye scanner versus noncontact specular microscopy and ultrasound pachymetry. J Cataract Refract Surg 32, 970–977, doi:10.1016/j.jcrs.2006.02.037 (2006).
Barkana, Y. et al. Central corneal thickness measurement with the Pentacam Scheimpflug system, optical low-coherence reflectometry pachymeter, and ultrasound pachymetry. J Cataract Refract Surg 31, 1729–1735, doi:10.1016/j.jcrs.2005.03.058 (2005).
Chen, Y. A., Hirnschall, N. & Findl, O. Evaluation of 2 new optical biometry devices and comparison with the current gold standard biometer. J Cataract Refract Surg 37, 513–517, doi:10.1016/j.jcrs.2010.10.041 (2011).
Santodomingo-Rubido, J., Mallen, E. A., Gilmartin, B. & Wolffsohn, J. S. A new non-contact optical device for ocular biometry. Br J Ophthalmol 86, 458–462, doi:10.1136/bjo.86.4.458 (2002).
Huang, J. et al. Comparison of anterior segment measurements with rotating Scheimpflug photography and partial coherence reflectometry. J Cataract Refract Surg 37, 341–348, doi:10.1016/j.jcrs.2010.08.044 (2011).
Holzer, M. P., Mamusa, M. & Auffarth, G. U. Accuracy of a new partial coherence interferometry analyser for biometric measurements. Br J Ophthalmol 93, 807–810, doi:10.1136/bjo.2008.152736 (2009).
Cruysberg, L. P. et al. Evaluation of the Lenstar LS 900 non-contact biometer. Br J Ophthalmol 94, 106–110, doi:10.1136/bjo.2009.161729 (2010).
Buckhurst, P . J. et al. A new optical low coherence reflectometry device for ocular biometry in cataract patients. Br J Ophthalmol 93, 949–953, doi:10.1136/bjo.2008.156554 (2009).
Goebels, S. et al. Comparison of 3 biometry devices in cataract patients. J Cataract Refract Surg 41, 2387–2393, doi:10.1016/j.jcrs.2015.05.028 (2015).
Huang J, et al. Repeatability and interobserver reproducibility of a new optical biometer based on swept-source optical coherence tomography and comparison with IOLMaster. Br J Ophthalmol 101, 493-498, doi:10.1136/bjophthalmol-2016-308352 (2017).
Bland, J. M. & Altman, D. G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1, 307–310, doi:10.1016/S0140-6736(86)90837-8 (1986).
Institution., B. S. Accuracy (Trueness and Precision) of Measurement Methods and Results: General Principles and Definitions. London: HMO BS ISO 5725 part 1 (1994).
Institution., B. S. Accuracy (Trueness and Precision) of Measurement Methods and Results: Basic Methods for the Determination of Repeatability and Reproducibility of a Standard Measurement Method. London: HMO BS ISO 5725 part 2 (1994).
Kongsap, P. Comparison of a new optical biometer and a standard biometer in cataract patients. Eye & Vision 3, 27, doi:10.1186/s40662-016-0059-1 (2016).
Kunert, K. S. et al. Repeatability and agreement in optical biometry of a new swept-source optical coherence tomography-based biometer versus partial coherence interferometry and optical low-coherence reflectometry. J Cataract Refract Surg 42, 76–83, doi:10.1016/j.jcrs.2015.07.039 (2016).
Huang, J. et al. Measurement of central corneal thickness with optical low-coherence reflectometry and ultrasound pachymetry in normal and post-femtosecond laser in situ keratomileusis eyes. Cornea 34, 204–208, doi:10.1097/ICO.0000000000000329 (2015).
Bayhan, H. A., Aslan Bayhan, S. & Can, I. Comparison of central corneal thickness measurements with three new optical devices and a standard ultrasonic pachymeter. International journal of ophthalmology 7, 302–308, doi:10.3980/j.issn.2222-3959.2014.02.19 (2014).
Huang, J. et al. Precision of a new Scheimpflug and Placido-disk analyzer in measuring corneal thickness and agreement with ultrasound pachymetry. Journal of cataract and refractive surgery 39, 219–224, doi:10.1016/j.jcrs.2012.10.034 (2013).
Bao, F. et al. Comparison and evaluation of central corneal thickness using 2 new noncontact specular microscopes and conventional pachymetry devices. Cornea 33, 576–581, doi:10.1097/ICO.0000000000000113 (2014).
Kohlhaas, M. et al. Effect of central corneal thickness, corneal curvature, and axial length on applanation tonometry. Arch Ophthalmol 124, 471–476, doi:10.1001/archopht.124.4.471 (2006).
Bolz, M., Prinz, A., Drexler, W. & Findl, O. Linear relationship of refractive and biometric lenticular changes during accommodation in emmetropic and myopic eyes. Br J Ophthalmol 91, 360–365, doi:10.1136/bjo.2006.099879 (2007).
Malyugin, B. E., Shpak, A. A. & Pokrovskiy, D. F. Accommodative changes in anterior chamber depth in patients with high myopia. J Cataract Refract Surg 38, 1403–1407, doi:10.1016/j.jcrs.2012.04.030 (2012).
Read, S. A., Collins, M. J., Woodman, E. C. & Cheong, S. H. Axial length changes during accommodation in myopes and emmetropes. Optometry and vision science: official publication of the American Academy of Optometry 87, 656–662, doi:10.1097/OPX.0b013e3181e87dd3 (2010).
Dominguez-Vicent, A., Monsalvez-Romin, D., Albarran-Diego, C., Sanchis-Jurado, V. & Montes-Mico, R. Changes in anterior chamber eye during accommodation as assessed using a Dual Scheimpflug system. Arq Bras Oftalmol 77, 243–249, doi:10.5935/0004-2749.20140062 (2014).
Dominguez-Vicent, A., Monsalvez-Romin, D., Del Aguila-Carrasco, A. J., Ferrer-Blasco, T. & Montes-Mico, R. Changes in the anterior chamber during accommodation assessed with a Scheimpflug system. J Cataract Refract Surg 40, 1790–1797, doi:10.1016/j.jcrs.2014.02.043 (2014).
Li, J. et al. Measurement agreement between a new biometer based on partial coherence interferometry and a validated biometer based on optical low-coherence reflectometry. J Cataract Refract Surg 42, 68–75, doi:10.1016/j.jcrs.2015.05.042 (2016).
Olsen, T. & Hoffmann, P. C constant: new concept for ray tracing-assisted intraocular lens power calculation. J Cataract Refract Surg 40, 764–773, doi:10.1016/j.jcrs.2013.10.037 (2014).
Thibos, L. N., Wheeler, W. & Horner, D. Power vectors: an application of Fourier analysis to the description and statistical analysis of refractive error. Optom Vis Sci 74, 367–375, doi:10.1097/00006324-199706000-00019 (1997).
Rozema, J. J., Wouters, K., Mathysen, D. G. & Tassignon, M. J. Overview of the repeatability, reproducibility, and agreement of the biometry values provided by various ophthalmic devices. Am J Ophthalmol 158(1111–1120), e1111, doi:10.1016/j.ajo.2014.08.014 (2014).
Huang, J. et al. Comparison of keratometry and white-to-white measurements obtained by Lenstar with those obtained by autokeratometry and corneal topography. Cont Lens Anterior Eye 38, 363–367, doi:10.1016/j.clae.2015.04.003 (2015).
Shin, M. C., Chung, S. Y., Hwang, H. S. & Han, K. E. Comparison of Two Optical Biometers. Optometry and vision science: official publication of the American Academy of Optometry 93, 259–265, doi:10.1097/OPX.0000000000000799 (2016).
Huang, J. et al. The effect of cycloplegia on the lenstar and the IOLMaster biometry. Optom Vis Sci 89, 1691–1696, doi:10.1097/ OPX.0b013e3182772f4f (2012).
Chen, W. et al. Scheimpflug-Placido topographer and optical low-coherence reflectometry biometer: repeatability and agreement. J Cataract Refract Surg 38, 1626–1632, doi:10.1016/j.jcrs.2012.04.031 (2012).
McAlinden C, et al. Axial length measurement failure rates with biometers using swept source optical coherence tomography compared to partial coherence interferometry and optical low-coherence interferometry. American journal of ophthalmology 173, 64–69, doi:10.1016/j.ajo.2016.09.019 (2017).
Hoffer, K. J. Definition of ACD. Ophthalmology 118, 1484, doi:10.1016/j.ophtha.2011.03.009 (2011).
Read, S. A. & Collins, M. J. Diurnal variation of corneal shape and thickness. Optom Vis Sci 86, 170–180, doi:10.1097/OPX.0b013e3181981b7e (2009).
Katz, J., Zeger, S. & Liang, K. Y. Appropriate statistical methods to account for similarities in binary outcomes between fellow eyes. Invest Ophthalmol Vis Sci 35, 2461–2465 (1994).
Huang, J. et al. A comprehensive comparison of central corneal thickness measurement. Optometry and vision science: official publication of the American Academy of Optometry 88, 940–949, doi:10.1097/OPX.0b013e31821ffe2c (2011).
Acknowledgements
Supported in part by the National Key Research and Development Program of China (2016YFC0100200, 2016YFC0100201); Foundation of Wenzhou City Science & Technology Bureau (J20140014, Y20150076, Y20150068); Health Bureau of Zhejiang Province (2016RCB013); Zhejiang Provincial & Ministry of Health Research Fund For Medical Sciences (WKJ-ZJ-1530); National Science and Technology Major Project (2014ZX09303301). The contribution of G.B. Bietti Foundation IRCCS was supported by the Italian Ministry of Health and Fondazione Roma.
Author information
Authors and Affiliations
Contributions
Concept and design R.G., H.C., Y.M., Q.W., J.H.; data collection J.Y., W.Z.; statistical expertise X.W., G.S.; literature research G.S., Y.M., X.W., J.H.; administrative, technical or logistic support Q.W., J.H.; analysis and interpretation R.G., G.S., Y.M., J.Y., W.Z., Q.W., J.H.; writing the article G.S., Q.W., J.H.; critical revision of the article R.G., H.C., G.S., Q.W., J.H.; final approval of the article R.G., H.C., G.S., Y.M., Q.W., J.H.; All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, R., Chen, H., Savini, G. et al. Comparison of ocular biometric measurements between a new swept-source optical coherence tomography and a common optical low coherence reflectometry. Sci Rep 7, 2484 (2017). https://doi.org/10.1038/s41598-017-02463-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02463-z
This article is cited by
-
Repeatability of a new swept-source optical coherence tomographer and agreement with other three optical biometers
Graefe's Archive for Clinical and Experimental Ophthalmology (2022)
-
Effect of orthokeratology on precision and agreement assessment of a new swept-source optical coherence tomography biometer
Eye and Vision (2020)
-
Comparison of composite and segmental methods for acquiring optical axial length with swept-source optical coherence tomography
Scientific Reports (2020)
-
Comparison of three optical biometers: IOLMaster 500, Lenstar LS 900 and Aladdin
International Ophthalmology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.