Abstract
Hearing loss is a global public health problem with a high prevalence, significantly impairing communication and leading to a decrease in the quality of life. The association between moderate alcohol consumption (MAC) and hearing impairment has been addressed in several studies with inconsistent results. The intent of our study is to clarify the correlation between MAC and the hearing threshold and further investigate the interplay between MAC and the hearing threshold categorized by gender. The study included 4,075 participants aged 20–69 years from the 1999–2004 data of National Health and Nutrition Examination Survey (NHANES). The associations among MAC, gender differences, and high-frequency and low-frequency hearing thresholds were analyzed. We found that current female drinkers with MAC tended to have lower hearing thresholds. There is a significant protective effect of MAC on hearing threshold shifts in the US adult population, especially in females. Our research was the first study to further indicate that there is a gender difference in the association between MAC and hearing impairment. In accordance with our results, if people drink, they should consume moderate rather than higher amounts, especially in women, which may result in a reduced risk of hearing loss.
Similar content being viewed by others
Introduction
Hearing loss is a highly prevalent sensory disorder and global public health problem. It significantly impairs communication and leads to a decrease in the quality of life because it negatively affects social interaction, leading to progressive isolation and withdrawal1. The widely known causes of hearing loss include aging, exposure to noise, ototoxic agents, inherent genetic determination, diabetes, hypertension, and cardiovascular disease2. By analyzing the possible contributing factors to hearing loss, many studies attempted to identify the possible modifiable factors that can potentially help to prevent or slow the development of hearing loss3,4,5,6,7. Notably, alcohol consumption seemed to have a protective effect on hearing impairment.
The most impressive effect of alcohol consumption is its protective association with cardiovascular disease. The accumulating evidence of consistent observational and mechanistic studies reveals that moderate alcohol consumption (MAC, 1 drink per day for women and 1–2 drinks per day for men) has beneficial effects on cardiovascular disease8. The plausible mechanisms about the protective effect of alcohol consumption on cardiovascular disease include increasing levels of plasma high-density lipoprotein (HDL) cholesterol9, 10, improving insulin sensitivity11, 12, the potential salutary effects of endothelial function13, 14, fibrinolysis/coagulation15, 16, and anti-inflammatory action17, 18.
The association between alcohol consumption and hearing impairment has also been reported in several research works. These studies appear to contradict one another; some of them noted a detrimental effect or positive association19, 20, some found a beneficial effect or negative association4, 21,22,23,24,25,26, and others observed no remarkable association27,28,29. Different study methods or purposes or different definitions of alcohol consumption might have been responsible for these inconsistent results. In general, the protective effect of alcohol consumption on hearing impairment was addressed, and the decreased protective effect or increased risk of hearing loss was noted regarding alcohol consumption at higher levels than moderate levels.
Gender differences in the consequences of alcohol use have been extensively studied. Women are more susceptible to the effects of alcohol consumption than men because women have higher blood alcohol concentrations after drinking equivalent doses of alcohol per kilogram of body weight30,31,32. Compared with men, women develop cirrhosis of the liver and more cognitive and motor impairment at lower levels of exposure to alcohol33. Gender differences in alcoholic cardiomyopathy, alcohol related behavioral or medical problems, and alcohol use and misuse in adults have also been reported34. Within the extensive research on alcohol consumption, comparatively little has focused on the gender differences in the association between MAC and hearing impairment. Among the studies indicating that MAC had a protective effect on hearing impairment, some did not find different effects between women and men4, 21, 23, while others did not analyze gender effects22, 24.
By studying the possible beneficial or risk factors for hearing impairment, we would be able to understand the etiologic factors of hearing loss, develop ideas for basic research, and make public health efforts for hearing preservation. Knowledge regarding the MAC, hearing impairment, and gender differences is rare. Given the theoretical positions taken for the study and the status of the field as briefly reviewed above, the aim of our study was to further clarify the interactions between MAC, hearing thresholds, and gender distribution using the National Health and Nutrition Examination Survey(NHANES) dataset.
Results
Characteristics of the study population
The present study included 4,075 participants obtained from the 1999–2004 NHANES dataset. The clinical and demographic characteristics of the study group were analyzed according to the patterns of alcohol consumption (Table 1). The current drinkers with MAC had a smaller waist circumference, lower fasting glucose levels, lower hearing threshold at low frequencies in the worse ear, lower frequencies of stroke, heart disease, and present use of ototoxic medication than lifetime abstainers and current drinkers with above moderate alcohol consumption (AMAC). The lifetime abstainers were younger, had lower uric acid and total cholesterol levels, lower hearing threshold at high frequencies in the worse ear, fewer males and non-Hispanic whites than those with the other three patterns of alcohol consumption.
Association between patterns of alcohol consumption and hearing thresholds
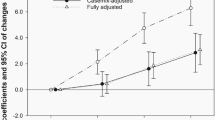
The results of patterns of alcohol consumption-based multiple linear regression analysis are presented in Table 2. Current drinkers with MAC tended to have lower hearing thresholds. The significant negative associations between MAC and the hearing thresholds, high-frequency pure-tone average (High-PTA) and low-frequency pure-tone average (Low-PTA), were noted in model 1–3. After adjusting with other covariates in model 3, the β coefficients of the High-PTA comparing the current drinkers with MAC to lifetime abstainers was −0.112 (p = 0.030); the β coefficients of the Low-PTA was −0.131 (p = 0.013).
Association between patterns of alcohol consumption and hearing thresholds in different genders
The relationships between patterns of alcohol consumption and hearing thresholds in different genders were analyzed and are presented in Table 3. Current female drinkers with MAC tended to have lower hearing thresholds. In model 1, model 2, and model 3, significant negative associations between MAC and hearing thresholds (High-PTA and Low-PTA) were observed. In model 3, the β coefficients of the High-PTA comparing the current female drinkers with MAC to lifetime abstainers was −0.154 (p = 0.013); the β coefficients of the Low-PTA was −0.150 (p = 0.034). However, no significant associations were observed in males.
Discussion
Based on analyzing the national representative sample of US adults, we found that current drinkers with MAC had significant negative associations regarding high-frequency and low-frequency hearing thresholds compared with lifetime abstainers but former drinkers and current drinkers with AMAC did not. That is, current drinkers with MAC had lower high-frequency and low-frequency hearing thresholds than lifetime abstainers. It is worth noting that this association was observed in female subjects but not in male. MAC seems to have a protective effect on hearing threshold shift only in women. To our best knowledge, ours was the first study to indicate a gender difference in the association between MAC and hearing impairment.
Several studies have suggested that alcohol consumption has a protective effect on hearing impairment. Compared to never drinkers, some studies reported that the protective effect was only noted in current drinkers with MAC (a U-shaped association was presented between alcohol consumption and hearing impairment with no risk for AMAC and lower risk for MAC)4, 21, 22, 24, and other studies report that the protective effect was observed in current drinkers with all categories, even above average or higher levels25, 26. The results of the Japanese epidemiological cross-sectional study with 496 subjects by Itoh et al.22 and the Blue Mountains Hearing Study with 2956 subjects in Australia by Gopinath et al.24 showed the protective effect of MAC. However, Itoh et al. did not differentiate high and low frequency hearing loss, Gopinath et al. only found significance in low frequency hearing loss, and these two studies did not analyze the gender effect. In another two cross-sectional studies by Popelka et al. in America21 and Fransen et al. in Europe4, they found a protective effect of MAC in both low and high frequency hearing loss, which were consistent with our results. However, the gender differences were not observed in their analysis. Our findings extended the scanty literature about MAC and hearing loss and further indicated the gender differences. Further larger studies or prospective surveys are still needed to explore the gender differences in the protective effect of MAC on hearing impairment more clearly.
The protective effects of alcohol consumption on hearing impairment may occur through several possible pathways or mechanisms. First, as far as we know, cardiovascular disease is associated with hearing loss2. Some studies proposed that the beneficial cardiovascular effects of alcohol consumption lead to the decreased risks of hearing loss4, 24, 25. Second, alcohol consumption may increase plasma HDL cholesterol concentrations8,9,10, contribute to better endothelial function13, 14, and reduce coagulation15, 16, which contribute to an optimal cochlear circulation and an intensifying anti-atherosclerotic condition and result in decreased hearing impairment. Third, alcohol consumption had anti-inflammatory action and neuroprotection17, 18, 35 that may help to enhance cellular survival of the cochlea. Finally, a U-shape association between alcohol consumption and hearing impairment was reported and the lack of enhanced benefits of higher levels of alcohol consumption or AMAC on hearing impairment is likely due to the adverse effects on other risk factors for hearing loss4, 21, 22, 24.
The tonotopic distribution of the cochlear hair cells leads to the base part of the cochlea responding to high frequencies and the apical part to low frequencies. We found a protective effect of MAC on the hearing threshold shift in both low and high frequency. It indicated this effect was influencing the whole cochlea. It was consistent with the strial hearing loss proposed by Schuknecht36, which is characterized by histopathologic findings with atrophy of the stria vascularis in the whole cochlea and by clinical results of decreased hearing sensitivity across the whole frequency spectrum. In other words, the protective effect of MAC may be related to the involvement of the stria vascularis, affecting the microvasculature of the cochlea. Notably, this is consistent with the possible mechanism about the protective effects of alcohol consumption that we described in the second point above; further study is required to investigate whether the stria vascularis of the whole cochlea is affected by alcohol consumption.
Lifestyle variables may also be confounding factors of the hearing protective effects between current drinkers with MAC and never drinkers. Emerging studies have reported that never drinkers had a higher body mass index, less regular exercise, lower vegetable consumption, and higher fat intake comparing with all drinkers37. Current drinkers with MAC were observed to pay more attention to their health using preventive care or improving their health behaviors than never and heavy drinkers38. Other confounding factors from MAC, such as socioeconomic status, have also been suggested39.
The gender differences in the association of MAC and hearing threshold shifts is likely due to innate biological differences. Women are more susceptible to the effects of alcohol consumption than men because women have higher blood alcohol concentrations after drinking equivalent doses of alcohol per kilogram of body weight30,31,32. There are several differences between men and women in the pharmacokinetics of alcohol. Women have lower gastric alcohol dehydrogenase; thus, the first stage of alcohol metabolism is decreased and more alcohol is absorbed into the blood stream30, 31. Women have a higher percentage of body fat32 and a lower percentage of body water than men40; thus, each drink is more concentrated in women’s blood circulation. Therefore, gender differences should always be considered when studying the effects of alcohol consumption.
There are some limitations in our study. First, causality cannot be claimed because of the cross-sectional design of our study. Second, we could not exclude potential recall bias for information about the frequency and quantity of past and current alcohol consumption and other personal and medical history data. Third, the questions in the Alcohol Use Questionnaire are about drinking alcoholic beverages, including liquor, wine, beer, and any other type of alcoholic beverage. The types of alcoholic beverages cannot be differentiated in our dataset. Fourth, there was no information about genetic or congenital hearing impairment in the NHANES data.
Conclusions
Our study highlighted that there is a significant protective effect of MAC on hearing threshold shifts in the US adult population, especially in female individuals. This study may lead to a better understanding of the interplay between gender differences, MAC, and hearing threshold shifts. If people drink, they should consume moderate rather than higher amounts, especially in women, which may result in a reduced risk of hearing loss. Further research is warranted to clarify the benefit of MAC on hearing threshold shifts and gender differences.
Methods
Description of NHANES and study population
NHANES is a special cross-sectional survey that combines personal interviews, standardized physical examinations, and numerous laboratory studies. It was directed by the Centers for Disease Control and Prevention’s National Center for Health Statistics and designed to gather information about the health, nutritional status, and health behaviors of the non-institutionalized civilian resident population of the United States. Starting in 1999, it became an ongoing annual survey; the data sets have been released every two years on the NHANES website.
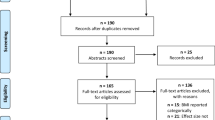
We limited our study subjects to 1999–2004 NHANES individuals who had completed both the personal questionnaires about alcohol consumption and audiometric examinations. Individuals were excluded if they had current or past occupational noise exposure and if their relevant data were incomplete. The final analytic data sets included 4,075 subjects aged 20–69 years, including demographic data, the results of physical investigations and laboratory tests, questionnaire information, and audiometric examinations.
Audiometric measurements
According to the 1999–2004 NHANES protocol, half of the included participants were randomly arranged for audiometric examinations after household personal interview and medical examinations. The participants were not included if they could not remove their hearing aids or bear with the headphones. Audiometric measurement was performed in a mobile examination center’s sound-isolated room by technicians who had been trained by a qualified audiologist from the National Institute for Occupational Safety and Health. Audiometric equipment included an interacoustic model AD226 audiometer with EARTone 3 A insert earphones and standard TDH-39P headphone. Each ear was tested from 500 to 8000 Hz with an intensity-range of −10 to 120 dB to determine the air-conduction pure tone hearing thresholds. The average of pure tone hearing thresholds at 3,000, 4,000, 6,000, and 8,000 Hz was defined as the high-frequency pure-tone average (High-PTA). The average of pure tone hearing thresholds at 500, 1,000, and 2,000 Hz was defined as the low-frequency pure-tone average (Low-PTA). The worse ear with the lower pure-tone average was chosen for the regression analysis41.
Definition of patterns of alcohol consumption
The Alcohol Use Questionnaire (ALQ) in NHANES was designed to collect information about the frequency and quantity of past and current alcohol consumption. The definition of a drink was an ounce of liquor, a 4-ounce glass of wine, or a 12-ounce beer. The variable of drinks per day (dkspd) was calculated by the ALQ data42. We classified individuals into the following four groups42,43,44: (I) lifetime abstainers- < 12 drinks in entire life; (II) former drinkers− ≧ 12 drinks in the past but none during the past 12 months; (III) current drinkers with MAC- < 0 to ≦2 dkspd in men and <0 to ≦ 1 dkspd in women during the past 12 months; and (IV) current drinkers with AMAC- > 2 dkspd in men and >1 dkspd in women during the past 12 months.
Assessment of covariates
Demographic data, including age, gender, race, and medical history were collected. Race was classified as non-Hispanic white, non-Hispanic black, or other. Individuals were considered to have diabetes if there was a physician’s diagnosis by self-report medical history, random and fasting glucose level greater than or equal to 200 and 126, respectively, and/or the use of diabetic medications. Heart disease was considered positive if an individual had a diagnosis of or had experienced congestive heart failure, coronary artery disease, myocardial infarction or angina. Stroke was ascertained by self-reports. The present use of ototoxic medication, including non-steroidal anti-inflammatory drugs, anticancer drugs, aminoglycoside, or loop diuretics was defined by self-reports. Waist circumference was determined at the top of the hip bone after breathing out normally to the nearest 0.1 cm. Abnormal otoscopy was recorded if there were abnormalities in screening otoscopic examination of the eardrums and ear canals before the audiometric measurement. Tympanometry was determined as abnormal if the compliance was less than or equal to 0.3 ml or middle ear peak pressure was less than or equal to −150 daPa using an audiometric tympanometer. Measurement of serum uric acid was determined using a Hitachi 737 analyzer. The level of total cholesterol was measured using a Hitachi 704 analyzer. The level of serum fasting glucose was measured using the Cobas Mira assay. All associated information about biospecimen collection and processing protocols is available on the NHANES website.
Statistical analysis
SPSS (version 18.0 for Windows; SPSS, Inc., Chicago, IL, USA) was used to perform the statistical analyses. The significant differences were defined as two-sided p-values < 0.05. Covariates, including age, waist circumference, uric acid, fasting glucose, and total cholesterol were managed as continuous variables and presented as the mean value with standard deviation. Other covariates, such as gender and race were treated as categorical variables and expressed as numbers with percentages. Logarithmic transformation was performed for the values of the PTA hearing thresholds to approximate a normal distribution. Our study used patterns of alcohol consumption-base analysis by dividing participants into four groups with the lifetime abstainers as the referent group. The linear regression model was used to evaluate the effect of patterns of alcohol consumption on the hearing threshold. A multiple-model analysis was performed for adjustments of covariates41. First, age, gender, and race were adjusted in model 1. Model 1 subsequently adjusted for waist circumference, uric acid, fasting glucose, and total cholesterol was model 2. Model 2 further adjusted for abnormal otoscopy and tympanometry, current use of ototoxic medication, history of diabetes, heart disease and stroke was model 3.
Ethics statement
The Research Ethics Review Board of National Center for Health Statistics approved the NHANES study and underwent annual review. All eligible subjects provided their written informed consent before participating the study. Because de-identified data were used in our study, it was exempt from IRB review.
References
Agrawal, Y., Platz, E. A. & Niparko, J. K. Prevalence of hearing loss and differences by demographic characteristics among US adults: data from the National Health and Nutrition Examination Survey, 1999–2004. Arch Intern Med 168, 1522–1530 (2008).
Yamasoba, T. et al. Current concepts in age-related hearing loss: epidemiology and mechanistic pathways. Hear Res 303, 30–38 (2013).
Gates, G. A., Cobb, J. L., D’Agostino, R. B. & Wolf, P. A. The relation of hearing in the elderly to the presence of cardiovascular disease and cardiovascular risk factors. Arch Otolaryngol Head Neck Surg 119, 156–161 (1993).
Fransen, E. et al. Occupational noise, smoking, and a high body mass index are risk factors for age-related hearing impairment and moderate alcohol consumption is protective: a European population-based multicenter study. J Assoc Res Otolaryngol 9, 264–276; discussion 261–263 (2008).
Nash, S. D. et al. The prevalence of hearing impairment and associated risk factors: the Beaver Dam Offspring Study. Arch Otolaryngol Head Neck Surg 137, 432–439 (2011).
Curhan, S. G., Eavey, R., Wang, M., Stampfer, M. J. & Curhan, G. C. Body mass index, waist circumference, physical activity, and risk of hearing loss in women. Am J Med 126(1142), e1141–1148 (2013).
Cruickshanks, K. J. et al. Smoking, central adiposity, and poor glycemic control increase risk of hearing impairment. J Am Geriatr Soc 63, 918–924 (2015).
Matsumoto, C., Miedema, M. D., Ofman, P., Gaziano, J. M. & Sesso, H. D. An expanding knowledge of the mechanisms and effects of alcohol consumption on cardiovascular disease. J Cardiopulm Rehabil Prev 34, 159–171 (2014).
Muth, N. D., Laughlin, G. A., von Muhlen, D., Smith, S. C. & Barrett-Connor, E. High-density lipoprotein subclasses are a potential intermediary between alcohol intake and reduced risk of cardiovascular disease: the Rancho Bernardo Study. Br J Nutr 104, 1034–1042 (2010).
Brien, S. E., Ronksley, P. E., Turner, B. J., Mukamal, K. J. & Ghali, W. A. Effect of alcohol consumption on biological markers associated with risk of coronary heart disease: systematic review and meta-analysis of interventional studies. BMJ 342, d636 (2011).
Davies, M. J. et al. Effects of moderate alcohol intake on fasting insulin and glucose concentrations and insulin sensitivity in postmenopausal women: a randomized controlled trial. JAMA 287, 2559–2562 (2002).
Sierksma, A. et al. Effect of moderate alcohol consumption on adiponectin, tumor necrosis factor-alpha, and insulin sensitivity. Diabetes Care 27, 184–189 (2004).
Suzuki, K. et al. Moderate alcohol consumption is associated with better endothelial function: a cross sectional study. BMC Cardiovasc Disord 9, 8 (2009).
Teragawa, H. et al. Effect of alcohol consumption on endothelial function in men with coronary artery disease. Atherosclerosis 165, 145–152 (2002).
Ridker, P. M., Vaughan, D. E., Stampfer, M. J., Glynn, R. J. & Hennekens, C. H. Association of moderate alcohol consumption and plasma concentration of endogenous tissue-type plasminogen activator. JAMA 272, 929–933 (1994).
Salem, R. O. & Laposata, M. Effects of alcohol on hemostasis. Am J Clin Pathol 123(Suppl), S96–105 (2005).
Albert, M. A., Glynn, R. J. & Ridker, P. M. Alcohol consumption and plasma concentration of C-reactive protein. Circulation 107, 443–447 (2003).
Imhof, A. et al. Effect of alcohol consumption on systemic markers of inflammation. Lancet 357, 763–767 (2001).
Spitzer, J. B. Auditory effects of chronic alcoholism. Drug Alcohol Depend 8, 317–335 (1981).
Rosenhall, U., Sixt, E., Sundh, V. & Svanborg, A. Correlations between presbyacusis and extrinsic noxious factors. Audiology 32, 234–243 (1993).
Popelka, M. M. et al. Moderate alcohol consumption and hearing loss: a protective effect. J Am Geriatr Soc 48, 1273–1278 (2000).
Itoh, A. et al. Smoking and drinking habits as risk factors for hearing loss in the elderly: epidemiological study of subjects undergoing routine health checks in Aichi, Japan. Public Health 115, 192–196 (2001).
Helzner, E. P. et al. Race and sex differences in age-related hearing loss: the Health, Aging and Body Composition Study. J Am Geriatr Soc 53, 2119–2127 (2005).
Gopinath, B. et al. The effects of smoking and alcohol consumption on age-related hearing loss: the Blue Mountains Hearing Study. Ear Hear 31, 277–282 (2010).
Dawes, P. et al. Cigarette smoking, passive smoking, alcohol consumption, and hearing loss. J Assoc Res Otolaryngol 15, 663–674 (2014).
Rigters, S. C. et al. Contributing Determinants to Hearing Loss in Elderly Men and Women: Results from the Population-Based Rotterdam Study. Audiol Neurootol 21(Suppl 1), 10–15 (2016).
Brant, L. J. et al. Risk factors related to age-associated hearing loss in the speech frequencies. J Am Acad Audiol 7, 152–160 (1996).
Curhan, S. G., Eavey, R., Shargorodsky, J. & Curhan, G. C. Prospective study of alcohol use and hearing loss in men. Ear Hear 32, 46–52 (2011).
Curhan, S. G., Eavey, R., Wang, M., Stampfer, M. J. & Curhan, G. C. Prospective study of alcohol consumption and self-reported hearing loss in women. Alcohol 49, 71–77 (2015).
Frezza, M. et al. High blood alcohol levels in women. The role of decreased gastric alcohol dehydrogenase activity and first-pass metabolism. N Engl J Med 322, 95–99 (1990).
Baraona, E. et al. Gender differences in pharmacokinetics of alcohol. Alcohol Clin Exp Res 25, 502–507 (2001).
Yue, Y. et al. Gender differences in the association between cigarette smoking, alcohol consumption and depressive symptoms: a cross-sectional study among Chinese adolescents. Sci Rep 5, 17959 (2015).
Nolen-Hoeksema, S. Gender differences in risk factors and consequences for alcohol use and problems. Clin Psychol Rev 24, 981–1010 (2004).
Erol, A. & Karpyak, V. M. Sex and gender-related differences in alcohol use and its consequences: Contemporary knowledge and future research considerations. Drug Alcohol Depend 156, 1–13 (2015).
Collins, M. A. et al. Alcohol in moderation, cardioprotection, and neuroprotection: epidemiological considerations and mechanistic studies. Alcohol Clin Exp Res 33, 206–219 (2009).
Schuknecht, H. F. & Gacek, M. R. Cochlear pathology in presbycusis. Ann Otol Rhinol Laryngol 102, 1–16 (1993).
Barefoot, J. C. et al. Alcoholic beverage preference, diet, and health habits in the UNC Alumni Heart Study. Am J Clin Nutr 76, 466–472 (2002).
Green, C. A. & Polen, M. R. The health and health behaviors of people who do not drink alcohol. Am J Prev Med 21, 298–305 (2001).
Rehm, J., Sempos, C. T. & Trevisan, M. Alcohol and cardiovascular disease–more than one paradox to consider. Average volume of alcohol consumption, patterns of drinking and risk of coronary heart disease–a review. J Cardiovasc Risk 10, 15–20 (2003).
Ely, M., Hardy, R., Longford, N. T. & Wadsworth, M. E. Gender differences in the relationship between alcohol consumption and drink problems are largely accounted for by body water. Alcohol Alcohol 34, 894–902 (1999).
Lin, Y. Y. et al. Secondhand Smoke is Associated with Hearing Threshold Shifts in Obese Adults. Sci Rep 6, 33071 (2016).
Churilla, J. R. et al. Association between alcohol consumption patterns and metabolic syndrome. Diabetes Metab Syndr 8, 119–123 (2014).
Tsai, J., Ford, E. S., Li, C. & Zhao, G. Past and current alcohol consumption patterns and elevations in serum hepatic enzymes among US adults. Addict Behav 37, 78–84 (2012).
Taylor, A. L., Denniston, M. M., Klevens, R. M., McKnight-Eily, L. R. & Jiles, R. B. Association of Hepatitis C Virus With Alcohol Use Among U.S. Adults: NHANES 2003–2010. Am J Prev Med 51, 206–215 (2016).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Y.-Y.L. and W.-L.C. Performed the experiments: Y.-Y.L., H.-C.C., W.-S.L., L.-W.W., C.-H.W., J.-C.L., T.-W.K. and W.-L.C. Analyzed the data: Y.-Y.L., H.-C.C.,W.-S.L., L.-W.W., C.-H.W., J.-C.L., T.-W.K. and W.-L.C. Contributed reagents/materials/analysis tools: Y.-Y.L., H.-C.C.,W.-S.L., L.-W.W., C.-H.W., J.-C.L., T.-W.K. and W.-L.C. Prepared Tables 1–3: Y.-Y.L., H.-C.C.,W.-S.L., L.-W.W., C.-H.W., J.-C.L., T.-W.K. and W.-L.C. Wrote the paper: Y.-Y.L. and W.-L.C. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lin, YY., Chen, HC., Lai, WS. et al. Gender Differences in the Association between Moderate Alcohol Consumption and Hearing Threshold Shifts. Sci Rep 7, 2201 (2017). https://doi.org/10.1038/s41598-017-02426-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02426-4
This article is cited by
-
Hearing loss and the COVID-19 pandemic
BMC Research Notes (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.