Abstract
The high prevalence of diabetes mellitus (DM) among multidrug resistant tuberculosis (MDR-TB) patients is a serious cause for concern. We conducted a meta-analysis to determine whether DM is an independent risk factor for MDR-TB. Electronic literature searches of the PubMed, Web of Science and EMBASE databases up to July 12, 2016 were conducted. The pooled adjusted odds ratio (OR) and 95% confidence intervals (CIs) were calculated using the random effects model with STATA 12.0 software. In total 13 studies, including 9289 individuals with TB, were included in this meta-analysis. Significant association between DM and MDR-TB (OR = 1.71; 95% CI = 1.32, 2.22) was identified. Subgroup analyses showed that: 1) Pooled OR was 1.25 (95% CI: 0.82–1.91) for cross-sectional studies, and was 2.14 (95% CI: 1.51–3.02) for longitudinal studies; 2) The pooled OR was 1.69 (95% CI:1.09–2.62) for primary MDR-TB, 1.94 (95% CI:1.42–2.65) for any MDR-TB, and 0.85 for secondary MDR-TB (95% CI: 0.29–2.54); 3) DM was significantly associated with MDR-TB in both Caucasian (OR = 2.26, 95% CI: 1.66–3.07) and Asian (OR = 1.40, 95% CI: 1.01–1.95) subgroups. No evidence of publication bias was identified. In conclusion, the pooling analysis indicated that DM was an independent risk factor for MDR-TB, especially for primary MDR-TB.
Similar content being viewed by others
Introduction
The emergence of MDR-TB (multidrug resistant tuberculosis), caused by M. tuberculosis (MTB) that is resistant to at least isoniazid and rifampicin, is posing a great threat to global public health. Despite effective preventive and therapeutic methods that have been actively promoted worldwide, it is still estimated that 3.3% of newly diagnosed tuberculosis (TB) cases and 20% of previously treated TB cases have MDR-TB according to WHO reports1. The treatment of patients infected with MDR-TB strains is extremely challenging due to the complexity of chemotherapy regimens and the toxicity of alternative drugs. Furthermore, treatment of MDR-TB imposes a huge financial burden on public health systems. However, compared with the cure rate of 96% in drug-susceptible TB, the cure rate of MDR-TB reaches only 54%, making it known as a fatal disease2, 3. Accordingly, identifying the risk factors associated with MDR-TB is of great significance, which may assist in the guidance of intervention measures, promote development of follow-up strategies in specific susceptible populations, and help decision-making in terms of resource allocation.
Several risk factors have been identified for MDR-TB4. Among them, previous treatment ranks the strongest and most frequent determinant of MDR-TB, which may be related to the selective pressure of suboptimal regimens or treatment interruptions4,5,6,7,8. Other factors include younger age, foreign born, human immunodeficiency virus (HIV) infection, smoking or other substance abuse, being a health care worker and so on9,10,11. However, as the association between most risk factors and MDR-TB differed in different regions and study designs varied, further insight into this area is required.
Recently, along with the convergence of the diabetes mellitus (DM) and TB epidemics, the high prevalence of DM among MDR-TB patients is a serious cause for concern, with a range of 10–23% of MDR-TB patients having DM12,13,14,15. Whether DM, usually accompanied with altered immunity, has an effect on MDR-TB transmission, as similar with other immunodeficiency related disease (e.g. HIV), is yet to be determined16. Findings from studies exploring the associations between DM and MDR-TB have been discordant and some studies did not consider potential confounding factors17,18,19. Thus we conducted a meta-analysis aimed to determine whether DM was an independent risk factor for MDR-TB.
Methods
Literature search
This review was registered in the International Prospective Register of Systematic Reviews PROSPERO (registration number: CRD42017057430). Electronic literature search of PubMed, Web of Science and EMBASE databases between the earliest available indexing dates to July 12, 2016 were conducted. The search strategy for PubMed was: (“Diabetes Mellitus”[Mesh] OR “diabetic”[tiab] OR “diabetes”[tiab]) AND “tuberculosis”[Mesh] AND (“Tuberculosis, Multidrug-Resistant”[Mesh] OR “drug resistance”[Mesh] OR “multidrug-resistant”[tiab] OR “multidrug resistant”[tiab] OR “multidrug resistance”[tiab] OR “drug-resistant”[tiab] OR “drug resistant”[tiab]). Web of Science was searched with the search strategy as follows: (“Diabetes Mellitus” OR “Diabetes Mellitus, Type 2” OR “diabetic” OR “diabetes”) AND “tuberculosis” AND (“multidrug-resistant” OR “multidrug resistant” OR “multidrug resistance” OR “drug-resistant” OR “drug resistant” OR “drug resistance”). EMBASE was searched with: (“Diabetes Mellitus”[Emtree] OR “diabetic”[tiab] OR “diabetes”[tiab]) AND “tuberculosis”[Emtree] AND (“drug resistant tuberculosis”[Emtree] OR “drug resistance”[Emtree] OR “multidrug-resistant”[tiab] OR “multidrug resistant”[tiab] OR “multidrug resistance”[tiab] OR “drug-resistant”[tiab] OR “drug resistant”[tiab]). Reference lists of all identified publications were reviewed to identify additional studies. No restrictions were set with respect to language in the entire search process.
Inclusion and exclusion criteria
Studies were considered eligible for inclusion based on the following criteria: (1) exploring the association between DM and MDR-TB with consideration of potential confounding factors; (2) designed as cohort, case-control, or cross-sectional studies; (3) adjusted odds ratio (OR) and 95% confidence intervals (CIs) were reported or could be calculated. In several studies, the adjusted OR was unavailable in multivariate analysis, due to non-significant statistical results during univariate analysis. Excluding those studies would bias the pooled result towards favoring DM as a risk factor. In these cases, we used a statistically non-significant crude OR instead of the unavailable adjusted OR.We excluded duplicated publications and conference abstracts.
Data extraction and definitions
Two authors (Qianqian Liu and Wenzhang Li) independently assessed the eligibility of all included studies. Disagreements were resolved by discussion. The following variables were extracted: title, name of first author, publication year, sample size, the country in which the study was conducted, study design, age, gender, MDR-TB type (primary or secondary or any), adjusted OR (or crude OR) and 95% CI, variables adjusted in multivariate regression model. MDR-TB is defined as MTB strains that show resistance at least to rifampicin and isoniazid. Secondary or primary MDR-TB was defined as patients who acquired MDR-TB with or without previously treatment, respectively.
Quality assessment
We evaluated the methodological quality of all studies in the meta-analysis using the Newcastle-Ottawa scale (NOS), which is composed of the following aspects: selection, comparability, and exposure (case-control or cross-sectional studies) or outcome (cohort studies). The maximum score was nine points. Studies with NOS score <3, 7> NOS score ≥3 and NOS score ≥7 were considered to be of poor, median, high quality, respectively20.
Statistical analysis
The χ 2 based Q statistics and I 2 test were used to assess the between-study heterogeneity (significant at p < 0.10 and/or I 2 > 50%). The pooled OR and 95% CIs were calculated using the random effects model. Subgroup analyses were performed by study design, ethnicity and MDR-TB type. Sensitivity analysis was also performed to explore the sources of heterogeneity. Publication bias was assessed by Begg rank correlation and Egger weighted regression test methods21. All statistical analyses were conducted by the metan program in STATA version 12.0 (StataCorp, College Station, Texas), using two-sided p values.
Results
Identification of eligible studies
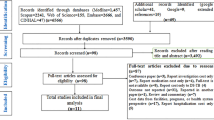
A total of 1257 studies were identified after the initial search (Fig. 1). After excluding over-lapping articles, the remaining 869 studies were screened by reading the titles and abstracts. 670 studies were further excluded for being irrelevant to the topic. We also excluded studies which were conducted in animals, review articles or case reports. Out of 31 studies reviewed in full, six were excluded for being conference abstracts and 12 were excluded because they did not considering using multivariable models to adjust for covariates. We ultimately included 13 studies in this meta-analysis10, 22,23,24,25,26,27,28,29,30,31,32,33.
Study characteristics
The characteristics of all included studies are presented in Table 1. These 13 studies were published between 2001 and 2015, including a total of 9289 individuals with TB, distributed in Asia (two in China, one in Saudi Arabia, one in Georgia, one in Bangladesh, one in Korea, one in Thailand), the Americas (three in Mexico, two in USA) and Europe (one in Spain). Among those studies, eight were designed as case-control studies, one was a cohort study and four were cross-sectional studies. Four studies clearly stated the MDR-TB type (primary or secondary), while the other nine studies did not classify patients according to history of anti-TB treatment.
Methodological quality
As shown in Table 1, the NOS scores of all included studies ranged from 6 to 9 points, with 12 out of 13 studies deemed to be of high quality. In terms of selection and outcome bias, all studies met all criteria. As for comparability bias, two studies did not adjust for age in their multivariate analyses. In assessing bias related to exposure, five studies obtained the risk factor data according to self report or medical record only, and seven studies did not report the non-response rate in both case and control groups.
Statistical results
The overall pooling result of all included studies is shown in Fig. 2 and there was statistical heterogeneity observed among studies (P = 0.02, I 2 = 46.8%). Meta-analysis indicated significant association between DM and MDR-TB, i.e., patients with DM were more likely to have MDR-TB (OR = 1.71; 95% CI = 1.32, 2.22). Sensitivity analysis revealed that most of the heterogeneity derived from one of Mi et al. studies, which focused upon the impact of DM as a risk factor on secondary MDR-TB. When we re-analyzed the data after excluding this study, the between-study heterogeneity was significantly reduced (P = 0.11, I 2 = 32.3%) and the result still confirmed that DM was an independent risk factor for MDR-TB (OR = 1.83, 95% CI: 1.45–2.31). The symmetric funnel plots shown in Fig. 3 and the lack of significance in the Egger’s test (P = 0.561) both indicated that there was no evidence of publication bias.
The subgroup analyses results by study design, MDR-TB type and ethnicity are presented in Table 2. Stratification of the data by study design showed that the OR was 1.25 (95% CI: 0.82–1.91) for cross-sectional studies, and was 2.14 (95% CI: 1.51–3.02) for longitudinal studies. When stratified by MDR-TB type, the pooled OR was 1.69 (95% CI: 1.09–2.62) for primary MDR-TB, 1.94 (95% CI:1.42–2.65) for any MDR-TB, however we did not find that DM was a risk factor for secondary MDR-TB (OR = 0.85, 95% CI: 0.29–2.54). DM was significantly associated with MDR-TB in both Caucasian (OR = 2.26, 95% CI: 1.66–3.07) and Asian (OR = 1.40, 95% CI: 1.01–1.95) subgroups. Compared with the overall pooling analysis, the heterogeneity reduced during subgroup analyses by MDR-TB type, ethnicity, and study design, especially in the Caucasian and primary MDR-TB subgroups.
Discussion
The growing prevalence of TB-DM comorbidity worldwide has provided a new challenge to clinical management and health systems control strategy34. It was observed that patients who have DM complicated with TB often experience delayed sputum culture conversion, increased risk of death and recurrence29, 35,36,37. What is more, the emergence of MDR-TB makes the adverse anti-TB treatment outcomes in TB-DM comorbidity even worse, which may increase treatment related economic burden, promote the transmission of MDR-TB, and even accelerate the generation of extensively drug-resistant-TB (XDR-TB). This meta-analysis addressed the association between DM and MDR-TB using 13 selected studies and demonstrated that complicating DM was a significant independent risk factor for MDR-TB, and the risk effect is robust regardless of ethnicity. We didn’t detect any evidence of publication bias. Our data provide a basis for the prioritization of early detection screening measures for MDR-TB among TB patients who are complicated with DM, which may be cost-effective for these patients38, 39. In addition, a more intensive anti-TB regimen and careful MDR-TB follow-up might also be suggested in those patients.
The reasons for a higher MDR rate in patients with TB-DM comorbidity are not thoroughly understood, however they may differ by different MDR type (primary or secondary). The combination of impaired immune system in DM and bacterial genetics might be a reasonable explanation for primary MDR. It has been reported that poor glucose control is often associated with dysfunction of phagocytosis, reactive oxygen species (ROS) production, chemotaxis and T-cell reaction in DM patients40. On the other hand, MDR strains are shown to be less virulent due to heterogeneous mutations and they are less likely to lead to secondary TB cases compared with drug sensitive strains41,42,43. Then the less fit MDR strains are more likely to flourish in immunocompromised DM patients, which lead to the higher primary MDR-TB in those patients. The situation seems to be more complex with regard to the mechanisms of secondary MDR-TB in DM. Possible explanations include higher mycobacterial burden, altered pharmacokinetics of anti-TB drugs and lower treatment adherence, which promote the selection of MDR strains by anti-TB drugs40, 44,45,46.
Sensitivity analyses showed that most of heterogeneity belonged to one study of Mi et al., which focused upon secondary MDR-TB. However, when we conducted the meta-analysis by excluding this research, we also arrived at consistent result. As heterogeneity reduced during subgroup analyses in this meta-analysis, we postulated that MDR-TB type, ethnicity, and perhaps study design may also contribute to heterogeneity. According to our subgroup analysis by MDR type, DM is an independent risk factor for primary MDR-TB with high homogeneity among included studies, while no association between DM and secondary MDR-TB was identified. Similar to our research, a previous meta-analysis also detected significant association between HIV (also being an immune impaired disease) and primary MDR-TB rather than secondary MDR-TB16. However, the small sample size of secondary MDR-TB in our meta-analysis might preclude us from drawing a reliable negative conclusion, and it needs to be further explored.
Although most of our included studies scored high quality using the NOS scale, there are still some aspects that need to be improved. Future studies should obtain information about the risk factors (e.g. the diagnosis of DM) according to objective laboratory data rather than basing on self-report or medical record only, which may otherwise lead to misclassification bias. Furthermore, it is necessary to completely report the non-response rate in both case and control groups, so that the existence of non-response bias can be ascertained.
Some limitations of this meta-analysis should be pointed out. First, in this meta-analysis we included data with different study designs, which may lead to heterogeneity of results. However, the similar positive association detected between DM and MDR-TB in the longitudinal study subgroup, which is more liable to prove a cause-and-effect relationship than cross-sectional studies, might strengthen our conclusion. Second, although we have strictly set the inclusion criteria to studies with multivariate analysis to adjust for potential confounding factors, we still cannot adjust the same covariates in all included studies, which may also weaken our conclusion. Third, the control group was from non-MDR TB patients in some studies while from drug susceptible TB patients in others, which may also lead to variability in the strength of association.
In conclusion, the pooling analysis indicated that DM was an independent risk factor for MDR-TB, especially for primary MDR-TB. In patients with DM-TB comorbidity, effective measures need to be implemented to promote early diagnosis of MDR-TB, and followed by intensive treatment and follow-up.
References
WHO. Global tuberculosis report 2015. Geneva, Switzerland: WHO, 2015. http://www.who.int/tb/publications/global_report/en/ (accessed July 1, 2016).
Narayanan, P. R. Evaluation of a non-rifampicin continuation phase (6HE) following thrice-weekly intensive phase for the treatment of new sputum positive pulmonary tuberculosis. The Indian journal of tuberculosis 54, 84–90 (2007).
Ahuja, S. D. et al. Multidrug resistant pulmonary tuberculosis treatment regimens and patient outcomes: an individual patient data meta-analysis of 9,153 patients. PLoS medicine 9, e1001300, doi:10.1371/journal.pmed.1001300 (2012).
Faustini, A., Hall, A. J. & Perucci, C. A. Risk factors for multidrug resistant tuberculosis in Europe: a systematic review. Thorax 61, 158–163, doi:10.1136/thx.2005.045963 (2006).
Caminero, J. A. Multidrug-resistant tuberculosis: epidemiology, risk factors and case finding. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 14, 382–390 (2010).
Espinal, M. A. et al. Determinants of drug-resistant tuberculosis: analysis of 11 countries. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 5, 887–893 (2001).
Liang, L. et al. Factors contributing to the high prevalence of multidrug-resistant tuberculosis: a study from China. Thorax 67, 632–638, doi:10.1136/thoraxjnl-2011-200018 (2012).
Skrahina, A. et al. Multidrug-resistant tuberculosis in Belarus: the size of the problem and associated risk factors. Bulletin of the World Health Organization 91, 36–45, doi:10.2471/BLT.12.104588 (2013).
Casal, M. et al. A case-control study for multidrug-resistant tuberculosis: risk factors in four European countries. Microbial drug resistance 11, 62–67, doi:10.1089/mdr.2005.11.62 (2005).
Suarez-Garcia, I. et al. Risk factors for multidrug-resistant tuberculosis in a tuberculosis unit in Madrid, Spain. European journal of clinical microbiology & infectious diseases: official publication of the European Society of Clinical Microbiology 28, 325–330, doi:10.1007/s10096-008-0627-y (2009).
O’Donnell, M. R. et al. High incidence of hospital admissions with multidrug-resistant and extensively drug-resistant tuberculosis among South African health care workers. Annals of internal medicine 153, 516–522, doi:10.7326/0003-4819-153-8-201010190-00008 (2010).
Singh, R., Gothi, D. & Joshi, J. Multidrug resistant tuberculosis: role of previous treatment with second line therapy on treatment outcome. Lung India 24, 54–57, doi:10.4103/0970-2113.44211 (2007).
Aragon, J., Litonjua, A., Tupasi, T. & Quela, I. Prevalence of type 2 diabetes among multi-drug resistant tuberculosis (MDR-TB) patients seen in Makati Medical Center under the directly observed therapy plus (DOTS PLUS) program. Philippine Journal of Internal Medicine 41, 7–10 (2003).
Tanrikulu, A. C., Hosoglu, S., Ozekinci, T., Abakay, A. & Gurkan, F. Risk factors for drug resistant tuberculosis in southeast Turkey. Trop Doct 38, 91–93, doi:10.1258/td.2007.070131 (2008).
Garcia, F., Solis, J., Calderon, J., Luque, E. & Zacarias, E. Prevalence of diabetes mellitus and related risk factors in an urban population. Revista de la Sociedad Peruana de Medicina Interna 20, 90–94 (2007).
Mesfin, Y. M., Hailemariam, D., Biadgilign, S. & Kibret, K. T. Association between HIV/AIDS and multi-drug resistance tuberculosis: a systematic review and meta-analysis. PloS one 9, e82235, doi:10.1371/journal.pone.0082235 (2014).
Chang, J. T. et al. Effect of type 2 diabetes mellitus on the clinical severity and treatment outcome in patients with pulmonary tuberculosis: a potential role in the emergence of multidrug-resistance. Journal of the Formosan Medical Association = Taiwan yi zhi 110, 372–381, doi:10.1016/S0929-6646(11)60055-7 (2011).
Magee, M. J. et al. Clinical characteristics, drug resistance, and treatment outcomes among tuberculosis patients with diabetes in Peru. International journal of infectious diseases: IJID: official publication of the International Society for Infectious Diseases 17, e404–412, doi:10.1016/j.ijid.2012.12.029 (2013).
Duangrithi, D. et al. Impact of diabetes mellitus on clinical parameters and treatment outcomes of newly diagnosed pulmonary tuberculosis patients in Thailand. International journal of clinical practice 67, 1199–1209, doi:10.1111/ijcp.12215 (2013).
Wells, G. A., Shea, B., O’Connell, D., Peterson, J., Welch, V., Losos, M. et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Available: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
Egger, M., Smith, G. D. & Phillips, A. N. Meta-analysis: principles and procedures. Bmj 315, 1533–1537, doi:10.1136/bmj.315.7121.1533 (1997).
Perez-Navarro, L. M., Fuentes-Dominguez, F. J. & Zenteno-Cuevas, R. Type 2 diabetes mellitus and its influence in the development of multidrug resistance tuberculosis in patients from southeastern Mexico. Journal of diabetes and its complications 29, 77–82, doi:10.1016/j.jdiacomp.2014.09.007 (2015).
Mi, F. et al. Is resistance to anti-tuberculosis drugs associated with type 2 diabetes mellitus? A register review in Beijing, China. Global health action 7, 24022, doi:10.3402/gha.v7.24022 (2014).
Gomez-Gomez, A. et al. Diabetes and Other Risk Factors for Multi-drug Resistant Tuberculosis in a Mexican Population with Pulmonary Tuberculosis: Case Control Study. Archives of medical research 46, 142–148, doi:10.1016/j.arcmed.2015.01.006 (2015).
Fisher-Hoch, S. P. et al. Type 2 diabetes and multidrug-resistant tuberculosis. Scandinavian journal of infectious diseases 40, 888–893, doi:10.1080/00365540802342372 (2008).
Hsu, A. H. et al. Diabetes is associated with drug-resistant tuberculosis in Eastern Taiwan. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 17, 354–356, doi:10.5588/ijtld.11.0670 (2013).
Magee, M. J. et al. Diabetes mellitus is associated with cavities, smear grade, and multidrug-resistant tuberculosis in Georgia. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 19, 685–692, doi:10.5588/ijtld.14.0811 (2015).
Rifat, M. et al. Development of multidrug resistant tuberculosis in Bangladesh: a case-control study on risk factors. PloS one 9, e105214, doi:10.1371/journal.pone.0105214 (2014).
Singla, R. et al. Influence of diabetes on manifestations and treatment outcome of pulmonary TB patients. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 10, 74–79 (2006).
Bashar, M., Alcabes, P., Rom, W. N. & Condos, R. Increased incidence of multidrug-resistant tuberculosis in diabetic patients on the Bellevue Chest Service, 1987 to 1997. Chest 120, 1514–1519, doi:10.1378/chest.120.5.1514 (2001).
Min, J., Park, K., Whang, S. & Kim, J. Risk Factors for Primary Multidrug Resistant Tuberculosis. Tuberculosis and Respiratory Diseases 59, 600–605, doi:10.4046/trd.2005.59.6.600 (2005).
Song, Q. S., Cai, C. K., Ren, Y. W. & Lu, X. W. Risk factors for MDR-TB and XDR-TB in Dalian patients. Jounal of Dalian Medical University 57, 45–48 (2015).
Jitmuang, A., Munjit, P. & Foongladda, S. Prevalence and Factors Associated with Multidrug-Resistant Tuberculosis at Siriraj Hospital, Bangkok, Thailand. The Southeast Asian journal of tropical medicine and public health 46, 697–706 (2015).
Ugarte-Gil, C. & Moore, D. A. Tuberculosis and diabetes co-morbidity: an unresolved problem. Revista peruana de medicina experimental y salud publica 31, 137–142 (2014).
Baker, M. A. et al. The impact of diabetes on tuberculosis treatment outcomes: a systematic review. BMC medicine 9, 81, doi:10.1186/1741-7015-9-81 (2011).
Leung, C. C. et al. Lower risk of tuberculosis in obesity. Archives of internal medicine 167, 1297–1304, doi:10.1001/archinte.167.12.1297 (2007).
Mi, F. et al. Diabetes mellitus and tuberculosis: pattern of tuberculosis, two-month smear conversion and treatment outcomes in Guangzhou, China. Tropical medicine & international health: TM & IH 18, 1379–1385, doi:10.1111/tmi.12198 (2013).
Djuretic, T. et al. Antibiotic resistant tuberculosis in the United Kingdom: 1993–1999. Thorax 57, 477–482, doi:10.1136/thorax.57.6.477 (2002).
Drobniewski, F. A., Watterson, S. A., Wilson, S. M. & Harris, G. S. A clinical, microbiological and economic analysis of a national service for the rapid molecular diagnosis of tuberculosis and rifampicin resistance in Mycobacterium tuberculosis. Journal of medical microbiology 49, 271–278, doi:10.1099/0022-1317-49-3-271 (2000).
Nijland, H. M. et al. Exposure to rifampicin is strongly reduced in patients with tuberculosis and type 2 diabetes. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 43, 848–854, doi:10.1086/507543 (2006).
Cohen, T. & Murray, M. Modeling epidemics of multidrug-resistant M. tuberculosis of heterogeneous fitness. Nature medicine 10, 1117–1121, doi:10.1038/nm1110 (2004).
Burgos, M., DeRiemer, K., Small, P. M., Hopewell, P. C. & Daley, C. L. Effect of drug resistance on the generation of secondary cases of tuberculosis. The Journal of infectious diseases 188, 1878–1884, doi:10.1086/379895 (2003).
Gagneux, S. et al. Impact of bacterial genetics on the transmission of isoniazid-resistant Mycobacterium tuberculosis. PLoS pathogens 2, e61, doi:10.1371/journal.ppat.0020061 (2006).
Jimenez-Corona, M. E. et al. Association of diabetes and tuberculosis: impact on treatment and post-treatment outcomes. Thorax 68, 214–220, doi:10.1136/thoraxjnl-2012-201756 (2013).
Milan-Segovia, R. C. et al. Relative bioavailability of rifampicin in a three-drug fixed-dose combination formulation. The international journal of tuberculosis and lung disease: the official journal of the International Union against Tuberculosis and Lung Disease 14, 1454–1460 (2010).
Wang, K. et al. Factors contributing to the high prevalence of multidrug-resistant tuberculosis among previously treated patients: a case-control study from China. Microbial drug resistance 20, 294–300, doi:10.1089/mdr.2013.0145 (2014).
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: Q.Q.L., W.Z.L., M.X.; Analyzed the data: Q.Q.L., W.Z.L., M.X., Y.F.C., X.M.D., C.D.W., L.N.H., Y.T., Y.L.F., C.M.T., J.Q.H.; Wrote the manuscript: Q.Q.L. and W.Z.L.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Q., Li, W., Xue, M. et al. Diabetes mellitus and the risk of multidrug resistant tuberculosis: a meta-analysis. Sci Rep 7, 1090 (2017). https://doi.org/10.1038/s41598-017-01213-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-01213-5
This article is cited by
-
Impact of hyperglycemia on tuberculosis treatment outcomes: a cohort study
Scientific Reports (2024)
-
Diabetes and infection: review of the epidemiology, mechanisms and principles of treatment
Diabetologia (2024)
-
Global burden of MDR-TB and XDR-TB attributable to high fasting plasma glucose from 1990 to 2019: a retrospective analysis based on the global burden of disease study 2019
European Journal of Clinical Microbiology & Infectious Diseases (2024)
-
Risk factors on healthcare-associated infections among tuberculosis hospitalized patients in China from 2001 to 2020: a systematic review and meta-analysis
BMC Infectious Diseases (2022)
-
New Developments and Insights in the Improvement of Mycobacterium tuberculosis Vaccines and Diagnostics Within the End TB Strategy
Current Epidemiology Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.