Abstract
LEAFY (LFY) is a plant-specific transcription factor, which is found in algae and all land plants. LFY homologs exert ancestral roles in regulating cell division and obtain novel functions to control floral identity. Isoetes L. is an ancient genus of heterosporous lycophytes. However, characters about LFY homologs in lycophytes remain poorly investigated. In this study, two LFY homologs, ILFY1 and ILFY2, were cloned from five Isoetes species, including I. hypsophila, I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. The full length of ILFY1 was 1449–1456 bp with an open reading frame (ORF) of 927–936 bp. The full length of ILFY2 was 1768 bp with ORF of 726 bp. Phylogenetic tree revealed that ILFY1 and ILFY2 were separated into two clades, and I. hypsophila were separated with the others. Expression analysis demonstrated that IsLFY1 and IsLFY2 for I. sinensis did not show functional diversity. The two transcripts were similarly accumulated in both vegetative and reproductive tissues and highly expressed in juvenile tissues. In addition, the IsLFY1 and IsLFY2 transgenic Arabidopsis similarly did not promote precocious flowering, and they were inactive to rescue lfy mutants. The results facilitate general understandings about the characteristics of LFY in Isoetes and evolutionary process.
Similar content being viewed by others
Introduction
Flowering is a common and critical event during the life cycle of angiosperm plants. During vegetative phase, apical meristems give rise to leaves and lateral shoots. Then plants switch from vegetative into reproductive development, and flowers are initiated which is controlled by floral meristem identity genes, including APETALA1(AP1) and LEAFY (LFY). LFY is a plant-specific transcript factor, which is found in algae and all land plants from mosses to angiosperms1,2,3. LFY in Arabidopsis thaliana is a master regulator for flower initiation and determining floral fate in lateral meristems4, 5. It regulates downstream genes to produce floral tissues according to the ABC model for floral organ identity6. Increasing copy number of the endogenous LFY promotes early flowering and terminates inflorescence apices with solitary flowers7, 8.
Roles of the LFY homologs have been investigated in numerous species4, 9,10,11. FLORICAULA (FLO) in Antirrhinum majus is expressed in bract primordia, sepal, petal, and carpel primordia10. However, RLF, a LFY homology in Oryza sativa, controls the branching of inflorescence12. The function of RLF for floral identity is partially lost, which has been replaced by another factors13. The unifoliata pea mutants alter in both floral development and leaf morphology14. It is generally considered that LFY homologs possess two functions in angiosperms, including conferring floral identity and promoting meristem growth15, 16. However, the two functions may be all prominent (such as in pea, maize and tobacco) or one function may be reduced and the other is obvious (such as in A. thaliana and O. sativa)4, 9, 14, 16. Another related paralog, NEEDLY is identified in gymnosperms and lost in angiosperms17. But only LFY in gymnosperms is orthologous with LFY in angiosperms. The LFY and NEEDLY paralogs in gymnosperms are specifically expressed in both vegetative and reproductive meristems18, 19. The LFY paralog provides more activity to rescue Arabidopsis lfy mutant phenotypes than NEEDLY 1, 18.
In addition, LFY homologs are also present in free-sporing land plants, including lycophytes, ferns or their allies and bryophytes1, 2, 20. Expressions of CrLFY1 and CrLFY2 in Ceratopteris richardii are prominent in shoot tips and circinate reproductive leaves20. But CrLFY2 only has some ability to rescue severe Arabidopsis lfy mutants, which is less than LFY homologs in gymnosperms1. Two PpLFY paralogs in the moss Physcomitrella patens are markedly expressed in gametophyte and sporophyte21. The PpLFY proteins play critical roles in controlling first zygotic cell division21. Whereas they are inactive to complement Arabidopsis lfy mutants1. Taken together, these observations imply that translation from the vegetative to reproductive development and directed induction of the floral homeotic MADS-box genes by LFY are established after the divergence of mosses and before the divergence of vascular plant lineage19, 20. LFY homologs probably regulate cell division, expansion and arrangement in free-sporing land plants, and they control both floral identity and cell division in seed plants16, 22. Additionally, LFY homolog is not specific to land plants and it is also found in algae, indicating ancestral roles predating land plants2, 3.
Isoetes L. is an ancient genus of heterosporous lycopsid23, 24. Phylogenetic analyses show that Isoetes is one of the earliest basal vascular plants, which can date back to the Devonian25, 26. Isoetes has approximately 200 species recognized by a strong reduced plant body23, 25, 27. Furthermore, this genus is the only survival of ancient taxa as the closest relatives of the famous tree lycopods23,24,25. The plants have fleshy corms with a range of spirally arranged microphylls and multiple dichotomizing roots along with median furrows of the corm28. Microsporangia and megasporangia are embedded in the bottom of fertile leaves. With the corms growing in girth, the leaves and sporangia are becoming mature from inside to outside29. It is prevalent to identify species and analyze phylogenetic relationships using second intron of LFY homologs30,31,32. Although Isoetes possesses an important position in phylogenetic evolution, characters and functions of LFY homologs for Isoetes (referred as ILFY) are still ambiguous.
In this present study, we cloned two ILFY paralogs, including ILFY1 and ILFY2. Totally, we identified the two genes in five Isoetes species, including I. hypsophila, I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. Roles of the two ILFY paralogs in I. sinensis (IsLFY1 and IsLFY2) were further investigated using the quantitative real-time PCR (qRT-PCR) and in situ hybridization assays. Moreover, we created transgenic Arabidopsis plants, which were constitutively expressed IsLFY1 and IsLFY2 under the control of the cauliflower mosaic virus 35S promoter, respectively. This comprehensive study will facilitate our understandings about the roles of LFY homologs in Isoetes and evolutionary process.
Results
Cloning of ILFY1 and ILFY2 sequences
The full-length of ILFY genes were cloned from five Isoetes species, including I. hypsophila, I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. In total, we isolated two ILFY paralogs, including ILFY1 and ILFY2, based on the 5′ and 3′ rapid amplifications of cDNA ends (RACE) systems. The full length of ILFY1 was 1449–1456 bp with an open reading frame (ORF) of 927–936 bp (Fig. 1). The full length of ILFY2 was 1768 bp with ORF of 726 bp. More detailed information about ILFY1 and ILFY2 was list in Table S1, including the full length cDNA, 5′UTR, open reading fragment (ORF), 3′UTR of the two homologs in the five species. The ORF sequences of ILFY1 and ILFY2 were identical for I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. ILFY1 for the four species occupied 94.86% identity with IhLFY1, and ILFY2 shared 98.68% identity with IhLFY2. Furthermore, The ORF fragments for ILFY1 and ILFY2 for I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis shared 63.41% identity. The ORF fragments for IhLFY1 and IhLFY2 displayed 62.57% identity. The corresponding sequences have been submitted into the GenBank database with the accession numbers from KX229755 to KX229764.
We also amplified and sequenced genomic ILFY1 and ILFY2 sequences in the five Isoetes species. Comparison analysis revealed that both ILFY1 and ILFY2 in the five Isoetes species contained three exons and two introns (Fig. 1). The length of first intron for ILFY1 was 319–325 bp and second intron was 957–992 bp in length. The first intron for ILFY2 was 200 bp in length and second intron was 914–926 bp in length.
Protein comparison and phylogenetic tree
The predicted ILFY1 and ILFY2 amino acid sequences were aligned with a diverse of LFY homologs downloaded from the NCBI database. The comparison analysis revealed that the N-terminal regions were partially conserved and C-terminal regions were highly conserved in evolutionary process (Fig. 2). Moreover, the clearly conserved C-terminal domains were comprised of two β-sheet and seven α-helix structures. Identical levels of amino acid sequences were shown in Table S2 through comparing deduced proteins of IhLFY1, IhLFY2, IsLFY1,and IsLFY2 with LFY in A. thaliana, PRLFY1 and NEEDLY in Pinus radiata, CRLFY2 in Ceratopteris richardii, SmLFY1 in Selaginella moellendorffii, and PpILFY1 in P. patens, respectively. A conserved histidine residue (His) is substituted by Asp in the C regions for PpILFY1 and PpILFY2 in P. patens 1. Nonetheless, The ILFY1 and ILFY2 proteins in the C regions consistently contained the conserved His residue, which was similar with that in numerous land plants1.
Sequences comparison of deduced LFY proteins. Deduced amino acid sequences of IhLFY1 and IhLFY2 for I. hypsophila, as well as IsLFY1 and IsLFY2 for I. sinensis were compared with LFY homologs, including LFY (Arabidopsis thaliana, AAA32826), FLO (Antirrhinum majus, AAA62574.1), PRFLL (Pinus radiata, O04116), NEEDLY (Pinus radiata, AAB68601.1), CRLFY2 (Ceratopteris richardii, BAB41070.2), SmLFY1 (Selaginella moellendorffii, XP_002978027), PpLFY1 (Physcomitrella patens, BAB60676.1), and PpLFY2 (Physcomitrella patens, BAB60677.1). Conservatively substituted and identical residues were depicted on a red background, and slightly conserved residues were on an orange background. N-terminal and C-terminal domains were overlined in blue and red, respectively. Second structures of the C-terminal domains including two β-sheet and seven α-helix, were indicated. Gaps were shown as dashes to maximize the alignments. The blue triangle represented a conspicuously different amino acid between P. patens and the other species.
To determine the phylogenetic relationships of ILFY1 and ILFY2, we constructed a phylogenetic tree of LFY amino acid sequences using the maximum likelihood method (Fig. 3). The phylogenetic tree showed that topology of LFY homologs was concordant with the species topology33, 34. The ILFY1 and ILFY2 paralogs in the five Isoetes species were separated into two clades. Moreover, the two paralogs in I. hypsophila were separated with the other four species. In addition, we also used nucleotide sequences of the conserved N-terminal and C-terminal regions to reveal the phylogenetic relationships of ILFY1 and ILFY2. The phylogenetic tree was similar with the phylogenetic tree based on the amino acid sequences (Figure S1). The ILFY1 and ILFY2 paralogs in the five Isoetes species were also separated into two clades and the two paralogs in I. hypsophila were separated with the other four species.
Phylogenetic tree of LFY homologs. The phylogenetic tree was constructed using the amino acid sequences of LFY homologs. Numbers above the branches represent bootstrap value, and bootstrap less than 50% was removed. Accession numbers for the LFY homologs in the dataset were listed in Table S5.
qRT-PCR
The expression levels of IsLFY1 and IsLFY2 were investigated using qRT-PCR approach, respectively. The materials included roots, corms, and each whorl of leaves, megasporangia, and microsporangia. Averagely, there were about 12 whorls for each plant, including the former 11 whorls of sporophylls and last whorl of immature leaves. We collected all whorls of sporophylls or immature leaves and sporangia, including the former 7 whorls of megasporangia and latter 4 whorls of microsporangia. Expression analysis revealed that the IsLFY1 transcripts were slightly expressed in the former 4 whorls of megasporophylls and megasporangia, and expression levels were increasing from the 5th to 7th whorls (Fig. 4). Moreover, the expression levels in the 7th whorl of megasporangia were the highest in all collected samples. Generally, the expression levels were higher in the 8th to 11th whorls of microsporophylls and microsporangia than that from the 1th to 5th whorls, respectively. Furthermore, expression level was higher in the 11th whorl of microsporangia than the other whorls of sporangia. For the 12th whorl for immature leaves, the expression levels were relatively higher than the other whorls of sporophylls. In addition, the expression levels at roots were prominently strong. For the IsLFY2 transcripts, expression levels in all tissues were similar with that of the IsLFY1 transcripts (Fig. 4). But IsLFY2 expressions were significantly higher than that of IsLFY1 at the corms. Similarly, the expression level of IsLFY2 was the highest in the 7th whorl of megasporangia, followed by the roots and 11th whorl of microsporangia.
Expression patterns of IsLFY1 and IsLFY2 using quantitative real-time PCR assays. The abbreviations L, Me, Mi, R, Co represent leaves, megasporangia, microsporangia, roots, and corms, respectively. The megasporangia were in the bottom of fertile leaves from 1th to 7th whorls, and microsporangia were from 8th to 11th whorls. The immature leaves were in the 12th whorl. Values represent the means ± SE. Asterisk indicated that the expression levels of IsLFY1 and IsLFY2 were significantly different with the P value less than 0.01.
In situ hybridization
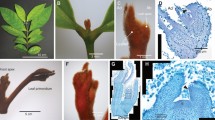
Expression patterns of the IsLFY1 and IsLFY2 transcripts were further characterized using in situ hybridization assays. A range of tissues were selected, containing roots, corms, and the 6th whorl of leaves and megasporangia, and 8th whorl of microsporangia. The IsLFY1 and IsLFY2 transcripts showed similar expression patterns in all examined tissues. The transcripts in roots were accumulated in all parts of roots except periderm (Figs 5a and 6a). Moreover, the two transcripts were significantly detected in endodermis, connective parts, and vascular bundles. Leaves of I. sinensis are quadrangular in outline and the two transcripts were detected all parts of the leaves, including parenchyma cells and vascular bundles (Figs 5b and 6b). Corm of I. sinensis is a short tuberous body35. In situ hybridization for longitudinal sections of the corms showed that the two transcripts were dramatically expressed at the zone of parenchyma cells which surrounded vascular bundles (Figs 5d and 6d). The expressions were slight in the cortical cells from transverse sections of the corm (Figs 5c and 6c). In addition, megasporangia and microsporangia are in the bottom of the sporophylls (Figure S2), and the spores are separated by trabeculae29. In general, a large number of microspores are located in microsporangia and limited megaspores are in megasporangia. IsLFY1 was strongly expressed in trabeculae or intine of the microsporangia, and partial microspores (Fig. 5e). IsLFY2 was also strongly expressed in trabeculae or intine of the microsporangia, whereas it is faintly detected in microspores (Fig. 6e). In addition, the two transcripts were expressed at high levels in trabeculae or intine of the megasporangia and expressed at a low level in megaspores (Figs 5f and 6f).
In situ hybridization of IsLFY1 expressions. (a) Transverse section of roots. (b) Transverse section of leaves. (c) Transverse section of corms. (d) Longitudinal section of corms. (e) Transverse section of microsporangia. (f) Transverse section of megasporangia. Scale bars were 300 μm for megasporangia and microsporangia, 200 μm for root, leaves, and 100 μm for corms.
In situ hybridization of IsLFY2 expressions. (a) Transverse section of roots. (b) Transverse section of leaves. (c) Transverse section of corms. (d) Longitudinal section of corms. (e) Transverse section of microsporangia. (f) Transverse section of megasporangia. Scale bars were 300 μm for megasporangia and microsporangia, 200 μm for root, leaves, and 100 μm for corms.
Arabidopsis transgenes
To investigate whether the roles of IsLFY1 and IsLFY2 were conserved in evolutionary process, we created transgenic Arabidopsis plants, which constitutively expressed IsLFY1 and IsLFY2 under the control of the cauliflower mosaic virus 35S promoter, respectively. The T1 seeds were selected using kanamycin resistance and resistance plants were grown under long-day conditions. In total, we isolated 30 IsLFY1 transgenic Arabidopsis lines, and only two accelerated flowering. Totally, 25 IsLFY2 transgenic lines were identified and only one showed precocious flowering. As shown in Table S3, the remaining transgenic plants did not differ significantly in appearance from the wild-type controls and they did not cause precocious flowering (Fig. 7a–d).
Phenotypic characters in Arabidopsis of constitutive IsLFY1 and IsLFY2 expressions. Figures from a to d are related to transgenes that the genes were crossed into wild Arabidopsis plants. Four-week IsLFY1 transgenic line (a), wild Arabidopsis plants (b), and IsLFY2 transgenic line (c). The IsLFY1 and IsLFY2 transgenic plants did not differ significantly in appearance from the wild-type plant. Flowers for the transgenic plants were consistent with the wild plants (d). Figures from e to j are related to transgenes that the genes were crossed into lfy-1 mutants. lfy-1 mutant (e). IsLFY1 transgenic line (f). IsLFY2 transgenic line (g). The IsLFY1 and IsLFY2 were inactive to rescue the lfy mutants. The transgenic plants and lfy mutant consistently showed that the early flowers were replaced by bracts, which subtended secondary inflorescences (h). The later flowers consisted of sepals and carpels (i and j).
In addition, we also created the transgenic plants that the full-length IsLFY1 and IsLFY2 cDNAs were transformed into severe lfy-1 Arabidopsis mutants under the control of the cauliflower mosaic virus 35S promoter, respectively. In total, we produced 6 IsLFY1 transgenic and 5 IsLFY2 transgenic lines (T1). The phenotypic characters were investigated from the F2 progeny. For lfy-1 mutants, early-arising flowers are replaced by bracts and completely transformed into inflorescence shoots (Fig. 7e). The later-arising flowers are abnormal without petals and stamens5. Whereas wild-type flowers include four sepals, four petals, two carpels, and six stamens (Fig. 7d). Among the 200 T1 lines examined, 30 were homozygous for lfy-1 mutants. However, only one IsLFY1 transgenic lines partially complement the lfy phenotype. The remaining plants showed inactive to rescue the lfy mutants (Fig. 7f). In addition, among the identified 28 transgenic lfy-1 mutants, only one IsLFY2 transgenic lines partially complement the lfy phenotype and the remaining plants similarly were inactive to rescue the lfy mutants (Fig. 7g). The early flowers were replaced by bracts, which subtended secondary inflorescences (Fig. 7h). The later flowers consisted of sepals and carpels (Fig. 7i,j). Phenotypes of the transgenic plants were indistinguishable from the lfy mutants (Table S4).
Discussion
Analysis of the deduced ILFY1 and ILFY2 protein in five Isoetes species showed that ILFY1 and ILFY2 amino acid sequences were common in I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. There was 94.86% identity for ILFY1 and 98.68% identity for ILFY2 in the four species compared with IhLFY1 and IhLFY2, respectively. Moreover, the ORF fragments of ILFY1 and ILFY2 shared 63.41% identity for the four species and 62.57% identity for I. hypsophila. The five Isoetes species in China display a space order of the distribution pattern, I. hypsophila-I. yunguiensis-I. sinensis-I. orientalis-I. taiwanensis, from high altitude to low altitude and from west to east distribution36, 37. Therefore, geographic isolation probably results in a slight diversity of the ILFY homologs in phylogenetic evolution. LFY homologs possess a markedly conserved C-terminal domain and partially conserved N-terminal domain1, 38. Comparison analysis revealed that ILFY1 and IFLY2 in the C-terminal regions were highly conserved in the evolutionary process. The clearly conserved C-terminal regions include DNA binding domain to regulate downstream genes, whereas the partially conserved N-terminal regions play key roles in forming LFY dimerization and higher complex with other TFs or chromatin remodelers39,40,41. Moreover, An amino acid substitution from His to Asp in the C-terminal domain causes PpLFY proteins inactive by binding a canonical LFY-binding domain38. Nonetheless, ILFY1 and IFLY2 consistently shared the conserved His, which were resembled that in vascular plants.
Information about the expression patterns of IsLFY1 and IsLFY2 suggested that functions for the two genes might be conserved, probably being redundant in both paralogs, despite differences outside the DNA binding domains17, 42. In general, there is a single copy in most angiosperms and more than two copies in nonflowering plants. Nonetheless, major subfunctionalization is not occurred for the additional copies20, 43. I. sinensis includes sporophylls and immature leaves, and the leaves are closely spirally arranged on the fleshy corms29. The megasporophylls are located outside of the corm, and micriosporophylls are growing inside of the corm. Moreover, the leaves are becoming mature from inside to outside, and the immature leaves are in the center of the corm28. Generally speaking, the IsLFY1 and IsLFY2 transcripts were consistently higher in microsporangia than megasporangia, and expression levels were higher in the inside microsporophylls than outside megasporophylls. Expression levels of the two genes were similarly highest in the 7th whorl of megasporophylls, followed by roots, the 11th whorls of microsporangia, and the transcripts were highly expressed in young tissues. Thus, the results demonstrated that the two ILFY paralogs played general roles in both reproductive and vegetative developments. In situ hybridization analysis further revealed that IsLFY1 and IsLFY2 were ubiquitously expressed in all active parts of leaves and roots, trabeculae and intine of the megasporangia or microsporangia, and the zone of parenchyma cells which surrounded vascular bundles of the corms. The cambium appears in parenchyma cells which surrounded vascular bundles of the corms and it is not differentiated. Moreover, the transcripts were significantly detected in endodermis of the roots, which are poorly differentiated29. The results probably indicated that IsLFY1 and IsLFY2 probably have an ancestral role in controlling cell division or arrangement and developmental process16, 21.
Constitutive overexpression of LFY in Arabidopsis promotes early flowering and reduces number of adult leaves4, 8. The transgenic plants transformed lateral shoots into terminal flowers. LFY is strongly expressed in floral primordia, and faintly expressed in cauline leaf primordia4. Moreover, constitutive overexpression of LFY homologs results in precocious flowering in most angiosperms and gymnosperms15, 17, 18. However, most of the IsLFY1 and IsLFY2 transgenic lines did not show precocious flowering relative to the wild-type plants, suggesting that the two paralogs were not functional orthologs with LFY 4, 17, 18. The early-arising flowers in severe lfy mutants are converted into leaves and lateral shoots, and the late-arising flowers are only composed of sepals and carpels4, 5, 44. Observation revealed that the 35S::IsLFY1 and 35S::IsLFY2 transgenes were similarly inactive to complement lfy mutants, which were consistent with PpLFY1 and PpLFY2 in P. patens. Previous research proposed that complementation ability is gradually increasing from mosses to angiosperms, and a continuum of nonneutral change is responsible for the functional changes1. Moreover, the gradual activity among nonflowering plants implies alteration of the DNA binding specificity of LFY homologs. It is necessary to further investigate the interaction targets of ILFY to elucidate the evolutionary process.
In summary, we cloned two ILFY paralogs in five Isoetes species, including I. hypsophila, I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. Expression patterns of IsLFY1 and IsLFY2 in I. sinensis demonstrated that the two genes did not have functional divergences. The two transcripts are expressed not only in reproductive tissues but also in vegetative tissues. Moreover, the transcripts were expressed at a high level in juvenile tissues, indicating ILFY genes control both vegetative and reproductive developments in Isoetes. In addition, overexpression of IsLFY1 and IsLFY2 in Arabidopsis similarly did not show precocious flowering compared with wild-type plants. Furthermore, the two paralogs were inactive to complement severe lfy mutant phenotypes, implying that the two homologs were not functional orthologs with LFY. Overall, the study provides important information for understanding the characters and functions of LFY homologs in Isoetes and evolutionary process.
Methods
Plant materials
Totally, five Isoetes species were collected in China and cultivated in a greenhouse of Wuhan University, including I. hypsophila, I. yunguiensis, I. sinensis, I. orientalis, and I. taiwanensis. The material I. hypsophila was collected from Daocheng in Sichuan Province, China (N29°29′; E100°14′); I. yunguiensis was collected from Hongfeng Lake in Sichuan Province, China (N26°29′; E106°58′); I. sinensis was collected from Xinan River in Zhejiang Province, China (29°28′N; 119°14′E); I. orientalis was collected from Songyang in Zhejiang Province, China (N28°47′; E119°12′); and I. taiwanensis was collected from Jinmen in Taiwan Province, China (N24°27 ′; E118° 23′).
RNA isolation and cloning of LFY homologs
Initially, approximately 0.15 g juvenile leaves from the five Isoetes species were immediately sampled and frozen by liquid nitrogen before RNA extraction, respectively. Then total RNA was isolated using RNAiso Plus (Takara, Da Lian, China), according to the manufacturer’s protocols. Total RNA (6 μg) was incubated at 37 °C for 15 min to eliminate genomic DNA using 1 μL RNase-free DNase I (Promega, Madison, WI, USA). The treated total RNA was reverse transcribed into single-strand cDNA using M-MLV Reverse Transcriptase (Promega, Madison, WI, USA) and Oligo(dT)18-adaptor primer. Partial ILFY segments were amplified using specific primers (5′TTCAAATGGGAGCCCAGAATACC3′ and 5′GACATTGATGCGTGTGCTGGAT3′), which were designed according to Saiko Himi et al. 20. The obtained cDNA initially was diluted 10-fold, and used as templates for polymerase chain reactions (PCR) amplification. The reaction mixture (20 μL) contained 2.0 μL 10 × buffer with 2 mM MgCl2, 0.25 pmol forward and reverse primers, 0.1 mM dNTP, 2.0 μL diluted cDNA, 12.6 μL sterile water and 0.5 μL Primer Star HS DNA polymerase (Takara, Dalian, China). The PCR conditions included initial denaturation step of 5 min at 95 °C, followed by 40 cycles of 95 °C for 30 s, 55 °C for 40 s, and finally 72 °C for 1 min. Then the PCR products were purified (QIAquick PCR cleanup kit), cloned into the pEASY-Blunt vector (TransGen Biotech Company, Beijing, China), and sequenced in both directions on an ABI 3730 DNA Sequencer using BigDye Terminator version 3.1 (Applied Biosystems).
To obtain full-length ORF of ILFY sequences in the five Isoetes species, we performed 5′ and 3′ RACE PCR using the 5′ RACE System for Rapid Amplification of cDNA Ends and 3′ RACE System for Rapid Amplification of cDNA Ends (Invitrogen™) Kits, respectively. The gene-specific primers for the 5′ RACE PCR were included (5′GATCTTTGCCTTTCCGTCTCATCCTCC3′ and (5′CCAAGCTCTGCAACCTTTGCTATTGTGC3′). The gene-specific primers for the 3′ RACE PCR were used (5′GGTCCCAGGGGATATGAATGGCAGAG3′ and 5′TGGAGGATGAGACGGAAAGGCAAAGA3′). Then the products for the 5′ and 3′ RACE PCR were purified and cloned into pEASY-Blunt Cloning vector, respectively. Eight positive clones were sequenced in both directions for each sample.
DNA isolation, amplification and sequencing
Total genomic DNA of the five Isoetes species was isolated from juvenile leaves using the CTAB method45. The two ILFY paralogs were amplified using specific primers (ILFY1: 5′ATGACTGAGCTGGGTTTCACCG3′ and 5′TTAGCTGCTTTCTTTGCTTTTTTCT3′; and ILFY2: 5′TCAAATGGGAGCCCAGA3′ and 5′TTAGCTGCACTCTTTACCCTT3′) respectively. The PCR program was an initial denaturation step of 5 min at 95 °C, followed by 30 cycles of 95 °C for 30 s, 57 °C for 50 s, and finally 72 °C for 2 min. The products were further purified and ligated with pEASY-Blunt Cloning vector. Three positive clones for each sample were sequenced in both directions for each sample.
Construction of a phylogenetic tree
LFY homologs were searched using the program BLAST X model in the NCBI database (http://www.ncbi.nlm.nih.gov/). The amino acid and nucleotide sequences were aligned using Clustal X 2.0 software46, including 36 LFY homologous sequences and 10 ILFY homologous sequences obtained in this study. The phylogenetic analysis was conducted based on the JTT substitution model and maximum likelihood approach implemented in the Molecular Evolutionary Genetics Analysis version 7.0 (MEGA 7.0) program47. Support for internal nodes was estimated based on 1000 bootstrap replicates, and bootstrap less than 50% was removed above the relevant branch48.
qRT-PCR
Given that I. sinensis is relatively widespread in China and easily adapts to the greenhouse environment in Wuhan University, I. sinensis was employed to investigate the expression patterns, including IsLFY1 and IsLFY2. The leaves and sporangia were arranged on corms in whorls, and they are becoming mature from inside to outside29. Each whorl of green leaves and sporangia was collected, respectively. All sampled materials were immediately frozen by liquid nitrogen for RNA extraction. In addition, we also sampled the materials of corms and roots for RNA isolation. Total RNA of all samples was isolated using RNAisoTM Plus (Takara, Da Lian, China). Then the isolated RNA was treated with RNase-free DNase I (Takara, Da Lian, China) for 45 min according to the manufacturer's protocols. Subsequently, 1 μg treated RNA was reverse-transcribed into single-strand cDNA using PrimerscriptTM One Step RT-PCR Kit Ver.2 (Takara, Da Lian, China). The qRT-PCR experiments for IsLFY1 and IsLFY2 were carried out on a CFX96 Real-time PCR system (Bio-Rad, Hercules, USA) with IsLFY1-specific primers (5′GATGCTCTTGATGTGCAAGC3′ and 5′TCACCACACAGATTGACTC3′) and IsLFY2-specific primers (5′GAAAGACCACCAAGGAAA3′ and 5′AAGCAGACAGTCGGAAAG3′), respectively. The first-strand cDNA initially was diluted 10-fold and used as templates in the qRT-PCR tests. The reaction mixture (25 μL) included 0.25 pmol forward and reverse primers, 12.5 μL 2 × SYBR premix (Takara, Da Lian, China), 2.5 μL diluted cDNA, and 7.0 μL sterile water. The thermocycling condition was an initial denaturation step of 5 min at 95 °C, followed by 40 cycles of 95 °C for 30 s, 60 °C for 15 s, and finally 72 °C for 30 s. A melting curve was plotted to check the specific products of amplification reactions at the end of the PCR cycling over the range 65–95 °C. Baseline and threshold cycle (Ct) were determined automatically by the Bio-Rad CFX Manager 2.1 software. The relative expression levels were calculated using 2−△△Ct method, and normalized to the geometric average of Ct values with Actin as an internal control24, 49. All experiments and analyses were performed in triplicate.
In situ hybridization
I. sinensis was further employed to investigate spatial-specific expression patterns of IsLFY1 and IsLFY2 using in situ hybridization assays. Samples were fixed using 4% paraformaldehyde buffers, including roots, corms, and the 6th whorl of sporophylls and megasporangia, and 8th whorl of microsporangia. In order to ensure the specific hybridization patterns of IsLFY1 and IsLFY2, the non-conserved regions of IsLFY1 and IsLFY2 were used to design the specific probes. Partial sequences of IsLFY1 were amplified using IsLFY1-specific primers (5′AATCTATGCTGGCTACTGG3′ and 5′TTGAGTCGCACGTCGTAT3′); and partial sequences of IsLFY2 were amplified using IsLFY2-specific primers (5′GGACTATGGTGTTAGGCTCT3′ and 5′CAGATGTCCCTGTATTTGC3′). The two amplification segments were introduced into the pGEM-T Vector (Promega, Madison, WI, USA), respectively. Antisense probes for the two paralogs were synthesized using SacII (Takara, Da Lian, China) and SP6 RNA polymerases (Promega, Madison, WI, USA). Sense strand controls for the IsLFY1 and IsLFY2 probes were synthesized using SacI (Takara, Da Lian, China) and T7 RNA polymerases (Promega, Madison, WI, USA). Both the sense and antisense probes of IsLFY1 and IsLFY2 were generated into single-stranded digoxigenin-11-UTP-labeled RNA probes, respectively. Tissues materials were fixed, embedded with paraplast, sectioned into 8 μM in thickness, and hybridized using the probes, described by Bechtold et al.50. All slides were photographed using an OLYMPUS X73 microscope.
Plasmids construction and Arabidopsis transgenes
The coding regions of IsLFY1 and IsLFY2 were ligated with the binary vector pBI121 with a cauliflower mosaic virus 35S promoter, respectively. Initially, BamHI and SmaI restriction fragments were added to the upstream of start codon sequences and downstream of stop codon sequences for the two genes, respectively. The introduced restriction fragments of IsLFY1 and IsLFY2 were generated by PCR amplification using a set of primers (IsLFY1: 5′CGGGATCCATGACTGAGCTGGGTTTCACCG3′ and 5′TCCCCCGGGTTAGCTGCTTTCTTTGCTTTTTTCT3′; and IsLFY2: 5′CGGGATCCATGACAGATTCAGAGATAGAAGAAC3′ and 5′TCCCCCGGGTTAGCTGCACTCTTTACCCTT3′). The amplified fragments were digested with BamHI and SmaI, and further cloned into the BamHI-SmaI sites of pBI121 binary vector. Then the constructed 35S::IsLFY1 and 35S::IsLFY2 were sequenced to identify the correct orientation.
The identified 35S::IsLFY1 and 35S::IsLFY2 were introduced into Agrobacterium tumefaciens strain GV3101, and further crossed into the Arabidopsis plants (Columbia ecotype) using the floral dip method51. In addition, the 35S::IsLFY1 and 35S::IsLFY2 were also crossed into lfy heterozygous Arabidopsis plants, respectively. The heterozygous lines were isolated from a bulk of lfy-1 mutant seeds, which were obtained from TAIR (http://www.arabidopsis.org/). All transgenic plants (T1) were selected on MS medium containing 50 mg/mL kanamycin. The 35 S::IsLFY1 and 35 S::IsLFY2 lines were identified using PCR amplifications through plating 200 F1 seeds, and homozygous lines (T2) were selected for phenotypic analysis. The seeds were kept for 3 days at 4 °C before sowing. All resistant and wild plants were grown at 23 °C under 16 h light/8 h dark.
References
Maizel, A. et al. The floral regulator LEAFY evolves by substitutions in the DNA binding domain. Science 308, 260–263 (2005). págs.
Sayou, C. et al. A promiscuous intermediate underlies the evolution of LEAFY DNA binding specificity. Science 343, 645–8 (2014).
Brunkard, J. O., Runkel, A. M. & Zambryski, P. C. Comment on “A promiscuous intermediate underlies the evolution of LEAFY DNA binding specificity”. Science 347, 621–621 (2015).
Weigel, D., Alvarez, J., Smyth, D. R., Yanofsky, M. F. & Meyerowitz, E. M. LEAFY controls floral meristem identity in Arabidopsis. Cell 69, 843–859 (1992).
Schultz, E. A. & Haughn, G. W. LEAFY, a Homeotic Gene That Regulates Inflorescence Development in Arabidopsis. Plant Cell 3, 771–781 (1991).
Coen, E. S. & Meyerowitz, E. M. The war of the whorls: genetic interactions controlling flower development. Nature 353, 31–37 (1991).
Weigel, D. & Nilsson, O. A developmental switch sufficient for flower initiation in diverse plants. Nature 377, 495–500 (1995).
Blázquez, M. A., Soowal, L. N., Lee, I. & Weigel, D. LEAFY expression and flower initiation in Arabidopsis. Development 124, 3835–3844 (1997).
Ahearn, K. P., Johnson, H. A., Weigel, D. & Wagner, D. R. NFL1, a Nicotiana tabacum LEAFY-like gene, controls meristem initiation and floral structure. Plant & Cell Physiology 42, 1130–1139 (2001).
Coen, E. S. et al. Floricaula: A homeotic gene required for flower development in antirrhinum majus. Cell 63, 1311–22 (1990).
Souer, E. et al. Genetic control of branching pattern and floral identity during Petunia inflorescence development. Development 125, 733–742 (1998).
Kyozuka, J., Konishi, S., Nemoto, K., Izawa, T. & Shimamoto, K. Down-regulation of RFL, the FLO/LFY homolog of rice, accompanied with panicle branch initiation. Proceedings of the National Academy of Sciences 95, 1979–1982 (1998).
Ohmori, S. et al. MOSAIC FLORAL ORGANS1, an AGL6-like MADS box gene, regulates floral organ identity and meristem fate in rice. The Plant Cell 21, 3008–3025 (2009).
Hofer, J. et al. UNIFOLIATA regulates leaf and flower morphogenesis in pea. Current Biology Cb 7, 581–587 (1997).
Molinero-Rosales, N. et al. FALSIFLORA, the tomato orthologue of FLORICAULA and LEAFY, controls flowering time and floral meristem identity. Plant Journal 20, 685–693 (1999).
Moyroud, E., Kusters, E., Monniaux, M., Koes, R. & Parcy, F. LEAFY blossoms. Trends in Plant Science 15, 346–352 (2010).
Mouradov, A. et al. NEEDLY, a Pinus radiata ortholog of FLORICAULA/LEAFY genes, expressed in both reproductive and vegetative meristems. Proceedings of the National Academy of Sciences 95, 6537–6542 (1998).
Kelly, A. J., Bonnlander, M. B. & Meekswagner, D. R. NFL, the tobacco homolog of FLORICAULA and LEAFY, is transcriptionally expressed in both vegetative and floral meristems. Plant Cell 7, 225–34 (1995).
Silva, C. S. et al . Evolution of the plant reproduction master regulators LFY and the MADS Transcription Factors: the role of protein structure in the evolutionary development of the flower. Frontiers in Plant Science 6 (2016).
Himi, S. et al. Evolution of MADS-Box Gene Induction by FLO/LFY Genes. Journal of Molecular Evolution 53, 387–393 (2001).
Tanahashi, T., Sumikawa, N., Kato, M. & Hasebe, M. Diversification of gene function: homologs of the floral regulator FLO/LFY control the first zygotic cell division in the moss Physcomitrella patens. Development 132, 1727–36 (2005).
Moyroud, E., Tichtinsky, G. & Parcy, F. The LEAFY floral regulators in Angiosperms: conserved proteins with diverse roles. Journal of Plant Biology 52, 177–185 (2009).
Pigg, K. B. Evolution of isoetalean lycopsids. Annals of the Missouri Botanical Garden 79, 589–612 (1992).
Yang, T. & Liu, X. Comparing photosynthetic characteristics of Isoetes sinensis Palmer under submerged and terrestrial conditions. Scientific Reports 5 (2015).
Pigg, K. B. Isoetalean Lycopsid Evolution: from the Devonian to the Present. American Fern Journal 91, 99–114 (2001).
Pryer, K. M. et al. Horsetails and ferns are a monophyletic group and the closest living relatives to seed plants. Nature 409, 618–622 (2001).
Li, Z., Han, Q., Chen, Y. & Li, W. Microsatellite primers in the endangered quillwort Isoetes hypsophila (Isoetaceae) and cross-amplification in I. sinensis. American Journal of Botany 99, e184–6 (2012).
Rury, P. M. A new and unique, mat-forming Merlin’s-grass (Isoetes) from Georgia. American Fern Journal 68, 99–108 (1978).
Stokey, A.G. The anatomy of Isoetes. Botanical Gazette, 311–335 (1909).
Taylor, W. C. et al. Phylogenetic relationships of Isoëtes (Isoëtaceae) in China as revealed by nucleotide sequences of the nuclear ribosomal ITS region and the second intron of a LEAFY homolog. American Fern Journal 94, 196–205 (2004).
Hoot, S. B. & Taylor, W. C. The utility of nuclear ITS, a LEAFY homolog intron, and chloroplast atpB-rbcL spacer region data in phylogenetic analyses and species delimitation in Isoetes. American Fern Journal 91, 166–177 (2001).
Hoot, S. B., Napier, N. S. & Taylor, W. C. Revealing unknown or extinct lineages within Isoetes (Isoetaceae) using DNA sequencesfrom hybrids. American Journal of Botany 91, 899–904 (2004).
YL, Q. et al. The deepest divergences in land plants inferred from phylogenomic evidence. Proceedings of the National Academy of Sciences of the United States of America 103, 15511–6 (2006).
Rensing, S. A. et al. The Physcomitrella Genome Reveals Evolutionary Insights into the Conquest of Land by Plants. Science 319, 64–69 (2008).
Karrfalt, E. E. & Eggert, D. A. The Comparative Morphology and Development of Isoetes L. II. Branching of the Base of the Corm in I. tuckermanii A. Br. and I. nuttallii A. Br. Botanical Gazette. 357–368 (1977).
Liu, X., Wang, J.-Y. & Wang, Q.-F. Current status and conservation strategies for Isoetes in China: a case study for the conservation of threatened aquatic plants. Oryx 39, 335–338 (2005).
Liu, X., Gituru, W. R. & Wang, Q. F. Distribution of basic diploid and polyploid species of Isoetes in East Asia. Journal of biogeography 31, 1239–1250 (2004).
Hamès, C. et al. Structural basis for LEAFY floral switch function and similarity with helix-turn-helix proteins. The EMBO journal 27, 2628–2637 (2008).
Gocal, G. F. et al. Evolution of floral meristem identity genes. Analysis of Lolium temulentum genes related to APETALA1 and LEAFY of Arabidopsis. Plant Physiology 125, 1788–1801 (2001).
Siriwardana, N. S. & Lamb, R. S. A conserved domain in the N-terminus is important for LEAFY dimerization and function in Arabidopsis thaliana. Plant Journal 71, 736–49 (2012).
Wu, M. F. et al. SWI2/SNF2 chromatin remodeling ATPases overcome polycomb repression and control floral organ identity with the LEAFY and SEPALLATA3 transcription factors. Proceedings of the National Academy of Sciences of the United States of America 109, 3576–81 (2012).
Vázquez‐Lobo, A. et al. Characterization of the expression patterns of LEAFY/FLORICAULA and NEEDLY orthologs in female and male cones of the conifer genera Picea, Podocarpus, and Taxus: implications for current evo-devo hypotheses for gymnosperms. Evolution & development 9, 446–459 (2007).
Frohlich, M. W. & Parker, D. S. The mostly male theory of flower evolutionary origins: from genes to fossils. Systematic Botany 25, 155–170 (2000).
Huala, E. & Sussex, I. M. LEAFY interacts with floral homeotic genes to regulate Arabidopsis floral development. The Plant Cell 4, 901–913 (1992).
Dellaporta, S. L., Wood, J. & Hicks, J. B. A plant DNA minipreparation: version II. Plant molecular biology reporter 1, 19–21 (1983).
Larkin, M. A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948 (2007).
Kumar, S., Stecher, G. & Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Molecular biology and evolution (2016).
Felsenstein, J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 783–791 (1985).
Yang, T. & Liu, X. Comparative Transcriptome Analysis of Isoetes Sinensis Under Terrestrial and Submerged Conditions. Plant Molecular Biology Reporter, 1–10 (2015).
Bechtold, N., Ellis, J. & Pelletier, G. In planta Agrobacterium mediated gene transfer by infiltration of adult Arabidopsis thaliana plants. Comptes rendus de l’Académie des sciences. Série 3, Sciences de la vie 316, 1194–1199 (1993).
Zhang, X., Henriques, R., Lin, S.-S., Niu, Q.-W. & Chua, N.-H. Agrobacterium-mediated transformation of Arabidopsis thaliana using the floral dip method. Nature protocols 1, 641–646 (2006).
Acknowledgements
The authors thank Lei Zhang for experimental assistance related to the material Arabidopsis, Zan Wu for assistance for constructing vectors. The research was supported by “National Natural Science Foundation of China” (30870168 and 31170203).
Author information
Authors and Affiliations
Contributions
T.Y. cultured the plant tissue and isolated RNA, performed the experiments, analyzed the data, and drafted the manuscript. M.D. performed partial experiments, X.L. and Y.G. revised the manuscript. All authors reviewed the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yang, T., Du, Mf., Guo, Yh. et al. Two LEAFY homologs ILFY1 and ILFY2 control reproductive and vegetative developments in Isoetes L.. Sci Rep 7, 225 (2017). https://doi.org/10.1038/s41598-017-00297-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-00297-3
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.