Abstract
The demand for emergency department (ED) services is increasing across the globe, particularly during the current COVID-19 pandemic. Clinical triage and risk assessment have become increasingly challenging due to the shortage of medical resources and the strain on hospital infrastructure caused by the pandemic. As a result of the widespread use of electronic health records (EHRs), we now have access to a vast amount of clinical data, which allows us to develop prediction models and decision support systems to address these challenges. To date, there is no widely accepted clinical prediction benchmark related to the ED based on large-scale public EHRs. An open-source benchmark data platform would streamline research workflows by eliminating cumbersome data preprocessing, and facilitate comparisons among different studies and methodologies. Based on the Medical Information Mart for Intensive Care IV Emergency Department (MIMIC-IV-ED) database, we created a benchmark dataset and proposed three clinical prediction benchmarks. This study provides future researchers with insights, suggestions, and protocols for managing data and developing predictive tools for emergency care.
Similar content being viewed by others
Introduction
Emergency Departments (ED) experience large volumes of patient flows and growing resource demands, particularly during the current COVID-19 pandemic1. This growth has caused ED crowding2 and delays in care delivery3, resulting in increased morbidity and mortality4. Prediction models5,6,7,8,9 provide opportunities for identifying high-risk patients and prioritizing limited medical resources. ED prediction models center on risk stratification, which is a complex clinical judgment based on factors such as patient’s likely acute course, availability of medical resources, and local practices10.
The widespread use of Electronic Health Records (EHR) has led to the accumulation of large amounts of data, which can be used to develop predictive models to improve emergency care11,12,13,14. Based on a few large-scale EHR databases, such as Medical Information Mart for Intensive Care III (MIMIC-III)15, eICU Collaborative Research Database16, and Amsterdam University Medical Centers Database (AmsterdamUMCdb)17, several prediction benchmarks have been established18,19,20. These public benchmarks standardized the process of transforming raw EHR data into readily usable data to construct prediction models. They have provided clinicians and methodologists with easily accessible and high-quality medical data, accelerating research and validation efforts21,22. These non-proprietary databases and open-source pipelines make it possible to reproduce and improve clinical studies in ways that would otherwise not be possible18. While there are some publicly available benchmarks, most pertain to intensive care settings, and there are no widely accepted clinical prediction benchmarks related to the ED. An ED-based public benchmark dataset would lower the entry barrier for new researchers, allowing them to focus on developing novel research ideas.
Machine learning has seen tremendous advances in recent years and has gained increasing popularity in the realm of ED-based prediction models23,24,25,26,27,28,29,30. These prediction models involve machine learning, deep learning, interpretable machine learning, and others. However, we have found that researchers often develop an ad-hoc model for one clinical prediction task at a time, using only one dataset23,24,25,26,27,28. There is a lack of comparative studies among different methods and models to predict the same ED outcome, undermining the generalizability of any single model. Generally, existing prediction models were developed on retrospective data without prospective validation in real-world clinical settings. Hence, there remains a need for prospective, comparative studies on accuracy, interpretability, and utility of risk models for ED. Using an extensive public EHR database, we aimed to standardize data preprocessing and establish a comprehensive ED benchmark dataset alongside comparable risk prediction models for three ED-based outcomes. It is expected to facilitate reproducibility and model comparison and accelerate progress toward utilizing machine learning in future ED-based studies.
In this paper, we proposed a public benchmark suite for the ED using a large EHR dataset and introduced three ED-based outcomes: hospitalization, critical outcomes, and 72-hour ED reattendance. We implemented and compared several popular methods for these clinical prediction tasks. We used data from the publicly available MIMIC IV Emergency Department (MIMIC-IV-ED) database31,32, which contains over 400,000 ED visit episodes from 2011 to 2019. Our code is open-source (https://github.com/nliulab/mimic4ed-benchmark) so that anyone with access to MIMIC-IV-ED can follow our data processing steps, create benchmarks, and reproduce our experiments. This study provides future researchers with insights, suggestions, and protocols to process the raw data and develop models for emergency care in an efficient and timely manner.
Methods
This section consists of three parts. First, we describe raw data processing, benchmark data generation, and cohort formation. Second, we introduce baseline models for three prediction tasks. Finally, we elaborate on the experimental setup and model performance evaluation.
Master data generation
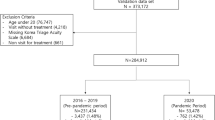
We use standardized terminologies as follows. Patients are referred to by their subject_id. Each patient has one or more ED visits, identified by stay_id in edstays.csv. If there is an inpatient stay following an ED visit, this stay_id could be linked with an inpatient admission, identified by hadm_id in edstays.csv. subject_id and hadm_id can also be traced back to the MIMIC-IV31 database to follow the patient throughout inpatient or ICU stay and patients’ future or past medical utilization, if needed. In the context of our tasks, we used edstays.csv as the root table and stay_id as the primary identifier. As a general rule, we have one stay_id for each prediction in our benchmark tasks. All raw tables were linked through extract_master_dataset.ipynb, illustrated in Fig. 1. The linkage was based on the root table, and merged through different identifiers, including stay_id (ED), subject_id, hadm_id, or stay_id (ICU). We extracted all high-level information and consolidated them into a master dataset (master_dataset.csv).
To construct the master dataset, we reviewed a number of prominent ED studies5,7,33,34,35 to identify relevant variables and outcomes. Moreover, we consulted clinicians and informaticians familiar with the raw data and ED operation to identify and confirm all ED-relevant variables. We excluded variables that were irrelevant, repeated, or largely absent. A list of high-level constructed variables is presented in Supplementary eTable 1, including patient history, variables collected at triage and during ED stay, and primary ED-relevant outcomes. The final master dataset includes 448,972 ED visits by 216,877 unique patients.
Data processing and benchmark dataset generation
The data processing workflow (data_general_processing.ipynb), illustrated in Fig. 2, begins with the master dataset generated previously to derive the benchmark dataset. In the first step, we filtered out all ED visits with patients under 18 years old and those without primary emergency triage class assignments. A total of 441,437 episodes remained after the filtering process.
The raw EHR data cannot be directly used for model building due to missing values, outliers, duplicates, or incorrect records caused by system errors or clerical mistakes. We addressed these issues with several procedures. For vital signs and lab tests, a value would be considered an outlier and marked as missing if it was outside the plausible physiological range as determined by domain knowledge, such as a value below zero or a SpO2 level greater than 100%. We followed the outlier detection procedure used in MIMIC-EXTRACT20, a well-known data processing pipeline for MIMIC-III. We utilized the thresholds available in the source code repository of Harutyunyan et al., where one set of upper and lower thresholds was used for filtering outliers. Any value that falls outside this range was marked as missing. Another set of thresholds was introduced to indicate the physiologically valid range, and any value that falls beyond this range was replaced by its nearest valid value. These thresholds were suggested by clinical experts based on domain knowledge.
For benchmarking purposes, we fixed a test set of 20% (n = 88,287) of ED episodes, covering 65,169 unique patients. Future researchers are encouraged to use the same test set for model comparisons and to interact with the test set as infrequently as possible. The training set consisted of the remaining 80% of ED episodes. The validation set can be derived from the training set if needed. Missing values (including outliers marked as missing and those initially absent) were imputed. In this project, we used the median values from the training set and other options are provided through our code repository. The same values were used for imputation on the test set.
ICD codes processing
In MMIC-IV, each hospital admission is associated with a group of ICD diagnosis codes (in diagnoses_icd.csv), indicating the patients’ comorbidities. We embedded the ICD codes within a time range (e.g., five years) from each ED visit into Charlson Comorbidity Index (CCI)36 and Elixhauser Comorbidity Index (ECI)37 according to the mapping proposed by Quan H et al.38. We adopted the codebase from Cates et al. and developed the neural network-based embedding with similar network structures to Med2Vec39.
Benchmark tasks
Following are three ED-relevant clinical outcomes. They are all of utmost importance to clinicians and hospitals due to their immense implications on costs, resource prioritization, and patients’ quality of life. Accurate prediction of these outcomes with the aid of big data and artificial intelligence has the potential to transform health services.
-
The hospitalization outcome is met with an inpatient care site admission immediately following an ED visit40,41,42. Patients who transitioned to ED observation were not considered hospitalized unless they were eventually admitted to the hospital. As hospital beds are limited, this outcome indicates resource utilization and may facilitate resource allocation efforts. The hospitalization outcome also suggests patient acuity, albeit in a limited way, since hospitalized patients represent a broad spectrum of disease severity.
-
The critical outcome34 is compositely defined as either inpatient mortality43 or transfer to an ICU within 12 hours. This outcome represents the critically ill patients who require ED resources urgently and may suffer from poorer health outcomes if care is delayed. Predicting the critical outcome at ED triage may enable physicians to allocate ED resources efficiently and intervene on high-risk patients promptly.
-
The ED reattendance outcome refers to a patient’s return visit to ED within 72 hours after their previous discharge from the ED. It is a widely used indicator of the quality of care and patient safety and is believed to represent patients who may not have been adequately triaged during their first emergency visit44.
Baseline methods
Various triage systems, including clinical judgment, scoring systems, regression, machine learning, and deep learning, were applied to the benchmark dataset and evaluated on each benchmark task, as detailed in Table 1. A five-level triage system, Emergency Severity Index (ESI)45, was assigned by a registered nurse based on clinical judgments. Level 1 is the highest priority, and level 5 is the lowest. Several scoring systems were also calculated, including the Modified Early Warning Score (MEWS)46, National Early Warning Score (NEWS, versions 1 and 2)47, Rapid Emergency Medicine Score (REMS)48, and Cardiac Arrest Risk Triage (CART)49. It is important to note that there are no neurological features (i.e., Glasgow Coma Scale) in the MIMIC-IV-ED dataset, which may lead to incomplete scores. Three machine learning methods – logistic regression (LR), random forest (RF), and gradient boosting (GB) – were benchmarked as well as deep learning methods multilayer perceptron (MLP)50, Med2Vec39, and long short-term memory (LSTM)51,52,53. These neural network structures are illustrated in Supplementary eFigure 1. We used the scikit-learn package54 with the default parameters for machine learning methods and Keras55 for deep learning methods. In addition, the interpretable machine learning method, AutoScore56,57,58,59, was implemented with its R software package60.
Experiments, settings, and evaluation
We conducted all experiments on a server equipped with an Intel Xeon W-2275 processor, 128GB of memory, and an Nvidia RTX 3090 GPU, and the running time at model training was recorded. Deep learning models were trained using the Adam optimizer and binary cross-entropy loss. The AutoScore method optimized the number of variables through a parsimony plot. As the implementation was only for demonstration purposes, Module 5 of the clinical fine-tuning process in AutoScore was not implemented. We conducted the receiver operating characteristic (ROC) and precision-recall curve (PRC) analysis to evaluate the performance of all prediction models. The area under the ROC curve (AUROC) and the area under the PRC (AUPRC) values were reported as an overall measurement of predictive performance. Model performance was reported on the test set, and 100 bootstrapped samples were applied to calculate 95% confidence intervals (CI). Furthermore, we computed the sensitivity and specificity measures under the optimal cutoffs, defined as the points nearest to the upper-left corner of the ROC curves.
Results
Baseline characteristics of the benchmark dataset
We compiled a master dataset comprising 448,972 ED visits of 216,877 unique patients. After excluding incomplete or pediatric visits, a total of 441,437 adult ED visits were finally included in the benchmark dataset. They were randomly split into 80% (353,150) training data and 20% (88,287) test data. Table 2 and Supplementary eTable 2 summarize the baseline characteristics of the entire cohort, stratified by outcomes. The average age of the patients was 52.8 years old, and 54.3% (n = 239,794) of them were females. Compared with other patients, those with critical outcomes displayed higher body temperature and heart rate, and were prescribed a greater amount of medication. Additionally, they were more likely to have fluid and electrolyte disorders, coagulopathy, cancer, cardiac arrhythmias, valvular disease, and pulmonary circulation disorders.
The outcome statistics for the benchmark data are presented in Table 3, demonstrating a balanced stratification of the training and test data. In the overall cohort, 208,976 (47.34%) episodes require hospitalization, 26,174 (5.93%) episodes have critical outcomes, and 15,299 (3.47%) result in 72-hour ED reattendance.
Variable importance and ranking
Following a descending order of variable importance obtained from RF, the top 10 variables selected for each predictive task are presented in Table 4. Vital signs show significant predictive value in all three tasks. Age is also among the top predictive variables for all tasks, underscoring the impact of aging on emergency care utilization. While the triage level (i.e., ESI) is highly related to the hospitalization and critical outcome, it is not relevant to 72-hour ED reattendance. Conversely, despite its lower importance for hospitalization and critical outcomes, ED length of stay becomes the top variable for 72-hour ED reattendance prediction. The previous health utilization variable seems to be a less important feature for ED-based tasks.
Benchmark task evaluation
Machine learning exhibited a higher degree of discrimination in predicting all three outcomes. Gradient boosting achieved an AUC of 0.880 (95% CI: 0.876–0.884) for the critical outcome and an AUC of 0.819 (95% CI: 0.817–0.822) for the hospitalization outcome. However, the corresponding performance for 72-hour ED reattendance was considerably lower. Compared with gradient boosting, deep learning could not achieve even higher performance. While traditional scoring systems did not show good discriminatory performance, interpretable machine learning-based AutoScore achieved an AUC of 0.846 (95% CI: 0.842–0.851) for critical outcomes with seven variables, and 0.793 (95% CI: 0.791–0.797) for hospitalization outcomes with 10 variables. Tables 5–7 and Supplementary eTable 3 present the performance of of a variety of machine learning and scoring systems on different prediction tasks assessed by various metrics on the test set. Moreover, they are also plotted in Fig. 3.
Bar plots comparing the performance of various prediction models based on three different outcomes. AUROC: The area under the receiver operating characteristic curve. AUPRC: The area under the precision-recall curve. CART: Cardiac Arrest Risk Triage. ESI: Emergency Severity Index. GB: Gradient boosting. LSTM: Long short-term memory. LR: Logistic regression. MEWS: Modified Early Warning Score. MLP: Multilayer perceptron. NEWS: National Early Warning Score. NEWS2: National Early Warning Score, Version 2. REMS: Rapid Emergency Medicine Score. RF: Random forest.
Discussion
This paper proposes standardized data benchmarks for future researchers who are interested in analyzing large-scale ED-based clinical data. Our study provides a pipeline to process raw data from the newly published MIMIC-IV-ED database and generates a benchmark dataset, the first of its kind in the ED context. The benchmark dataset contains approximately half a million ED visits, and is highly accessible by researchers who plan to replicate our experiments or further build upon our work. Additionally, we demonstrated several clinical prediction models (e.g., machine learning and clinical scoring systems) on routinely available information using this benchmark dataset for three ED-relevant outcomes: hospitalization, critical outcome, and ED reattendance. Our benchmark dataset also supports linkage to the main MIMIC-IV database, allowing researchers to analyze a patient’s clinical course from the time of ED presentation through the hospital stay.
Our study showed that machine learning models demonstrated higher predictive accuracy, consistent with the previous studies9,19,61. Complex deep learning62 models such as Med2Vec and LSTM did not perform better than simpler models. These results suggest that overly complex models do not necessarily improve performance with relatively low-dimensional ED data. Furthermore, predictions made by black-box machine learning have critical limitations in clinical practice63,64, particularly for decision-making in emergency care. Although machine learning models outperform in terms of predictive accuracy, the lack of explainability makes it challenging for frontline physicians to understand how and why the model reaches a particular conclusion. In contrast, scoring systems combine just a few variables using simple arithmetic and have a more explicit clinical representation56. This transparency allows doctors to understand and trust model outputs more easily and contributes to the validity and acceptance of clinical scores in real-world settings65,66. In our experiments, predefined scoring systems were unable to achieve satisfactory accuracy. However, AutoScore-based data-driven scoring systems complemented them with much higher accuracy while maintaining the advantages of the point-based scores7.
The primary goals of ED prediction models are to identify high-risk patients accurately and to allocate limited resources efficiently. While physicians can generally determine the severity of a patient’s acute condition, their decisions necessarily contain subjective influences that depend on the healthcare context and practitioner’s knowledge. Objective predictive systems can outperform expert intuition40 in making multi-criteria decisions by taking away interpersonal variation between healthcare practitioners41. This could be a potentially valuable tool for emergency physicians who have to constantly multitask67, especially in the complex ED environment where decisions must be made based on heuristics and dynamic changes68. This study explores data-driven methods to provide an objective assessment for three ED-relevant risk triaging tasks based on large-scale public EHRs. Several previous studies34,69,70 have also demonstrated that objective electronic predictive triage systems provide more accurate differentiation for patients with regards to clinical outcomes compared with traditional subjective clinical assessment. In addition, the openly accessible nature of the models makes them suitable for reproducibility and improvement. The scientific research community can make full use of the benchmark data and the prediction benchmark in future research.
Three ED-based clinical outcomes were explored in this study with clinical significance. Accurate prediction of those three outcomes could help optimize ED resources with timely care delivery and mitigate ED delayed care problems. Our hospitalization prediction model can give an idea of the likelihood of hospitalization at the time of triage to the patients and staff, even before a physician is assigned to examine the patient40,41. Identifying patients who might end up with critical illness or death could potentially differentiate high-risk patients from more-stable patients and efficiently allocate finite ED resources5,6,7. Predicting ED reattendance could also allow providers to reconsider patient’s discharge plans and provide optimal care for those who had been prematurely discharge71. In addition, these three outcomes are interrelated yet represent distinct groups of predictors. Prediction models of hospitalization and critical outcomes share a similar set of predictors, whereas ED reattendances depend on various other variables. Although understanding personal risk or prognosis has great value, it is more important to realize the full potential of these prediction models in improving emergency care in clinical practice. In the future, focus should be shifted to filling the implementation gap by considering the model’s actionability and real-world utility72.
From a data science perspective, this study contributes to the scientific community by standardizing research workflows and reducing barriers of entry18 for both clinicians and data scientists engaged in ED research. In the future, researchers may use this data pipeline to process raw MIMIC-IV-ED data. They may also develop new models and evaluate them against our ED-based benchmark tasks and prediction models. Additionally, our pipeline does not focus exclusively on ED data; we also provide linkages to the MIMIC-IV main database73,74,75,76 with all ICU and inpatient episodes. Data scientists interested in extracting ED data as additional variables and linking them to the other settings of the MIMIC-IV database can exploit our framework to streamline their research without consulting different ED physicians. With the help of this first large-scale public ED benchmark dataset and data processing pipeline, researchers can conduct high-quality ED research without needing a high level of technical proficiency.
This study has several limitations. First, although the study is based on an extensive database, it is still a single-center study. The performance of different methods used in this study may differ in other healthcare settings. Nevertheless, the proposed clinical prediction pipeline could still be used as a reference for future big data research in ED. Furthermore, examining whether models trained on the benchmark data generalize to other clinical datasets would be interesting. Second, the benchmark dataset established in this study is based on EHR data extracted from the hospital’s patient portal with routinely collected variables, where certain potential risk factors, such as socioeconomic status, critical first look, and neurological features, were not recorded. For example, some health utilization data such as intubation and resuscitation have been proven to be predictive of overall mortality and should have been included in our model. Furthermore, neurological features like the Glasgow Coma Scale (GCS)77 score, were not available in the MIMIC-IV-ED database. These features are considered significant predictors in the ED setting and could have greatly increased the performance of our models. In addition, the dataset lacks sufficient information to detect out-of-hospital deaths, which may introduce bias into our predictions. Lastly, simple median imputation was employed to handle missing vital signs in the raw data, potentially obscuring data structures that could have been captured by more sophisticated methods. Future researchers utilizing our data pipeline should attempt to apply more advanced techniques for dealing with missing values. Despite these limitations, the data processing pipeline can be leveraged widely when new researchers wish to conduct ED research using the MIMIC-IV-ED database.
Data availability
The data that support the findings of this study are available from the MIMIC-IV database31: https://physionet.org/content/mimiciv/1.0/ and MIMIC-IV-ED database32: https://physionet.org/content/mimic-iv-ed/1.0/.
Code availability
The code used to analyze the data in the current study is available at: https://github.com/nliulab/mimic4ed-benchmark.
References
Jeffery, M. M. et al. Trends in Emergency Department Visits and Hospital Admissions in Health Care Systems in 5 States in the First Months of the COVID-19 Pandemic in the US. JAMA internal medicine 180, 1328–1333, https://doi.org/10.1001/jamainternmed.2020.3288 (2020).
Morley, C., Unwin, M., Peterson, G. M., Stankovich, J. & Kinsman, L. Emergency department crowding: A systematic review of causes, consequences and solutions. PLoS One 13, e0203316, https://doi.org/10.1371/journal.pone.0203316 (2018).
Huang, Q., Thind, A., Dreyer, J. F. & Zaric, G. S. The impact of delays to admission from the emergency department on inpatient outcomes. BMC Emerg Med 10, 16, https://doi.org/10.1186/1471-227X-10-16 (2010).
Sun, B. C. et al. Effect of emergency department crowding on outcomes of admitted patients. Ann Emerg Med 61, 605–611 e606, https://doi.org/10.1016/j.annemergmed.2012.10.026 (2013).
Raita, Y. et al. Emergency department triage prediction of clinical outcomes using machine learning models. Crit Care 23, 64, https://doi.org/10.1186/s13054-019-2351-7 (2019).
Iversen, A. K. S. et al. A simple clinical assessment is superior to systematic triage in prediction of mortality in the emergency department. Emerg Med J 36, 66–71, https://doi.org/10.1136/emermed-2016-206382 (2019).
Xie, F. et al. Development and Assessment of an Interpretable Machine Learning Triage Tool for Estimating Mortality After Emergency Admissions. JAMA Netw Open 4, e2118467, https://doi.org/10.1001/jamanetworkopen.2021.18467 (2021).
Liu, N. et al. Heart rate n-variability (HRnV) and its application to risk stratification of chest pain patients in the emergency department. BMC Cardiovasc Disord 20, 168, https://doi.org/10.1186/s12872-020-01455-8 (2020).
Nguyen, M. et al. Developing machine learning models to personalize care levels among emergency room patients for hospital admission. J Am Med Inform Assoc 28, 2423–2432, https://doi.org/10.1093/jamia/ocab118 (2021).
Schull, M. J., Ferris, L. E., Tu, J. V., Hux, J. E. & Redelmeier, D. A. Problems for clinical judgement: 3. Thinking clearly in an emergency. CMAJ 164, 1170–1175 (2001).
Ward, M. J. et al. The effect of electronic health record implementation on community emergency department operational measures of performance. Ann. Emerg. Med. 63, 723–730, https://doi.org/10.1016/j.annemergmed.2013.12.019 (2014).
Walker, K., Dwyer, T. & Heaton, H. A. Emergency medicine electronic health record usability: where to from here. Emergency Medicine Journal 38, 408, https://doi.org/10.1136/emermed-2021-211384 (2021).
Ang, Y. et al. Development and validation of an interpretable clinical score for early identification of acute kidney injury at the emergency department. Scientific Reports 12, 7111, https://doi.org/10.1038/s41598-022-11129-4 (2022).
Liu, N. et al. Leveraging Large-Scale Electronic Health Records and Interpretable Machine Learning for Clinical Decision Making at the Emergency Department: Protocol for System Development and Validation. JMIR Res Protoc 11, e34201, https://doi.org/10.2196/34201 (2022).
Johnson, A. E. et al. MIMIC-III, a freely accessible critical care database. Sci Data 3, 160035, https://doi.org/10.1038/sdata.2016.35 (2016).
Pollard, T. J. et al. The eICU Collaborative Research Database, a freely available multi-center database for critical care research. Sci Data 5, 180178, https://doi.org/10.1038/sdata.2018.178 (2018).
Thoral, P. J. et al. Sharing ICU Patient Data Responsibly Under the Society of Critical Care Medicine/European Society of Intensive Care Medicine Joint Data Science Collaboration: The Amsterdam University Medical Centers Database (AmsterdamUMCdb) Example. Crit Care Med 49, e563–e577, https://doi.org/10.1097/CCM.0000000000004916 (2021).
Harutyunyan, H., Khachatrian, H., Kale, D. C., Ver Steeg, G. & Galstyan, A. Multitask learning and benchmarking with clinical time series data. Sci Data 6, 96, https://doi.org/10.1038/s41597-019-0103-9 (2019).
Purushotham, S., Meng, C., Che, Z. & Liu, Y. Benchmarking deep learning models on large healthcare datasets. J Biomed Inform 83, 112–134, https://doi.org/10.1016/j.jbi.2018.04.007 (2018).
Wang, S. et al. MIMIC-Extract: a data extraction, preprocessing, and representation pipeline for MIMIC-III. Proceedings of the ACM Conference on Health, Inference, and Learning, 222–235, https://doi.org/10.1145/3368555.3384469 (2020).
Roy, S. et al. Multitask prediction of organ dysfunction in the intensive care unit using sequential subnetwork routing. Journal of the American Medical Informatics Association 28, 1936–1946, https://doi.org/10.1093/jamia/ocab101 (2021).
Coombes, C. E., Coombes, K. R. & Fareed, N. A novel model to label delirium in an intensive care unit from clinician actions. BMC Medical Informatics and Decision Making 21, 97, https://doi.org/10.1186/s12911-021-01461-6 (2021).
Wardi, G. et al. Predicting Progression to Septic Shock in the Emergency Department Using an Externally Generalizable Machine-Learning Algorithm. Ann. Emerg. Med. 77, 395–406, https://doi.org/10.1016/j.annemergmed.2020.11.007 (2021).
Kang, S. Y. et al. Predicting 30-day mortality of patients with pneumonia in an emergency department setting using machine-learning models. Clin Exp Emerg Med 7, 197–205, https://doi.org/10.15441/ceem.19.052 (2020).
Sarasa Cabezuelo, A. Application of Machine Learning Techniques to Analyze Patient Returns to the Emergency Department. J Pers Med 10, https://doi.org/10.3390/jpm10030081 (2020).
Tsai, C. M. et al. Using Machine Learning to Predict Bacteremia in Febrile Children Presented to the Emergency Department. Diagnostics (Basel) 10, https://doi.org/10.3390/diagnostics10050307 (2020).
Kuo, Y. H. et al. An Integrated Approach of Machine Learning and Systems Thinking for Waiting Time Prediction in an Emergency Department. Int J Med Inform 139, 104143, https://doi.org/10.1016/j.ijmedinf.2020.104143 (2020).
Hunter-Zinck, H. S., Peck, J. S., Strout, T. D. & Gaehde, S. A. Predicting emergency department orders with multilabel machine learning techniques and simulating effects on length of stay. J Am Med Inform Assoc 26, 1427–1436, https://doi.org/10.1093/jamia/ocz171 (2019).
Chee, M. L. et al. Artificial Intelligence Applications for COVID-19 in Intensive Care and Emergency Settings: A Systematic Review. Int J Environ Res Public Health 18, https://doi.org/10.3390/ijerph18094749 (2021).
Parker, C. A. et al. Predicting hospital admission at the emergency department triage: A novel prediction model. Am J Emerg Med 37, 1498–1504, https://doi.org/10.1016/j.ajem.2018.10.060 (2019).
Johnson, A. et al. MIMIC-IV (version 1.0). PhysioNet https://doi.org/10.13026/a3wn-hq05 (2021).
Johnson, A. et al. MIMIC-IV-ED (version 1.0). PhysioNet https://doi.org/10.13026/as7t-c445 (2021).
Dickson, S. J. et al. Agreement and validity of electronic patient self-triage (eTriage) with nurse triage in two UK emergency departments: a retrospective study. Eur J Emerg Med, https://doi.org/10.1097/MEJ.0000000000000863 (2021).
Levin, S. et al. Machine-Learning-Based Electronic Triage More Accurately Differentiates Patients With Respect to Clinical Outcomes Compared With the Emergency Severity Index. Ann Emerg Med 71, 565–574 e562, https://doi.org/10.1016/j.annemergmed.2017.08.005 (2018).
Dugas, A. F. et al. An Electronic Emergency Triage System to Improve Patient Distribution by Critical Outcomes. J Emerg Med 50, 910–918, https://doi.org/10.1016/j.jemermed.2016.02.026 (2016).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40, 373–383, https://doi.org/10.1016/0021-9681(87)90171-8 (1987).
Elixhauser, A., Steiner, C., Harris, D. R. & Coffey, R. M. Comorbidity measures for use with administrative data. Med Care 36, 8–27, https://doi.org/10.1097/00005650-199801000-00004 (1998).
Quan, H. et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care 43, 1130–1139, https://doi.org/10.1097/01.mlr.0000182534.19832.83 (2005).
Choi, E. et al. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining. 1495–1504.
Cameron, A., Rodgers, K., Ireland, A., Jamdar, R. & McKay, G. A. A simple tool to predict admission at the time of triage. Emergency Medicine Journal 32, 174, https://doi.org/10.1136/emermed-2013-203200 (2015).
Kraaijvanger, N. et al. Development and validation of an admission prediction tool for emergency departments in the Netherlands. Emerg Med J 35, 464–470, https://doi.org/10.1136/emermed-2017-206673 (2018).
Mowbray, F., Zargoush, M., Jones, A., de Wit, K. & Costa, A. Predicting hospital admission for older emergency department patients: Insights from machine learning. Int J Med Inform 140, 104163, https://doi.org/10.1016/j.ijmedinf.2020.104163 (2020).
Xie, F. et al. Novel model for predicting inpatient mortality after emergency admission to hospital in Singapore: retrospective observational study. BMJ Open 9, e031382, https://doi.org/10.1136/bmjopen-2019-031382 (2019).
Chan, A. H. et al. Characteristics of patients who made a return visit within 72 hours to the emergency department of a Singapore tertiary hospital. Singapore Med J 57, 301–306, https://doi.org/10.11622/smedj.2016104 (2016).
Eitel, D. R., Travers, D. A., Rosenau, A. M., Gilboy, N. & Wuerz, R. C. The emergency severity index triage algorithm version 2 is reliable and valid. Academic Emergency Medicine 10, 1070–1080 (2003).
Subbe, C. P., Kruger, M., Rutherford, P. & Gemmel, L. Validation of a modified early warning score in medical admissions. QJM 94, 521–526 (2001).
Royal College of, P. National early warning score (NEWS) 2. Standardising the assessment of acute-illness severity in the NHS (2017).
Olsson, T., Terent, A. & Lind, L. Rapid Emergency Medicine score: a new prognostic tool for in-hospital mortality in nonsurgical emergency department patients. J Intern Med 255, 579–587, https://doi.org/10.1111/j.1365-2796.2004.01321.x (2004).
Churpek, M. M. et al. Derivation of a cardiac arrest prediction model using ward vital signs. Crit Care Med 40, 2102–2108, https://doi.org/10.1097/CCM.0b013e318250aa5a (2012).
Hinton, G. E. Connectionist learning procedures. Artificial Intelligence 40, 185–234, https://doi.org/10.1016/0004-3702(89)90049-0 (1989).
Baytas, I. M. et al. in Proceedings of the 23rd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining 65–74 (Association for Computing Machinery, Halifax, NS, Canada, 2017).
Maragatham, G. & Devi, S. LSTM Model for Prediction of Heart Failure in Big Data. J Med Syst 43, 111, https://doi.org/10.1007/s10916-019-1243-3 (2019).
Lu, W., Ma, L., Chen, H., Jiang, X. & Gong, M. A Clinical Prediction Model in Health Time Series Data Based on Long Short-Term Memory Network Optimized by Fruit Fly Optimization Algorithm. IEEE Access 8, 136014–136023, https://doi.org/10.1109/ACCESS.2020.3011721 (2020).
Pedregosa, F. et al. Scikit-learn: Machine learning in Python. the Journal of machine Learning research 12, 2825–2830 (2011).
Gulli, A. & Pal, S. Deep learning with Keras. (Packt Publishing Ltd, 2017).
Xie, F., Chakraborty, B., Ong, M. E. H., Goldstein, B. A. & Liu, N. AutoScore: A Machine Learning-Based Automatic Clinical Score Generator and Its Application to Mortality Prediction Using Electronic Health Records. JMIR Med Inform 8, e21798, https://doi.org/10.2196/21798 (2020).
Xie, F. et al. AutoScore-Survival: Developing interpretable machine learning-based time-to-event scores with right-censored survival data. Journal of Biomedical Informatics 125, 103959, https://doi.org/10.1016/j.jbi.2021.103959 (2022).
Yuan, H. et al. AutoScore-Imbalance: An interpretable machine learning tool for development of clinical scores with rare events data. J Biomed Inform 129, 104072, https://doi.org/10.1016/j.jbi.2022.104072 (2022).
Ning, Y. et al. A novel interpretable machine learning system to generate clinical risk scores: An application for predicting early mortality or unplanned readmission in a retrospective cohort study. PLOS Digital Health 1, e0000062, https://doi.org/10.1371/journal.pdig.0000062 (2022).
Xie, F. et al. Package ‘AutoScore’: An Interpretable Machine Learning-Based Automatic Clinical Score Generator. (2021).
Sadeghi, R., Banerjee, T. & Romine, W. Early hospital mortality prediction using vital signals. Smart Health 9-10, 265–274, https://doi.org/10.1016/j.smhl.2018.07.001 (2018).
Xie, F. et al. Deep learning for temporal data representation in electronic health records: A systematic review of challenges and methodologies. Journal of Biomedical Informatics 126, 103980, https://doi.org/10.1016/j.jbi.2021.103980 (2022).
Hsu, W. & Elmore, J. G. Shining Light Into the Black Box of Machine Learning. J Natl Cancer Inst 111, 877–879, https://doi.org/10.1093/jnci/djy226 (2019).
Rudin, C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nature Machine Intelligence 1, 206–215 (2019).
Alam, N. et al. The impact of the use of the Early Warning Score (EWS) on patient outcomes: A systematic review. Resuscitation 85, 587–594, https://doi.org/10.1016/j.resuscitation.2014.01.013 (2014).
Gerry, S. et al. Early warning scores for detecting deterioration in adult hospital patients: systematic review and critical appraisal of methodology. BMJ 369, m1501, https://doi.org/10.1136/bmj.m1501 (2020).
Hargrove, J. & Nguyen, H. B. Bench-to-bedside review: outcome predictions for critically ill patients in the emergency department. Crit Care 9, 376–383, https://doi.org/10.1186/cc3518 (2005).
Franklin, A. et al. Opportunistic decision making and complexity in emergency care. Journal of Biomedical Informatics 44, 469–476, https://doi.org/10.1016/j.jbi.2011.04.001 (2011).
Chang, Y.-H. et al. Machine learning–based triage to identify low-severity patients with a short discharge length of stay in emergency department. BMC Emergency Medicine 22, 88, https://doi.org/10.1186/s12873-022-00632-6 (2022).
Xie, F. et al. Development and validation of an interpretable machine learning scoring tool for estimating time to emergency readmissions. EClinicalMedicine 45, 101315, https://doi.org/10.1016/j.eclinm.2022.101315 (2022).
Hong, W. S., Haimovich, A. D. & Taylor, R. A. Predicting 72-hour and 9-day return to the emergency department using machine learning. JAMIA Open 2, 346–352, https://doi.org/10.1093/jamiaopen/ooz019 (2019).
Seneviratne, M. G., Shah, N. H. & Chu, L. Bridging the implementation gap of machine learning in healthcare. BMJ Innovations 6, 45, https://doi.org/10.1136/bmjinnov-2019-000359 (2020).
Mandyam, A., Yoo, E. C., Soules, J., Laudanski, K. & Engelhardt, B. E. COP-E-CAT: cleaning and organization pipeline for EHR computational and analytic tasks. Proceedings of the 12th ACM Conference on Bioinformatics, Computational Biology, and Health Informatics, Article 5, https://doi.org/10.1145/3459930.3469536 (2021).
Meng, C., Trinh, L., Xu, N., Enouen, J. & Liu, Y. Interpretability and fairness evaluation of deep learning models on MIMIC-IV dataset. Scientific Reports 12, 1–28 (2022).
Gupta, M. et al. An Extensive Data Processing Pipeline for MIMIC-IV. arXiv preprint arXiv:2204.13841 (2022).
Johnson, A. E. W., Stone, D. J., Celi, L. A. & Pollard, T. J. The MIMIC Code Repository: enabling reproducibility in critical care research. Journal of the American Medical Informatics Association 25, 32–39, https://doi.org/10.1093/jamia/ocx084 (2018).
Sternbach, G. L. The Glasgow coma scale. The Journal of emergency medicine 19, 67–71 (2000).
Author information
Authors and Affiliations
Contributions
N.L. and F.X. conceived and designed the study. F.X., J.Z., J.W.L., M.T., S.L. and L.S.R. analyzed the data. All authors interpreted the data and results. F.X., J.Z., J.W.L., M.T., S.L., M.L.C., F.G. and N.L. drafted the manuscript. All authors critically revised the manuscript for intellectual content. F.X. and J.Z. contributed equally to this work. F.G. and N.L. jointly supervised the study. All authors had access to all the data in the study and had final responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xie, F., Zhou, J., Lee, J.W. et al. Benchmarking emergency department prediction models with machine learning and public electronic health records. Sci Data 9, 658 (2022). https://doi.org/10.1038/s41597-022-01782-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-022-01782-9
This article is cited by
-
Machine learning models for predicting unscheduled return visits to an emergency department: a scoping review
BMC Emergency Medicine (2024)