Abstract
Vertebrate replication forks arrested at interstrand DNA cross-links (ICLs) engage the Fanconi anemia pathway to incise arrested forks, ‘unhooking’ the ICL and forming a double strand break (DSB) that is repaired by homologous recombination (HR). The FANCP product, SLX4, in complex with the XPF (also known as FANCQ or ERCC4)–ERCC1 endonuclease, mediates ICL unhooking. Whether this mechanism operates at replication fork barriers other than ICLs is unknown. Here, we study the role of mouse SLX4 in HR triggered by a site-specific chromosomal DNA–protein replication fork barrier formed by the Escherichia coli-derived Tus–Ter complex. We show that SLX4–XPF is required for Tus–Ter-induced HR but not for error-free HR induced by a replication-independent DSB. We additionally uncover a role for SLX4–XPF in DSB-induced long-tract gene conversion, an error-prone HR pathway related to break-induced replication. Notably, Slx4 and Xpf mutants that are defective for Tus–Ter-induced HR are hypersensitive to ICLs and also to the DNA–protein cross-linking agents 5-aza-2′-deoxycytidine and zebularine. Collectively, these findings show that SLX4–XPF can process DNA–protein fork barriers for HR and that the Tus–Ter system recapitulates this process.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Source data are provided with this paper. All data supporting the findings of this study are available from the corresponding author upon request.
References
Berti, M., Cortez, D. & Lopes, M. The plasticity of DNA replication forks in response to clinically relevant genotoxic stress. Nat. Rev. Mol. Cell Biol. 21, 633–651 (2020).
Cortez, D. Replication-coupled DNA repair. Mol. Cell 74, 866–876 (2019).
Quinet, A., Tirman, S., Cybulla, E., Meroni, A. & Vindigni, A. To skip or not to skip: choosing repriming to tolerate DNA damage. Mol. Cell 81, 649–658 (2021).
Crossley, M. P., Bocek, M. & Cimprich, K. A. R-loops as cellular regulators and genomic threats. Mol. Cell 73, 398–411 (2019).
Semlow, D. R., Zhang, J., Budzowska, M., Drohat, A. C. & Walter, J. C. Replication-dependent unhooking of DNA interstrand cross-links by the NEIL3 glycosylase. Cell 167, 498–511.e14 (2016).
Hodskinson, M. R. et al. Alcohol-derived DNA crosslinks are repaired by two distinct mechanisms. Nature 579, 603–608 (2020).
Huang, J. et al. The DNA translocase FANCM/MHF promotes replication traverse of DNA interstrand crosslinks. Mol. Cell 52, 434–446 (2013).
Huang, J. et al. Remodeling of interstrand crosslink proximal replisomes is dependent on ATR, FANCM, and FANCD2. Cell Rep. 27, 1794–1808.e5 (2019).
Semlow, D. R. & Walter, J. C. Mechanisms of vertebrate DNA interstrand cross-link repair. Annu. Rev. Biochem. 90, 107–135 (2021).
Niraj, J., Färkkilä, A. & D’Andrea, A. D. The fanconi anemia pathway in Cancer. Annu. Rev. Cancer Biol. 3, 457–478 (2019).
Taylor, A. M. R. et al. Chromosome instability syndromes. Nat. Rev. Dis. Primers 5, 64 (2019).
Garaycoechea, J. I. et al. Alcohol and endogenous aldehydes damage chromosomes and mutate stem cells. Nature 553, 171–177 (2018).
Langevin, F., Crossan, G. P., Rosado, I. V., Arends, M. J. & Patel, K. J. Fancd2 counteracts the toxic effects of naturally produced aldehydes in mice. Nature 475, 53–58 (2011).
Rosado, I. V., Langevin, F., Crossan, G. P., Takata, M. & Patel, K. J. Formaldehyde catabolism is essential in cells deficient for the Fanconi anemia DNA-repair pathway. Nat. Struct. Mol. Biol. 18, 1432–1434 (2011).
O’Brien, P. J., Siraki, A. G. & Shangari, N. Aldehyde sources, metabolism, molecular toxicity mechanisms, and possible effects on human health. Crit. Rev. Toxicol. 35, 609–662 (2005).
Raschle, M. et al. Mechanism of replication-coupled DNA interstrand crosslink repair. Cell 134, 969–980 (2008).
Wu, R. A. et al. TRAIP is a master regulator of DNA interstrand crosslink repair. Nature 567, 267–272 (2019).
Wang, S., Wang, R., Peralta, C., Yaseen, A. & Pavletich, N. P. Structure of the FA core ubiquitin ligase closing the ID clamp on DNA. Nat. Struct. Mol. Biol. 28, 300–309 (2021).
Shakeel, S. et al. Structure of the Fanconi anaemia monoubiquitin ligase complex. Nature 575, 234–237 (2019).
Xue, X., Sung, P. & Zhao, X. Functions and regulation of the multitasking FANCM family of DNA motor proteins. Genes Dev. 29, 1777–1788 (2015).
Alcón, P. et al. FANCD2-FANCI is a clamp stabilized on DNA by monoubiquitination of FANCD2 during DNA repair. Nat. Struct. Mol. Biol. 27, 240–248 (2020).
Tan, W. et al. Monoubiquitination by the human Fanconi anemia core complex clamps FANCI:FANCD2 on DNA in filamentous arrays. eLife 9, e54128 (2020).
Amunugama, R. et al. Replication fork reversal during DNA interstrand crosslink repair requires CMG unloading. Cell Rep. 23, 3419–3428 (2018).
Kim, Y. et al. Regulation of multiple DNA repair pathways by the Fanconi anemia protein SLX4. Blood 121, 54–63 (2013).
Klein Douwel, D. et al. XPF-ERCC1 acts in unhooking DNA interstrand crosslinks in cooperation with FANCD2 and FANCP/SLX4. Mol. Cell 54, 460–471 (2014).
Bogliolo, M. et al. Mutations in ERCC4, encoding the DNA-repair endonuclease XPF, cause Fanconi anemia. Am. J. Hum. Genet. 92, 800–806 (2013).
Marín, M. et al. Functional comparison of XPF missense mutations associated to multiple DNA repair disorders. Genes 10, 60 (2019).
Hodskinson, M. R. et al. Mouse SLX4 is a tumor suppressor that stimulates the activity of the nuclease XPF-ERCC1 in DNA crosslink repair. Mol. Cell 54, 472–484 (2014).
Manandhar, M., Boulware, K. S. & Wood, R. D. The ERCC1 and ERCC4 (XPF) genes and gene products. Gene 569, 153–161 (2015).
Zhang, J. & Walter, J. C. Mechanism and regulation of incisions during DNA interstrand cross-link repair. DNA Repair 19, 135–142 (2014).
Duxin, J. P. & Walter, J. C. What is the DNA repair defect underlying Fanconi anemia? Curr. Opin. Cell Biol. 37, 49–60 (2015).
Willis, N. A. et al. BRCA1 controls homologous recombination at Tus/Ter-stalled mammalian replication forks. Nature 510, 556–559 (2014).
Willis, N. A. et al. Mechanism of tandem duplication formation in BRCA1-mutant cells. Nature 551, 590–595 (2017).
Nik-Zainal, S. et al. Landscape of somatic mutations in 560 breast cancer whole-genome sequences. Nature 534, 47–54 (2016).
Menghi, F. et al. The tandem duplicator phenotype is a prevalent genome-wide cancer configuration driven by distinct gene mutations. Cancer Cell 34, 197–210.e5 (2018).
Scully, R., Panday, A., Elango, R. & Willis, N. A. DNA double-strand break repair-pathway choice in somatic mammalian cells. Nat. Rev. Mol. Cell Biol. 20, 698–714 (2019).
Panday, A. et al. FANCM regulates repair pathway choice at stalled replication forks. Mol. Cell. 81, 2428–2444.e6 (2021).
Lambert, S. et al. Homologous recombination restarts blocked replication forks at the expense of genome rearrangements by template exchange. Mol. Cell 39, 346–359 (2010).
Wyatt, H. D., Laister, R. C., Martin, S. R., Arrowsmith, C. H. & West, S. C. The SMX DNA repair tri-nuclease. Mol. Cell 65, 848–860.e11 (2017).
Fekairi, S. et al. Human SLX4 is a Holliday junction resolvase subunit that binds multiple DNA repair/recombination endonucleases. Cell 138, 78–89 (2009).
Svendsen, J. M. et al. Mammalian BTBD12/SLX4 assembles a Holliday junction resolvase and is required for DNA repair. Cell 138, 63–77 (2009).
Saito, T. T., Youds, J. L., Boulton, S. J. & Colaiácovo, M. P. Caenorhabditis elegans HIM-18/SLX-4 interacts with SLX-1 and XPF-1 and maintains genomic integrity in the germline by processing recombination intermediates. PLoS Genet. 5, e1000735 (2009).
Andersen, S. L. et al. Drosophila MUS312 and the vertebrate ortholog BTBD12 interact with DNA structure-specific endonucleases in DNA repair and recombination. Mol. Cell 35, 128–135 (2009).
Guervilly, J. H. & Gaillard, P. H. SLX4: multitasking to maintain genome stability. Crit. Rev. Biochem. Mol. Biol. 53, 475–514 (2018).
Young, S. J. & West, S. C. Coordinated roles of SLX4 and MutSβ in DNA repair and the maintenance of genome stability. Crit. Rev. Biochem. Mol. Biol. 56, 157–177 (2021).
Young, S. J. et al. MutSβ stimulates Holliday junction resolution by the SMX complex. Cell Rep. 33, 108289 (2020).
Kim, Y. et al. Mutations of the SLX4 gene in Fanconi anemia. Nat. Genet. 43, 142–146 (2011).
Lachaud, C. et al. Distinct functional roles for the two SLX4 ubiquitin-binding UBZ domains mutated in Fanconi anemia. J. Cell Sci. 127, 2811–2817 (2014).
Katsuki, Y. et al. RNF168 E3 ligase participates in ubiquitin signaling and recruitment of SLX4 during DNA crosslink repair. Cell Rep. 37, 109879 (2021).
Stoepker, C. et al. SLX4, a coordinator of structure-specific endonucleases, is mutated in a new Fanconi anemia subtype. Nat. Genet. 43, 138–141 (2011).
Crossan, G. P. et al. Disruption of mouse Slx4, a regulator of structure-specific nucleases, phenocopies Fanconi anemia. Nat. Genet. 43, 147–152 (2011).
Yamamoto, K. N. et al. Involvement of SLX4 in interstrand cross-link repair is regulated by the Fanconi anemia pathway. Proc. Natl Acad. Sci. USA 108, 6492–6496 (2011).
Kosicki, M., Tomberg, K. & Bradley, A. Repair of double-strand breaks induced by CRISPR-Cas9 leads to large deletions and complex rearrangements. Nat. Biotechnol. 36, 765–771 (2018).
Jüttermann, R., Li, E. & Jaenisch, R. Toxicity of 5-aza-2′-deoxycytidine to mammalian cells is mediated primarily by covalent trapping of DNA methyltransferase rather than DNA demethylation. Proc. Natl Acad. Sci. USA 91, 11797–11801 (1994).
Zhou, L. et al. Zebularine: a novel DNA methylation inhibitor that forms a covalent complex with DNA methyltransferases. J. Mol. Biol. 321, 591–599 (2002).
Natsume, T., Kiyomitsu, T., Saga, Y. & Kanemaki, M. T. Rapid protein depletion in human cells by auxin-inducible degron tagging with short homology donors. Cell Rep. 15, 210–218 (2016).
Nishimura, K. et al. A super-sensitive auxin-inducible degron system with an engineered auxin-TIR1 pair. Nucleic Acids Res. 48, e108 (2020).
Chung, H. K. et al. Tunable and reversible drug control of protein production via a self-excising degron. Nat. Chem. Biol. 11, 713–720 (2015).
Wang, X. et al. Role of DNA replication proteins in double-strand break-induced recombination in Saccharomyces cerevisiae. Mol. Cell. Biol. 24, 6891–6899 (2004).
Duxin, J. P., Dewar, J. M., Yardimci, H. & Walter, J. C. Repair of a DNA-protein crosslink by replication-coupled proteolysis. Cell 159, 346–357 (2014).
Larsen, N. B. et al. Replication-coupled DNA-protein crosslink repair by SPRTN and the proteasome in Xenopus egg extracts. Mol. Cell 73, 574–588.e7 (2019).
Sparks, J. L. et al. The CMG helicase bypasses DNA-protein cross-links to facilitate their repair. Cell 176, 167–181.e21 (2019).
Stingele, J., Bellelli, R. & Boulton, S. J. Mechanisms of DNA-protein crosslink repair. Nat. Rev. Mol. Cell Biol. 18, 563–573 (2017).
Wu, W. et al. RTEL1 suppresses G-quadruplex-associated R-loops at difficult-to-replicate loci in the human genome. Nat. Struct. Mol. Biol. 27, 424–437 (2020).
Di Marco, S. et al. RECQ5 helicase cooperates with MUS81 endonuclease in processing stalled replication forks at common fragile sites during mitosis. Mol. Cell 66, 658–671.e8 (2017).
Garribba, L. et al. Folate stress induces SLX1- and RAD51-dependent mitotic DNA synthesis at the fragile X locus in human cells. Proc. Natl Acad. Sci. USA 117, 16527–16536 (2020).
Puget, N., Knowlton, M. & Scully, R. Molecular analysis of sister chromatid recombination in mammalian cells. DNA Repair 4, 149–161 (2005).
Willis, N. A. & Scully, R. Measurement of homologous recombination at stalled mammalian replication forks. Methods Mol. Biol. 2153, 329–353 (2021).
Willis, N. A., Panday, A., Duffey, E. E. & Scully, R. Rad51 recruitment and exclusion of non-homologous end joining during homologous recombination at a Tus/Ter mammalian replication fork barrier. PLoS Genet. 14, e1007486 (2018).
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 3, 1101–1108 (2008).
Acknowledgements
We thank J. Walter and K. J. Patel for helpful discussions and expert advice. This work was supported by grants R01GM134425 (to R.S.) and R01CA204127 (to A.S.), and by AACR fellowship 19-40-12-PAND (to A.P.). A.S. is a Howard Hughes Faculty Scholar.
Author information
Authors and Affiliations
Contributions
R.E., A.P., F.P.L., K.N., N.A.W. and E.E.D. conducted the experiments. R.E., A.S. and R.S. designed the experiments. R.E. and R.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Structural & Molecular Biology thanks Katharina Schlacher and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editors: Beth Moorefield and Carolina Perdigoto, in collaboration with the Nature Structural & Molecular Biology team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Characterization of Slx4∆125/∆ cells.
a. RT-qPCR analysis of Slx4 mRNA normalized to Gapdh mRNA using the 2–ΔCT method in three independent experiments. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3), Error bars: standard deviation (s.d.) Statistical analysis using Student’s t-test. b and c. Ratio of Tus/Ter-induced (b) and I-SceI-induced (c) LTGC: total HR in Slx4+/+ clones and Slx4∆125/∆ clones. Data shows mean values, n = 6. Error bars: s.e.m. Analysis by ANOVA. P-value *p < 0.05 and ****p < 0.0001 d. CRISPR/Cas9 with dual sgRNA targeting to generate Slx4+/– hemizygous cells. The Slx4– allele contains a 19.3 kb deletion between exons 2 and 15. Red half arrowheads: PCR and sequencing primers specific to Exons 2 and 15. Predicted PCR product sizes for Slx4+ (wild type) allele shown. e. Gel shows PCR products detected using gDNA from Slx4+/+ and Slx4+/– clones. Note the 444 bp PCR product formed across the 19.3 kb deletion between exons 2 and 15 in Slx4+/– cells. f. DNA sequencing chromatogram of PCR products from (e). g. RT qPCR analysis of Slx4 mRNA in Slx4+/+ and Slx4+/– cells. Data shows mean of three independent experiments (n = 3) normalized to Gapdh mRNA using the 2–Δ∆CT method and analyzed by unpaired Student’s t-test. Error bars: s.d.. h. and i. Quantification of colony formation of Slx4+/+ vs. Slx4+/– clones in the presence of MMC (h), Olaparib (i). Data shows mean of three biologically independent replicates, n = 3. Error bars: s.d. j. Tus/Ter-induced and I-SceI-induced repair frequencies in Slx4+/+ and Slx4+/– cells. Data shows mean values of five biologically independent replicates, n = 5. Error bars: s.e.m. Statistical analysis using ANOVA.
Extended Data Fig. 2 The SLX4 UBZ domain promotes resistance to DPC-inducing drugs.
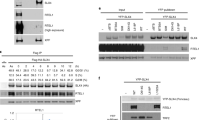
a. Strategy for in-frame deletion of UBZ domain-encoding regions of Slx4. Red half-arrow heads: genotyping PCR primers. Gel shows PCR products using gDNA from Slx4+/– and Slx4∆UBZ/– clones. Sequencing chromatogram shows in-frame breakpoint of Slx4∆UBZ allele. b. RT-qPCR analysis of mRNA encoding UBZ, MLR, SAP and SBD domains in Slx4+/– and Slx4∆UBZ/–clones. Data normalized to Gapdh mRNA using the 2–ΔCT method, mRNA expression in Slx4∆UBZ/– samples were normalized to Slx4+/– of the same experiment. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3). Analysis by unpaired Student’s t-test (n = 3). Error bars: standard deviation. P = 0.0125 for mRNA expression of the UBZ region in Slx4+/– compared to Slx4∆UBZ/–. No significant differences were observed in expression levels of all other domains. c-h. Quantification of colony formation of Slx4+/–, Slx4∆UBZ/– and Brca1-∆exon11 clones in the presence of MMC (c), Olaparib (d), 5-aza-2′-deoxycytidine (e) Zebularine (f), Hydroxyurea (g) and Camptothecin (h). Data shows mean of values of three biologically independent replicates, n = 3. Analysis using Student’s t-test. P-value *p < 0.05 and ***p < 0.001. Red asterisks refer to comparison between Slx4+/– and Slx4∆UBZ/– clones; blue asterisks denote comparison between Slx4+/– and Brca1-∆exon11 clones and blue bracket with red asterisks denotes comparison between Slx4∆UBZ/– and Brca1-∆exon11 clones. Error bars: s.d. i. RT-qPCR analysis of N-terminal Flag-tagged wild-type full-length human SLX4 and UBZ 4 C > A expression plasmids 48 h after transfection. Data normalized to Gapdh mRNA using the 2–ΔCT method and compared to empty vector (EV) control. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3). Error bar: s.d. j. Western blot analysis of the chromatin-bound fraction of various human 3xFLAG-SLX4 full-length and UBZ 4 C > A transiently expressed for 48 hours and blotted with anti-FLAG antibody. k. Western blot analysis of whole cell extract (WCE) and chromatin-bound fraction of clones of Slx4+/– and Slx4∆UBZ/– tagged with a dual degron containing a C-terminal 8xHA tag and untagged hemizygote cells (+/–). Asterisk (*) denotes non-specific bands.
Extended Data Fig. 3 The SLX4 UBZ domain mediates I-SceI-induced LTGC.
a, b. Tus/Ter-induced LTGC (a) and LTGC/Total HR ratio (b) in Slx4+/– and Slx4∆UBZ/– clones. Data shows mean values of five biologically independent replicates, n = 5. Error bars: s.e.m. Analysis by one-way ANOVA. P-value: ****p < 0.0001. Error bars: s.e.m. c. Impact of exogenous hSLX4 on Tus/Ter-induced LTGC in two independent Slx4+/– and two independent Slx4∆UBZ/– clones. EV: empty vector. 4 C > A: hSLX4 with inactivated UBZ motifs. Data shows mean values of six biologically independent replicates, n = 6. Error bars: s.e.m. Analysis by unpaired Student’s t-test. Error bars: s.e.m. d and e. I-SceI-induced LTGC (d) and LTGC/Total HR ratio (e) in Slx4+/– and Slx4∆UBZ/– clones. Data shows mean values of five biologically independent replicates, n = 5. P-value: ****p < 0.0001 Error bars: s.e.m. Analysis by one-way ANOVA. Error bars: s.e.m. f. Impact of exogenous hSLX4 on I-SceI-induced LTGC in two independent Slx4+/– and two independent Slx4∆UBZ/– clones. EV: empty vector. 4 C > A: hSLX4 with inactivated UBZ motifs. Data shows mean values of six biologically independent replicates, n = 6. Error bars: s.e.m. Analysis by unpaired Student’s t-test. P-value: *p < 0.05. Error bars: s.e.m.
Extended Data Fig. 4 Characterization of Slx4∆SAP/– and Slx4∆SBD/– mutant clones.
a. Strategy for in-frame deletion of SAP domain-encoding region of Slx4. Red half-arrow heads: genotyping PCR primers. Gels show PCR products using gDNA from Slx4+/– and Slx4∆SAP/– clones. Sequencing chromatogram shows in-frame breakpoint of Slx4∆SAP allele. b. RT-qPCR analysis of UBZ, MLR, SAP and SBD domains encoding mRNA in Slx4+/– and Slx4∆SAP/–clones. Data normalized to Gapdh mRNA using the 2–ΔCT method, mRNA expression in Slx4∆SAP/– were normalized to Slx4+/–. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3). Analysis by unpaired Student’s t-test. Error bars: s.d. P = 0.0478 for mRNA expression of the SAP region in Slx4+/– compared to Slx4∆SAP/–. No significant differences were observed in expression levels of all other domains. c. Anti-HA immunoblot of two independent HA-degron tagged Slx4∆SAP/– and Slx4+/– isogenic cell lines. The parental Slx4+/– untagged cell line was used as a control. d and e. Tus/Ter-induced LTGC (d) and I-SceI-induced LTGC (e) in Slx4+/– and Slx4∆SAP/– clones. Data shows mean values of five biologically independent replicates, n = 5. Error bars: s.e.m. Analysis by ordinary one-way ANOVA. Error bars: s.e.m. f. Strategy for in-frame deletion of SBD domain-encoding region of Slx4. Red half-arrow heads: genotyping PCR primers. Gels show PCR products using gDNA from Slx4+/– and Slx4∆SBD/– clones. Sequencing chromatogram shows in-frame breakpoint of Slx4∆SBD allele. g. RT-qPCR analysis of UBZ, MLR, SAP and SBD domains encoding mRNA in Slx4+/– and Slx4∆SBD/–clones. Data normalized to Gapdh mRNA using the 2–ΔCT method, mRNA expression in Slx4∆SBD/– was normalized to Slx4+/– of the same experiment. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3). Analysis by unpaired Student’s t-test (n = 3). Error bars: standard deviation. P = 0.048 for mRNA expression of the SAP region in Slx4+/– compared to Slx4∆SBD/–. No significant differences were observed in expression levels of all other domains. h. Anti-HA immunoblot of two independent HA-degron tagged Slx4∆SBD/– and Slx4+/– isogenic cell lines. The parental Slx4+/– untagged cell line was used as a control. i and j. Tus/Ter-induced LTGC (i) and I-SceI-induced LTGC (j) in Slx4+/– and Slx4∆SBD/– clones. Data shows mean values of five biologically independent replicates, n = 5. Error bars: s.e.m. Analysis by ordinary one-way ANOVA. Error bars: s.e.m.
Extended Data Fig. 5 Slx4∆SAP/– and Slx4∆SBD/– mutant clones are resistant to DPC-inducing drugs.
a-f. Quantification of colony formation of Slx4+/– vs. Slx4∆SAP/– clones in the presence of MMC (a), Olaparib (b), 5-aza-2′-deoxycytidine (c), Zebularine (d) Hydroxyurea (e) and Camptothecin (f). Data show mean of three biologically independent replicates normalized to untreated samples (n = 3). Error bars: s.d. g-l. Quantification of colony formation of Slx4+/– vs. Slx4∆SBD/– clones in the presence of MMC (g), Olaparib (h), 5-aza-2′-deoxycytidine (i), Zebularine (j) Hydroxyurea (k) and Camptothecin (l). Data show mean of three biologically independent replicates normalized to untreated samples (n = 3). Error bars: s.d.
Extended Data Fig. 6 Characterization of Slx4∆MLR/– mutant clones.
a. Strategy for in-frame deletion of MLR domain-encoding region of Slx4. Red half-arrow heads: genotyping PCR primers. Gels show PCR products using gDNA from Slx4+/– and Slx4∆MLR/– clones. Sequencing chromatogram shows in-frame breakpoint of Slx4∆MLR allele. b. RT-qPCR analysis of UBZ, MLR, SAP and SBD domains encoding mRNA in Slx4+/– and Slx4∆MLR/–clones. Data normalized to Gapdh mRNA using the 2–ΔCT method, mRNA expression in Slx4∆MLR/– were normalized to Slx4+/– of the same experiment. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3). Analysis by unpaired Student’s t-test (n = 3). Error bars: standard deviation (s.d.). P = 0.01 for mRNA expression of the MLR region in Slx4+/– compared to Slx4∆MLR/–. No significant differences were observed in expression levels of all other domains. c Anti-HA immunoblot of two independent HA-degron tagged Slx4∆MLR/– and Slx4+/– isogenic cell lines. The parental Slx4+/– untagged cell line was used as a control. d-g. Quantification of cell survival assays of Slx4+/– and Slx4∆MLR/– clones in the presence of MMC (d), Olaparib (e) 5-aza-2′-deoxycytidine (f) Zebularine (g), Hydroxyurea (h) and Camptothecin (i). Data show mean values of three biologically independent replicates, n = 3. P-value: *p < 0.05. Analysis using unpaired Student’s t-test. Error bars: s.d. j. Representative images of metaphase spreads showing chromatid breaks (black arrows) and radial chromosomes (black arrowheads) in cells either untreated or treated with 20 ng/mL MMC for 12 h in Slx4∆SAP/– and Slx4∆SBD/– cells. k. and l. Quantitation of the number of breaks (k) and radial chromosomes (l) per metaphase nucleus in Slx4 mutant cells shown. Treated and untreated cells were harvested at the same time and each sample represented one independent experiment, n = 1. Data shows mean values of breaks or radials per genotype. Analysis using unpaired Student’s t-test. P-value: ****p < 0.0001.
Extended Data Fig. 7 The SLX4 MLR domain contributes to I-SceI-induced LTGC.
a, b. Tus/Ter-induced LTGC (a) and I-SceI-induced LTGC (b) in Slx4+/– and Slx4∆MLR/– clones. Data shows mean values of five biologically independent replicates, n = 5. P-value: **p < 0.01. Error bars: s.e.m. Analysis by ordinary one-way ANOVA. Error bars: s.e.m. c and d. Impact of exogenous hSLX4 alleles (see Fig. 5c) on Tus/Ter-induced LTGC (c) and on I-SceI-induced LTGC (d) in Slx4+/– and Slx4∆UBZ/– clones. EV: empty vector. 4 C > A: hSLX4 with inactivated UBZ motifs. Data shows mean values of six biologically independent replicates, n = 6. P-value: *p < 0.05; **p < 0.01; ***p < 0.00. Error bars: s.e.m. Analysis by unpaired Student’s t-test.
Extended Data Fig. 8 Characterization of Xpf+/– and Xpf∆Nuc/– cells.
a. CRISPR/Cas9 with dual sgRNA strategy for generation of Xpf+/– hemizygous cells. The Xpf– allele contains a 42.3 kb deletion between the 5’UTR and exon 11. Red half arrowheads: PCR and sequencing primers specific to 5’UTR and Exon 11. Predicted PCR product sizes for Xpf+ (wild type) allele shown. b. Gel shows PCR products using gDNA from Xpf+/+ and Xpf+/– clones. Note the 440 bp PCR product formed across the 42.3 kb deletion between the 5’UTR and exon 11 in Xpf+/– cells. c. RT qPCR analysis of mRNA in Xpf+/+ and Xpf+/– cells. Data shows mean of three independent experiments (n = 3) normalized to Gapdh mRNA using the 2–Δ∆CT method and analyzed by unpaired Student’s t-test. Error bars: s.d. d. Western blot of whole cell lysates in Xpf+/+ or Xpf+/– clones. siLuc and siErcc1 were compared in Xpf+/– cell line 48 hours after transfection to validate specificity of the Ercc1 band. e. and f. Quantification of colony formation of Xpf+/+ vs. Xpf+/– clones in the presence of MMC (e), Olaparib (f). Data show mean values of three biologically independent replicates, n = 3. Error bars: s.d. g. Tus/Ter-induced and I-SceI-induced HR frequencies in Xpf+/+ or Xpf+/– clones. Data shows mean values of four biologically independent replicates, n = 4. Error bars: s.e.m. Analysis by unpaired Student’s t-test. Error bars: s.e.m h. RT-qPCR analysis of mRNA encoding the XPF helicase, nuclease and Hh2h domains in 4 isogenic Xpf +/– clones and 4 Xpf∆Nuc/– clones. Data normalized to Gapdh mRNA using the 2–ΔCT method, mRNA expression in Xpf∆Nuc/– was normalized to Xpf +/– of the same experiment. Each data point is an average of three technical replicates. Data shows mean of three independent biological replicates (n = 3). Analysis by unpaired Student’s t-test. Error bars: s.d. P = 0.0153 for mRNA expression of the Nuclease region in Xpf+/− compared to Xpf∆Nuc/–. No significant differences were observed in expression levels of all other domains. i. Western blot analysis of ERCC1 in whole cell extract (WCE). *: non-specific band. β-tubulin: Loading control. j. Western blot analysis of ERCC1 in chromatin fraction of Xpf +/– and Xpf∆Nuc/– clones. H3: Histone H3 loading control. Note presence of ERCC1 signal in in-frame deleted Xpf∆Nuc/– clone #1, and absence of ERCC1 signal in frame-shifted Xpf∆Nuc/– clones #2, 11 and 13 k. Cell cycle distribution of Xpf+/– clone #6 and Xpf∆Nuc/– clone #1, either untreated or treated with 20, 30, 40 or 50 ng/mL of MMC. Data shows mean values of three biologically independent replicates, n = 3. P-value: **p < 0.01; ***p < 0.001; ****p < 0.0001. Error bars: s.e.m. Untreated samples were compared using the unpaired Student’s t-test; MMC-treated groups were compared by one-way ANOVA. l and m. Tus/Ter-induced LTGC (l) and I-SceI induced LTGC (m) in Xpf+/– and Xpf∆Nuc/– clones. Data shows mean values of six biologically independent replicates, n = 6. Error bars: s.e.m. Analysis by ordinary one-way ANOVA. P-value: ****p < 0.0001.
Supplementary information
Source data
Source Data Fig. 1
Unprocessed gels.
Source Data Fig. 1
Numerical source data.
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 2
Numerical source data.
Source Data Fig. 4
Numerical source data.
Source Data Fig. 5
Unprocessed gels.
Source Data Fig. 6
Unprocessed gels.
Source Data Fig. 6
Numerical source data.
Source Data Extended Data Fig. 1
Unprocessed gels.
Source Data Extended Data Fig. 1
Numerical source data.
Source Data Extended Data Fig. 2
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 2
Numerical source data.
Source Data Extended Data Fig. 4
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 5
Numerical source data.
Source Data Extended Data Fig. 6
Unprocessed Western blots and/or gels.
Source Data Extended Data Fig. 6
Numerical source data.
Source Data Extended Data Fig. 8
Unprocessed western blots and/or gels.
Source Data Extended Data Fig. 8
Numerical source data.
Rights and permissions
About this article
Cite this article
Elango, R., Panday, A., Lach, F.P. et al. The structure-specific endonuclease complex SLX4–XPF regulates Tus–Ter-induced homologous recombination. Nat Struct Mol Biol 29, 801–812 (2022). https://doi.org/10.1038/s41594-022-00812-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-022-00812-9
This article is cited by
-
Control of DNA replication in vitro using a reversible replication barrier
Nature Protocols (2024)
-
Inactivating TDP2 missense mutation in siblings with congenital abnormalities reminiscent of fanconi anemia
Human Genetics (2023)