Abstract
Pituitary adenylate cyclase-activating polypeptide (PACAP) is a pleiotropic neuropeptide hormone. The PACAP receptor PAC1R, which belongs to the class B G-protein-coupled receptors (GPCRs), is a drug target for mental disorders and dry eye syndrome. Here, we present a cryo-EM structure of human PAC1R bound to PACAP and an engineered Gs heterotrimer. The structure revealed that transmembrane helix TM1 plays an essential role in PACAP recognition. The extracellular domain (ECD) of PAC1R tilts by ~40° compared with that of the glucagon-like peptide-1 receptor (GLP-1R) and thus does not cover the peptide ligand. A functional analysis demonstrated that the PAC1R ECD functions as an affinity trap and is not required for receptor activation, whereas the GLP-1R ECD plays an indispensable role in receptor activation, illuminating the functional diversity of the ECDs in class B GPCRs. Our structural information will facilitate the design and improvement of better PAC1R agonists for clinical applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data Availability
Source data for Figs. 3c and 5b,c,e and Extended Data Fig. 1a are available with the online version of this paper. Values of the fluorescence intensities in Extended Data Fig. 1b,c are not shown, because each data point is shown in the graphs. Atomic coordinates for the PAC1R−mini-Gsβ1γ2−Nb35 complex have been deposited in the Protein Data Bank under PDB 6LPB. The associated electron microscopy data have been deposited in the Electron Microscopy Data Bank and Electron Microscopy Public Image Archive, under accession codes EMD-0940 and EMPIAR-10351.
References
Miyata, A. et al. Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem. Biophys. Res. Commun. 164, 567–574 (1989).
Vaudry, D. et al. Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol. Rev. 61, 283–357 (2009).
Hashimoto, H., Ishihara, T., Shigemoto, R., Mori, K. & Nagata, S. Molecular cloning and tissue distribution of a receptor for pituitary adenylate cyclase-activating polypeptide. Neuron 11, 333–342 (1993).
Doan, N.-D. et al. Design and in vitro characterization of PAC1/VPAC1-selective agonists with potent neuroprotective effects. Biochem. Pharmacol. 81, 552–561 (2011).
Ressler, K. J. et al. Post-traumatic stress disorder is associated with PACAP and the PAC1 receptor. Nature 470, 492–497 (2011).
Nakamachi, T. et al. PACAP suppresses dry eye signs by stimulating tear secretion. Nat. Commun. 7, 12034 (2016).
Castro, M., Nikolaev, V. O., Palm, D., Lohse, M. J. & Vilardaga, J.-P. Turn-on switch in parathyroid hormone receptor by a two-step parathyroid hormone binding mechanism. Proc. Natl Acad. Sci. USA 102, 16084–16089 (2005).
Zhao, L.-H. et al. Differential requirement of the extracellular domain in activation of class B G protein-coupled receptors. J. Biol. Chem. 291, 15119–15130 (2016).
Kumar, S., Pioszak, A., Zhang, C., Swaminathan, K. & Xu, H. E. Crystal structure of the PAC1R extracellular domain unifies a consensus fold for hormone recognition by class B G-protein coupled receptors. PLoS One 6, e19682 (2011).
Inoue, A. et al. Illuminating G-protein-coupling selectivity of GPCRs. Cell 177, 1933–1947.e25 (2019).
García-Nafría, J., Lee, Y., Bai, X., Carpenter, B. & Tate, C. G. Cryo-EM structure of the adenosine A2A receptor coupled to an engineered heterotrimeric G protein. Elife 7, e35946 (2018).
Wootten, D., Simms, J., Miller, L. J., Christopoulos, A. & Sexton, P. M. Polar transmembrane interactions drive formation of ligand-specific and signal pathway-biased family B G protein-coupled receptor conformations. Proc. Natl Acad. Sci. USA. 110, 5211–5216 (2013).
García-Nafría, J. & Tate, C. G. Cryo-EM structures of GPCRs coupled to Gs, Gi and Go. Mol. Cell Endocrinol. 488, 1–13 (2019).
Dautzenberg, F. M. et al. N-terminal splice variants of the type I PACAP receptor: isolation, characterization and ligand binding/selectivity determinants. J. Neuroendocrinol. 11, 941–949 (2001).
Inooka, H. et al. Conformation of a peptide ligand bound to its G-protein coupled receptor. Nat. Struct. Biol. 8, 161–165 (2001).
Zhao, L.-H. et al. Structure and dynamics of the active human parathyroid hormone receptor-1. Science 364, 148–153 (2019).
Zhang, H. et al. Structure of the glucagon receptor in complex with a glucagon analogue. Nature 553, 106–110 (2018).
Jazayeri, A. et al. Crystal structure of the GLP-1 receptor bound to a peptide agonist. Nature 546, 254–258 (2017).
Zhang, Y. et al. Cryo-EM structure of the activated GLP-1 receptor in complex with a G protein. Nature 546, 248–253 (2017).
Liang, Y.-L. et al. Cryo-EM structure of the active, Gs-protein complexed, human CGRP receptor. Nature 561, 492–497 (2018).
Bailey, R. J. & Hay, D. L. Agonist-dependent consequences of proline to alanine substitution in the transmembrane helices of the calcitonin receptor. Br. J. Pharmacol. 151, 678–687 (2007).
Conner, A. C. et al. A key role for transmembrane prolines in calcitonin receptor-like receptor agonist binding and signalling: implications for family B G-protein-coupled receptors. Mol. Pharmacol. 67, 20–31 (2005).
Venkatakrishnan, A. J. et al. Molecular signatures of G-protein-coupled receptors. Nature 494, 185–194 (2013).
Liang, Y.-L. et al. Phase-plate cryo-EM structure of a class B GPCR–G-protein complex. Nature 546, 118–123 (2017).
Hilger, D. et al. Structural insights into ligand efficacy and activation of the glucagon receptor. Preprint at bioRxiv https://doi.org/10.1101/660837 (2019).
Shihoya, W. et al. Activation mechanism of endothelin ETB receptor by endothelin-1. Nature 537, 363–368 (2016).
Shihoya, W. et al. X-ray structures of endothelin ETB receptor bound to clinical antagonist bosentan and its analog. Nat. Struct. Mol. Biol. 24, 758–764 (2017).
Shihoya, W. et al. Crystal structures of human ETB receptor provide mechanistic insight into receptor activation and partial activation. Nat. Commun. 9, 4711 (2018).
Nagiri, C. et al. Crystal structure of human endothelin ETB receptor in complex with peptide inverse agonist IRL2500. Commun. Biol. 2, 236 (2019).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. Elife 7, e42166 (2018).
Zivanov, J., Nakane, T. & Scheres, S. H. W. A Bayesian approach to beam-induced motion correction in cryo-EM single-particle analysis. IUCrJ. 6, 5–17 (2019).
Liang, Y.-L. et al. Phase-plate cryo-EM structure of a biased agonist-bound human GLP-1 receptor–Gs complex. Nature 555, 121–125 (2018).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. Sect. D Biol. Crystallogr. 60, 2126–2132 (2004).
Wang, R. Y.-R. et al. Automated structure refinement of macromolecular assemblies from cryo-EM maps using Rosetta. Elife 5, e17219 (2016).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D. Biol. Crystallogr. 66, 213–221 (2010).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D. Biol. Crystallogr. 66, 12–21 (2010).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Schrage, R. et al. The experimental power of FR900359 to study Gq-regulated biological processes. Nat. Commun. 6, 10156 (2015).
Acknowledgements
We thank R. Danev and M. Kikkawa for setting up the cryo-EM infrastructure, K. Ogomori for technical assistance and K. Yamashita for model building. We also thank A. Inoue (Tohoku University, Japan) for technical assistance. This work was supported by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)) from the Japan Agency for Medical Research and Development (AMED) under grant no. JP19am01011115 (support no. 1109), AMED grants: the PRIME JP18gm5910013 (A.I.) and the LEAP JP18gm0010004 (A.I. and J.A.), JSPS KAKENHI grant nos. 16H06294 (O.N.), 17J30010 (W.S.), 30809421 (W.S.), 17K08264 (A.I.), 17H05000 (T.N.) and the National Institute of Biomedical Innovation.
Author information
Authors and Affiliations
Contributions
K.K. expressed and purified the mini-Gs heterotrimer and performed the complex formation, grid preparation and cryo-EM observation. W.S. designed the experiments, purified the receptor, established the preparation method for the mini-Gs heterotrimer and Nb35 and refined the structure. T.N. performed the cryo-EM data collection and single-particle analysis. A.I., F.M.N.K. and J.A. performed and oversaw the cell-based assays. The manuscript was mainly prepared by W.S., K.K. and A.I., with assistance from T.N. and O.N.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Katarzyna Marcinkiewicz was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
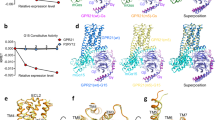
Extended Data Fig. 1 Functional characterization of mutant PAC1 receptors.
a, PACAP-induced Gs activation, measured by the NanoBiT-G-protein dissociation assay. Cells transiently expressing the NanoBiT-Gs, along with the indicated PAC1R construct, were treated with PACAP (1–38) and the change in the luminescent signal was measured. The dashed lines in the panels of ∆C and G389A indicate the Gs dissociation signal of the WT receptor. Concentration-response data are displayed as means ± s.e.m. (standard error of the mean) from four independent experiments. b, Cell surface expression of the PAC1R constructs. Cells transiently expressing the indicated FLAG epitope-tagged PAC1R constructs were labeled with an anti-FLAG tag antibody along with an Alexa488-conjugated secondary antibody, and the fluorescent signals from individual cells were measured by a flow cytometer. For the measurement of the WT-L, the plasmid encoding the WT receptor was reduced by 40-fold. c, Cell surface expression of the GLP-1R constructs, measured as in b. Data for graph in panel a are available as Source data.
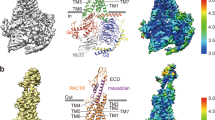
Extended Data Fig. 2 Cryo-EM analysis.
Flow chart of the cryo-EM data processing for the PACAP–PAC1R–mini-Gs complex, including particle projection selection, classification, and 3D density map reconstruction. Details are provided in the Methods section.
Extended Data Fig. 3 Map and model quality.
a, The cryo-EM density map and model are shown for PACAP, including all seven transmembrane α-helices, ECD, and α5 of Gαs. b, Dimer interface. The complexes are shown as ribbon representations, colored as in Fig. 1a. The side chains of V3185.48 and M3225.52 are shown as sticks. Two complexes form an anti-parallel dimer with C2 symmetry in the detergent micelles. This dimer does not reflect the physiological condition but is produced during the sample preparation. The molecular packing of the two complexes in the dimer class is mediated through only a weak hydrophobic contact between V3185.48 and M3225.52. Therefore, the dimerization minimally affects the conformation of the Gs-complexed PAC1R structure.
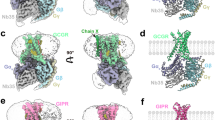
Extended Data Fig. 4 Comparison of peptide binding modes in class B GPCRs.
a-e, Ligand binding interactions with the TMDs in the class B GPCR structures (a, PAC1R, b, GCGR (5YQZ), c, PTH1R (6NBF), d, GLP-1R (5VAI), and e, CLR (6E3Y)). Hydrogen bonding interactions are indicated by black dashed lines. f-j, Relative positions of the peptide ligands and ECDs in the class B GPCR structures (f, PAC1R, g, GCGR, h, PTH1R, i, GLP-1R, and j, CLR).
Extended Data Fig. 5 G-protein coupling interface.
Complete interactions between PAC1R and the mini-Gs heterotrimer.
Extended Data Fig. 6 Structural comparison of PAC1R and PTH1R.
a–d, Surface representations of PAC1R (a) and three conformations of PTH1R (b–d) (PDB 6NBF, 6NBH, and 6NBI), viewed from the extracellular side. The receptors are shown as ribbon representations with transparent surfaces. The major conformation of PTH1R is colored orange-red, and the other conformations are colored light orange.
Supplementary information
Supplementary Information
Supplementary Table 1 and Supplementary Fig. 1.
Source data
Source Data Fig. 3
Raw data for Fig. 3c
Source Data Fig. 5
Raw data for Fig. 5b,c,e
Source Data Extended Data Fig. 1
Raw data for Extended Data Fig. 1a
Rights and permissions
About this article
Cite this article
Kobayashi, K., Shihoya, W., Nishizawa, T. et al. Cryo-EM structure of the human PAC1 receptor coupled to an engineered heterotrimeric G protein. Nat Struct Mol Biol 27, 274–280 (2020). https://doi.org/10.1038/s41594-020-0386-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-020-0386-8
This article is cited by
-
Class B1 GPCR activation by an intracellular agonist
Nature (2023)
-
Structure of the active Gi-coupled human lysophosphatidic acid receptor 1 complexed with a potent agonist
Nature Communications (2022)
-
Understanding VPAC receptor family peptide binding and selectivity
Nature Communications (2022)
-
A distinctive ligand recognition mechanism by the human vasoactive intestinal polypeptide receptor 2
Nature Communications (2022)
-
GPCR Intracellular Loop Regulation of Beta-Arrestin-Mediated Endosomal Signaling Dynamics
Journal of Molecular Neuroscience (2022)