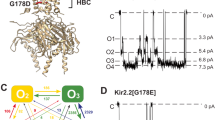
Abstract
Allosteric proteins transition among different conformational states in a ligand-dependent manner. Upon resolution of a protein’s individual states, one can determine the probabilities of these states, thereby dissecting the energetic mechanisms underlying their conformational changes. Here we examine individual regulator of conductance to K+ (RCK) domains that form the regulatory module of the Ca2+-activated MthK channel. Each domain adopts multiple conformational states differing on an ångström scale. The probabilities of these different states of the domain, assessed in different Ca2+ concentrations, allowed us to fully determine a six-state model that is minimally required to account for the energetic characteristics of the Ca2+-dependent conformational changes of an RCK domain. From the energetics of this domain, we deduced, in the framework of statistical mechanics, an analytic model that quantitatively predicts the experimentally observed Ca2+ dependence of the channel’s open probability.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data and materials described here will be made available upon reasonable request.
References
Jiang, Y. et al. Crystal structure and mechanism of a calcium-gated potassium channel. Nature 417, 515–522 (2002).
Ye, S., Li, Y., Chen, L. & Jiang, Y. Crystal structures of a ligand-free MthK gating ring: insights into the ligand gating mechanism of K+ channels. Cell 126, 1161–1173 (2006).
Pau, V. P. et al. Structure and function of multiple Ca2+-binding sites in a K+ channel regulator of K+ conductance (RCK) domain. Proc. Natl Acad. Sci. USA 108, 17684–17689 (2011).
Hill, A. V. The possible effects of the aggregation of the molcules of hemoglobin on its dissociation curves. J. Physiol. XL, iv–vii (1910).
Zadek, B. & Nimigean, C. M. Calcium-dependent gating of MthK, a prokaryotic potassium channel. J. Gen. Physiol. 127, 673–685 (2006).
Pau, V. P., Barca-Heidemann, K. & Rothberg, B. S. Allosteric mechanism of Ca2+ activation and H+-inhibited gating of the MthK K+ channel. J. Gen. Physiol. 135, 509–526 (2010).
Monod, J., Wyman, J. & Changeux, J. P. On the nature of allosteric transitions: a plausible model. J. Mol. Biol. 12, 88–118 (1965).
Marks, T. N. & Jones, S. W. Calcium currents in the A7r5 smooth muscle-derived cell line. An allosteric model for calcium channel activation and dihydropyridine agonist action. J. Gen. Physiol. 99, 367–390 (1992).
Zagotta, W. N., Hoshi, T. & Aldrich, R. W. Shaker potassium channel gating. III: evaluation of kinetic models for activation. J. Gen. Physiol. 103, 321–362 (1994).
Sakmann, B. & Neher, E. Single-Channel Recording (Plenum Press, New York, 1995).
Miller, C. Ion Channel Reconstitution (Plenum Press, New York, 1986).
Lewis, J. H. & Lu, Z. Resolution of ångström-scale protein conformational changes by analyzing fluorescence anisotropy. Nat. Struct. Mol. Biol. https://doi.org/10.1038/s41594-019-0274-2 (2019).
Smith, F. J., Pau, V. P., Cingolani, G. & Rothberg, B. S. Crystal structure of a Ba2+-bound gating ring reveals elementary steps in RCK domain activation. Structure 20, 2038–2047 (2012).
Lewis, J. H. & Lu, Z. Integrating spatiotemporal features of a ligand-regulated, multi-state allosteric protein. Nat. Struct. Mol. Biol. https://doi.org/10.1038/s41594-019-0276-0 (2019).
Acknowledgements
We thank Y. Zhou for technical support; Y. Jiang and R. MacKinnon for providing the cDNA of MthK; V. Pau and B. Rothberg for sharing their published data for comparison; and P. De Weer, T. Hoshi and B. Salzberg for critiques of our manuscript at different stages of its development. This study was supported by the grant no. GM055560 from the National Institute of General Medical Sciences of the National Institutes of Health to Z.L.
Author information
Authors and Affiliations
Contributions
J.H.L. and Z.L. designed the study; J.H.L. performed experiments, developed analytical tools, and analyzed the data, with the input from Z.L.; J.H.L. and Z.L. interpreted the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Inês Chen was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Crystal structure of the regulatory module of the MthK channel.
Ca2+ ions bind at three different sites, C1, C2 and C3, in individual RCK domains (PDB: 3RBZ).
Supplementary Figure 2 Comparison of models assuming different RCK conformations underlying the channel’s open states.
The three po-[Ca2+] curves calculated by assuming all-S2 species (blue, po), all-S1 (orange, PoS1), or both all-S1 and all-S2 species together (black, poS1,S2) underlie the open states. The black and blue curves are too close to be readily distinguished. For simplicity, gK was not included.
Supplementary information
Supplementary Information
Supplementary Figures 1–2, Supplementary Tables 1–2, Supplementary Notes 1–4
Rights and permissions
About this article
Cite this article
Lewis, J.H., Lu, Z. Energetics of ångström-scale conformational changes in an RCK domain of the MthK K+ channel. Nat Struct Mol Biol 26, 808–815 (2019). https://doi.org/10.1038/s41594-019-0275-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-019-0275-1
This article is cited by
-
Integrating spatiotemporal features of a ligand-regulated, multi-state allosteric protein
Nature Structural & Molecular Biology (2019)
-
Resolution of ångström-scale protein conformational changes by analyzing fluorescence anisotropy
Nature Structural & Molecular Biology (2019)