Abstract
The regulated exocytotic release of neurotransmitter and hormones is accomplished by a complex protein machinery whose core consists of SNARE proteins and the calcium sensor synaptotagmin-1. We propose a mechanism in which the lipid membrane is intimately involved in coupling calcium sensing to release. We found that fusion of dense core vesicles, derived from rat PC12 cells, was strongly linked to the angle between the cytoplasmic domain of the SNARE complex and the plane of the target membrane. We propose that, as this tilt angle increases, force is exerted on the SNARE transmembrane domains to drive the merger of the two bilayers. The tilt angle markedly increased following calcium-mediated binding of synaptotagmin to membranes, strongly depended on the surface electrostatics of the membrane, and was strictly coupled to the lipid order of the target membrane.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
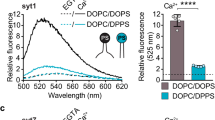
Source data for Figs. 2, 3a,b,d,e, and 4 are available with the paper online. The custom code used to acquire and analyze the data, as well other data in this study, is available from the corresponding author upon reasonable request.
References
Sabatini, B. L. & Regehr, W. G. Timing of neurotransmission at fast synapses in the mammalian brain. Nature 384, 170–172 (1996).
Schneggenburger, R. & Neher, E. Intracellular calcium dependence of transmitter release rates at a fast central synapse. Nature 406, 889–893 (2000).
Südhof, T. C. Neurotransmitter release: the last millisecond in the life of a synaptic vesicle. Neuron 80, 675–690 (2013).
Südhof, T. C. A molecular machine for neurotransmitter release: synaptotagmin and beyond. Nat. Med. 19, 1227–1231 (2013).
Jahn, R. & Fasshauer, D. Molecular machines governing exocytosis of synaptic vesicles. Nature 490, 201–207 (2012).
Jahn, R. & Scheller, R. H. SNAREs—engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 7, 631–643 (2006).
Weber, T. et al. SNAREpins: minimal machinery for membrane fusion. Cell 92, 759–772 (1998).
Geppert, M. et al. Synaptotagmin I: a major Ca2+ sensor for transmitter release at a central synapse. Cell 79, 717–727 (1994).
Stein, A., Radhakrishnan, A., Riedel, D., Fasshauer, D. & Jahn, R. Synaptotagmin activates membrane fusion through a Ca2+-dependent trans interaction with phospholipids. Nat. Struct. Mol. Biol. 14, 904–911 (2007).
Kiessling, V. et al. Rapid fusion of synaptic vesicles with reconstituted target SNARE membranes. Biophys. J. 104, 1950–1958 (2013).
Kyoung, M., Zhang, Y., Diao, J., Chu, S. & Brunger, A. T. Studying calcium-triggered vesicle fusion in a single vesicle-vesicle content and lipid-mixing system. Nat. Protoc. 8, 1–16 (2013).
Lee, H. K. et al. Dynamic Ca2+-dependent stimulation of vesicle fusion by membrane-anchored synaptotagmin 1. Science 328, 760–763 (2010).
Tucker, W. C., Weber, T. & Chapman, E. R. Reconstitution of Ca2+-regulated membrane fusion by synaptotagmin and SNAREs. Science 304, 435–438 (2004).
Malsam, J. et al. Complexin arrests a pool of docked vesicles for fast Ca2+-dependent release. EMBO J. 31, 3270–3281 (2012).
Südhof, T. C. & Rothman, J. E. Membrane fusion: grappling with SNARE and SM proteins. Science 323, 474–477 (2009).
Rizo, J. & Xu, J. The synaptic vesicle release machinery. Annu. Rev. Biophys. 44, 339–367 (2015).
Ledesma, M. D., Martin, M. G. & Dotti, C. G. Lipid changes in the aged brain: effect on synaptic function and neuronal survival. Prog. Lipid Res. 51, 23–35 (2012).
Ma, C., Su, L., Seven, A. B., Xu, Y. & Rizo, J. Reconstitution of the vital functions of Munc18 and Munc13 in neurotransmitter release. Science 339, 421–425 (2013).
Kreutzberger, A. J. B. et al. Reconstitution of calcium-mediated exocytosis of dense-core vesicles. Sci. Adv. 3, e1603208 (2017).
Liang, B., Dawidowski, D., Ellena, J. F., Tamm, L. K. & Cafiso, D. S. The SNARE motif of synaptobrevin exhibits an aqueous-interfacial partitioning that is modulated by membrane curvature. Biochemistry 53, 1485–1494 (2014).
Liang, B., Kiessling, V. & Tamm, L. K. Prefusion structure of syntaxin-1A suggests pathway for folding into neuronal trans-SNARE complex fusion intermediate. Proc. Natl. Acad. Sci. USA 110, 19384–19389 (2013).
Zdanowicz, R. et al. Complexin binding to membranes and acceptor t-SNAREs explains its clamping effect on fusion. Biophys. J. 113, 1235–1250 (2017).
Gong, J. et al. C-terminal domain of mammalian complexin-1 localizes to highly curved membranes. Proc. Natl. Acad. Sci. USA 113, E7590–E7599 (2016).
Dawidowski, D. & Cafiso, D. S. Allosteric control of syntaxin 1a by Munc18-1: characterization of the open and closed conformations of syntaxin. Biophys. J. 104, 1585–1594 (2013).
Cremona, O. & De Camilli, P. Phosphoinositides in membrane traffic at the synapse. J. Cell. Sci. 114, 1041–1052 (2001).
Holz, R. W. & Axelrod, D. Localization of phosphatidylinositol 4,5-P2 important in exocytosis and a quantitative analysis of chromaffin granule motion adjacent to the plasma membrane. Ann. NY Acad. Sci. 971, 232–243 (2002).
Milosevic, I. et al. Plasmalemmal phosphatidylinositol-4,5-bisphosphate level regulates the releasable vesicle pool size in chromaffin cells. J. Neurosci. 25, 2557–2565 (2005).
Martin, T. F. Role of PI(4,5)P2 in vesicle exocytosis and membrane fusion. Subcell. Biochem. 59, 111–130 (2012).
Kiessling, V. & Tamm, L. K. Measuring distances in supported bilayers by fluorescence interference-contrast microscopy: polymer supports and SNARE proteins. Biophys. J. 84, 408–418 (2003).
Lambacher, A. & Fromherz, P. Luminescence of dye moleculars on oxidized silicon and fluorescence interference contrast (FLIC) microscopy of biomembranes. J. Opt. Soc. Am. B 19, 1435–1453 (2002).
Kreutzberger, A. J., Liang, B., Kiessling, V. & Tamm, L. K. Assembly and comparison of plasma membrane SNARE acceptor complexes. Biophys. J. 110, 2147–2150 (2016).
Stein, A., Weber, G., Wahl, M. C. & Jahn, R. Helical extension of the neuronal SNARE complex into the membrane. Nature 460, 525–528 (2009).
Lai, A. L., Tamm, L. K., Ellena, J. F. & Cafiso, D. S. Synaptotagmin 1 modulates lipid acyl chain order in lipid bilayers by demixing phosphatidylserine. J. Biol. Chem. 286, 25291–25300 (2011).
Gaffaney, J. D., Dunning, F. M., Wang, Z., Hui, E. & Chapman, E. R. Synaptotagmin C2B domain regulates Ca2+-triggered fusion in vitro: critical residues revealed by scanning alanine mutagenesis. J. Biol. Chem. 283, 31763–31775 (2008).
Rizo, J. & Rosenmund, C. Synaptic vesicle fusion. Nat. Struct. Mol. Biol. 15, 665–674 (2008).
Pérez-Lara, Á. et al. PtdInsP2 and PtdSer cooperate to trap synaptotagmin-1 to the plasma membrane in the presence of calcium. eLife 5, e15886 (2016).
Park, Y. et al. Synaptotagmin-1 binds to PIP2-containing membrane but not to SNAREs at physiological ionic strength. Nat. Struct. Mol. Biol. 22, 815–823 (2015).
Zhou, Q. et al. Architecture of the synaptotagmin–SNARE machinery for neuronal exocytosis. Nature 525, 62–67 (2015).
Wang, S., Li, Y. & Ma, C. Synaptotagmin-1 C2B domain interacts simultaneously with SNAREs and membranes to promote membrane fusion. eLife 5, e14211 (2016).
McMahon, H. T., Kozlov, M. M. & Martens, S. Membrane curvature in synaptic vesicle fusion and beyond. Cell 140, 601–605 (2010).
Hui, E., Johnson, C. P., Yao, J., Dunning, F. M. & Chapman, E. R. Synaptotagmin-mediated bending of the target membrane is a critical step in Ca2+-regulated fusion. Cell 138, 709–721 (2009).
Wagner, M. L. & Tamm, L. K. Reconstituted syntaxin1a/SNAP25 interacts with negatively charged lipids as measured by lateral diffusion in planar supported bilayers. Biophys. J. 81, 266–275 (2001).
van den Bogaart, G. et al. Membrane protein sequestering by ionic protein-lipid interactions. Nature 479, 552–555 (2011).
Honigmann, A. et al. Phosphatidylinositol 4,5-bisphosphate clusters act as molecular beacons for vesicle recruitment. Nat. Struct. Mol. Biol. 20, 679–686 (2013).
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997).
Veatch, S. L. et al. Critical fluctuations in plasma membrane vesicles. ACS Chem. Biol. 3, 287–293 (2008).
Wang, J. et al. Calcium sensitive ring-like oligomers formed by synaptotagmin. Proc. Natl. Acad. Sci. USA 111, 13966–13971 (2014).
Shin, O. H. et al. Munc13 C2B domain is an activity-dependent Ca2+ regulator of synaptic exocytosis. Nat. Struct. Mol. Biol. 17, 280–288 (2010).
Snead, D., Wragg, R. T., Dittman, J. S. & Eliezer, D. Membrane curvature sensing by the C-terminal domain of complexin. Nat. Commun. 5, 4955 (2014).
Braun, D. & Fromherz, P. Fluorescence interferometry of neuronal cell adhesion on microstructured silicon. Phys. Rev. Lett. 81, 5241–5244 (1998).
Braun, D. & Fromherz, P. Fluorescence interference-contrast microscopy of cell adhesion on oxidized silicon. Appl. Phys., A Mater. Sci. Process 65, 341–348 (1997).
Crane, J. M., Kiessling, V. & Tamm, L. K. Measuring lipid asymmetry in planar supported bilayers by fluorescence interference contrast microscopy. Langmuir 21, 1377–1388 (2005).
Pabst, S. et al. Selective interaction of complexin with the neuronal SNARE complex. Determination of the binding regions. J. Biol. Chem. 275, 19808–19818 (2000).
Katti, S. et al. Non-native metal ion reveals the role of electrostatics in synaptotagmin 1–membrane interactions. Biochemistry 56, 3283–3295 (2017).
Domanska, M. K., Kiessling, V., Stein, A., Fasshauer, D. & Tamm, L. K. Single vesicle millisecond fusion kinetics reveals number of SNARE complexes optimal for fast SNARE-mediated membrane fusion. J. Biol. Chem. 284, 32158–32166 (2009).
Wagner, M. L. & Tamm, L. K. Tethered polymer-supported planar lipid bilayers for reconstitution of integral membrane proteins: silane-polyethyleneglycol-lipid as a cushion and covalent linker. Biophys. J. 79, 1400–1414 (2000).
Kalb, E., Frey, S. & Tamm, L. K. Formation of supported planar bilayers by fusion of vesicles to supported phospholipid monolayers. Biochim. Biophys. Acta 1103, 307–316 (1992).
van den Ent, F. & Löwe, J. RF cloning: a restriction-free method for inserting target genes into plasmids. J. Biochem. Biophys. Methods 67, 67–74 (2006).
Kiessling, V., Crane, J. M. & Tamm, L. K. Transbilayer effects of raft-like lipid domains in asymmetric planar bilayers measured by single molecule tracking. Biophys. J. 91, 3313–3326 (2006).
Acknowledgements
We thank A. Lambacher and P. Fromherz for providing FLIC analysis software and for their help with the production of FLIC substrates. This work was supported by US NIH grant P01 GM72694 to L.K.T., D.S.C., B.L., and V.K.
Author information
Authors and Affiliations
Contributions
V.K. performed and analyzed all of the sdFLIC experiments. A.J.B.K. performed and analyzed all of the DCV fusion experiments. B.L. and S.B.N. prepared protein samples. P.S. prepared shRNA for SytKD DCVs. V.K. designed the work. V.K., D.S.C., J.D.C., and L.K.T. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 SdFLIC example images and evaluation of different SNARE complexes in bPC/bPE/bPS/bPIP2/cholesterol.
a–c, Example sdFLIC images (second column) with a yellow grid of 12 × 12 oxides that were evaluated, extracted intensity data from one set of 16 oxides and FLIC fit curve (third column), and example histogram of fit results (fourth column) obtained from one sample under one condition for Syx*192/SNAP-25 (a), Syx*192/SNAP-25/Syb(1-96) (b), and Syx*192(183–288)/SNAP-25/Syb (c). Images were rotated and scaled for printing and show an area of 201 × 201 μm2.
Supplementary Figure 2 SdFLIC example images and evaluation for Syx*192/SNAP-25/Syb(1-96) in different lipid environments.
a–l, Example sdFLIC images with a yellow grid of 12 × 12 oxides that were evaluated, extracted intensity data from one set of 16 oxides and FLIC fit curve, and example histogram of fit results obtained from one sample under one condition for Syx*192/SNAP-25/Syb(1-96). Images in a–f were taken before and after the addition of Ca2+/C2AB in different lipid headgroup environments, as described in Fig. 1d and indicated in each panel. Images in g–l were taken in different lipid environments, as described in Fig. 2a and indicated in each panel. Images were rotated and scaled for printing and show an area of 201 × 201 μm2.
Supplementary Figure 3 SdFLIC example images and evaluation for Syx*192/SNAP-25/Syb(1-96) in different lipid environments (continued).
a–g, Example sdFLIC images with a yellow grid of 12 × 12 oxides that were evaluated, extracted intensity data from one set of 16 oxides and FLIC fit curve, and example histogram of fit results obtained from one sample under one condition for Syx*192/SNAP-25/Syb(1-96). Images were taken in different lipid environments, as described in Fig. 2a (a–d) and Fig. 2c (e–g), and as indicated in each panel. Images were rotated and scaled for printing and show an area of 201 × 201 μm2.
Supplementary Figure 4 SdFLIC and DCV fusion results obtained with different SNARE complex constructs and supported membrane control.
a, sdFLIC results from Syx*/SNAP-25/Syb(1-96) in bPC/bPE/bPS/bPIP2/cholesterol, labeled at residues 192 and 249 for comparison in EDTA (solid bars) and after addition of Ca2+/C2AB (open bars). b, sdFLIC results from Syb*28(1-96) after it was added to reconstituted Syx1a/SNAP-25 acceptor complex30 in different lipid environments as indicated in EDTA (solid bars) and after addition of Ca2+/C2AB (open bars). c, sdFLIC results from Syb*28(1-96) after binding to reconstituted Syx1a/SNAP-25 acceptor complex at different bPS concentrations with and without 1 mol% PIP2 as indicated. d, Distance of the lipid-bilayer surface from substrate as determined by FLIC in EDTA and after addition of Ca2+/C2AB. e, sdFLIC results from Syx*192/SNAP-25/Syb(1-96) in bPC/bPE/bPS/bPIP2/cholesterol in EDTA (solid bar) and after addition of C2AB/EDTA, Ca2+ and EDTA in successive order. f, sdFLIC and g, fusion probabilities when complexin, Munc18, or both have been added to form the ‘trigger-ready’ primed prefusion state19. When complexin is added to Syx*/SNAP-25/Syb(1-96), the complex moves slightly further away from the membrane. Adding Ca2+/C2AB straightens the complex in the same way as without complexin. When complexin is added to acceptor complex in the supported membrane, it inhibits Syb binding and DCV docking in the absence of Ca2+ (ref. 22). In the presence of Ca2+/C2AB, Syt-deficient DCVs dock and proceed to fusion with the supported membrane, similar to the WT DCV fusion in the presence of Ca2+ (ref. 19). When Munc18 and soluble Syb2 (Syb1-96) is added to reconstituted Syx*192/SNAP-25 (Fig. 1c), the measured distance of Syx*192 within this complex increases by more than 6 nm. When Ca2+/C2AB is added to this in the membrane assembled complex, its structure becomes more upright although the distance does not increase to the same height as in the isolated preassembled SNARE complex. Fusion of SytKD-DCVs in the absence of Ca2+ is increased by Munc18 and is further stimulated by Ca2+/C2AB. The height of Syx*192 within a complex that has been assembled by adding Munc18, complexin, and Syb1-96 to Syx*192/SNAP-25, is raised to 9.5 nm. When Ca2+/C2AB is added, the distance of Syx*192 from the membrane increases further to 11.6 nm, about the same as that of the cis-SNARE complex. Adding Munc18 and complexin at the same time to Syx1a/SNAP-25 in the supported membrane results in a ‘trigger competent’ prefusion state that allows DCV docking in the absence of Ca2+ without significant fusion19. Adding Ca2+/C2AB to the docked DCVs after a 15-min incubation time increases the fusion probability of the SytKD-DCVs from almost zero to 58%. Note that when multiprotein complexes are assembled on the membrane, the measured distance of Syx*192 most likely represents an average originating from different multiprotein assemblies. All data represent means from at least five independent experiments. Error bars represent s.e.m.
Supplementary Figure 5 SNARE and Ca2+/C2AB-dependent fusion in proteo-liposome/DCV fusion assay and Syx*192-membrane distance changes induced by C2AB-WT or the C2AB-KAKA mutant with and without PIP2.
a–d, Example (NBD) fluorescence dequenching curves after WT-DCVs have been added to Syx/SNAP-25-containing proteoliposomes in EDTA (black curves) or in the presence of Ca2+/C2AB. Syx/SNAP-25 is reconstituted into DP (a), PO (b), DO (c), or brain lipids (d). e, Average fluorescence increase due to NBD dequenching after proteoliposome/DCV fusion in EDTA (solid bars) and in the presence of Ca2+/C2AB. The lipid headgroup composition is PC/PE/PS/PIP2/cholesterol (34/30/15/1/20) in all cases, and the acyl chain composition is as indicated. Averages represent means from three repeats, and error bars represent s.d. f, Syx*192-membrane distance changes in response to C2AB-WT with (red) and without (orange) 1 mol% PIP2 and C2AB-KAKA with (light blue) and without (dark blue) 1 mol% PIP2. Data points represent means from at least five independent experiments. Error bars represent s.e.m.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5
Rights and permissions
About this article
Cite this article
Kiessling, V., Kreutzberger, A.J.B., Liang, B. et al. A molecular mechanism for calcium-mediated synaptotagmin-triggered exocytosis. Nat Struct Mol Biol 25, 911–917 (2018). https://doi.org/10.1038/s41594-018-0130-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41594-018-0130-9
This article is cited by
-
Mechanisms of SNARE proteins in membrane fusion
Nature Reviews Molecular Cell Biology (2024)
-
Synaptotagmin-7 outperforms synaptotagmin-1 to promote the formation of large, stable fusion pores via robust membrane penetration
Nature Communications (2023)
-
VAMP726 and VAMP725 regulate vesicle secretion and pollen tube growth in Arabidopsis
Plant Cell Reports (2023)
-
Ebola virus glycoprotein interacts with cholesterol to enhance membrane fusion and cell entry
Nature Structural & Molecular Biology (2021)
-
Conserved arginine residues in synaptotagmin 1 regulate fusion pore expansion through membrane contact
Nature Communications (2021)