Abstract
Besides generating vision, light modulates various physiological functions, including mood. While light therapy applied in the daytime is known to have anti-depressive properties, excessive light exposure at night has been reportedly associated with depressive symptoms. The neural mechanisms underlying this day–night difference in the effects of light are unknown. Using a light-at-night (LAN) paradigm in mice, we showed that LAN induced depressive-like behaviors without disturbing the circadian rhythm. This effect was mediated by a neural pathway from retinal melanopsin-expressing ganglion cells to the dorsal perihabenular nucleus (dpHb) to the nucleus accumbens (NAc). Importantly, the dpHb was gated by the circadian rhythm, being more excitable at night than during the day. This indicates that the ipRGC→dpHb→NAc pathway preferentially conducts light signals at night, thereby mediating LAN-induced depressive-like behaviors. These findings may be relevant when considering the mental health effects of the prevalent nighttime illumination in the industrial world.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study, besides what has been included in this manuscript (including the supplemental section), are available from the corresponding authors (T.X.) upon request.
Code availability
The customized codes used to generate the results are available from the corresponding authors upon reasonable request.
References
LeGates, T. A., Fernandez, D. C. & Hattar, S. Light as a central modulator of circadian rhythms, sleep and affect. Nat. Rev. Neurosci. 15, 443–454 (2014).
Vandewalle, G., Maquet, P. & Dijk, D. J. Light as a modulator of cognitive brain function. Trends Cogn. Sci. 13, 429–438 (2009).
Bais, B. et al. Bright light therapy in pregnant women with major depressive disorder: study protocol for a randomized, double-blind, controlled clinical trial. BMC Psychiatry 16, 381 (2016).
Golden, R. N. et al. The efficacy of light therapy in the treatment of mood disorders: a review and meta-analysis of the evidence. Am. J. Psychiatry 162, 656–662 (2005).
Lam, R. W. et al. Efficacy of bright light treatment, fluoxetine, and the combination in patients with nonseasonal major depressive disorder: a randomized clinical trial. JAMA Psychiatry 73, 56–63 (2016).
Bedrosian, T. A. & Nelson, R. J. Influence of the modern light environment on mood. Mol. Psychiatry 18, 751–757 (2013).
Zielinska-Dabkowska, K. M. Make lighting healthier. Nature 553, 274–276 (2018).
Scott, A. J., Monk, T. H. & Brink, L. L. Shiftwork as a risk factor for depression: a pilot study. Int J. Occup. Environ. Health 3, S2–S9 (1997).
Obayashi, K., Saeki, K., Iwamoto, J., Ikada, Y. & Kurumatani, N. Exposure to light at night and risk of depression in the elderly. J. Affect Disord. 151, 331–336 (2013).
Min, J. Y. & Min, K. B. Outdoor light at night and the prevalence of depressive symptoms and suicidal behaviors: a cross-sectional study in a nationally representative sample of Korean adults. J. Affect Disord. 227, 199–205 (2018).
Brzezinski, A. Melatonin in humans. N. Engl. J. Med 336, 186–195 (1997).
Fonken, L. K., Aubrecht, T. G., Melendez-Fernandez, O. H., Weil, Z. M. & Nelson, R. J. Dim light at night disrupts molecular circadian rhythms and increases body weight. J. Biol. Rhythms 28, 262–271 (2013).
Stevens, R. G. Working against our endogenous circadian clock: breast cancer and electric lighting in the modern world. Mutat. Res 680, 106–108 (2009).
Lyall, L. M. et al. Association of disrupted circadian rhythmicity with mood disorders, subjective wellbeing, and cognitive function: a cross-sectional study of 91 105 participants from the UK Biobank. Lancet Psychiatry 5, 507–514 (2018).
Vandewalle, G. et al. Spectral quality of light modulates emotional brain responses in humans. Proc. Natl Acad. Sci. USA 107, 19549–19554 (2010).
Fernandez, D. C. Light affects mood and learning through distinct retina-brain pathways. Cell 175, 71–84.e18 (2018).
Berson, D. M., Dunn, F. A. & Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 295, 1070–1073 (2002).
Do, M. T. & Yau, K. W. Intrinsically photosensitive retinal ganglion cells. Physiol. Rev. 90, 1547–1581 (2010).
Fu, Y. et al. Intrinsically photosensitive retinal ganglion cells detect light with a vitamin A-based photopigment, melanopsin. Proc. Natl Acad. Sci. USA 102, 10339–10344 (2005).
Hattar, S. et al. Central projections of melanopsin-expressing retinal ganglion cells in the mouse. J. Comp. Neurol. 497, 326–349 (2006).
Dunlap, J. C. Molecular bases for circadian clocks. Cell 96, 271–290 (1999).
Lowrey, P. L. & Takahashi, J. S. Genetics of the mammalian circadian system: photic entrainment, circadian pacemaker mechanisms, and posttranslational regulation. Annu Rev. Genet 34, 533–562 (2000).
Buhr, E. D. & Van Gelder, R. N. Local photic entrainment of the retinal circadian oscillator in the absence of rods, cones, and melanopsin. Proc. Natl Acad. Sci. USA 111, 8625–8630 (2014).
Zhang, J., Wang, H., Wu, S., Liu, Q. & Wang, N. Regulation of reentrainment function is dependent on a certain minimal number of intact functional ipRGCs in rd mice. J. Ophthalmol. 2017, 6804853 (2017).
Zingg, B. et al. AAV-mediated anterograde transsynaptic tagging: mapping corticocollicular input-defined neural pathways for defense behaviors. Neuron 93, 33–47 (2017).
Hu, H. Reward and aversion. Annu Rev. Neurosci. 39, 297–324 (2016).
Russo, S. J. & Nestler, E. J. The brain reward circuitry in mood disorders. Nat. Rev. Neurosci. 14, 609–625 (2013).
Hannibal, J. Neurotransmitters of the retino-hypothalamic tract. Cell Tissue Res 309, 73–88 (2002).
Whitt, J. P., Montgomery, J. R. & Meredith, A. L. BK channel inactivation gates daytime excitability in the circadian clock. Nat. Commun. 7, 10837 (2016).
Flourakis, M. et al. A conserved bicycle model for circadian clock control of membrane excitability. Cell 162, 836–848 (2015).
Rothen, N. & Meier, B. Time of day affects implicit memory for unattended stimuli. Conscious Cogn. 46, 1–6 (2016).
Russo, K. A. et al. Circadian control of the female reproductive axis through gated responsiveness of the RFRP-3 system to VIP signaling. Endocrinology 156, 2608–2618 (2015).
Goto, M., Oshima, I., Tomita, T. & Ebihara, S. Melatonin content of the pineal gland in different mouse strains. J. Pineal. Res. 7, 195–204 (1989).
Roseboom, P. H. et al. Natural melatonin ‘knockdown’ in C57BL/6J mice: rare mechanism truncates serotonin N-acetyltransferase. Brain Res Mol. Brain Res 63, 189–197 (1998).
Commons, K. G., Cholanians, A. B., Babb, J. A. & Ehlinger, D. G. The rodent forced swim test measures stress-coping strategy, not depression-like behavior. ACS Chem. Neurosci. 8, 955–960 (2017).
Reardon, S. Depression researchers rethink popular mouse swim tests. Nature 571, 456–457 (2019).
Molendijk, M. L. & de Kloet, E. R. Immobility in the forced swim test is adaptive and does not reflect depression. Psychoneuroendocrinology 62, 389–391 (2015).
Yang, Y. et al. Ketamine blocks bursting in the lateral habenula to rapidly relieve depression. Nature 554, 317–322 (2018).
Oishi, Y. et al. Slow-wave sleep is controlled by a subset of nucleus accumbens core neurons in mice. Nat. Commun. 8, 734 (2017).
Xu, Q. et al. A mouse model mimicking human first night effect for the evaluation of hypnotics. Pharm. Biochem Behav. 116, 129–136 (2014).
Menard, C. et al. Social stress induces neurovascular pathology promoting depression. Nat. Neurosci. 20, 1752–1760 (2017).
Huang, J. et al. A neuronal circuit for activating descending modulation of neuropathic pain. Nat. Neurosci. 22, 1659–1668 (2019).
Acknowledgements
The authors thank L.Q. Luo, X.K. Chen, Z.L. Qiu and X.H. Xu for their critical reading of the manuscript and their insightful advice. We also thank Y. Cai and X.Y. Zhou for their intellectual input and technical support. We thank members of the Neuroscience Pioneer Club for insightful discussions throughout the course of this study. This work was funded by the Strategic Priority Research Program of the Chinese Academy of Science (grant no. XDA16020603 to T.X.), the National Natural Science Foundation of China (grant nos. 81790644, 81925009 and 91432104 to T.X., 61890953 to J.B., 31601134 and 61727811 to H.Z., 91432104 to J.-T.C. and 81900855 to M.Z.), the Anhui Provincial Natural Science Foundation (grant no. 1708085QC57 to H.Z., 1808085MH289 to M.Z. and 1908085MC66 to J.-J.M.), the National Key Basic Research Program of China (grant no. 2016YFA0400900 to T.X.), the CAS priority research program (grant nos. XDPB10 and XDB02010000 to T.X.), the Innovative Program (grant no. ZT00102901 to H.Z.), the User with Excellence Program of Hefei Science Center CAS (grant no. 2019HSC-UE018 to T.X.), the Fundamental Research Funds for the Central Universities(grant no. WK2070000174 to T.X., M.Z. and Y.-Q. M.) and The Key Program in the Youth Elite Support Plan in Universities of Anhui Province (grant no. gxyqZD2018020 to Q.X.).
Author information
Authors and Affiliations
Contributions
T.X. conceived the idea. T.X., H.Z. and K.A. designed the study. K.A. performed all viral tracing, immunostaining and behavioral experiments with the help of Y.M., Y.-F.L. and J.-T.C. Y.-Q.M. and Y.-M.S. helped with tracing experiments. J.B. helped with setting up the actogram recording system. H.Z. and K.A. performed all electrophysiological recordings. J.-W.S. helped with in vivo photometry recordings and J.-J.M. analyzed photometry data. Q.X. performed the EEG recordings and analyzed the EEG data with the help of K.A. K.A. and H.Z. analyzed all other data and generated figures. T.X., H.Z, and K.A. interpreted the results with critical inputs from Y.-G.Y., M.Z. and Z.Z. H.Z., K.A. and T.X. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Mario Penzo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Supporting information related to Fig. 1.
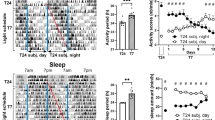
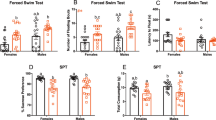
A) Immunostaining of Per1 in the SCN at different circadian times. Experiments were independently repeated 4 times for ZT 1 and 5, and 3 times for ZT 9, 13, 17, and 21. Scale: 50 μm. B) Development of depressive-like behavior, as measured in the FST, during chronic exposure to LAN. The immobility time (mean ± SD) progressively increased after 2 weeks (wks) under LAN conditions compared to the baseline before LAN treatment (Pre), and remained significantly elevated after 3 wks (n = 8 mice). Data obtained at 3 wks was plotted in Fig. 1. One-way ANOVA followed by Sidak’s test: F (3, 28) = 5.3, p = 0.0051. Pre vs 3wks, p = 0.0097; Pre vs 4wks, p = 0.0083. C-D) Persistence of 3wk LAN-induced depressive-like phenotype after 3wks, 6wks, and 9wks of recovery. LAN was given during the first 3 wks and removed afterwards. The immobility of LAN exposed animals (n = 10 mice) remained significantly higher than baseline at 6wks (or 3 wks of recovery), and returned to baseline level at 9-12 wks (or 6-9 wks of recovery, respectively). Repeated Measure One-way ANOVA with Tukey’s test: F (1.99, 17.91) = 23.71, p < 0.0001, Pre vs 3wks, p = 0.0008; Pre vs 6wks, p = 0.0030; 3wks vs 6wks, p = 0.1162, ns; 3wks vs 9wks, p = 0.0077; 3wks vs 12wks, p = 0.0005. Sucrose preference only recovered after 6wks (or 3wks of recovery). Repeat Measure One-way ANOVA with Tukey’s test: F(1.63, 14.67)=15.96, p = 0.0004. Pre vs 3wks, p = 0.0052; 3wks vs 6wks, p = 0.9034; 3wks vs 9wks, p = 0.1504; 3wks vs 12wks, p = 0.0003. E) Retinal projections to the pHb in WT and ipRGC-ablated animals. Retinal projections were visualized using intravitreally injected CTB-555. In WT animals, RGC terminals in the pHb were observed (n = 3 mice). Whereas in Opn4-Cre::rosa-DTA (DTA) animals, in which ipRGCs were ablated, retinal innervation to the pHb was completely eliminated (n = 3 mice). Scale: 100 μm. F) Retinal innervations of the SCN in WT and ipRGC-ablated animals. RGC terminals within the SCN were visualized using intravitreally-administered CTB-555.In WT animals, typical dense innervation of the SCN was observed (n = 3 mice). In DTA animals, the retinal innervation of the SCN was substantially diminished but not completely eliminated (n = 3 mice). The scarce innervation may come from regular RGCs or few residual ipRGCs, and was likely adequate for normal photo-entrainment of DTA animals. Bar: 100 μm. G) 3wks of LAN exposure did not affect overall locomotor activity level. Locomotor response to novelty (mean ± SD) was assessed in an open field apparatus (n = 10 mice) before (pre) and after (post) 3wk LAN exposure, both during the light phase (D) or the dark phase (N). One-way ANOVA: F (2.482, 22.34) = 0.8581, p = 0.4586. H) The effect of LAN was examined using FST conducted at night in the dark. LAN exposure for 3 wks (post/blue, in comparison to the baseline (pre/pink) before LAN) induced an elevation of immobility (mean ± SD) even when assessed during the dark phase. N = 10 mice, P = 0.0002, t = 5.85 df=9, two-tailed paired t-test. I) Total water consumption (mean ± SD) expressed as the ratio of total water consumption (water+sucrose) in SPT after LAN exposure (post) over that before LAN exposure (pre), corresponding to experiments in Figs. 1i, 1e, 1m, 1q, and 1u. None was significantly changed after LAN, with p values of 0.0532, 0.6930, 0.9683, 0.1770, and 0.0640, respectively, as determined by the two-tailed paired t-test comparing total consumptions pre vs. post within each group. **: p < 0.01; ***: p < 0.001.
Extended Data Fig. 2 Supporting information related to Fig. 2.
A) Schematic diagram of anterograde tracing with intravitreally-injected tracers. B) Retinal projections to the pHb area revealed by intravitreally-injected CTB-555 (b’) and HSV (b”), and ipRGC projections to the pHb revealed by AAV2-DIO-Channelrhodopsin (ChR2)-eYFP administered in Opn4-Cre mice (b”’). All tracing experiments were repeated at least three time. Scale: 100 μm. C) Schematic diagram of retrograde tracing from the pHb to retina. D) Retrogradely labeled (CTB, red) M1 ipRGCs (melanopsin), with dendrites stratified solely in the OFF sublamina of the retinal inner plexiform layer. Scale: 25 μm. E) Quantification of retrograde tracing results (contra: eye contralateral to the injection side; ipsi: ipsilateral eye; CTB: CTB only; Mela: CTB/melanopsin co-stained; n = 5 mice; contra: 18 cells; ipsi: 7 cells; Contra/mela: 75 cells; ipsi/mela: 6 cells). F) Subtype analysis of pHb-projecting ipRGCs judged from the stratification pattern (data were pooled from 4 retinae due to the difficulty of finding clearly stained dendrites and the cell body to which they belong on the same section. M1: 22 cells; M2: 2 cells; M3: 4 cells; Dislocated (DL): 2 cells). G) Fluorescence from CTB-555 at the injection site (pHb). Scale: 100 μm. H) Example of a M2 ipRGC that was stratified (arrowhead) in the ON sublamina of the retinal inner plexiform layer (IPL). Scale: 25 μm. I) Example of a M3 ipRGC that was stratified in both ON and OFF layers (arrowheads). Scale: 25 μm. J) Example of a dislocated ipRGC that appeared on the amacrine cell layer. Experiments in G-I were independently repeated four times in two mice. Scale: 25 μm.
Extended Data Fig. 3 Additional supporting information related to Fig. 2.
A) Histology for the optogenetic experiments in Fig. 2g–2i. Scale: 200 μm. B) Total water consumption (in grams/g, expressed as mean ± SD) corresponding to experiments in Figs. 2d and 2g, with p values of 0.4896, 0.2337 0.7212, and 0.9695, for the sham (n = 10 mice), lesion (n = 9 mice), viral (n = 19 mice), and ctrl (n = 13 mice) groups, respectively, as determined by two-sided paired t-test. C) Upregulation of cFos in the pHb elicited by the optostimulation of ipRGC terminals. Expression of cFos was induced by optostimulation of ipRGCs axonal terminals within the pHb. Selective expression of ChR2 in ipRGCs was achieved via intravitreal injection of AAV2/2-EF1α-DIO-ChR2-eYFP in Opn4-Cre mice (n = 5 sections). In sham-treated mice (littermates that received virus injection but no local opto-stimulation), minimum expression of cFos was observed (n = 6 sections). Parallel treated control mice in which virus injection was omitted did not respond to local optostimulation with elevated cFos expression (n = 7 sections). Bars: 50 μm. D) Schematic diagram of simultaneous retrograde tracing from NAc and mPFC using CTB-555 and CTB-488, respectively. E) Fluorescence at the injection sites: mPFC (green) and NAc (red). Experiments were independently repeated six times. Scale: 150 μm. F) Retrogradely-labelled NAc-projecting dpHb neurons (red dots) and mPFC-projecting vpHb neurons (blue dots) with rare overlaps (yellow dots). n = 6 mice. Data were expressed as mean ± SD. Scale: 100 μm.
Extended Data Fig. 4 Nested analysis of data presented in Figs. 3e and 3g.
A) Data presented in Fig. 3e were re-analyzed using nested ANOVA. Quantification of action potentials (mean ± SEM) generated by current injections in NAc-projecting dpHb neurons (Day: n = 10 neurons from 3 mice; Night: n = 20 neurons from 5 mice; Dark/day: 11 neurons from 4 mice; Light/nigh: 13 neurons from 4 mice). Nested ANOVA with Tukey’s test revealed significant difference among groups at: 60 pA: p = 0.0111, F (3,50) = 4.104 (*); 70 pA: p = 0.0050, F (3,50) = 4.825 (**); 80 pA: p = 0.0034, F (3,50) = 5.176 (**); 90pA: p = 0.0027, F (3,50) = 5.391 (**); ‡: “Day” significantly different from “Night” and “Light/night” significantly different from “dark/day”. B) Data presented in Fig. 3g were re-analyzed using nested two-sided t-test. Circadian variations in the intrinsic excitability of NAc-projecting dpHb neurons after 3 wk LAN exposure. n = 18 neurons from 4 mice for “Day (post 3w LAN)” and 22 neurons from 5 mice for “night (post 3w LAN)”. Data were plotted as mean ± SEM. #: Significant difference determined by nested t-test at 80 pA (p = 0.0296; t = 2.260, df=38) and 90 pA (p = 0.0258, t = 2.320, df=38). Data of “Day (no LAN)” and “Night (no LAN)” were replotted from A in light gray for easy comparison. Difference between Day (no LAN) and Night (no LAN) was annotated by *.
Extended Data Fig. 5 Supporting information related to Figs. 4–5.
A) Changes in total water consumption (mean ± SD) expressed as the ratio of the total water volume (water+sucrose) in the SPT after LAN exposure to that before LAN exposure, corresponding to experiments in 4e (n = 7 mice for mCherry and n = 8 mice for Tet-tox), 4f (n = 6 mice for mCherry/CNO, n = 7 mice for hM4D/PBS, and n = 9 mice for hM4D/CNO), and 4n (n = 9 mice for eGFP and n = 7 mice for Tet-tox). None was significantly altered by LAN exposure with p values of 0.0583, 0.6833, 0.1663, 0.9858, 0.3895, 0.2829, and 0.9468, respectively, as determined by two-tailed paired t-test comparing total consumptions pre vs. post within each group. B) (Related to experiments in Fig. 5a–d) Retrograde trans-synaptic tracing with RV revealed presynaptic inputs from IGL to NAc-projecting pHb neurons. The inter-geniculate leaflet (IGL, outlined) was densely innervated by ipRGCs. AAV2/2-EF1α-DIO-ChR2-eYFP was intravitreally injected to selectively express eYFP in ipRGCs, and intense labeling within the IGL was observed. dLGN: dorsal lateral geniculate nucleus. vLGN: ventral lateral geniculate nucleus. RV-labeled cells (arrowheads) were observed in the IGL. On average 26 ± 22 IGL neurons were observed on each side of each animal from a total of 6 mice. Bar: 100 μm.
Extended Data Fig. 6 Heat-maps for the photometric experiments in Fig. 5.
Heat-maps showing the day/night responses of all 16 clusters (8 NAc-projecting dpHb neuron clusters and 8 mPFC-projecting vpHb neuron clusters) in the photometry experiments. Within the same pair (day vs. night), heat-maps were drawn on the same color-scale for a clearer comparison between day and night responses. Across different pairs, the scales were different as marked on the side.
Extended Data Fig. 7 Supporting information related to Fig. 6.
A) Histology for the optogenetic manipulation of the dpHb-NAc projection in experiment 6d, e, f, and h. Trace of the optic fiber was outlined by the dotted line. Verification was independently performed 8 times. Scale: 100 μm. B) Histology showing NAc-projecting dpHb neurons expressing hM3D (red) and cFos (green) after i.p. injection of 1 mg/kg CNO, corresponding to experiments 6b, c, and g. Experiments were independently performed nine times. Scale: 100 μm. C) Changes in the total water consumption (mean ± SD) expressed as the ratio of the total water volume (water+sucrose) in the SPT after various stimulations (chemogenetic, optogenetic, or chronic CNO) to that during baseline period. Corresponding to experiments in Fig. 6b: n = 7 mice for hM3D, p = 0.6721; n = 8 mice for mCherry, p = 0.2153. Corresponding to experiments in Fig. 6c, n = 8 mice for hM3D/Day, p = 0.1846. Corresponding to experiments in Fig. 6d, n = 8 mice for ChR2, p = 0.3375; n = 7 mice for mCherry, p = 0.3517, all determined by the two-tailed paired t-test comparing total consumptions pre vs. post within each group, indicating no change before vs. after stimulations. Corresponding to experiments in Fig. 6j, n = 10 mice for GFP/CNO, p = 0.0204 (*); n = 10 mice for 3D/water, p = 0.1327; and n = 10 mice for 3D/CNO, p = 0.5638, all determined by the two-tailed paired t-test comparing total consumptions pre vs. post within each group, indicating a slight increase of total water consumption in the GFP/CNO group after CNO administration. *: p < 0.05.
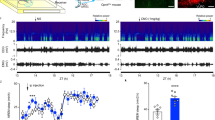
Extended Data Fig. 8 EEG recordings of mice during and after LAN exposure.
A) Diagram of strategy for inhibitory manipulation of the dpHb-NAc projection. Mice were randomly allocated to receiving injections of either DIO-Tet-tox (experimental group) or DIO-GFP (control group) into the pHb area. B) Experimental procedures for the EEG recordings during and after LAN exposure. Animals were allowed to recover for 3wks after stereotaxic injections of the viruses to ensure adequate habituation as well as virus expression, followed by EEG setup including electrode implantation and test runs. EEG was recorded during the last several days of LAN, after the termination of LAN exposure. C-D) Sleep-wake architecture of animals during LAN exposure. For both GFP (C, n = 3 mice) and Tet-tox (D, n = 3 mice) groups, the presence of light during the dark phase elevated the percentage of slow-wave sleep (blue open circle, mean ± SEM) and REM sleep (purple square, plotted to the right Y axis, mean ± SEM) but suppressed wakefulness (orange circle, mean ± SEM) in both GFP and Tet-tox mice. E-F) Both GFP and Tet-tox animals (n = 3 mice in each group) resumed normal sleep-wake architecture after the termination of 3wk LAN exposure. Data were expressed as mean ± SEM. G) FST of GFP and Tet-tox animals after LAN treatment. GFP mice (n = 5 mice, green) exhibited significantly higher immobility (mean ± SD) than Tet-tox animals (n = 4 mice, brown). p = 0.0142, t = 3.407 df=6.046, two-sided unpaired t-test with Welch’s correction. H) Sucrose preference (mean ± SD) of GFP mice was statistically lower than Tet-tox mice. p = 0.0003, t = 6.993 df=6.344, two-sided unpaired t-test with Welch’s correction. *: p < 0.05; ***: p < 0.001.
Supplementary information
Supplementary Information
Supplementary Fig. 1.
Rights and permissions
About this article
Cite this article
An, K., Zhao, H., Miao, Y. et al. A circadian rhythm-gated subcortical pathway for nighttime-light-induced depressive-like behaviors in mice. Nat Neurosci 23, 869–880 (2020). https://doi.org/10.1038/s41593-020-0640-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-020-0640-8
This article is cited by
-
CRF regulates pain sensation by enhancement of corticoaccumbal excitatory synaptic transmission
Molecular Psychiatry (2024)
-
The cognitive impact of light: illuminating ipRGC circuit mechanisms
Nature Reviews Neuroscience (2024)
-
A Comprehensive Overview of the Neural Mechanisms of Light Therapy
Neuroscience Bulletin (2024)
-
Potential Role of Bmal1 in Lipopolysaccharide-Induced Depression-Like Behavior and its Associated "Inflammatory Storm"
Journal of Neuroimmune Pharmacology (2024)
-
Light at night and cause-specific mortality risk in Mainland China: a nationwide observational study
BMC Medicine (2023)