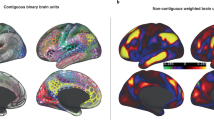
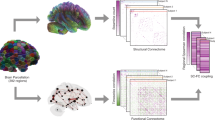
Abstract
Cognition and behavior emerge from brain network interactions, such that investigating causal interactions should be central to the study of brain function. Approaches that characterize statistical associations among neural time series—functional connectivity (FC) methods—are likely a good starting point for estimating brain network interactions. Yet only a subset of FC methods (‘effective connectivity’) is explicitly designed to infer causal interactions from statistical associations. Here we incorporate best practices from diverse areas of FC research to illustrate how FC methods can be refined to improve inferences about neural mechanisms, with properties of causal neural interactions as a common ontology to facilitate cumulative progress across FC approaches. We further demonstrate how the most common FC measures (correlation and coherence) reduce the set of likely causal models, facilitating causal inferences despite major limitations. Alternative FC measures are suggested to immediately start improving causal inferences beyond these common FC measures.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Valdes-Sosa, P. A., Roebroeck, A., Daunizeau, J. & Friston, K. Effective connectivity: influence, causality and biophysical modeling. Neuroimage 58, 339–361 (2011).
Ramsey, J. D. et al. Six problems for causal inference from fMRI. Neuroimage 49, 1545–1558 (2010).
Mill, R. D., Ito, T. & Cole, M. W. From connectome to cognition: The search for mechanism in human functional brain networks. Neuroimage 160, 124–139 (2017).
Cole, M. W., Yang, G. J., Murray, J. D., Repovš, G. & Anticevic, A. Functional connectivity change as shared signal dynamics. J. Neurosci. Methods 259, 22–39 (2016).
Smith, S. M. The future of FMRI connectivity. Neuroimage 62, 1257–1266 (2012).
Friston, K. J. Functional and effective connectivity: a review. Brain Connect. 1, 13–36 (2011).
Horwitz, B. The elusive concept of brain connectivity. Neuroimage 19, 466–470 (2003).
Korzybski, A. Science and Sanity: An Introduction to Non-Aristotelian Systems and General Semantics. (Institute of General Semantics, 1933).
Yeo, B. T. T. et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 106, 1125–1165 (2011).
Power, J. D. et al. Functional network organization of the human brain. Neuron 72, 665–678 (2011).
Power, J. D. & Petersen, S. E. Control-related systems in the human brain. Curr. Opin. Neurobiol. 23, 223–228 (2013).
Smith, S. M. et al. Correspondence of the brain’s functional architecture during activation and rest. Proc. Natl Acad. Sci. USA 106, 13040–13045 (2009).
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L. & Petersen, S. E. Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 59, 2142–2154 (2012).
Van Dijk, K. R. A., Sabuncu, M. R. & Buckner, R. L. The influence of head motion on intrinsic functional connectivity MRI. Neuroimage 59, 431–438 (2012).
Satterthwaite, T. D. et al. Impact of in-scanner head motion on multiple measures of functional connectivity: relevance for studies of neurodevelopment in youth. Neuroimage 60, 623–632 (2012).
Mehler, D.M.A. & Kording, K.P. The lure of causal statements: rampant mis-inference of causality in estimated connectivity. Preprint at arXiv https://arxiv.org/abs/1812.03363 (2018).
Pearl, J. A probabilistic calculus of actions. in Uncertainty Proceedings 1994 (eds. de Mantaras, R. L. & Poole, D.) 454–462 (Morgan Kaufmann, 1994).
Pearl, J. & Mackenzie, D. The Book of Why: The New Science of Cause and Effect. (Basic Books, 2018).
Pearl, J., Glymour, M. & Jewell, N. P. Causal Inference in Statistics: A Primer. (Wiley, 2016).
Robins, J. A new approach to causal inference in mortality studies with a sustained exposure period—application to control of the healthy worker survivor effect. Math. Model. 7, 1393–1512 (1986).
Spirtes, P., Glymour, C. & Scheines, R. Causation, Prediction, and Search. (MIT Press, 2000).
Marinescu, I. E., Lawlor, P. N. & Kording, K. P. Quasi-experimental causality in neuroscience and behavioural research. Nat. Hum. Behav. 2, 891–898 (2018).
Hume, D. An enquiry concerning human understanding (originally published 1748). in The Clarendon Edition of the Works of David Hume: An Enquiry Concerning Human Understanding (eds. Beauchamp, T. L., Hume, D. & Beauchamp, T. L.) 134–198 (Oxford University Press, 2000).
Pearl, J., Robins, J. M. & Greenland, S. Confounding and collapsibility in causal inference. Stat. Sci. 14, 29–46 (1999).
Friston, K. J. Functional and effective connectivity in neuroimaging: a synthesis. Hum. Brain Mapp. 2, 56–78 (1994).
Friston, K. J. et al. Psychophysiological and modulatory interactions in neuroimaging. Neuroimage 6, 218–229 (1997).
Roebroeck, A., Formisano, E. & Goebel, R. Mapping directed influence over the brain using Granger causality and fMRI. Neuroimage 25, 230–242 (2005).
Klahr, D. & Dunbar, K. Dual space search during scientific reasoning. Cogn. Sci. 12, 1–48 (1988).
Lee, H. S., Betts, S. & Anderson, J. R. Learning problem-solving rules as search through a hypothesis space. Cogn. Sci. 40, 1036–1079 (2016).
Smith, S. M. et al. Network modelling methods for FMRI. Neuroimage 54, 875–891 (2011).
Mill, R. D., Bagic, A., Bostan, A., Schneider, W. & Cole, M. W. Empirical validation of directed functional connectivity. Neuroimage 146, 275–287 (2017).
Wang, H. E. et al. A systematic framework for functional connectivity measures. Front. Neurosci. 8, 405 (2014).
Illari, P. M. & Williamson, J. What is a mechanism? Thinking about mechanisms across the sciences. Eur. J. Philos. Sci. 2, 119–135 (2012).
Hutchison, R. M. et al. Dynamic functional connectivity: promise, issues, and interpretations. Neuroimage 80, 360–378 (2013).
Lurie, D. et al. On the nature of resting fMRI and time-varying functional connectivity. Preprint at PsyArXiv https://doi.org/10.31234/osf.io/xtzre (2018).
Smith, S. M. et al. A positive-negative mode of population covariation links brain connectivity, demographics and behavior. Nat. Neurosci. 18, 1565–1567 (2015).
Schultz, D. H. & Cole, M. W. Higher intelligence is associated with less task-related brain network reconfiguration. J. Neurosci. 36, 8551–8561 (2016).
Cole, M. W., Yarkoni, T., Repovs, G., Anticevic, A. & Braver, T. S. Global connectivity of prefrontal cortex predicts cognitive control and intelligence. J. Neurosci. 32, 8988–8999 (2012).
Friston, K. J., Harrison, L. & Penny, W. Dynamic causal modelling. Neuroimage 19, 1273–1302 (2003).
Frässle, S. et al. A generative model of whole-brain effective connectivity. Neuroimage 179, 505–529 (2018).
Honey, C. J., Kötter, R., Breakspear, M. & Sporns, O. Network structure of cerebral cortex shapes functional connectivity on multiple time scales. Proc. Natl Acad. Sci. USA 104, 10240–10245 (2007).
Lohmann, G., Erfurth, K., Müller, K. & Turner, R. Critical comments on dynamic causal modelling. Neuroimage 59, 2322–2329 (2012).
Lewontin, R. C. The Genetic Basis of Evolutionary Change. (Columbia University Press, 1974).
Hodgkin, A. L. & Huxley, A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. (Lond.) 117, 500–544 (1952).
Hines, M. L. & Carnevale, N. T. The NEURON simulation environment. Neural Comput. 9, 1179–1209 (1997).
Goodman, D. & Brette, R. Brian: a simulator for spiking neural networks in python. Front. Neuroinform. 2, 5 (2008).
Ramsey, J. D., Hanson, S. J. & Glymour, C. Multi-subject search correctly identifies causal connections and most causal directions in the DCM models of the Smith et al. simulation study. Neuroimage 58, 838–848 (2011).
Hyttinen, A., Plis, S., Järvisalo, M., Eberhardt, F. & Danks, D. Causal discovery from subsampled time series data by constraint optimization. JMLR 52, 216–227 (2016).
Schubert, N. et al. 3D reconstructed cyto-, muscarinic M2 receptor, and Fiber architecture of the rat brain registered to the Waxholm Space Atlas. Front. Neuroanat. 10, 51 (2016).
Eickhoff, S. B. et al. A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25, 1325–1335 (2005).
Craddock, R. C., James, G. A., Holtzheimer, P. E. III, Hu, X. P. & Mayberg, H. S. A whole brain fMRI atlas generated via spatially constrained spectral clustering. Hum. Brain Mapp. 33, 1914–1928 (2012).
Buzsáki, G., Anastassiou, C. A. & Koch, C. The origin of extracellular fields and currents—EEG, ECoG, LFP and spikes. Nat. Rev. Neurosci. 13, 407–420 (2012).
Potjans, T. C. & Diesmann, M. The cell-type specific cortical microcircuit: relating structure and activity in a full-scale spiking network model. Cereb. Cortex 24, 785–806 (2014).
Logothetis, N. K. The neural basis of the blood-oxygen-level-dependent functional magnetic resonance imaging signal. Philos. Trans. R. Soc. Lond. B 357, 1003–1037 (2002).
Buxton, R. B., Wong, E. C. & Frank, L. R. Dynamics of blood flow and oxygenation changes during brain activation: the balloon model. Magn. Reson. Med. 39, 855–864 (1998).
Birn, R. M., Saad, Z. S. & Bandettini, P. A. Spatial heterogeneity of the nonlinear dynamics in the FMRI BOLD response. Neuroimage 14, 817–826 (2001).
Jellema, W. T. et al. Heterogeneity and prediction of hemodynamic responses to dobutamine in patients with septic shock. Crit. Care Med. 34, 2392–2398 (2006).
Tarantini, S., Tran, C. H. T., Gordon, G. R., Ungvari, Z. & Csiszar, A. Impaired neurovascular coupling in aging and Alzheimer’s disease: Contribution of astrocyte dysfunction and endothelial impairment to cognitive decline. Exp. Gerontol. 94, 52–58 (2017).
Handwerker, D. A., Ollinger, J. M. & D’Esposito, M. Variation of BOLD hemodynamic responses across subjects and brain regions and their effects on statistical analyses. Neuroimage 21, 1639–1651 (2004).
Di, X., Kannurpatti, S. S., Rypma, B. & Biswal, B. B. Calibrating BOLD fMRI activations with neurovascular and anatomical constraints. Cereb. Cortex 23, 255–263 (2013).
Rangaprakash, D., Wu, G.-R., Marinazzo, D., Hu, X. & Deshpande, G. Hemodynamic response function (HRF) variability confounds resting-state fMRI functional connectivity. Magn. Reson. Med. 80, 1697–1713 (2018).
Calhoun, V. D., Stevens, M. C., Pearlson, G. D. & Kiehl, K. A. fMRI analysis with the general linear model: removal of latency-induced amplitude bias by incorporation of hemodynamic derivative terms. Neuroimage 22, 252–257 (2004).
Schoffelen, J.-M. & Gross, J. Source connectivity analysis with MEG and EEG. Hum. Brain Mapp. 30, 1857–1865 (2009).
Salimi-Khorshidi, G. et al. Automatic denoising of functional MRI data: combining independent component analysis and hierarchical fusion of classifiers. Neuroimage 90, 449–468 (2014).
Sochat, V. et al. A robust classifier to distinguish noise from fMRI independent components. PLoS One 9, e95493 (2014).
Acharjee, P. P., Phlypo, R., Wu, L., Calhoun, V. D. & Adali, T. Independent vector analysis for gradient artifact removal in concurrent EEG-fMRI Data. IEEE Trans. Biomed. Eng. 62, 1750–1758 (2015).
Du, Y. et al. Artifact removal in the context of group ICA: A comparison of single-subject and group approaches. Hum. Brain Mapp. 37, 1005–1025 (2016).
Glasser, M. F. et al. Using temporal ICA to selectively remove global noise while preserving global signal in functional MRI data. Neuroimage 181, 692–717 (2018).
Buibas, M. & Silva, G. A. A framework for simulating and estimating the state and functional topology of complex dynamic geometric networks. Neural Comput. 23, 183–214 (2011).
Fuentes, L., Aldana, J.F. & Troya, J.M. GENESIS: an object-oriented framework for simulation of neural network models. in Artificial Neural Nets and Genetic Algorithms (eds. Pearson, D. W., Steele, N. C. & Albrecht, R. F.) 321–324 (Springer Vienna, 1995).
Ritter, P., Schirner, M., McIntosh, A. R. & Jirsa, V. K. The virtual brain integrates computational modeling and multimodal neuroimaging. Brain Connect. 3, 121–145 (2013).
Deco, G., Jirsa, V. K., Robinson, P. A., Breakspear, M. & Friston, K. The dynamic brain: from spiking neurons to neural masses and cortical fields. PLOS Comput. Biol. 4, e1000092 (2008).
David, O., Cosmelli, D. & Friston, K. J. Evaluation of different measures of functional connectivity using a neural mass model. Neuroimage 21, 659–673 (2004).
Gourévitch, B., Bouquin-Jeannès, R. L. & Faucon, G. Linear and nonlinear causality between signals: methods, examples and neurophysiological applications. Biol. Cybern. 95, 349–369 (2006).
Wang, Y., Katwal, S., Rogers, B., Gore, J. & Deshpande, G. Experimental validation of dynamic granger causality for inferring stimulus-evoked sub-100 ms timing differences from fMRI. IEEE Trans. Neural Syst. Rehabil. Eng. 25, 539–546 (2017).
Nee, D. E. & D’Esposito, M. Causal evidence for lateral prefrontal cortex dynamics supporting cognitive control. eLife 6, e28040 (2017).
Wheeler, M. E., Petersen, S. E. & Buckner, R. L. Memory’s echo: vivid remembering reactivates sensory-specific cortex. Proc. Natl Acad. Sci. USA 97, 11125–11129 (2000).
David, O. et al. Identifying neural drivers with functional MRI: an electrophysiological validation. PLoS Biol. 6, 2683–2697 (2008).
Smith, V. A., Yu, J., Smulders, T. V., Hartemink, A. J. & Jarvis, E. D. Computational inference of neural information flow networks. PLOS Comput. Biol. 2, e161 (2006).
Ryali, S. et al. Combining optogenetic stimulation and fMRI to validate a multivariate dynamical systems model for estimating causal brain interactions. Neuroimage 132, 398–405 (2016).
Lee, J. H. Informing brain connectivity with optogenetic functional magnetic resonance imaging. Neuroimage 62, 2244–2249 (2012).
Power, J. D., Schlaggar, B. L. & Petersen, S. E. Recent progress and outstanding issues in motion correction in resting state fMRI. Neuroimage 105, 536–551 (2015).
Power, J. D. et al. Methods to detect, characterize, and remove motion artifact in resting state fMRI. Neuroimage 84, 320–341 (2014).
Cole, M. W. et al. Task activations produce spurious but systematic inflation of task functional connectivity estimates. Neuroimage 189, 1–18 (2019).
Mumford, J. A. & Ramsey, J. D. Bayesian networks for fMRI: a primer. Neuroimage 86, 573–582 (2014).
Friston, K., Moran, R. & Seth, A. K. Analysing connectivity with Granger causality and dynamic causal modelling. Curr. Opin. Neurobiol. 23, 172–178 (2013).
Aertsen, A. M., Gerstein, G. L., Habib, M. K. & Palm, G. Dynamics of neuronal firing correlation: modulation of “effective connectivity”. J. Neurophysiol. 61, 900–917 (1989).
Cole, M. W., Ito, T., Bassett, D. S. & Schultz, D. H. Activity flow over resting-state networks shapes cognitive task activations. Nat. Neurosci. 19, 1718–1726 (2016).
Barnett, L. & Seth, A. K. The MVGC multivariate Granger causality toolbox: a new approach to Granger-causal inference. J. Neurosci. Methods 223, 50–68 (2014).
Bishop, C. M. Pattern Recognition and Machine Learning. (Springer, New York, 2016).
Rebane, G. & Pearl, J. The recovery of causal poly-trees from statistical data. Proceedings of the Third Workshop on Uncertainty in AI 222–228 (1987).
Schiefer, J. et al. From correlation to causation: Estimating effective connectivity from zero-lag covariances of brain signals. PLoS Comput. Biol. 14, e1006056 (2018).
Ramsey, J., Glymour, M., Sanchez-Romero, R. & Glymour, C. A million variables and more: the Fast Greedy Equivalence Search algorithm for learning high-dimensional graphical causal models, with an application to functional magnetic resonance images. Int. J. Data Sci. Anal. 3, 121–129 (2017).
Sanchez-Romero, R. et al. Estimating feedforward and feedback effective connections from fMRI time series: assessments of statistical methods. Netw. Neurosci. 3, 274–306 (2019).
Acknowledgements
The authors acknowledge the following support: National Institutes of Health grant R01MH107549 to L.Q.U.; National Institutes of Health grants R01MH109520 and R01AG055556 to M.W.C.; National Institutes of Health grants P20GM103472, R01EB020407, and NSF National Science Foundation grant 1539067 to V.C. The content is solely the responsibility of the authors and does not necessarily represent the official views of any of the funding agencies. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Reid, A.T., Headley, D.B., Mill, R.D. et al. Advancing functional connectivity research from association to causation. Nat Neurosci 22, 1751–1760 (2019). https://doi.org/10.1038/s41593-019-0510-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-019-0510-4
This article is cited by
-
Causation in neuroscience: keeping mechanism meaningful
Nature Reviews Neuroscience (2024)
-
Orthogonal neural encoding of targets and distractors supports multivariate cognitive control
Nature Human Behaviour (2024)
-
Consistent causal inference from time series with PC algorithm and its time-aware extension
Statistics and Computing (2024)
-
Identifying oscillatory brain networks with hidden Gaussian graphical spectral models of MEEG
Scientific Reports (2023)
-
Locating causal hubs of memory consolidation in spontaneous brain network in male mice
Nature Communications (2023)