Abstract
In vivo gene editing in post-mitotic neurons of the adult brain may be a useful strategy for treating neurological diseases. Here, we develop CRISPR–Cas9 nanocomplexes and show they were effective in the adult mouse brain, with minimal off-target effects. Using this system to target Bace1 suppressed amyloid beta (Aβ)-associated pathologies and cognitive deficits in two mouse models of Alzheimer’s disease. These results broaden the potential application of CRISPR–Cas9 systems to neurodegenerative diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The analysis of WGS, WES, and Digenome-seq data reported in this paper has been deposited in the NCBI SRA database (SRR5576254, SRR5576255, SRR8443475, SRR8443476, SRR5576256, SRR5576257, SRR8450353, SRR8450354).
References
Selkoe, D. J. Science 298, 789–791 (2002).
Thies, W. H. Alzheimers Dement. 11, 727–729 (2015).
Small, D. H., Mok, S. S. & Bornstein, J. C. Nat. Rev. Neurosci. 2, 595–598 (2001).
Fukumoto, H., Cheung, B. S., Hyman, B. T. & Irizarry, M. C. Arch. Neurol. 59, 1381–1389 (2002).
Serrano-Pozo, A., Frosch, M. P., Masliah, E. & Hyman, B. T. Cold Spring Harb. Perspect. Med. 1, a006189 (2011).
Yang, L. B. et al. Nat. Med. 9, 3–4 (2003).
Roberds, S. L. et al. Hum. Mol. Genet. 10, 1317–1324 (2001).
Wang, R. et al. Curr. Alzheimer Res. 9, 606–615 (2012).
Kang, E. L., Biscaro, B., Piazza, F. & Tesco, G. J. Biol. Chem. 287, 42867–42880 (2012).
Yin, H. et al. Nat. Biotechnol. 34, 328–333 (2016).
Long, C. et al. Science 351, 400–403 (2016).
Yin, H. et al. Nat. Biotechnol. 32, 551–553 (2014).
Tabebordbar, M. et al. Science 351, 407–411 (2016).
Nelson, C. E. et al. Science 351, 403–407 (2016).
Doran, A. G. et al. Genome Biol. 17, 167 (2016).
Oakley, H. et al. J. Neurosci. 26, 10129–10140 (2006).
Ma, H. et al. Nature 548, 413–419 (2017).
Saito, T. et al. Nat. Neurosci. 17, 661–663 (2014).
Kim, D. et al. Nat. Methods 12, 237–243 (2015).
Kim, E. et al. Nat. Commun. 8, 14500 (2017).
Kim, H. Y. et al. Nat. Commun. 6, 8997 (2015).
Swiech, L. et al. Nat. Biotechnol. 33, 102–106 (2015).
Yang, Y. et al. Nat. Biotechnol. 34, 334–338 (2016).
Acknowledgements
This work was supported by the National Research Foundation of Korea funded by the Ministry of Education, Science, and Technology (grant nos. NRF-2017M3A9C6029306 and 2016R1A2B2014195), Korea Health Technology R&D Project, Ministry of Health & Welfare (grant no. HI16C1176).
Author information
Authors and Affiliations
Contributions
H.P. conceived and designed the study, performed the experiments, and analyzed and interpreted the data. J.O., G.S., Y.C., S.K., S.B., H.K., J.S., H.C., J.Y., and J.K. performed the experiments and/or analyzed the data. B.C. designed the study and analyzed the data. M.L., W.J., C.L., Y.O., and J.K. conceived and designed the study, analyzed and interpreted the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information Nature Neuroscience thanks Ralph Nixon and other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Cas9 nanocomplex formation for the combined delivery of Cas9 proteins and sgRNAs.
(a) Schematic representation of the CRISPR-Cas9 nanocomplex delivery system. i) Formation of CRISPR-Cas9 nanocomplex. ii) Delivery of Cas9 nanocomplexes into in vivo brain. iii) Gene editing in in vivo post-mitotic neurons with Cas9 nanocomplex. (b) Gel retardation assay for R7L10 peptide binding to Cas9 and sgRNA. A fixed amount of sgRNA/Cas9 (0.6ug) was mixed with increasing amounts of the R7L10 peptide in deionized water. The weight ratio of Cas9 nanocomplexes for 1:1, 1:2, 1:3, 1:4, and 1:5 is the molar ratios of 1:23, 1:47, 1:70, 1:93, and 1:116, respectively. (c) Fluorescence quenching assay in Supplementary Fig 1b. Data are expressed as mean ± SEM, n = 3 per group. (d) Heparin competition assay of Cas9 nanocomplex. Cas9 nanocomplex was incubated in the presence of indicated heparin for 1hour. (e) Fluorescence quenching assay in Supplementary Fig 1d. Data are expressed as mean ± SEM, n = 3 per group. (f) Gel retardation assay for R7L10 peptide binding to Cas9 and sgRNA at different incubation periods. sgRNA/Cas9 (0.6ug) was mixed with the R7L10 peptide with 1:3 weight ratio (1:70 moral ratio). (g) Particle size of Cas9 nanocomplex with a various weight ratio of Cas9/sgRNA: R7L10. Data represent mean ± SEM (n = 3 per group). (h) Zeta potential of Cas9 nanocomplex with a various weight ratio of Cas9/sgRNA: R7L10. Data represent mean ± SEM (n = 3 per group). (i) (Upper panel) TEM images of Cas9/sgRNA (left panel) and Cas9 nanocomplex (right panel) (n = 3). (Lower panel) AFM images of Cas9/sgRNA complex (left panel) and Cas9 nanocomplex (right panel) (n = 30 per group). (j) Cytotoxic effects of Cas9 nanocomplex was monitored in mouse fibroblasts. Cell death was quantified by MTT assay. The value indicates R7L10 concentration (1Cas9 RNP: 3R7L10=0.14μM:10μM) Data represent mean ± SEM of three independent experiments. (k) Western blot analysis of Cas9 proteins at 1-4 weeks post-injection of Cas9 nanocomplex in wild-type mouse brain. (n = 3)
Supplementary Figure 2 Cas9 nanocomplex-mediated gene targeting in vitro.
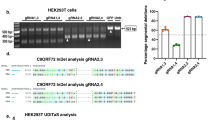
(a) InDel % derived from Surveyor assay in Fig. 1d. Cas9 nanocomplex (mouse Th and Bace1 sgRNA) was treated to mouse primary neurons. Data represent mean ± SEM, n = 3. (b) Deep sequencing of on-target InDel frequencies at the TH loci in Cas9 nanocomplex-treated human cells. (c) Deep sequencing of off-target InDel frequencies of top-three THsgRNA off-targets in Cas9 nanocomplex-treated human cells. (d) Surveyor assay of inDel mutations induced by Cas9 nanocomplex (Bace1 sgRNA and Th sgRNA) in mouse primary neurons. Data represent mean ± SEM. Three independent experiments were performed. (e) Surveyor assay of indel mutations induced by TH sgRNA Cas9 nanocomplex in hESCs (H9), hiPSCs (HSP0076), HEK293T, and hNSCs. Data represent mean ± SEM. Three independent experiments were performed.
Supplementary Figure 3 Homology-directed recombination via Cas9 nanocomplex.
(a) Schematic illustration depicting targeting strategy for the human TH locus. Primers used for PCR analysis are indicated by red and yellow arrows. Puro: puromycin resistance gene. (b) Representative image of TH PCR products in targeted neural stem cell-derived from human neurons. Genomic DNA from the targeted neural stem cell was amplified with primers shown in (a). (upper panel) The upper bands indicate the PCR fragments amplified with the knockin donor (about 3.1kb) and the lower bands indicate the PCR fragments without knockin donor (about 500 bp). (lower panel) The PCR fragments were amplified by the primers based on 5'arm and donor sequences (right) and 3’arm and donor sequences (left). (c) Sanger sequencing of GFP/Puro knock-in in human TH locus. Red colored sequences indicated GFP/Puro knock-in sequence by Cas9 (human TH sgRNA) nanocomplex. (d) Morphology of targeted human neuron stem cells (upper left) and differentiated neurons (upper right). Immunofluorescence of GFP in targeted human neurons. (e) Quantitative real-time PCR analysis of neuronal genes in targeted differentiated neurons. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (NSC vs. human neurons control, p = 0.001; NSC vs. Cas9 treated human neuron, p = 0.001 from all groups). Images in b, and d are representative of ≥ 3 similar experiments.
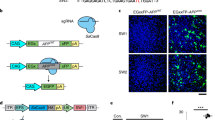
Supplementary Figure 4 Cas9 nanocomplex gene targeting in in vivo mouse brain.
(a) Schematic diagram showing the strategy for in vivo gene targeting by CRISPR-Cas9 nanocomplex. Cas9 nanocomplexes were injected into the cerebral cortex of Tau-eGFP and midbrains of Pitx3-eGFP knock-in (KI) adult mice (8 weeks old). (b) Quantification of Western blot analysis in Fig. 1g. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (Pitx3-eGFP control vs. Cas9 treated Pitx3-eGFP, p = 0.001; Tau-eGFP control vs. Cas9 treated Tau-eGFP, p = 0.001). (c) Expression of GFP in Cas9 nanocomplexes (GFPsgRNA) treated Pitx3-eGFP KI mice brain. (d) Quantification of GFP+ cells in control and Cas9 nanocomplex injected Pitx3-eGFP KI mouse and Tau-eGFP KI mouse brain. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test. Three brains slices with 40 μm thickness around the targeted region for each group were counted (Pitx3-eGFP, p = 0.001; Tau-eGFP, p = 0.001). (e) Fluorescence intensity of Bace1 and Th staining from immunohistochemistry data in Fig. 1f. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (Bace1, p = 0.001; Th, p = 0.001). (f) Immunohistochemistry for Th (green) and DAPI (White) in the Cas9 nanocomplex (ThsgRNA)-treated mouse midbrain. (g) Percentage of Th-immunoreactive cells in the midbrain nuclei. Data were expressed as mean ± SEM. **p < 0.01, ANOVA with Tukey’s post hoc test. Three independent experiments were performed with different mice (Cas9nano. ThsgRNA vs. Control, p = 0.001; Cas9nano. ThsgRNA vs. Sham, p = 0.001; Cas9nano. ThsgRNA vs. Control sgRNA Cas9nano., p = 0.001). (h) Surveyor assay for Cas9 nanocomplex mediated cleavage of Bace1 and Th in mouse brain. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (Bace1, p = 0.0018; Th, p = 0.0014). Images in c, and f are representative of ≥ 3 similar experiments.
Supplementary Figure 5 Immunogenicity assessment of Cas9 nanocomplex in vivo mouse brain.
(a) Immunohistochemistry for Gfap (green), Iba1 (red) and DAPI (blue) at 1day, 2eeks, 4weeks and 3months after injection of Cas9 nanocomplex in WT mouse hippocampus. (b) Immunohistochemistry for Gfap (green), Iba1 (red) and DAPI (blue) at 8weeks and 12weeks post-injection of Cas9 nanocomplex in 5XFAD mouse hippocampus. (c) Quantification of Gfap and Iba1 immunohistochemistry in Cas9 nanocomplex injected WT mouse. Data were expressed as mean ± SEM, n = 3. ANOVA with Tukey’s post hoc test (Gfap: Control vs. 1day, p = 0.942; Control vs. 2weeks, p = 0.974; Control vs. 4weeks, p = 0.96; Control vs. 3months, p = 0.999), (Iba1: Control vs. 1day, p = 0.861; Control vs. 2weeks, p = 0.989; Control vs. 4weeks, p = 0.993; Control vs. 3months, p = 0.999). (d) Quantification of Gfap and Iba1 immunohistochemistry in Cas9 nanocomplex injected 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. one-way analysis of variance (ANOVA) with Tukey’s post hoc test (Gfap: Control vs. 8weeks, p = 0.198; Control vs. 12weeks, p = 0.987), (Iba1: Control vs. 8weeks, p = 0.668; Control vs. 12weeks, p = 0.876). Images in a, and b are representative of ≥ 3 similar experiments.
Supplementary Figure 6 Cytotoxicity of Cas9 nanocomplex in vivo mouse brain.
(a) Immunohistochemistry for NeuN (green), Caspase3 (red) and DAPI (white) in Cas9 nanocomplex injected WT mouse hippocampus. Three independent experiments were performed. (b) Quantification of caspase3 immunohistochemistry in Supplementary fig. 10a. Data were expressed as mean ± SEM, n = 3. two-sided Student’s t-test (p = 0.37). (c) Measurement of blood urea nitrogen (BUN) at 1hour, 4hours, 24hours, 3weeks and 3months after post-injection of cas9 nanocomplex. Data were expressed as mean ± SEM, n = 3. Different letters above the error bars indicate statistical differences determined by ANOVA with Tukey’s post hoc test. p < 0.05 (Control vs. 1hour, p = 0.097; Control vs. 4hours, p = 0.006; Control vs. 24hours, p = 0.043; Control vs.3weeks, p = 0.961; Control vs. 3months, p = 0.821). (d) The concentration of Tnf-α and Il-12 were determined by ELISA in Cas9 nanocomplex treated mice. Data were expressed as mean ± SEM, n = 3. (e) mRNA expression of microglia marker including Itgam, Cd68, and Cd86 at 1weeks, 4weeks, 2months and 3months after post-injection of Cas9 nanocomplex. Data were expressed as mean ± SEM, n = 3.
Supplementary Figure 7 Aβ-associated pathologies and cognitive deficits in Cas9–Bace1 nanocomplex-treated 5XFAD mouse.
(a) Schematic diagrams of the in vivo genome targeting with Cas9 nanocomplexes for Alzheimer’s disease therapy. (b) Sanger sequencing of indel mutations induced by Bace1 sgRNA Cas9 nanocomplex in 6-months-old 5XFAD mouse hippocampus. (c) Quantification of western blot analysis on Fig. 2b. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (App, p = 0.391; Bace1, p = 0.001). (d) Western blot analysis of App, App CTFs (C99 and C83) and Actin in the hippocampus of Cas9 nanocomplex treated wild-type mouse and 5XFAD mouse. Quantification of the App and C99 western blot analysis (right panel). App C-terminal antibody is used to detect both App and App CTFs. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test. (e) Off-target indel frequencies of top-seven Bace1 off-targets (OF1~OF7) by deep sequencing in Cas9 nanocomplex treated mouse brain. Potential off-target sites were searched by the Cas-OFFinder software. (f) Immunohistochemistry for ThT (Aβ plaques depositions) in Cas9 nanocomplex (Bace1sgRNA) treated wild-type mouse hippocampus. (g) Immunohistochemistry for Aβ42 (red), ThT (green; referred to Aβ plaque) and DAPI (blue) in Cas9 nanocomplex (Bace1sgRNA) injected 6-months-old 5XFAD mouse hippocampus. (h) A number of Aβ plaque and Aβ42 positive cells in Cas9 nanocomplex injected 6 months old 5XFAD mouse hippocampus. Data are presented as mean ± SEM, n = 3. (i) DAB staining with Aβ42 in the hippocampus of Cas9 nanocomplex treated wild-type mice and 5XFAD mice. (j) A number of Aβ42 and Aβ42 plaque area of hippocampus and cortex region in cas9 nanocomplex treated WT and 5XFAD mice. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (5xFAD control vs. Cas9 treated 5XFAD in Hippocampus Aβ plaque number (%), p = 0.001; 5XFAD control vs. Cas9 treated 5XFAD in Hippocampus Aβ plaque area (%), p = 0.001; 5XFAD control vs. Cas9 treated 5XFAD in Cortex Aβ plaque number(%), p = 0.008; 5XFAD control. vs. Cas9 treated 5xFAD in Cortex Aβ plaque area(%), p = 0.001). (k) Y-maze test of Cas9 nanocomplex (Bace1sgRNA) treated 5XFAD Alzheimer’s disease mice. Y-maze test performed at age-matched WT control mouse, WT sham control, 5XFAD mouse sham control and 5XFAD mice treated with Cas9 nanocomplex. Data were expressed as mean ± SEM. *p < 0.05, ANOVA with Tukey’s post hoc test (5XFAD control vs. Cas9 treated 5XFAD, p = 0.02). (n = 9 for each group). Images in f, g, and i are representative of ≥ 3 similar experiments.
Supplementary Figure 8 The number of cleavage sites captured by Digenome-seq analysis on Cas9 nanocomplex (Bace1 sgRNA)-treated mice.
OF*, Off-target; OT*, On-target.
Supplementary Figure 9 Integrative genomics viewer presentation showing Bace1 potential off-target sites.
11 off-target sites were captured by Digenome-seq analysis.
Supplementary Figure 10
Integrative genomics viewer images showing the potential off-target sites captured by Cas9-OFFinder (http://www.rgenome.net/cas-offinder).
Supplementary Figure 11 Multiple deliveries of Cas9 nanocomplex in hippocampal areas.
(a) Immunohistochemistry for NeuN (green), Bace1 (red) and DAPI (white) in multiple Cas9 nanocomplex injected mouse brain. Top panel; control Cas9 nanocomplex injection (One time injection at CA3 region), second panel; Bace1sgRNA-Cas9 nancomplex injection (One time injection at CA3 region), third panel; Bace1sgRNA- Cas9 nanocomplex injections (one time injection at CA3 and CA1 regions), fourth panel; Bace1sgRNA-Cas9 nanocomplex injection (Two time injections at CA3 region). (b) Quantification of Bace1 immunofluorescence intensity and cell numbers for Bace1. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (Bace1 intensity: Control vs. 1injection+CA3, and 1injection.+CA1, and CA3, p = 0.001; 2 injection vs. 1injection+CA3, p = 0.001; 2injection+CA3 vs. 1injection+CA3, p = 0.001), (% of Bace1+ cells: Control vs. 1injection+CA3, p = 0.001 ; Control vs. 1injection+CA1, and CA3, p = 0.002; 2injection vs. 1injection+CA3, p = 0.015 ; 2injection+CA3 vs. 1injection+CA3, p = 0.011). (c) Immunohistochemistry for ThT 5XFAD mouse. (d) A number of total Aβ plaques in multiple Cas9 nanocomplex injected 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (Control vs. 1injection+CA3, p = 0.001; Control vs. 1injection+CA1, and CA3, p = 0.001; 2injection+CA3 vs. 1injection+CA3, p = 0.007; 2injection+CA3 vs. 1injection+CA1, and CA3, p = 0.027). (e) Levels of Aβ42 measured by ELISA in multiple Cas9 nanocomplex injected 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (Control vs. 1injection+CA3, p = 0.087; Control vs. 1injection+CA1, and CA3, p = 0.008; Control vs. 2inject.ion+CA3, p = 0.033). (f) Alteration in Y-maze in multiple Cas9 nanocomplex injected 5XFAD mouse. Data were expressed as mean ± SEM. *p < 0.05, ANOVA with Tukey’s post hoc test. (n = 5 for each group). Images in a, and c are representative of ≥ 3 similar experiments.
Supplementary Figure 12 Aβ-associated pathologies and cognitive deficits in control Cas9 nanocomplex and Bace1 inhibitor-treated 5XFAD mouse.
(a) Immunohistochemistry for NeuN (green), Bace1 (red) and DAPI (white) in Cas9 nanocomplex (Control sgRNA)-injected 5XFAD mouse hippocampus. (b) Immunohistochemistry for ThT in Cas9 nanocomplex treated WT mouse and 5XFAD mouse. (c) A number of total Aβ plaques in Cas9 nanocomplex (Control sgRNA)-injected mouse. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (Control vs. 5XFAD control, p = 0.001; Control vs. Cas9 treated 5XFAD, p = 0.001). (d) Levels of Aβ42 measured by ELISA in Cas9 nanocomplex (Control sgRNA)-injected 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. two-sided Student’s t-test (p = 0.355). (e) Y-maze test of Cas9 nanocomplex (Control sgRNA)-injected 5xFAD mouse. The percentage of alteration in y-maze was measured. Data were expressed as mean ± SEM. **p < 0.01, ANOVA with Tukey’s post hoc test (WT control vs. 5XFAD control, p = 0.001; WT control vs. Cas9 treated 5XFAD, p = 0.001; 5XFAD control. vs. Cas9 treated 5XFAD, p = 0.376). (n = 5 for each group). (f) Schematic diagrams of Bace1 inhibitor (β-Secretase Inhibitor IV, 565788) treatment in 5XFAD mice. (g) Immunohistochemistry for ThT in Bace1 inhibitor-treated WT and 5XFAD mouse hippocampus. (h) A number of total Aβ plaques in Bace1 inhibitor-treated WT and 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. **p < 0.01, ANOVA with Tukey’s post hoc test (Cas9 treated 5XFAD vs. 5XFAD control, p = 0.001; Cas9 treated 5XFAD vs. 1/week inhibitor, p = 0.001; Cas9 treated 5XFAD vs. daily inhibitor, p = 0.001). (i) Levels of Aβ42 measured by ELISA in Bace1 inhibitor-treated 5XFAD mouse. Data were expressed as mean ± SEM. Different letters (a, b, and c) above the error bars indicate statistical differences determined by ANOVA with Tukey’s post hoc test. p < 0.05 (Cas9 treated 5XFAD vs. 5XFAD control, p = 0.033; Cas9 treated 5XFAD vs. 1/week inhibitor, p = 0.110; Cas9 treated 5XFAD vs. daily inhibitor, p = 0.175). (n = 3). (j) Y-maze test of Bace1 inhibitor-treated 5XFAD Alzheimer’s disease mice. Y-maze test performed with WT, Bace1 inhibitor-treated WT mouse, 5XFAD mouse, and Bace1 inhibitor-treated 5XFAD mouse. Data were expressed as mean ± SEM. Different letters (a, b, and c) above the error bars indicate statistical differences determined by ANOVA with Tukey’s post hoc test. p < 0.01 (WT mouse vs. 5XFAD control, p = 0.001; WT mouse vs. 5XFAD 1/week inhibitor, p = 0.001; WT mouse vs. 5XFAD daily inhibitor, p = 0.001; 5XFAD cas9. vs. 5XFAD con., p = 0.001; 5XFAD cas9. vs. 5XFAD 1/week inhibitor, p = 0.001; 5XFAD cas9. vs. 5XFAD daily inhibitor, p = 0.012). (n = 6 for each group). Images in a, b, and g are representative of ≥ 3 similar experiments.
Supplementary Figure 13 Longitudinal effects of Cas9 nanocomplex-injected 5XFAD mouse model.
(a) Immunostaining for ThT (Aβ plaques depositions) in Bace1sgRNA Cas9 nanocomplex injected WT mouse. (b) Hippocampus immunohistochemistry for ThT in Cas9 nanocomplex injected 5XFAD mice (6 months old) on 8 and 12 weeks post-injection. (c) A number of total Aβ plaques in Bace1 sgRNA Cas9 nanocomplex injected WT and 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (5XFAD at 8 weeks control vs. Cas9 treated 5XFAD at 8 weeks, p = 0.001; 5XFAD at 12 weeks control vs. Cas9 treated 5XFAD at 12 weeks, p = 0.001). (d) Levels of Aβ42 measured by ELISA in Cas9 nanocomplex injected 5XFAD mouse. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (5XFAD at 8 weeks control vs. Cas9 treated 5XFAD at 8 weeks, p = 0.001; 5XFAD at12 weeks control vs. Cas9 treated 5XFAD at 12 weeks, p = 0.001). (e) Y-maze test of Cas9 nanocomplex treated 5XFAD mouse. Y-maze test performed with WT control, sham control and 5XFAD mice with Cas9 nanocomplex. Data were expressed as mean ± SEM. *p < 0.05, ANOVA with Tukey’s post hoc test (5XFAD control vs. Cas9 treated 5XFAD at 8 weeks, p = 0.031; 5XFAD control. vs. Cas9 treated 5XFAD at 12weeks, p = 0.041), (n = 6 for each group). (f, g) Associative long-term spatial reference memory was measured with water maze. (f) Escape latency and distance moved during 60 seconds in water maze trial test. Data represent mean ± SEM. (n = 6 for each group). (g) Trained mice in 4 days of the trial test were tested for 60 sec. Control and Cas9 nanocomplex treated 5XFAD mice was shown as % of quadrant occupancy time during training. Upper panel presented as visited area and bottom panel presented quantification of quadrant occupancy analysis. Data were expressed as mean ± SEM. *p < 0.05, ANOVA with Tukey’s post hoc test (5XFAD control vs. Cas9 treated 5XFAD at 8week, p = 0.47). (n = 6 for each group). All behavior tests were conducted by a person blinded to the animal groups. Images in a, and b are representative of ≥ 3 similar experiments.
Supplementary Figure 14 Aβ-associated pathologies and cognitive deficits in Cas9–Bace1 nanocomplex-treated APPNL-G-F/NL-G-F knock-in mice model.
(a) Immunohistochemistry for ThT (Aβ plaques depositions) in cas9 nanocomplex injected WT mouse and 4month old AppNL-G-F/NL-G-F mouse. (b) A number of total Aβ plaques in mouse hippocampus. Data were expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (p = 0.008). (c) Immunohistochemistry for NeuN (green), Bace1 (red) and DAPI (white) in Cas9 nanocomplex WT and AppNL-G-F/NL-G-F. (d) Levels of Aβ42 measured by ELISA in Cas9 nanocomplex WT and AppNL-G-F/NL-G-F mouse. Data are expressed as mean ± SEM, n = 3. **p < 0.01, two-sided Student’s t-test (p = 0.001). (e) Y-maze test of Cas9 nanocomplex injected AppNL-G-F/NL-G-F mouse. The percentage of alteration in y-maze was measured. Data were expressed as mean ± SEM. *p < 0.05, ANOVA with Tukey’s post hoc test (App control vs. Cas9 treated App, p = 0.03). (n = 5 for each group). (f,g) Associative long-term spatial reference memory was measured with water maze. (f) Escape latency and distance moved during the 60s in water maze test. Data were expressed as mean ± SEM (n = 5 for each group). (g) Trained mice were tested for the 60s. Red color indicates the most frequently visited areas and the location of the platform is presented as a circle in the target region (upper panel). Data represent quantification of quadrant occupancy analysis. Data were expressed as mean ± SEM. Different letters (a, and b) above the error bars indicate statistical differences determined by ANOVA with Tukey’s post hoc test. p < 0.05 (App control vs. Cas9 treated App, p = 0.242). (n = 5 for each group). Images in a, and c are representative of ≥ 3 similar experiments.
Supplementary information
Rights and permissions
About this article
Cite this article
Park, H., Oh, J., Shim, G. et al. In vivo neuronal gene editing via CRISPR–Cas9 amphiphilic nanocomplexes alleviates deficits in mouse models of Alzheimer’s disease. Nat Neurosci 22, 524–528 (2019). https://doi.org/10.1038/s41593-019-0352-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-019-0352-0
This article is cited by
-
CRISPR base editing-mediated correction of a tau mutation rescues cognitive decline in a mouse model of tauopathy
Translational Neurodegeneration (2024)
-
Engineering self-deliverable ribonucleoproteins for genome editing in the brain
Nature Communications (2024)
-
Acoustically targeted noninvasive gene therapy in large brain volumes
Gene Therapy (2024)
-
Application of CRISPR/Cas9 System in the Treatment of Alzheimer’s Disease and Neurodegenerative Diseases
Molecular Neurobiology (2024)
-
Techniques for investigating lncRNA transcript functions in neurodevelopment
Molecular Psychiatry (2023)